Preparation of MgAl2O4 Porous Ceramics for High-Temperature Flue Gas Filtration Application by In-Situ Decomposition Method
Jiarong Li 1 Lei Zhang 2 Xiaojie Ji 1 Chenran Zhang 1 Jianbiao Kong 2 Zheng Zhang 1,3 Wenying Zhou 1,* Degang Zhao 1,*
Received: 29 January 2026 Revised: 24 February 2026 Accepted: 13 March 2026 Published: 23 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Although energy sources such as wind and solar power have developed rapidly, fossil fuels still remain the main source of energy generation. However, their combustion emits large amounts of high-temperature flue gases [1,2] containing acid corrosive gases (CO, CO2, SO2) [3] and dust particles (PM), which can destroy the ecological environment [4,5,6]. To reduce such environmental pollution, porous ceramics are used as filters to filter the flue gas before emission. Generally, SiC porous ceramics with their exceptional thermal stability [7] and substantial specific surface area are suitable for filtering flue. However, high-temperature flue gas can erode SiC ceramics, thereby reducing their service life. Furthermore, the susceptibility of SiC ceramics to oxidation limits their operating temperature and makes them prone to failure. Hence, it is worth investigating alternative materials to produce advanced porous ceramics with excellent inoxidizability, greater chemical stability, and improved filtration performance. Compared with SiC, MgAl2O4 offers high melting point, high temperature stability, and good corrosion resistance, making it a promising candidate for high-temperature filtration applications [2,8,9,10].
Gas permeability is one of the most important properties of porous ceramic filters and is affected by porosity, pore size, and pore connectivity [11]. To introduce pores into the ceramic, the pore-forming agent method, gel-casting method, and freeze-drying [12] have been applied. Kumar et al. [11] prepared Al2O3 based ceramic filters by adding a pore-forming agent method with characteristics of an apparent porosity of 39–70%, a compressive strength of 6–82 MPa, and a relatively high gas permeability coefficient of 0.38 × 10−10–9.15 × 10−10 m2. Liu et al. [13] use gel-casting method and freeze-drying method to prepare the mullite porous ceramics with a high apparent porosity of 56.04–75.34%, a compressive strength of 0.61–3.03 MPa, and a gas permeability coefficient of 1.11 × 10−10–4.73 × 10−11 m2. Based on the above analysis, various porous ceramics with a high apparent porosity have been fabricated by these methods. However, the high porosity results in a low compressive strength. Compared with the above method, the in-situ decomposition method, which relies on the decomposition of raw materials, offers a more uniform pore size distribution, thereby helping to preserve the compressive strength of porous ceramic filters. For instance, Zhou et al. [14] prepared MgAl2O4 porous ceramics via the decomposition of bauxite and magnesite, with characteristics of a density of 3.43 g/cm3 and a flexural strength of 93.5 MPa. Wang et al. [15] prepared MgAl2O4 porous ceramics by the in-situ decomposition method, achieving an optimal sample with an apparent porosity of 50%, a flexural strength of 35.06 MPa, and a nitrogen gas flux of 3057 m3·m−2·h−1 under the gas pressure of 0.1 MPa. It can therefore be concluded that the in-situ decomposition method is an effective method for improving the strengths of MgAl2O4 porous ceramic.
EDTA-MgNa2 is an organic magnesium compound that decomposes to generate more gas during sintering. When used for the in-situ decomposition, it provides a new opportunity to enhance the pore interconnectivity between pores. During the sintering process, EDTA-MgNa2 also releases Na+ ions that react with impurity ions in the raw materials to form a transitional liquid phase, thereby promoting the sintering reaction. Combined with the in-situ decomposition method, this approach enables the formation of uniformly sized pores, ultimately optimizing the balance between compressive strength and gas permeability. Based on the above analysis, preparing porous ceramics via the in-situ decomposition method using EDTA-MgNa2 as a raw material is expected to yield high-temperature porous MgAl2O4 ceramic with high compressive strength and high gas permeability.
In this study, a MgAl2O4 porous ceramic filter was prepared via the in-situ decomposition method, using α-Al2O3, MgO, and EDTA-MgNa2 as raw materials. The influence of EDTA-MgNa2 content on the phase composition, microstructure, apparent porosity, pore size distribution, thermal expansion coefficient, mechanical properties, and gas permeability of MgAl2O4 porous ceramics was investigated to verify the feasibility of MgAl2O4 as a high-temperature flue gas filter.
2. Materials and Methods
2.1. Raw Materials
α-Al2O3 (purity ≥ 99.99%, D50 = 4.00 μm, Shandong Higiant High-Purity Alumina Technology Co., Ltd., Binzhou, China), MgO (purity ≥ 99.90%, D50 = 5.00 μm, Hebei Zhuyan Alloy Materials Co., Ltd., Xingtai, China), and EDTA-MgNa2 (Shandong Smatts Agricultural Science and Technology Co., Ltd., Weifang, China) were used as the main material. C2H5OH (Tianjin Fuyu Chemical Co., Ltd., Tianjin, China) was chosen as a binder.
2.2. Specimen Preparation
In order to form stoichiometric MgAl2O4 spinel, the weight ratio of Al2O3 and MgO was chosen as 2.53:1. Firstly, the raw materials were mixed with ethyl alcohol using a mortar and pestle for 5 min according to the designed compositions described in Table 1. EDTA-MgNa2 with the contents of 0.0 wt%, 2.5 wt%, 5.0 wt%, 7.5 wt%, or 10.0 wt% was introduced, and the samples were named as M1, M2, M3, M4, M5. Dry mixtures were pressed uniaxially in a hydraulic press at 20 MPa to φ30 × H10 mm and φ20 × H20 mm. The green samples were then dried in an oven at 110 °C for 24 h. Finally, the samples were placed in a muffle furnace with an air atmosphere and sintered at a heating rate of 5 °C/min. During the sintering process, they were pre-fired at 700 °C for 3 h, then sintered at 1500 °C for 3 h, and allowed to cool down to room temperature at a rate of 5 °C/min inside the furnace.
Table 1. Sample codes with different compositions.
|
Sample Codes |
Al2O3 (wt%) |
MgO (wt%) |
EDTA-MgNa2 (wt%) |
|---|---|---|---|
|
M1 |
71.7 |
28.3 |
0.0 |
|
M2 |
70.1 |
27.4 |
2.5 |
|
M3 |
68.5 |
26.5 |
5.0 |
|
M4 |
66.9 |
25.6 |
7.5 |
|
M5 |
65.4 |
24.6 |
10.0 |
2.3. Characterizations
Thermal analysis of the EDTA-MgNa2 was detected using a simultaneous TG-DSC apparatus (STA449, NETZSCH, Selb, Germany). The phase compositions of samples were determined via X-ray diffraction (Rigaku Smart Lab 9kW, Rigaku, Tokyo, Japan) with Cu Kα radiation in the 2θ range of 5–90° and a scanning speed of 2°/min. The microstructure was studied by field emission scanning electron microscopy (QUANTA 250 FEG, FEI Company, Hillsboro, OR, USA). The pore size distribution was examined via mercury porosimetry (AutoPore Ⅴ 9600, Micromeritics, Norcross, GA, USA). The bulk density and apparent porosity were determined by the Archimedes method using water as the medium.
The linear change of the sample before and after calcination was determined by the following equation:
|
$$\text{Linear change} = \frac{\text{l}_2 \,- \,\text{l}_1}{\text{l}_1} \times 100\%$$ |
(1) |
where l1 and l2 are the diameters of the samples before and after sintering.
The weight change of the sample before and after calcination was determined by the following equation:
|
$$\text{Weight change} = \frac{\text{m}_1 \,-\, \text{m}_2}{\text{m}_1} \times 100\%$$ |
(2) |
where m1 and m2 are the weights of the samples before and after sintering.
The compressive strength was assessed using a universal testing machine (CMT5305, MTS Systems (China) Co., Ltd., Shanghai, China). The sample was cylindrical with 20 mm in diameter and 20 mm in height. Thermal expansion analysis was carried out in a thermomechanical analyzer (TMAPT1000, LINSEIS, Selb, Germany) from room temperature to 600 °C. The theoretical liquid phases were calculated by the Equilib mode of the thermo-chemical software Factsage 6.2.
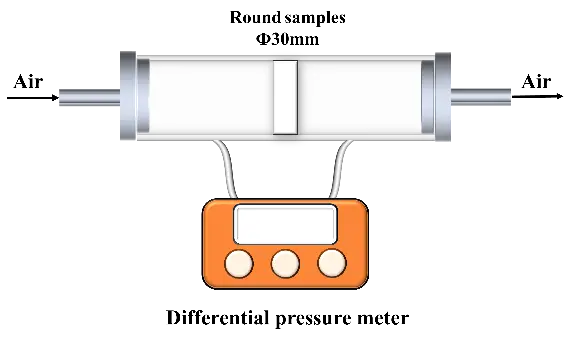
As shown in Figure 1, a self-made pressure drop tester was used to measure the pressure drop of a gas across the ceramics. The device pipe’s inner diameter and the test samples’ diameter were 30 mm each. The tests were carried out at the gas velocity of 10–19 m/s. According to previous research [16,17,18,19], the gas permeability coefficient and the ceramic permeance were calculated by using Equation (3) and Equation (4):
|
```latex\text{ψ}\text{ }\text{=}\text{ }\frac{\text{ηQt}}{\text{AΔP}}``` |
(3) |
where ψ is the permeability coefficient (m2), η is the dynamic viscosity of air, Q is the gas flow rate (m3/s), t is the thickness of the sample (m), A is the filter area (m2), and ΔP is the pressure drop (Pa).
|
```latex\text{Permeance}\text{ }\text{=}\text{ }\frac{\text{Flux}}{\text{ΔPA}}\text{ }\text{=}\text{ }\frac{\text{n}}{\text{ΔPA}}``` |
(4) |
The term “permeance” refers to gas permeance (mol·m−2·s−1·Pa−1), while n is the flow rate of the gas (mol·s−1).
3. Results and Discussion
3.1. Thermal Stability of EDTA-MgNa2
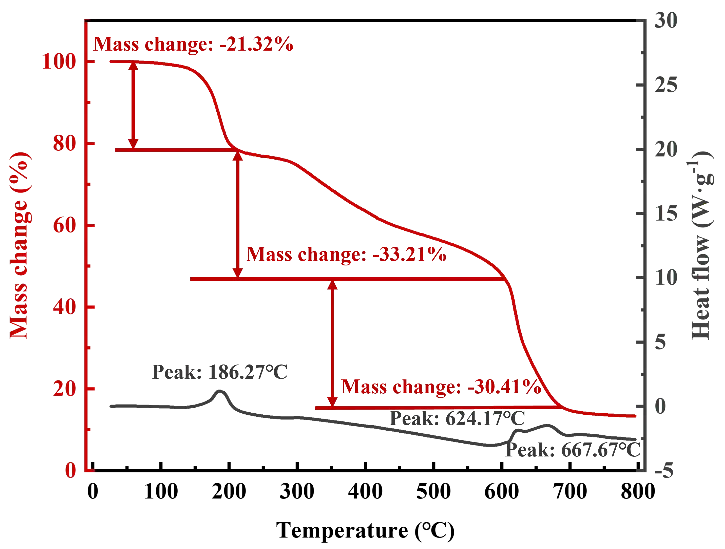
In order to determine the heat treatment process, the dehydration and thermal stability of EDTA-MgNa2 were investigated through the TG-DSC analysis, as depicted in Figure 2. The DSC curve can be divided into three stages. In the first stage, with the temperatures ranging from ambient to 186.27 °C, the mass of specimens was reduced to 79%, due to the vaporization of the physisorbed water. In the second stage, when the temperature increased to 624.17 °C, EDTA-MgNa2 decomposed, releasing gases such as CO and CO2 [20], and formed MgCO3 and Na2CO3, as shown in Equation (5).
|
```latex{\text{C}}_{\text{10}}{\text{H}}_{\text{12}}\text{Mg}{\text{N}}_{\text{2}}{\text{Na}}_{\text{2}}{\text{O}}_{\text{8}}\left(\text{s}\right)\text{ }\text{=}\text{ }\text{MgC}{\text{O}}_{\text{3}}\left(\text{s}\right)\text{ }\text{+}\text{ }\text{N}{\text{a}}_{\text{2}}\text{C}{\text{O}}_{\text{3}}\left(\text{s}\right)\text{ }\text{+}\text{ }\text{C}{\text{O}}_{\text{2}}\left(\text{g}\right)\text{ }\text{+}\text{ }{\text{H}}_{\text{2}}\text{O}\left(\text{g}\right)\text{ }\text{+}\text{ }\text{N}{\text{O}}_{\text{x}}\left(\text{g}\right)``` |
(5) |
Further increasing the temperature to 667.67 °C, MgCO3 decomposed into MgO and CO2, as depicted in Equation (6).
|
```latex\text{MgC}{\text{O}}_{\text{3}}\left(\text{s}\right)\text{ }\text{=}\text{ }\text{MgO}\left(\text{s}\right)\text{ }\text{+}\text{ }\text{C}{\text{O}}_{\text{2}}\left(\text{g}\right)``` |
(6) |
Upon further heating to 700 °C, the mass of MgO (the decomposition products of EDTA-MgNa2) remained stable, with a mass residual rate of 15%. Therefore, since the decomposition of EDTA-MgNa2 is completed at 700 °C, a holding step was chosen to be carried out at this temperature to ensure complete decomposition of EDTA-MgNa2.
3.2. Phase Compositions, Microstructures, and Pore Size Distributions of MgAl2O4 Ceramics
Figure 3 shows the XRD pattern of MgAl2O4 samples with different EDTA-MgNa2 contents, indicating that the main phase of these ceramics was MgAl2O4. It is well established that Al2O3 and MgO can react in situ to form MgAl2O4 (Equation (7)). In this study, MgO originates from two sources: the MgO present in the raw materials and the MgO derived from the decomposition of EDTA-MgNa2 (Figure 2). During the sintering process, both types of MgO can react with the Al2O3 in the raw materials to form MgAl2O4.
|
```latex\text{MgO}\left(\text{s}\right)\text{ }\text{+}\text{ }{\text{Al}}_{\text{2}}{\text{O}}_{\text{3}}\left(\text{s}\right)\text{ }\text{=}\text{ }\text{Mg}{\text{Al}}_{\text{2}}{\text{O}}_{\text{4}}\left(\text{s}\right)``` |
(7) |
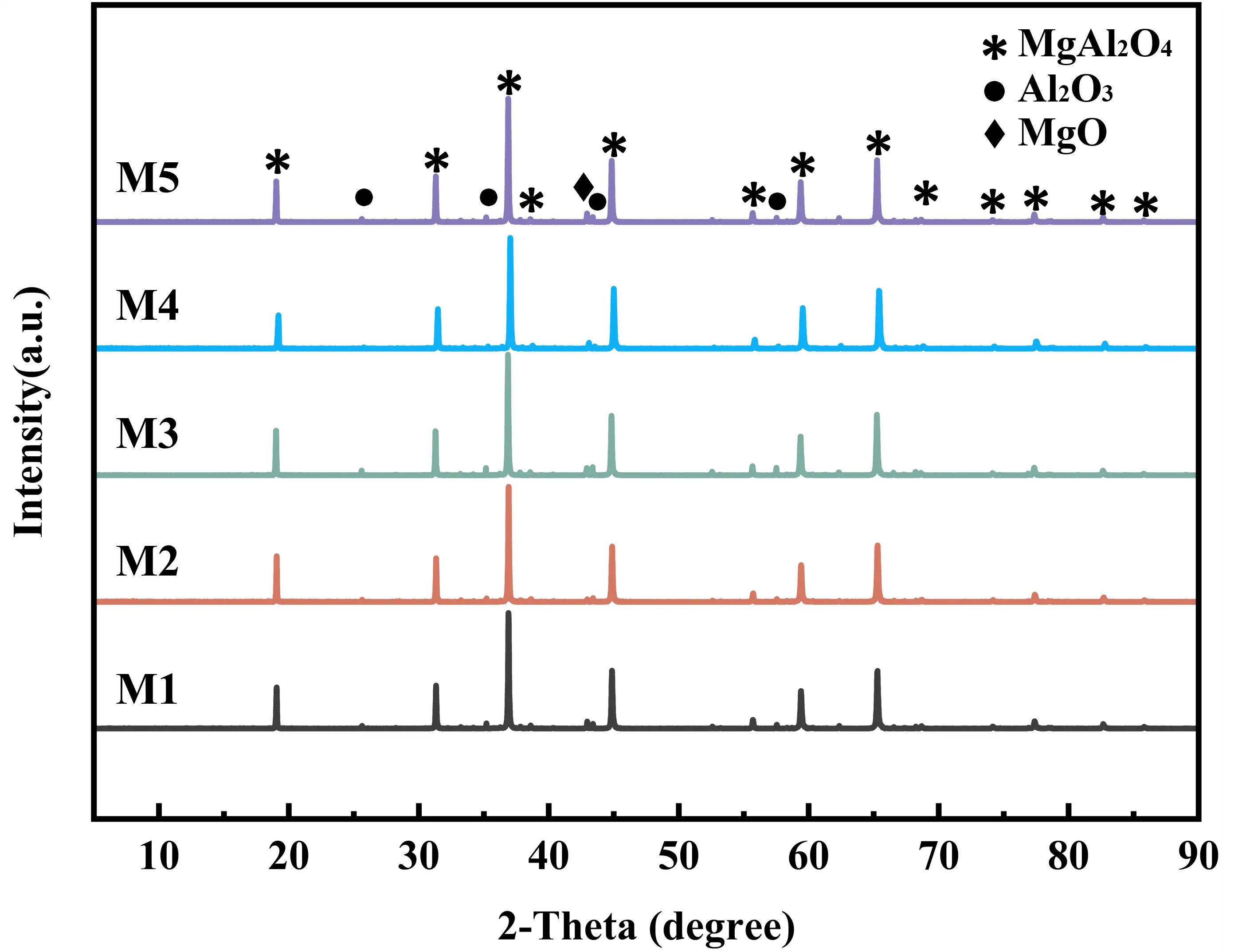
Figure 3. XRD pattern of the MgAl2O4 porous ceramics fired at 1500 °C with different EDTA-MgNa2 contents.
Figure 4 shows the micromorphology of fired samples M1–M5 with different EDTA-MgNa2 contents. Combined with EDS, the irregular particles are confirmed as MgAl2O4 grains, consistent with the XRD results shown in Figure 3. As shown in Figure 4a1–e1, the grain growth mechanism of MgAl2O4 can be described as follows: Initially, Al2O3 and MgO react to form MgAl2O4. As the reaction proceeds, more MgAl2O4 grains are formed and accumulated, resulting in larger grains until they reach the critical size. Since the MgO derived from the decomposition of EDTA-MgNa2 has higher reactivity, increasing the EDTA-MgNa2 content leads to a greater number of fine grains.

Figure 4. SEM images of the MgAl2O4 porous ceramics after firing at 1500 °C with different EDTA-MgNa2 contents: (a) M1, (b) M2, (c) M3, (d) M4, (e) M5; EDS elemental analysis of point 1 in (a2): (f,g).
The SEM images of polished sample M5 sintered at 1500 °C are given in Figure 5. As shown in Figure 5a, the matrix is mainly MgAl2O4, with a large number of uniformly distributed pores. Additionally, a few regions exhibiting a multi-particle packing structure surrounded by MgAl2O4 and annular pores (Figure 5b) are observed. By combining Figure 5d–g, it reveals that this region with multi-particle packing structure corresponds to partially reacted MgO, and there are pores formed by the decomposition of EDTA-MgNa2 between the MgO particles.
The pore structure parameters of MgAl2O4 porous ceramics are depicted in Figure 6. Analysis of Figure 6a reveals a unimodal distribution of the pore size in the porous ceramics with different EDTA-MgNa2 contents, indicating that the pore size of the samples is uniformly distributed. As shown in Figure 6a, with the EDTA-MgNa2 content increasing from 0 to 10 wt%, the pore size of MgAl2O4 porous ceramics shows an overall upward trend. The prominent peak value of the M5 sample reaches 4.09 μm, and a large pore size is conducive to enhancing the gas permeability of MgAl2O4 porous ceramics. As illustrated in Figure 6b, higher EDTA-MgNa2 content resulted in an upward shift in the cumulative pore volume curve, indicating that samples containing higher EDTA-MgNa2 content have better pore structure. The steep curve in the range of 0.70–7.20 μm reflects a rapid increase in cumulative pore volume, indicating that most pores fall within this size range, and a large and uniform pore size distribution is conducive to gas passing and flue gas filtration.
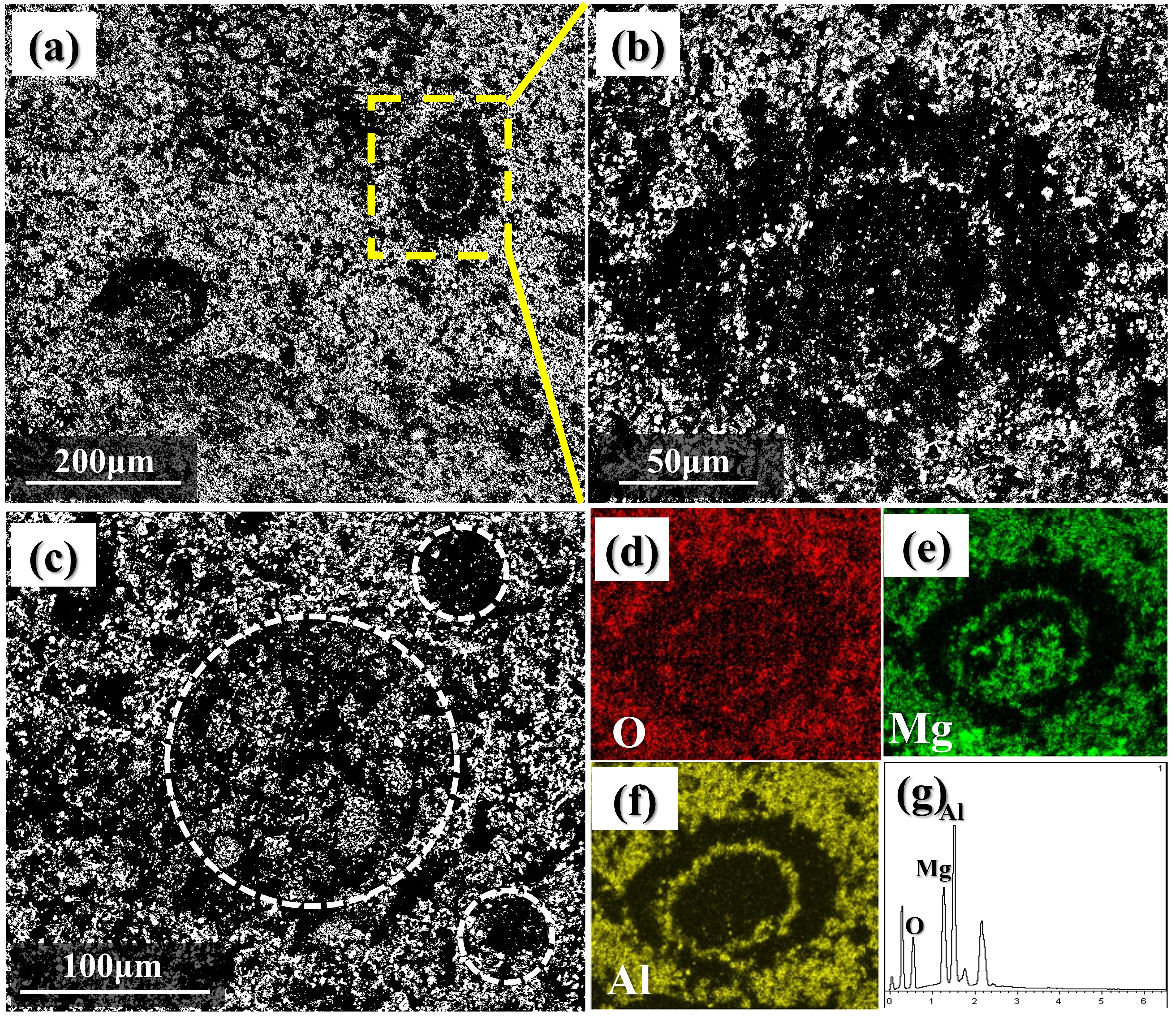
Figure 5. Microstructure of a polished sample M5 after firing at 1500 °C (a–c) SEM images; (d–g) element mapping analysis corresponding to O, Mg, and Al elements.
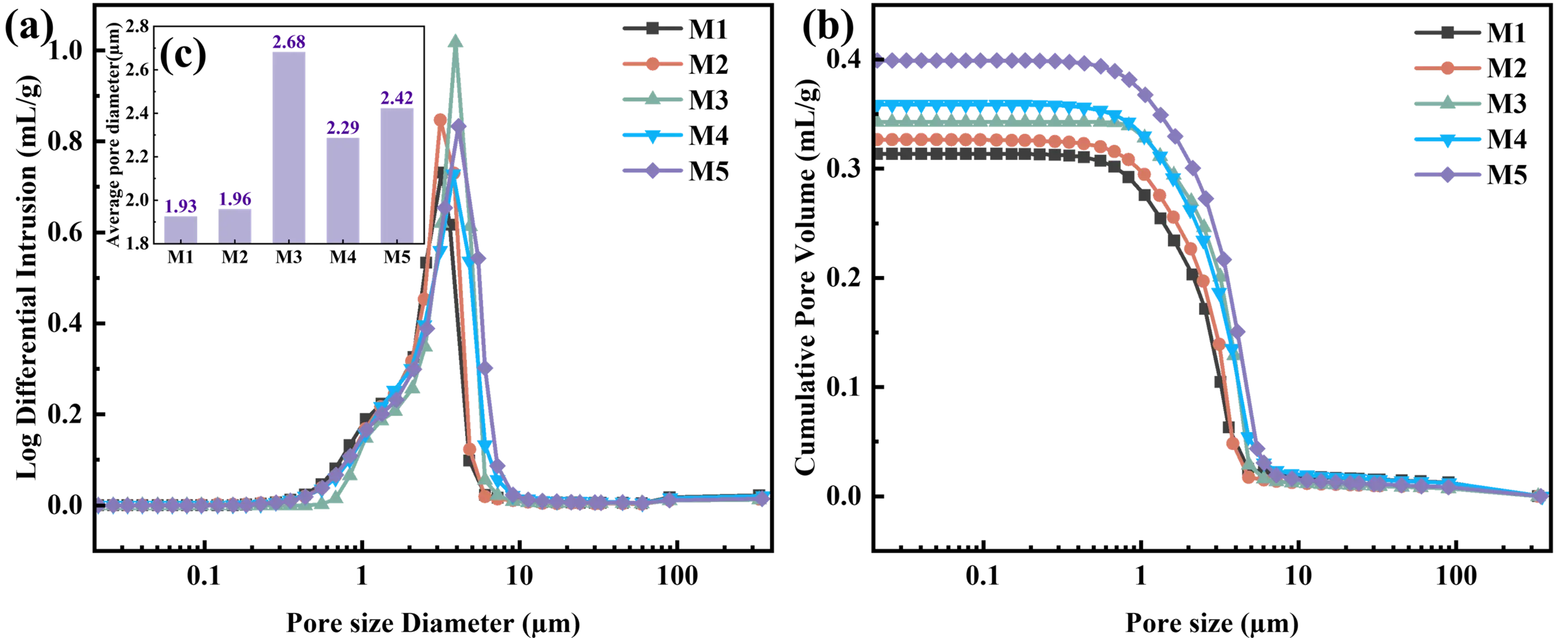
Figure 6. (a,b) Pore structure parameters and (c) average pore size of the MgAl2O4 porous ceramics after firing at 1500 °C with different EDTA-MgNa2 contents.
Figure 7 shows the weight change and linear change of the MgAl2O4 porous ceramics with different EDTA-MgNa2 contents. As shown in Figure 7, with the increase in EDTA-MgNa2 content from 0.0 to 10.0 wt%, the weight change gradually increased from 1.32% to 10.57%, due to the decomposition of EDTA-MgNa2 and release of gas [20]. The linear change of MgAl2O4 porous ceramics indicates that all samples exhibit thermal expansion characteristics, mainly due to the formation of spinel, which is accompanied by volume expansion [15,21].
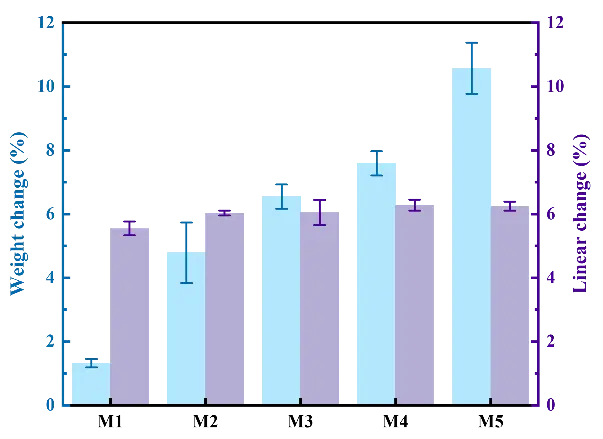
Figure 7. Weight change and linear change of the MgAl2O4 porous ceramics after firing at 1500 °C with different EDTA-MgNa2 contents.
3.3. Physical Properties
Figure 8 illustrates the apparent porosity and bulk density of MgAl2O4 porous ceramics with different EDTA-MgNa2 contents. The results showed that the apparent porosity of MgAl2O4 porous ceramics showed an upward trend from 47.27% to 56.28%, and the bulk density showed a down trend from 1.72 g·cm−3 to 1.45 g·cm−3 with increasing EDTA-MgNa2 content. Then, by combining the apparent porosity with the linear change in Figure 6, it can be found that the MgAl2O4 spinel formation is beneficial for the apparent porosity due to the volume expansion. And as mentioned above, the EDTA-MgNa2 is evenly distributed between the particles. After sintering, a large number of pores formed due to the decomposition of EDTA-MgNa2. Thus, the MgAl2O4 porous ceramics prepared by this in-situ decomposition method exhibited high porosity, an important condition for achieving high gas permeability.
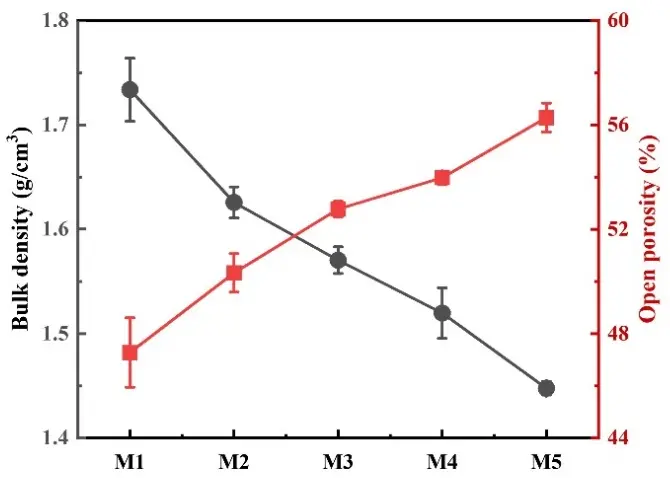
Figure 8. Apparent porosity and bulk density of the MgAl2O4 porous ceramics after firing at 1500 °C with different EDTA-MgNa2 contents.
Figure 9 displays the compressive strength of MgAl2O4 porous ceramics with different EDTA-MgNa2 contents. As observed, the compressive strength of MgAl2O4 porous ceramic decreased gradually from 35.56 to 10.93 MPa with increasing EDTA-MgNa2 content. Compressive strength is a crucial index of material mechanical properties, while porosity is a key factor that is usually negatively correlated with material strength. The relationship between the two parameters can be described by the Gibson-Ashby model, as expressed by Equation (8) [19,22,23]:
|
```latex\text{σ}\text{∝}{\text{σ}}_{\text{s}}{\text{(1}\text{ }\,-\,\text{ }\text{P)}}^{\text{m}}``` |
(8) |
where σ and σs represent the compressive strength of porous and dense material, respectively, P represents the porosity, and m is a constant.
As shown in Figure 8 and Equation (8), the compressive strength is negatively correlated with porosity (Figure 9b), indicating that higher apparent porosity reduces the load-bearing area of the material, thereby decreasing the compressive strength (Figure 9a). Thus, the porosity of the MgAl2O4 porous ceramics increased, and the strength decreased. However, the MgAl2O4 porous ceramics produced by the in-situ decomposition method still possess sufficient compressive strength to enable their industrial applications for high-temperature gas filtration.
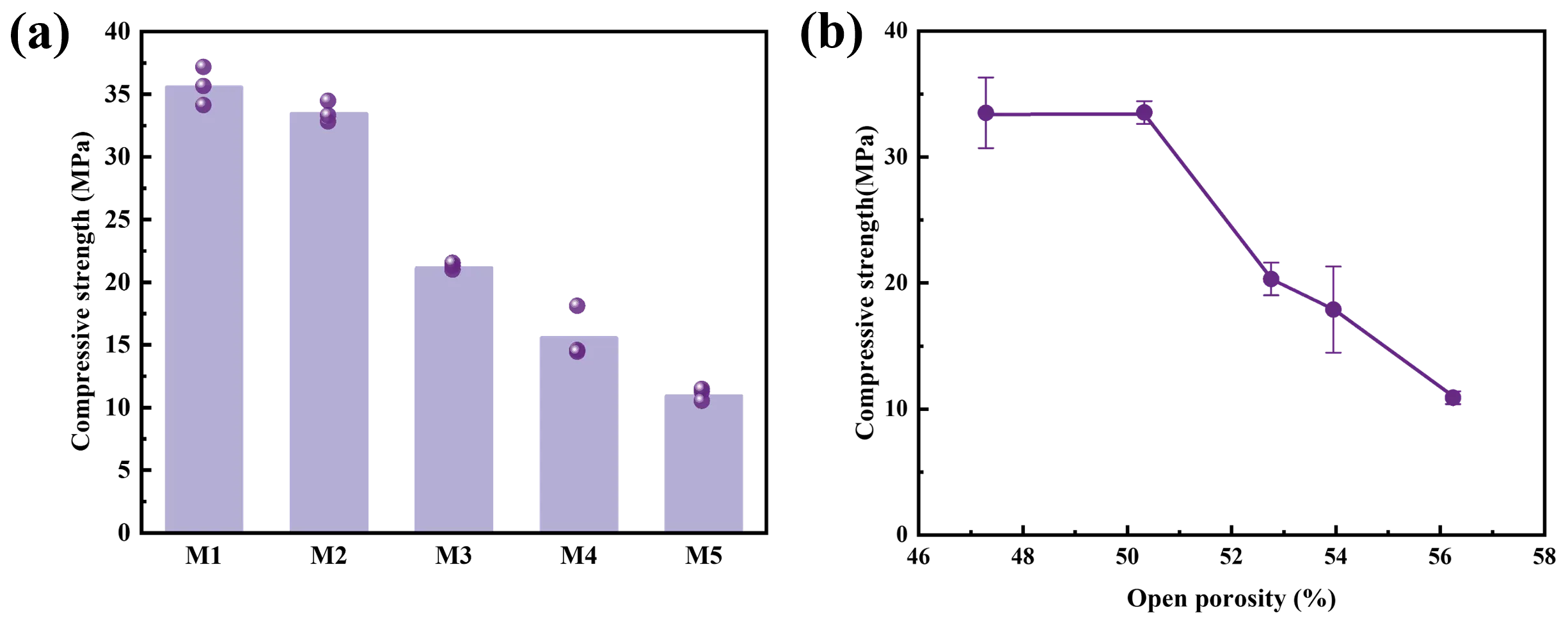
Figure 9. (a) Compressive strength; (b) linear relationship between compressive strength and apparent porosity of the MgAl2O4 porous ceramics after firing at 1500 °C with different EDTA-MgNa2 contents.
Figure 10 presents the coefficients of thermal expansion (CTE) of MgAl2O4 porous ceramics prepared with varying EDTA-MgNa2 contents. It is evident from the figure that the CTE of the MgAl2O4 porous ceramic increased gradually with the increasing test temperature, due to the average distance between atoms in the lattice increasing with the increase of temperature [24,25]. At 600 °C, as the content of EDTA-MgNa2 increases from 0.0 to 2.5 wt% and from 5.0 to 7.5 wt%, the CTE values of the MgAl2O4 porous ceramic increase. This is because MgAl2O4 itself expands during heating. Although pores in the sample can absorb the displacement of some solid particles caused by heating, the poor connectivity of the pores limits their ability to improve the CTE. As the content of EDTA-MgNa2 increases from 2.5 to 5.0 wt% and from 7.5 to 10.0 wt%, the CTE values of the MgAl2O4 porous ceramics decrease. This is attributed to the greater number of pores in the sample, which absorb the displacements of some solid particles caused by heating, thereby reducing the CTE. As a result, the MgAl2O4 porous ceramic with 10.0 wt% EDTA-MgNa2 shows the lowest CTE of 7.93 × 10−6/K.
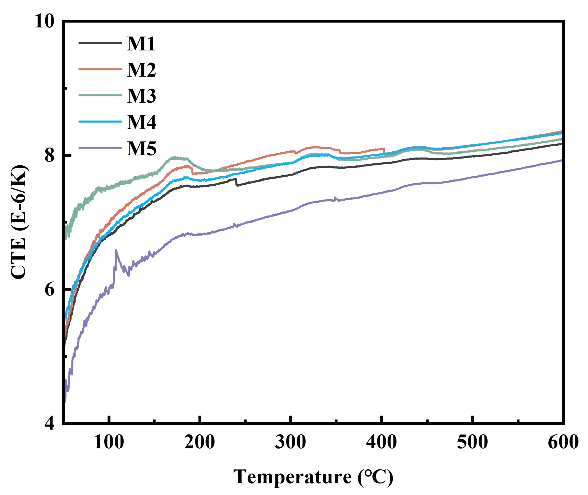
Figure 10. Coefficients of thermal expansion (CTE) of the MgAl2O4 porous ceramics after firing at 1500 °C with different EDTA-MgNa2 contents.
3.4. Gas Permeability
Pressure drop is an important parameter to characterize the gas permeance of ceramic filters, which is caused by the energy loss of gas flow in the pipeline. Figure 11a shows the relationship between EDTA-MgNa2 content and the pressure drop at flow velocities of 10–19 m/s. As shown in Figure 11a, the results indicate that the pressure drop gradually increased with increasing gas velocity, because resistance is generated as gas molecules collide with each other due to internal friction as they flow through a pipeline [26]. This resistance reduces the gas velocity, thereby increasing resistance to gas flow and leading to a rise in the pressure drop. However, as the gas velocity increases, the pressure drop of the M5 sample generally remains at a lower value, which is conducive to enhancing the gas permeability of MgAl2O4 porous ceramic in practical applications. As gas velocity increased to 19 m/s, the pressure drop of MgAl2O4 porous ceramic first increased and then decreased. The reason why the M2 sample has a higher pressure drop than the M1 sample is attributed to the presence of isolated small pores in the M2 sample, which will lead to longer and more tortuous paths in the sample when gas flows through. Subsequently, the number of interconnected pores in MgAl2O4 porous ceramic gradually improves, leading to a decreasing trend in pressure drop. Ultimately, the M5 sample achieved the minimum pressure drop of 3.39 hPa.
Figure 11b shows the gas permeability coefficient and gas permeance values of the MgAl2O4 porous ceramics with different EDTA-MgNa2 contents at a gas flow velocity of 19 m/s. It can be observed that, as fundamental performance parameters of filter materials, the gas permeability coefficient and gas permeance indicate that all MgAl2O4 porous ceramics exhibit good gas permeability, consistent with the gas permeability coefficient reported in the literature [27]. Among them, the M5 sample had the maximum gas permeability coefficient and gas permeance of 8.84 × 10−9 m2 and 1.52 mol·m−2·s−1·Pa−1, respectively, consistent with the phenomenon that the pressure drop decreases to 3.39 hPa. In summary, the content of EDTA-MgNa2 can improve the gas permeability while maintaining the high filtration precision of MgAl2O4 porous ceramics, and the M5 sample is the optimal one.
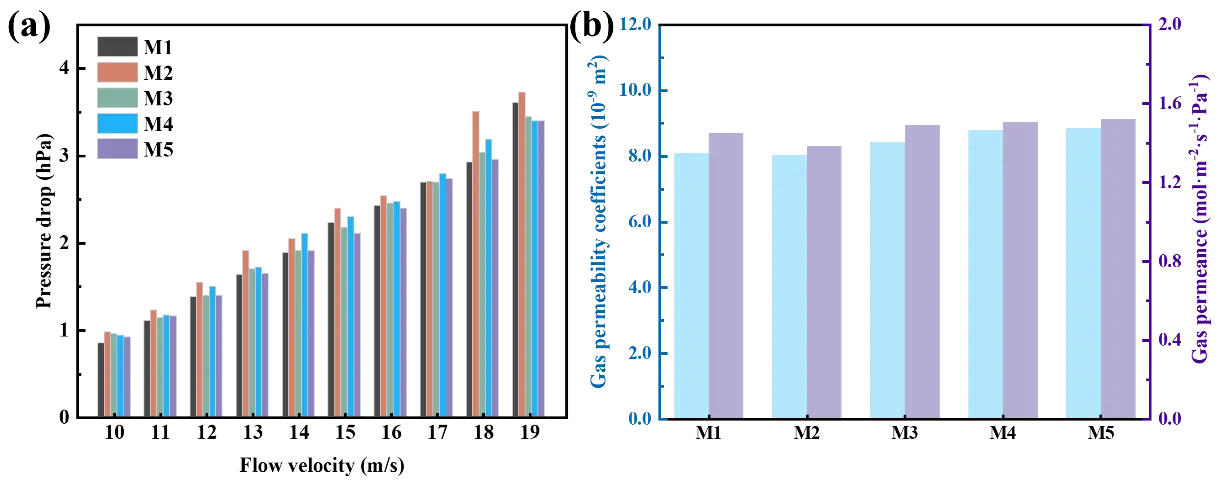
Figure 11. (a) Pressure drop at gas velocities of 10 m/s to 19 m/s; (b) gas permeability coefficient and gas permeance at a gas velocity of 19 m/s for MgAl2O4 porous ceramics after firing at 1500 °C with different EDTA-MgNa2 contents.
4. Discussion
The above results indicate that the MgAl2O4 porous ceramics with high gas permeability are successfully prepared using MgO, Al2O3, and EDTA-MgNa2 as raw materials through the in-situ decomposition method. The process will be discussed in three stages, as illustrated in Figure 12, focusing on EDTA-MgNa2 decomposition, spinel formation, and pore structure.
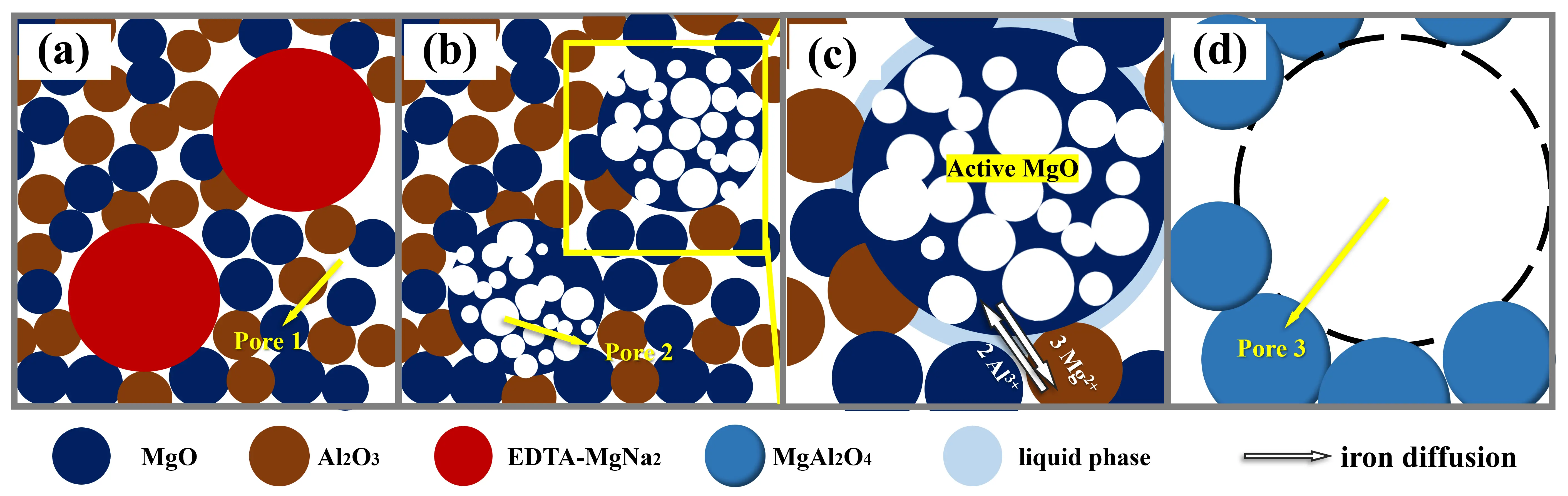
Figure 12. Schematic diagram of the sintering mechanism of MgAl2O4 porous ceramics: (a) initial process; (b) decomposition process; (c,d) sintering process.
(a) Initial Stage: As shown in Figure 12a, the green body primarily consists of large MgO and Al2O3 particles, and smaller EDTA-MgNa2 particles. Pores (Pore 1) are observed in the green body, arising from particle packing during the dry pressing process.
(b) Decomposition Stage: During the heating process, the organic backbone of EDTA-MgNa2 decomposes, and the C, H, and N elements react with oxygen to generate gases such as CO and CO2, which are subsequently released. When the temperature reaches about 700 °C, EDTA-MgNa2 particles completely decompose and form MgO grains with numerous pores (Pore 2), which may be the main reason for the significant increase in apparent porosity as the content of EDTA-MgNa2 increases (Figure 8). As shown in Figure 12b, the presence of pores isolates most particles from one another (Figure 5b), thereby providing more interfacial area that serves as nucleation sites.
(c,d) Sintering Stage: As the sintering temperature increases, the liquid phase forms on MgO particles due to the presence of impurities in the raw materials and the Na+ released from the EDTA-MgNa2 decomposition. The effects of temperature and EDTA-MgNa2 content on the liquid phase content of samples are shown in Figure 13. As can be seen, the liquid phase content increases gradually with higher EDTA-MgNa2 content, reaching 2.85 wt% at 1500 °C when the EDTA-MgNa2 content is 10 wt%. This increase in liquid phase accelerates the mutual diffusion of Mg2+ and Al3+, thereby promoting the formation and deposition of MgAl2O4 spinel.
To achieve the electric neutrality during the formation of spinel, 2-mol Al3+ diffuses into the MgO side to produce 1 mol MgAl2O4, whereas 3-mol Mg2+ diffuses into the Al2O3 side [28,29]. As shown in Figure 5a,b, the presence of MgAl2O4 rims around those pores indicates that they were previously occupied by MgO particles, which led to a quicker formation of MgAl2O4 on the Al2O3 side, thereby triggering the Kirkendall effect and creating Kirkendall voids (Pore 3 and Figure 5c) [29,30]. Due to the different rate of ion diffusion, neck bonds will appear between particles, leading to agglomeration (Figure 4), which helps to reduce the negative impact of increased apparent porosity on the compressive strength of MgAl2O4 porous ceramic. In turn, during the sintering stage, MgAl2O4 grains will rearrange, and pores between particles will merge and grow. This phenomenon is conducive to improving the pore size (Figure 6) and ultimately enhancing the gas permeability of MgAl2O4 porous ceramic (Figure 10).
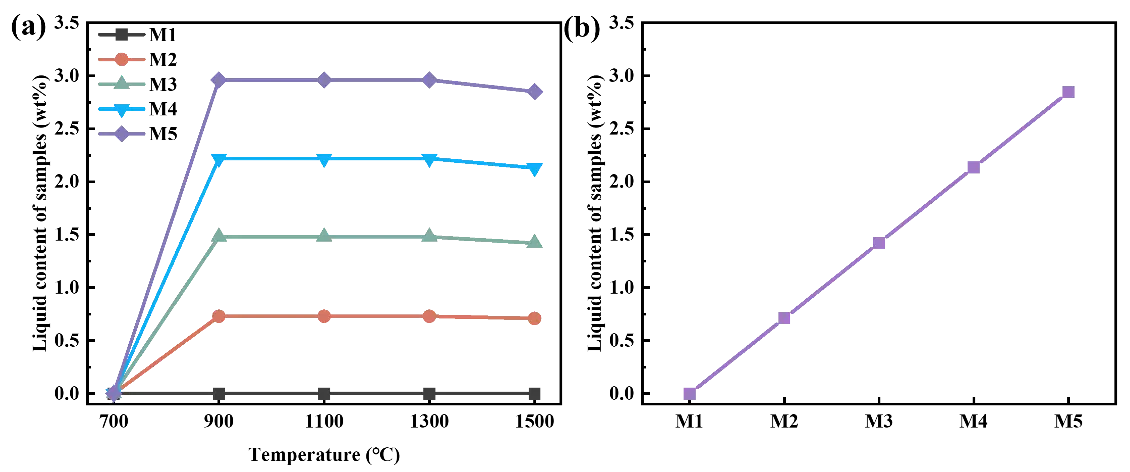
Figure 13. (a) Liquid contents of samples with different EDTA-MgNa2 at different temperatures; (b) linear relationship between liquid content of samples and EDTA-MgNa2 content at 1500 °C.
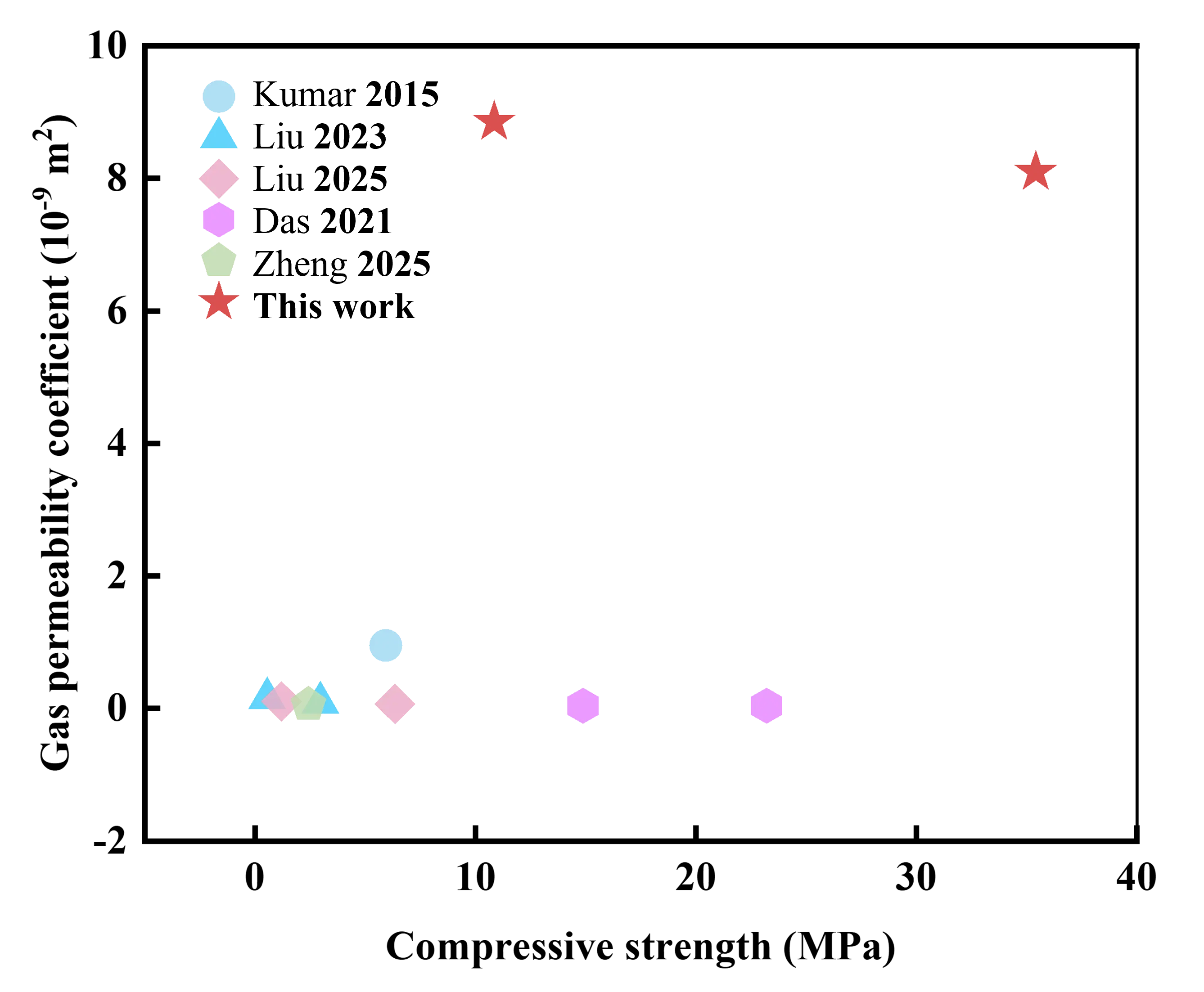
When compared to various porous ceramic filters (Figure 14 and Table 2), the MgAl2O4 porous ceramic filters developed in this work exhibit an outstanding overall balance of gas permeability and compressive strength. Notably, the compressive strength reaches 10.93 MPa, the highest among all compared samples, indicating superior structural integrity and resistance to mechanical stress, which are critical in harsh, high-temperature filtration environments. In addition, the material shows a gas permeability coefficient of 8.84 × 10−9 m2, surpassing many porous ceramic filters. Overall, this finding facilitates further optimization of the gas permeance in high-temperature flue gas filters.
Table 2. Comparison of relevant data in different literature.
5. Conclusions
To enhance the gas permeability and achieve high compressive strength of MgAl2O4 porous ceramics, EDTA-MgNa2 was introduced as a pore former and sintering aid by the in-situ decomposition method. The effect of the content of EDTA-MgNa2 on the gas permeability and performance of ceramics was investigated. The conclusions reached are as follows.
- (1)
-
The MgAl2O4 porous ceramic filters of 0–10 wt% exhibit a pressure drop of 0.84–3.72 hPa, gas permeability coefficient of 8.04 × 10−9–8.84 × 10−9 m2, gas permeability of 1.38–1.52 mol·m−2·s−1·Pa−1, compressive strength of 35.56–10.93 MPa, and apparent porosity of 47.27–56.28%.
- (2)
-
Increasing the EDTA-MgNa2 content generates more pores in the sample, thereby increasing porosity and, consequently, enhancing the gas permeability coefficient. Meanwhile, the formation of neck bonds between particles mitigates the decrease in compressive strength caused by increased porosity.
- (3)
-
Simultaneously, the increase of open porosity is able to improve the pore connectivity, making the gas permeability of MgAl2O4 porous ceramics higher (8.04 × 10−9–8.84 × 10−9 m2).
- (4)
-
The optimal product is the ceramics prepared with 10 wt% EDTA-MgNa2, which has a high gas permeability coefficient of 8.84 × 10−9 m2, a compressive strength of 10.93 MPa, and a high apparent porosity of 56.28%.
Acknowledgments
The authors gratefully acknowledge the financial support provided by the Taishan Scholar Program of Shandong Province, Shandong Postdoctoral Science Foundation, Leader of Scientific Research Studio Program of Jinan, University of Jinan Disciplinary Cross-Convergence Construction Projects 2023, the Natural Science Foundation of Shandong Province, the Jinan City-School Integration Development Strategy Project, and the Shandong Provincial Key Research and Development Program.
Author Contributions
Methodology, J.L.; Investigation, J.L.; Writing—Original Draft Preparation, J.L.; Data Curation, J.L., C.Z.; Writing—Review & Editing, W.Z., Z.Z.; Supervision, L.Z., J.K., X.J.; Project Administration, W.Z.; Funding Acquisition, D.Z.
Ethics Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data are available from the corresponding author on reasonable request.
Funding
This research was funded by the Taishan Scholar Program of Shandong Province (No. tsqn202306225), Shandong Postdoctoral Science Foundation (No. SDBX2023025), Leader of Scientific Research Studio Program of Jinan (No. 2021GXRC082), University of Jinan Disciplinary Cross-Convergence Construction Projects 2023 (Nos. XKJC-202301 and XKJC-202311), the Natural Science Foundation of Shandong Province (Nos. ZR2024QE044 and ZR2025QC605), the Jinan City-School Integration Development Strategy Project (Nos. JNSX2023015 and JNSX2023018), and the Shandong Provincial Key Research and Development Program (2025CXGC020107).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Asadi M, Larki I, Forootan MM, Ahmadi R, Farajollahi M. Long-Term Scenario Analysis of Electricity Supply and Demand in Iran: Time Series Analysis, Renewable Electricity Development, Energy Efficiency and Conservation. Sustainability 2023, 15, 4618. DOI:10.3390/su15054618 [Google Scholar]
- Lee YY, Li IC, Kogularasu S, Huang BW, Wang YF, Masimukku S, et al. Advanced oxide-stabilized zirconia ceramics for flue gas filtration in air purification systems. J. Hazard. Mater. Adv. 2025, 17, 100539. DOI:10.1016/j.hazadv.2024.100539 [Google Scholar]
- Li P, Lin ZG, Du HB, Feng T, Zuo J. Do environmental taxes reduce air pollution? Evidence from fossil-fuel power plants in China. J. Environ. Manag. 2021, 295, 113112. DOI:10.1016/j.jenvman.2021.113112 [Google Scholar]
- Nacken M, Ma L, Heidenreich S, Verpoort F, Baron GV. Development of a catalytic ceramic foam for efficient tar reforming of a catalytic filter for hot gas cleaning of biomass-derived syngas. Appl. Catal. B Environ. 2012, 125, 111–119. DOI:10.1016/j.apcatb.2012.05.027 [Google Scholar]
- Chen YS, Hsiau SS, Syu JR, Chang YL. Clean coal technology on hot gas clean-up process with a moving granular bed filter. Fuel 2019, 248, 136–142. DOI:10.1016/j.fuel.2019.03.071 [Google Scholar]
- Park JH, Ahn JW, Kim KH, Son YS. Historic and futuristic review of electron beam technology for the treatment of SO2 and NOx in flue gas. Chem. Eng. J. 2019, 355, 351–366. DOI:10.1016/j.cej.2018.08.103 [Google Scholar]
- Chen Y, Yu C, Cheng X, Wang R, Deng C, Ding J, et al. Advances in Sintering Technologies for SiC Ceramics: Mechanisms, Challenges, and Industrial Applications. High-Temp. Mater. 2025, 2, 10013. DOI:10.70322/htm.2025.10013 [Google Scholar]
- Gu QL, Ng TCA, Bao YP, Ng HY, Tan SC, Wang J. Developing better ceramic membranes for water and wastewater Treatment: Where microstructure integrates with chemistry and functionalities. Chem. Eng. J. 2022, 428, 130456. DOI:10.1016/j.cej.2021.130456 [Google Scholar]
- Henning LM, Abdullayev A, Vakifahmetoglu C, Simon U, Bensalah H, Gurlo A, et al. Review on Polymeric, Inorganic, and Composite Materials for Air Filters: From Processing to Properties. Adv. Energy Sustain. Res. 2021, 2, 2100005. DOI:10.1002/aesr.202100005 [Google Scholar]
- Jia C, Liu YB, Li L, Song JN, Wang HY, Liu ZL, et al. A Foldable All-Ceramic Air Filter Paper with High Efficiency and High-Temperature Resistance. Nano Lett. 2020, 20, 4993–5000. DOI:10.1021/acs.nanolett.0c01107 [Google Scholar]
- Kumar A, Mohanta K, Kumar D, Parkash O. Low cost porous alumina with tailored gas permeability and mechanical properties prepared using rice husk and sucrose for filter applications. Microporous Mesoporous Mater. 2015, 213, 48–58. DOI:10.1016/j.micromeso.2015.04.004 [Google Scholar]
- Xiao CF, Han B. Preparation of porous silicon nitride ceramics by freeze drying. J. Mater. Res. Technol. 2019, 8, 6202–6208. DOI:10.1016/j.jmrt.2019.10.014 [Google Scholar]
- Liu ZL, Yuan L, Tian C, Peng ZJ, Zhang DY, Wen TP, et al. High gas permeability and directional channels of mullite porous ceramics prepared by pectin-based freeze-drying method. Int. J. Appl. Ceram. Technol. 2023, 20, 3552–3564. DOI:10.1111/ijac.14446 [Google Scholar]
- Zhou Y, Ye DC, Wu YQ, Zhang CX, Bai W, Tian YM, et al. Low-cost preparation and characterization of MgAl2O4 ceramics. Ceram. Int. 2022, 48, 7316–7319. DOI:10.1016/j.ceramint.2021.11.196 [Google Scholar]
- Wang F, Ye JK, He G, Liu GH, Xie ZP, Li JT. Preparation and characterization of porous MgAl2O4 spinel ceramic supports from bauxite and magnesite. Ceram. Int. 2015, 41, 7374–7380. DOI:10.1016/j.ceramint.2015.02.044 [Google Scholar]
- Deng WX, Yu XH, Sahimi M, Tsotsis TT. Highly permeable porous silicon carbide support tubes for the preparation of nanoporous inorganic membranes. J. Membr. Sci. 2014, 451, 192–204. DOI:10.1016/j.memsci.2013.09.059 [Google Scholar]
- Han F, Zhong ZX, Yang Y, Wei W, Zhang F, Xing WH, et al. High gas permeability of SiC porous ceramics reinforced by mullite fibers. J. Eur. Ceram. Soc. 2016, 36, 3909–3917. DOI:10.1016/j.jeurceramsoc.2016.06.048 [Google Scholar]
- Xing ZH, Hu YH, Xiang DP, Ma YP. Porous SiC-mullite ceramics with high flexural strength and gas permeability prepared from photovoltaic silicon waste. Ceram. Int. 2020, 46, 1236–1242. DOI:10.1016/j.ceramint.2019.09.052 [Google Scholar]
- Liu ZL, Gao J, Tian C, Zhang DY, Peng ZJ, Zhang LH, et al. Preparation and study of mullite ceramic fibre porous membranes with near-net shape forming and high gas permeability. Ceram. Int. 2025, 51, 2626–2638. DOI:10.1016/j.ceramint.2024.11.246 [Google Scholar]
- Esteban M, Vizcaíno MCP, Vílchez FG. The thermal behaviour of ethylenediaminetetracetic acid and its sodium salts. Thermochim. Acta 1983, 62, 257–265. DOI:10.1016/0040-6031(83)85045-X [Google Scholar]
- II WPW, Stubican VS. Interdiffusion in the System MgO-MgAl2O4. J. Am. Ceram. Soc. 1971, 54, 349–352, DOI:10.1111/j.1151-2916.1971.tb12312.x [Google Scholar]
- Li CW, Han Y, Wu LH, Chen K, An LN. Fabrication and properties of porous anorthite ceramics with modelling pore structure. Mater. Lett. 2017, 190, 95–98. DOI:10.1016/j.matlet.2016.12.131 [Google Scholar]
- Wu LH, Li CW, Chen YF, Wang CA. Seed assisted in-situ synthesis of porous anorthite/mullite whisker ceramics by foam-freeze casting. Ceram. Int. 2021, 47, 11193–11201. DOI:10.1016/j.ceramint.2020.12.244 [Google Scholar]
- Ma GS, Xia L, Zhang T, Zhong B, Yang H, Xiong L, et al. Permeability and thermal expansion properties of porous LAS ceramic prepared by gel-casting method. J. Eur. Ceram. Soc. 2020, 40, 3462–3468. DOI:10.1016/j.jeurceramsoc.2020.03.056 [Google Scholar]
- Roy S. Thermal properties of porous ceramics. Open Ceram. 2025, 24, 100867, DOI:10.1016/j.oceram.2025.100867 [Google Scholar]
- Nacken M, Heidenreich S, Hackel M, Schaub G. Catalytic activation of ceramic filter elements for combined particle separation, NOx removal and VOC total oxidation. Appl. Catal. B Environ. 2007, 70, 370–376. DOI:10.1016/j.apcatb.2006.02.030 [Google Scholar]
- Xu XH, Liu X, Wu JF, Zhang C, Tian KZ, Yu JQ. Effect of preparation conditions on gas permeability parameters of porous SiC ceramics. J. Eur. Ceram. Soc. 2021, 41, 3252–3263. DOI:10.1016/j.jeurceramsoc.2021.01.015 [Google Scholar]
- Carter RE. Mechanism of Solid-state Reaction Between Magnesium Oxide and Aluminum Oxide and Between Magnesium Oxide and Ferric Oxide. J. Am. Ceram. Soc. 1961, 44, 116–120. DOI:10.1111/j.1151-2916.1961.tb13724.x [Google Scholar]
- Yan JJ, Yan W, Chen Z, Nath M, Liao N, Li GQ, et al. A strategy for controlling microstructure and mechanical properties of microporous spinel (MgAl2O4) aggregates from magnesite and Al(OH)3. J. Alloys Compd. 2022, 896, 163088. DOI:10.1016/j.jallcom.2021.163088 [Google Scholar]
- Salomão R, Arruda CC, Pandolfelli VC, Fernandes L. Designing high-temperature thermal insulators based on densification-resistant in situ porous spinel. J. Eur. Ceram. Soc. 2021, 41, 2923–2937. DOI:10.1016/j.jeurceramsoc.2020.12.014 [Google Scholar]
- Das D, Kayal N. Permeability and dust filtration behaviour of porous SiC ceramic candle filter. Mater. Today Proc. 2021, 39, 1235–1240. DOI:10.1016/j.matpr.2020.04.090 [Google Scholar]
- Zheng DZ, Hu YW, Liang SO, Wang ZM. Enhancing high-temperature gas filtration performance of mullite fiber-based porous ceramics by optimizing the fiber-to-colloid ratio. J. Alloys Compd. 2025, 1036, 181632. DOI:10.1016/j.jallcom.2025.181632 [Google Scholar]