Found 7 results
Open Access
Review
24 March 2025Recent Advances and Challenges in Engineering Metabolic Pathways and Cofactor Regeneration for Enhanced n-Butanol Biosynthesis
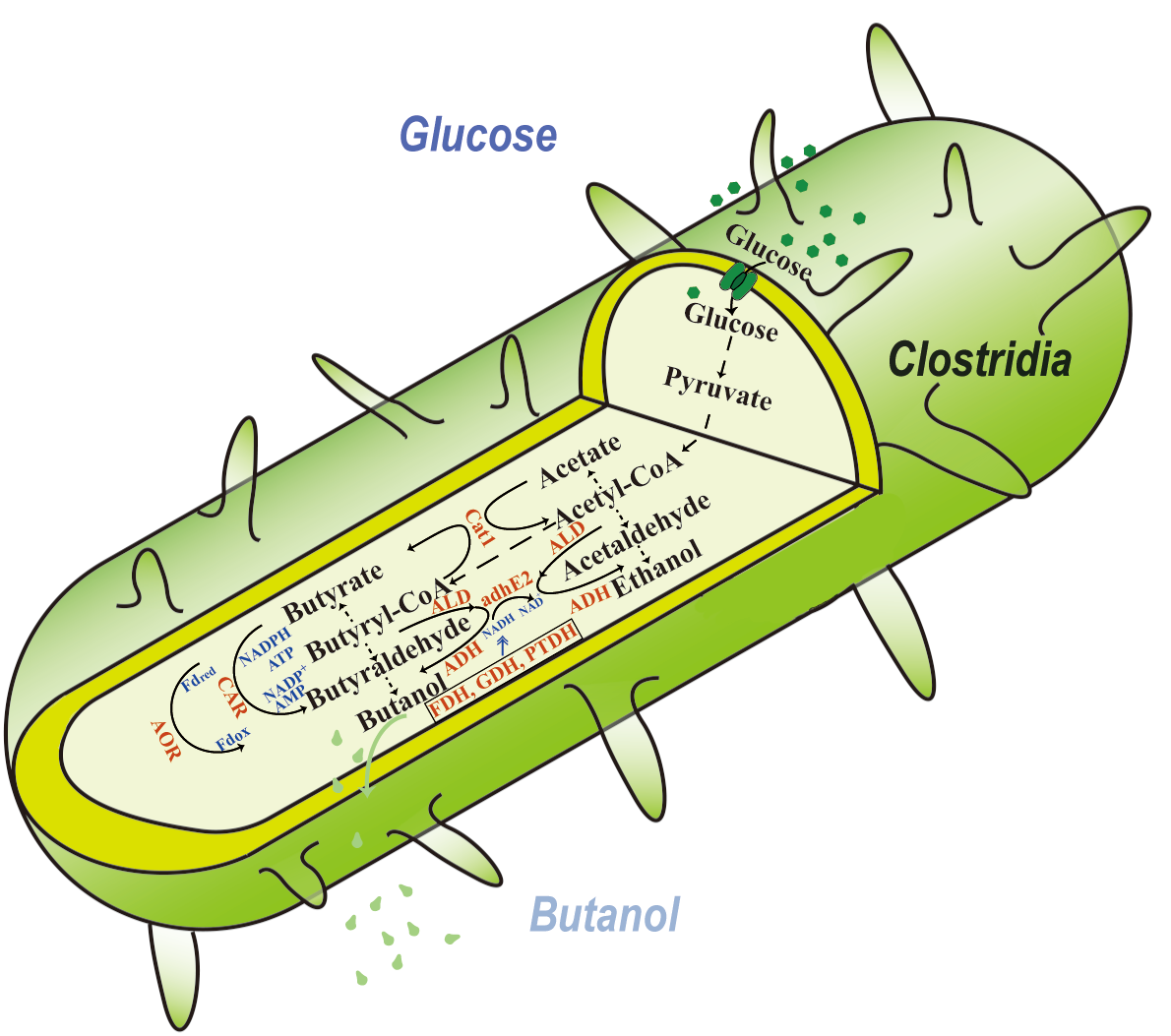
The biological production of n-butanol has seen renewed interest due to the need for the production of sustainable aviation fuel, for which n-butanol serves as a direct precursor. However, biological production of this alcohol is still limited by the fermentation’s low titers and low yields. Many approaches have been taken to increase n-butanol production, such as using alternative host organisms, utilizing heterologous enzymes for acid reduction and cofactor regeneration, and protein engineering of critical enzymes in the n-butanol production metabolic pathway. This review highlights key achievements made in each of these areas and shows the potential for these approaches in increasing n-butanol production. The review closes by pinpointing the challenges and limitations in these approaches and recommends that the ultimate approach to n-butanol production should inevitably utilize noncanonical redox cofactors to drive metabolic flux for butanol biosynthesis from glucose.
Open Access
Article
20 March 2025Metabolic Engineering and Genome-Wide Adaptive Evolution for Efficient Reduction of Glycerol in Industrial Saccharomyces cerevisiae
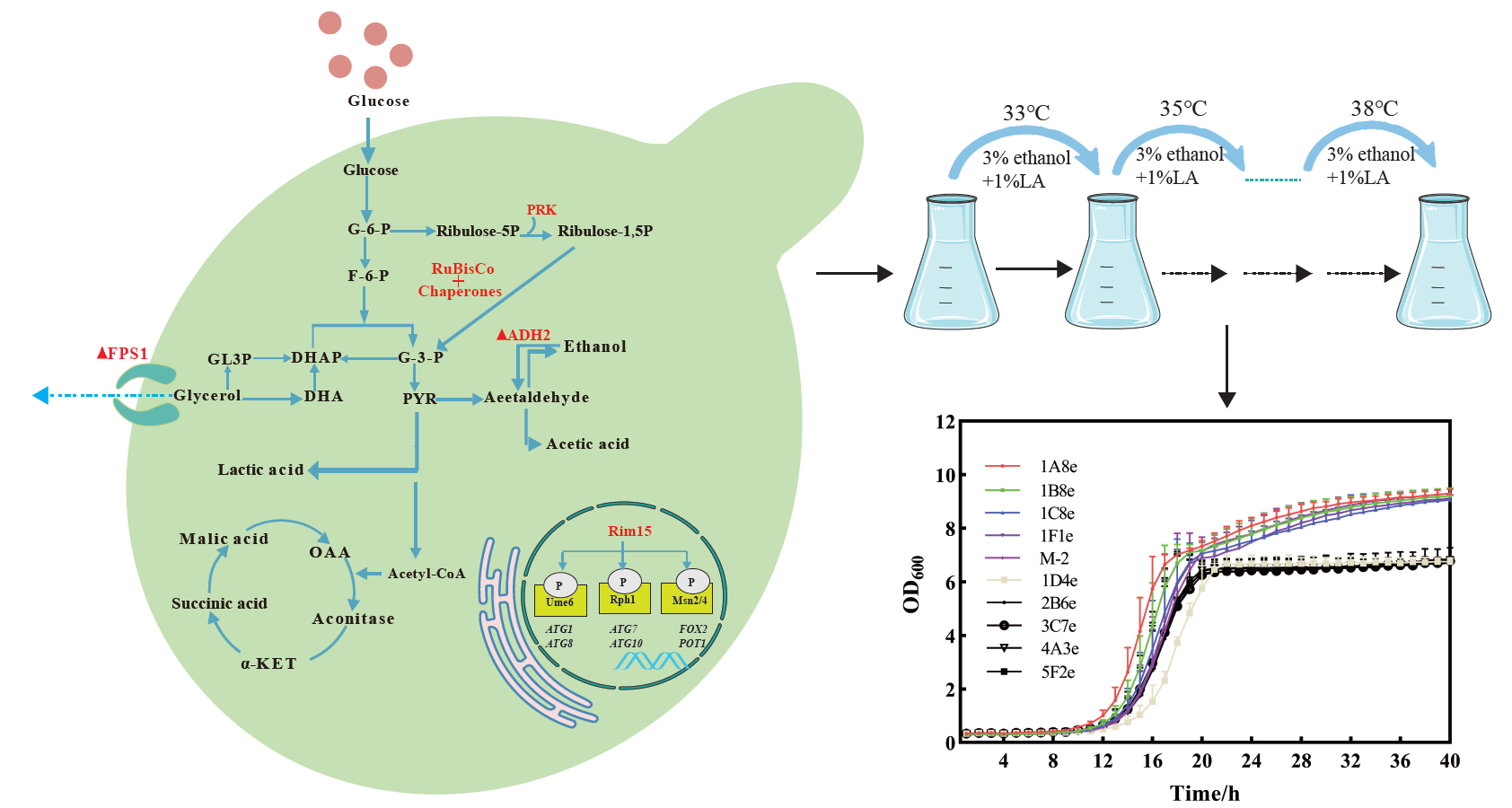
The production of glycerol as a major by-product during yeast-based bioethanol fermentation arises directly from the need to re-oxidize excess NADH, which reduces conversion efficiency. In this study, an optimized Cas9-based genome editing method was performed to develop a mixotrophic CO2-fixing industrial Saccharomyces cerevisiae by heterologous expression of ribulose-1,5-bisphosphate carboxylase-oxygenase (RuBisCO form Pseudomonas sp.) and phosphoribulokinase (PRK form Spinach). Additionally, the gene encoding alcohol dehydrogenase (ADH2) responsible for converting ethanol to acetaldehyde was deleted, while the great wall-family protein kinase Rim15 gene was overexpressed to facilitate the reduction in glycerol content. The resulting CO2-fixing yeast M-2 led to a 21.5% reduction of the by-product glycerol in corn mash fermentation cultures at 39 ℃. Moreover, we established a novel gene mutators mediated genome-wide mutations system that accumulates distinct mutations in the industrial S. cerevisiae strains under the stress conditions to improve the robustness in the S. cerevisiae strains efficiently.
Open Access
Review
13 February 2024Development and Perspective of Production of Terpenoids in Yeast
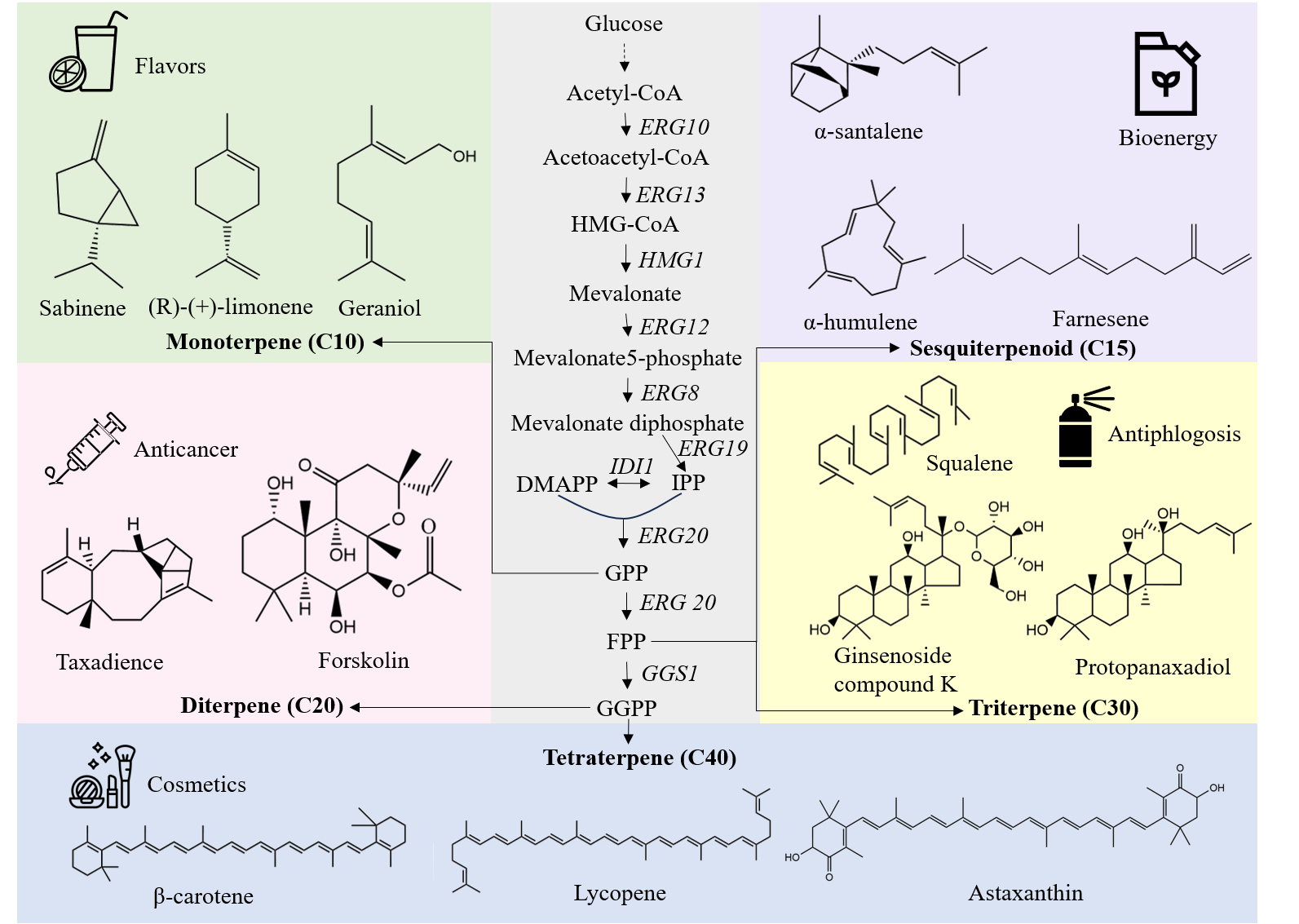
Terpenoids are a large class of secondary metabolites known for their remarkable diverse biological activities, making them widely utilized in the pharmaceutical, food, cosmetic, biofuel and agricultural fields. However, the current production of terpenoids heavily relies on plant extraction and chemical synthesis, which brings about concerns regarding infield, environmental and ecological issues. With the advancements in metabolic engineering and emerging synthetic biology tools, it is now possible to sustainably produce these high value-added terpenoids using microbial chassis. Among them, yeast has emerged as a promising candidate for the heterologous biosynthesis of terpenoids due to its inherent advantages, including robustness, safety, and the availability of sufficient precursor. This review focuses on the diverse strategies employed to enable terpenoids production in yeasts. These strategies encompass metabolic engineering approaches to optimize the mevalonate pathway, protein engineering techniques to improve terpenoid biosynthesis, the applications of organelles compartmentalization, high throughput screening and global approaches for the development of efficient cell factories. Furthermore, this review discusses the future prospects and challenges associated with yeast-based terpenoid production, while also emphasizing guidelines for future studies in this field.
Open Access
Article
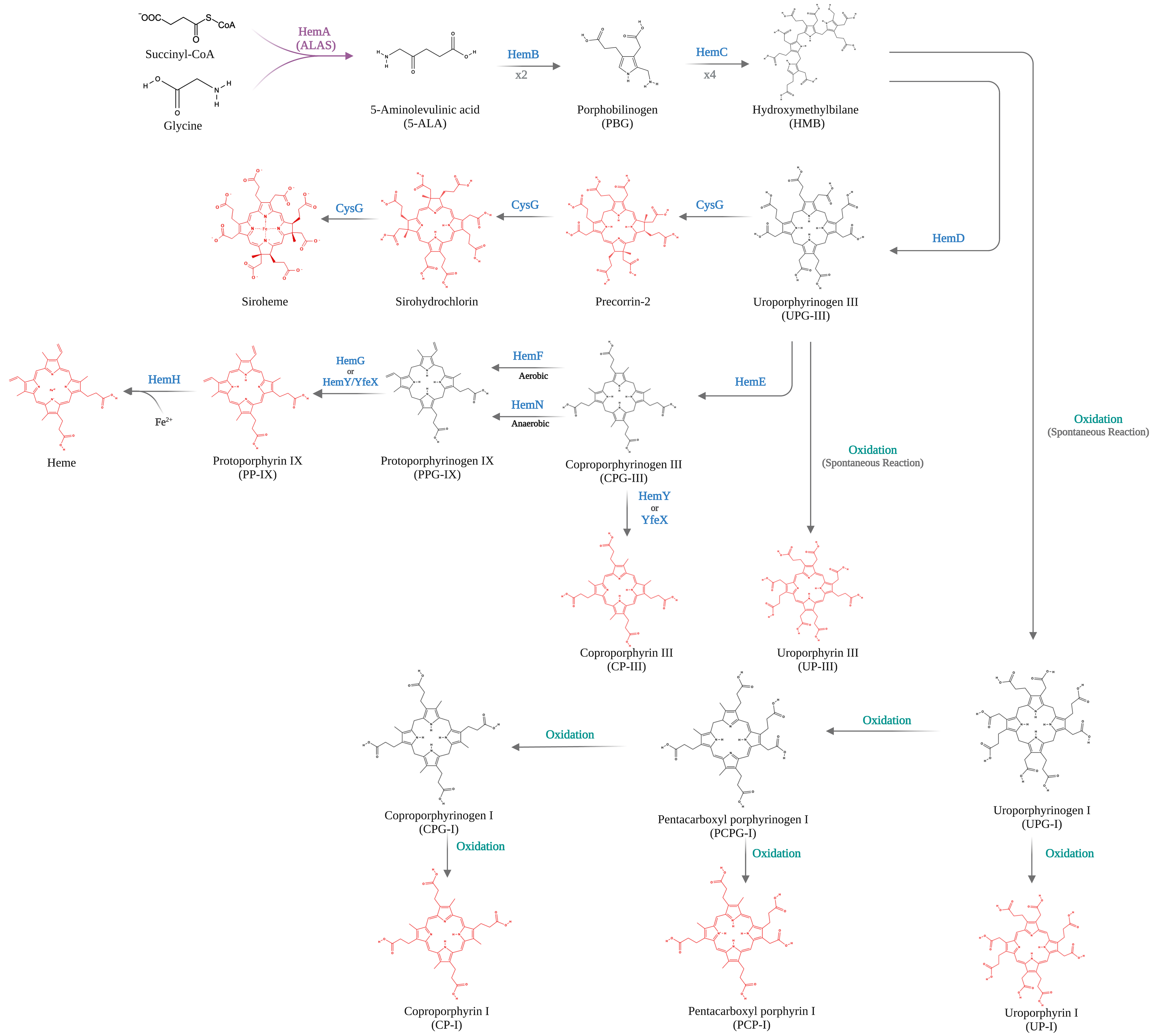
06 February 2024Bio-Based Production of Uroporphyrin in Escherichia coli
Uroporphyrin (UP) is a porphyrin compound with medical applications and a key precursor for heme biosynthesis. However, there is no biosynthetic strategy for UP production. In this study, we present a novel bioprocess for enhanced production of UP in engineered Escherichia coli. We first implemented the Shemin/C4 pathway heterologously in an E. coli strain with an enlarged intracellular pool of succinyl-CoA. Using a plasmid with the trc promoter regulating the expression of a synthesized gene operon, the effects of key pathway genes, including hemA, hemB, hemC, and hemD, on UP biosynthesis were characterized. By cultivating the resulting engineered E. coli strains in a batch bioreactor with 30 g/L glycerol under aerobic conditions, up to 901.9 mg/L UP was produced. Most of the synthesized UP was extracellularly secreted with a high purity more than 80 wt%, facilitating its downstream purification. The study paves the way for large-scale bio-based production of UP using synthetic biology and metabolic engineering strategies.
Open Access
Review
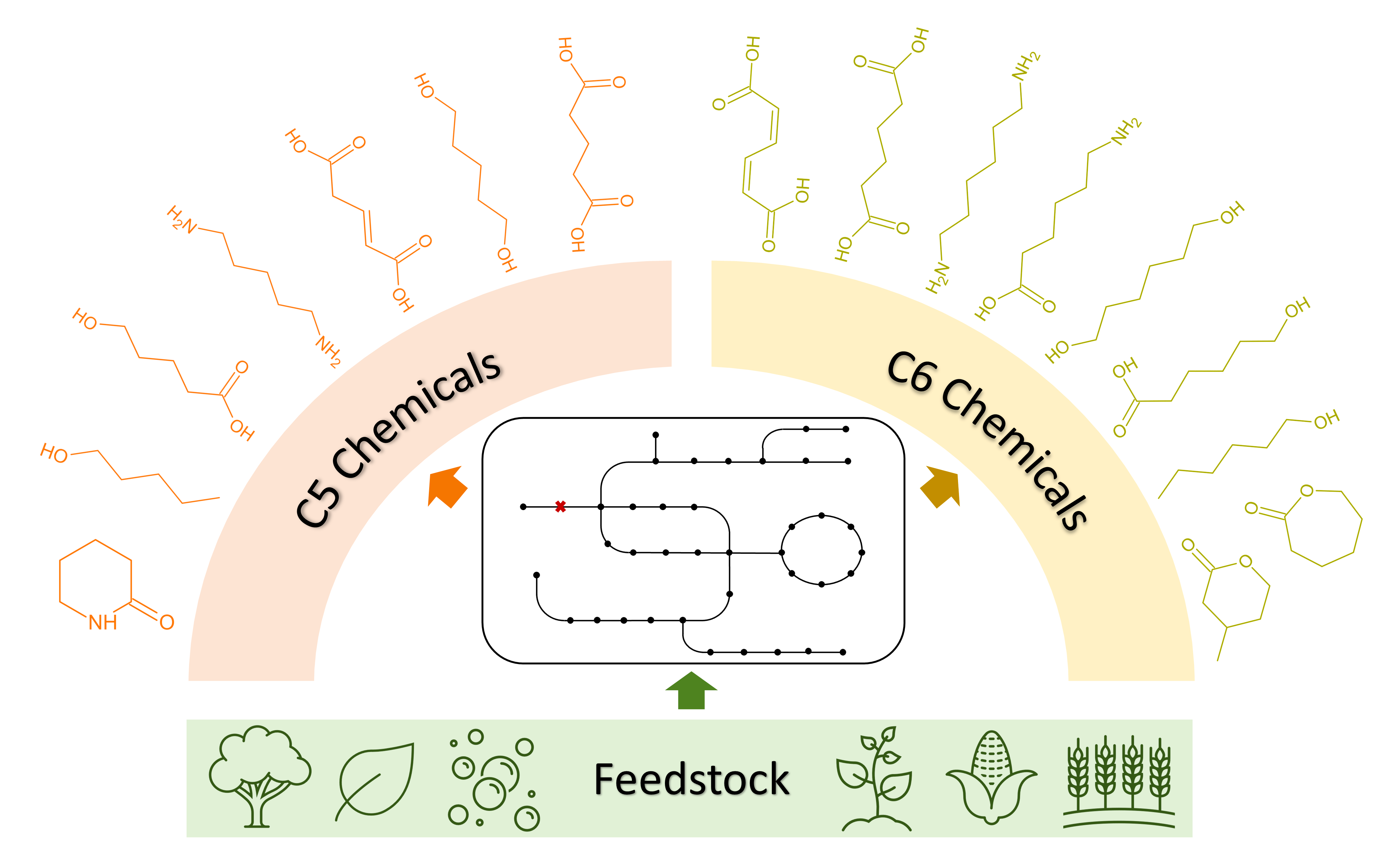
31 October 2023Metabolic Engineering of Microorganisms Towards the Biomanufacturing of Non-Natural C5 and C6 Chemicals
Five-carbon (C5) and six-carbon (C6) chemicals are essential components in the manufacturing of a variety of pharmaceuticals, fuels, polymers, and other materials. However, the predominant reliance on chemical synthesis methods and unsustainable feedstock sources has placed significant strain on Earth’s finite fossil resources and the environment. To address this challenge and promote sustainability, significant efforts have been undertaken to re-program microorganisms through metabolic engineering and synthetic biology approaches allowing for bio-based manufacturing of these compounds. This review provides a comprehensive overview of the advancements in microbial production of commercially significant non-natural C5 chemicals, including 1-pentanol, 1,5-pentanediol, cadaverine, δ-valerolactam, glutaric acid, glutaconic acid, and 5-hydroxyvaleric acid, as well as C6 chemicals, including cis, cis-muconic acid, adipic acid, 1,6-hexamethylenediamine, 6-aminocaproic acid, β-methyl-δ-valerolactone, 1-hexanol, ε-caprolactone, 6-hydroxyhexanoic acid, and 1,6-hexanediol.
Open Access
Article
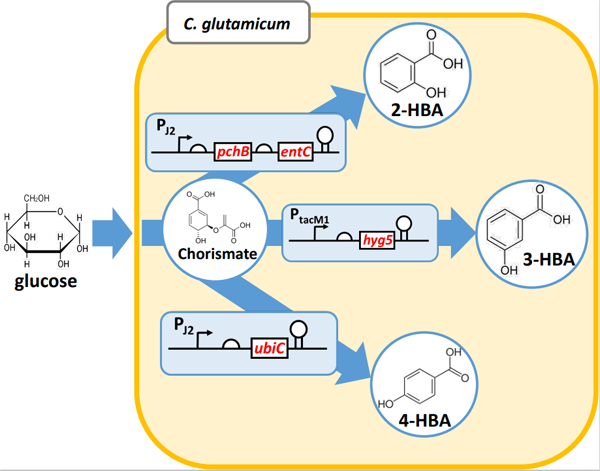
19 July 2023Hydroxybenzoic Acid Production Using Metabolically Engineered Corynebacterium glutamicum
Hydroxybenzoic acids (HBAs), including 4-HBA, 3-HBA, and 2-HBA, are valuable platform chemicals for production of commodity materials and fine chemicals. Herein, we employed metabolic engineering techniques to enhance the production of these HBAs in Corynebacterium glutamicum ATCC 13032. Our approach augmented the shikimate pathway and eliminated genes associated with HBA degradation, particularly phenol 2-monooxygenase encoded by cg2966. Increased titers of 3-HBA and 4-HBA were achieved via selection of suitable promoters for 3-hydroxybenzoate synthase and chorismate pyruvate lyase. A tac-M1 promoter was suitable for chorismate pyruvate lyase expression and 8.3 g/L of 4-HBA production was achieved. Efficient production of 2-HBA was enabled by maintaining a balanced expression of isochorismate synthase and isochorismate pyruvate lyase. Consequently, strains KSD5-tacM1-H and KSD5-J2-PE exhibited production levels of 19.2 g/L of 3-HBA and 12.9 g/L of 2-HBA, respectively, using 1 L jar fermenter containing 80 g/L of glucose. Therefore, this engineered strain platform holds significant potential for production of other valuable products derived from chorismate.
Open Access
Review
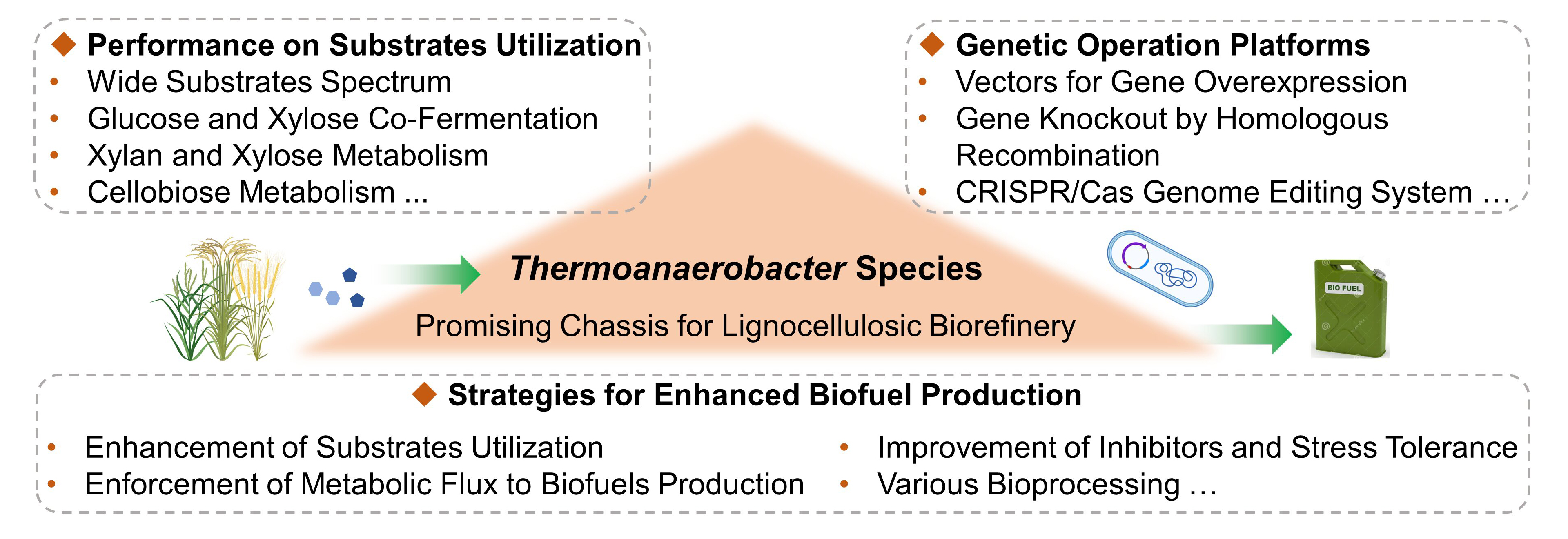
15 March 2023Thermoanaerobacter Species: The Promising Candidates for Lignocellulosic Biofuel Production
Thermoanaerobacter species, which have broad substrate range and high operating temperature, can directly utilize lignocellulosic materials for biofuels production. Compared with the mesophilic process, thermophilic process shows greater prospects in consolidated bioprocessing (CBP) due to its relatively higher efficiency of lignocellulose degradation and lower risk of microbial contamination. Additionally, thermophilic conditions can reduce cooling costs, and further facilitate downstream product recovery. This review comprehensively summarizes the advances of Thermoanaerobacter species in lignocellulosic biorefinery, including their performance on substrates utilization, and genetic modification or other strategies for enhanced biofuels production. Furthermore, bottlenecks of sugar co-fermentation, metabolic engineering, and bioprocessing are also discussed.