Beneficial Effects of Food Containing Lactononadecapeptide on Memory Function in Elderly Japanese Subjects—A Randomized, Double-Blind, Placebo-Controlled Study
Shizuo Yamada 1,* Michiyo Shirai 1 Fumiya Nakamura 2 Kazuhito Ohsawa 2 Manabu Watanabe 3 Michiko Koga 3
Received: 08 December 2025 Revised: 20 January 2026 Accepted: 02 March 2026 Published: 13 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Due to societal aging in Japan, the incidence of diseases accompanied by cognitive and memory impairments, such as dementia, is increasing. Alzheimer’s disease (AD), one type of dementia, has an unclear etiology; however, since patients have reduced levels of acetylcholine, a neurotransmitter in the brain, an impairment in the function of cholinergic neurons has been proposed as one of the underlying causes. The development of brain function-improving foods that may be safely consumed and taken preventively is anticipated [1,2]. Among the different types of nutrients found in the diet, milk fat globules, particularly polar lipids, are very important for cognition due to their abundance in the brain [3]. Research on the preventive effects of milk and dairy products on cognitive decline has increased in recent years. Liu et al. [4] showed that long-term intake of Tibetan fermented milk exerted beneficial effects on intestinal flora composition, which correlated with cognitive improvements in amyloid precursor protein (APP)/presenilin-1 (PS1) transgenic mice and appeared to help prevent and treat AD-induced cognitive decline. Furthermore, a prospective cohort study (Hisayama study) on Japanese subjects showed that increases in the intake of milk and dairy products reduced the risk of dementia, particularly AD [5].
Milk contains numerous peptides that are involved in crucial physiological functions, including hormone secretion, immune defenses, nutrient uptake, and neurotransmission [6]. Supplementation with a proline-rich polypeptide complex for 12 months improved cognitive dysfunction in AD patients with mild to moderate dementia [7]. Ohsawa et al. [8,9] demonstrated that Calpis sour milk whey, cultured milk fermented with Lactobacillus helveticus and Saccharomyces cerevisiae, was effective against scopolamine-induced memory impairment in mice, and the main active component was identified as the 19-residue peptide, Asn-Ile-Pro-Pro-Leu-Thr-Gln-Thr-Pro-Val-Val-Val-Pro-Pro-Phe-Leu-Gln-Pro-Glu (NIPPLTQTPVVVPPFLQPE, lactononadecapeptide (LNDP)). They also showed that supplementation with L. helveticus-fermented milk containing LNDP for eight weeks improved both attention and delayed memory in healthy middle-aged subjects [10]. Therefore, food containing several milligrams of LNDP may effectively prevent neurodegenerative disorders, such as AD, and enhance learning and memory in humans. The present study investigated the effects of food containing LNDP on memory function in elderly Japanese subjects using the Rey-Osterrieth complex figure (ROCF) test [11,12].
2. Materials and Methods
2.1. Test Food
The test food was consumed over 24 weeks from 16 May 2017, to 21 March 2018. The test food was provided by Asahi Group Holdings, Ltd., and consisted of test food containing 2.4 mg LNDP as a daily dose in tablet form, called Surasura Care® (Asahi Group Foods, Ltd., Tokyo, Japan). The placebo food consisted of dextrin without LNDP. The safety of the test food was previously confirmed in a 12-week long-term human consumption study (576 mg/day), a 4-week human overdose study (2880 mg/day), a chromosome aberration test using Chinese hamster lung-derived fibroblasts (CHL/IU), a single-dose toxicity study on rats (2000 mg/kg), and a 4-week repeated dose toxicity study on rats (1000 mg/kg/day) [8,9,10]. The test food and placebo food were both administered at a dose of 4 tablets once daily for 24 weeks.
2.2. Study Design
The present study was conducted as a randomized, double-blind, placebo-controlled, parallel-group comparison. The study protocol was approved by the Research Review Committee of the University of Shizuoka on 3 February 2017 (No. 28-45), and the study was conducted after public disclosure on 19 April 2017, via UMIN-ICDR (University Hospital Medical Information Network; UMIN000027064). The study was conducted in accordance with the Helsinki Declaration and the “Clinical Research Act and its Enforcement Regulations”, ensuring the adequate consideration of the human rights, safety, and welfare of the study participants. Study participants were recruited at medical institutions and local community organizations, and written informed consent was obtained from participants after fully explaining the study content. The study period began on 15 May 2017, with participant recruitment, and the observation period ended on 23 March 2018. No changes were made to the study protocol.
2.3. Study Participants
The present study included 114 elderly individuals who were patients at the following facilities: Seirei Welfare Foundation Hamana Lake Eden (Hamamatsu), Shunko Kai Community Hospital Koga Hospital (Yaizu), Seishu Kai Takido Hospital (Shizuoka), and Shuuji Kai Hagiwara Hospital (Shizuoka), as well as elderly individuals residing in the local community who were able to participate in the study. The target sample size was calculated based on a similar study on the primary outcome measure (figure reproduction, a subitem of the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) with an intergroup difference of 5.5, SD = 10, α = 0.05, and power = 0.8. In consideration of drop-outs and withdrawals, an additional 10 subjects were added, resulting in 114 subjects.
2.4. Selection Criteria
-
-
Provision of written informed consent from subjects prior to participation in the study
-
-
Participants aged 65 years or older who are aware of their own forgetfulness
-
-
The ability to consume 90% or more of the test food during the trial period
-
-
The ability to maintain a diary during the consumption period
-
-
Participants with a Mini-Mental State Examination (MMSE) score of 24 to 27
2.5. Exclusion Criteria
-
-
Individuals at risk of allergic reactions to the study product component (milk)
-
-
Individuals with dementia
-
-
Individuals who have undergone hospitalization, surgery, or treatment for stroke, subarachnoid hemorrhage, cerebral infarction, cerebral hemorrhage, or traumatic brain injury
-
-
Individuals with a history of epilepsy or diabetes, or currently receiving treatment for these conditions
-
-
Individuals currently taking medications that may affect cognitive function (e.g., antipsychotics, anxiolytics, antidepressants, antiparkinsonian agents, and anticonvulsants)
-
-
Individuals currently participating in or planning to participate in other clinical trials at the start of this trial
-
-
Other individuals deemed ineligible by the trial investigator
2.6. Randomization and Blinding
Study participants were 113 healthy elderly individuals, excluding one who did not meet the inclusion criteria after providing informed consent, who were randomly assigned to the placebo and test food groups using a stratified block randomization method by medical institutions. The allocation table was sealed and stored until the end of the trial, after which it was opened.
2.7. Evaluation Methods
Evaluation tests were conducted at the start of consumption and after 24 weeks. The study was terminated if a participant withdrew consent or ceased consumption of the test food. Additionally, any complications or adverse events reported by participants were recorded. Furthermore, if participants were taking medications, newly designated over-the-counter drugs, traditional Chinese medicines, health foods, or supplements unrelated to memory function prior to the start of the study, their dosage and administration were maintained throughout the study period.
2.7.1. Primary Endpoints
The primary outcome measures were the total scores for the copy and immediate recall (3-min) components of the ROCF test administered before test food/placebo intake and 24 weeks later [11,12]. The ROCF test consists of an abstract and complex figure composed of 34 line segments and one circle. The test comprises a copying task, in which participants freely copy the Rey Complex Figure, and an immediate recall task, in which participants draw a figure from memory 3 min after viewing the original. The scoring method uses a 36-point scale, with 2 points being awarded if the shape and position are accurate, 1 point if the shape is accurate, but the position is inaccurate, 1 point if the shape is inaccurate, but the position is accurate, 0.5 points if the shape and position are both inaccurate, but the figure is recognized as drawn, and 0 points if the figure is not drawn at all. Inter-rater reliability was set at 85% agreement between two evaluators. When discrepancies arose in the evaluation results, the evaluators discussed and selected the final score.
2.7.2. Secondary Evaluation Items
Secondary evaluation items included the total score of the Mini-Mental State Examination-Japanese version (MMSE-J) before test food/placebo intake and after 24 weeks, the total score of the Category Fluency Test (CFT), the total score of the Symbol Digit Modalities Test (SDMT), a self-made Visual Analogue Scale (VAS) method memory loss questionnaire, and a self-made 6-item questionnaire.
-
-
MMSE-J is a method for measuring cognitive function that assesses the severity of cognitive impairment at a specific point in time and tracks changes in cognitive function over time. It consists of 11 categories: orientation (time and place), memory, attention and calculation, reproduction, naming, repetition, comprehension, reading, writing, and drawing, with a maximum score of 30 points [13]. A score of 23 or lower indicates a possibility of dementia (sensitivity 83.9%, specificity 83.5%), while a score of 24 to 27 suggests mild cognitive impairment [14] (sensitivity 68.7%, specificity 78.8%).
-
-
CFT, Verbal fluency test is a language fluency task (Word fluency test and Verbal fluency test). The CFT is a test where participants are asked to name as many common nouns (animals) as possible within 60 s. It is associated with impaired cognitive functions, such as language function, executive function, and information processing speed, in early-stage frontotemporal dementia and AD [15,16].
-
-
The SDMT is a tool for quantitatively assessing attention as part of the Clinical Assessment for Attention, a standard attention assessment method. It is considered to reflect the central executive functions of working memory in cognitive psychology [17,18,19]. The SDMT consists of 110 tasks that evaluate the “distributive attention” required to allocate attention simultaneously to multiple pieces of information. Participants are presented with a table containing nine symbols and numbers and are instructed to fill in the numbers corresponding to the symbols within a time limit of 90 s.
-
-
The self-made VAS method memory loss questionnaire is a questionnaire that measures memory loss using the VAS method for the following items: “Unable to recall people’s names”, “Unable to recall the names of objects”, “Unable to recall what one intended to do”, “Unable to recall what one intended to say”, “Memory of events from a few hours ago”, “Forgetting appointments”, “Forgetting items”, and “Forgetting where items were placed”. Responses are categorized as “Can recall” or “Always unable to recall”.
2.7.3. Safety Evaluation Items
General examinations included height, weight, blood pressure, and pulse rate. Blood tests include the white blood cell count, red blood cell count, hemoglobin, the hematocrit, platelet count, AST (GOT), ALT (GPT), γ-GTP, total bilirubin, ALP, CHE, LDH, CPK, total protein, albumin, total cholesterol, HDL cholesterol, LDL cholesterol, triglycerides, blood urea nitrogen, creatinine, uric acid, blood glucose, HbA1c, sodium, chloride, potassium, magnesium, calcium, and iron. A urinalysis included protein qualitative, glucose qualitative, urobilinogen qualitative, bilirubin qualitative, hematuria qualitative, ketone body qualitative, specific gravity, and pH. General examinations, blood tests, urine tests, and cognitive function tests were conducted twice: at the start of and 24 weeks after test food/placebo intake. Safety evaluation items included adverse events, side effects, the number and details of serious adverse events and serious side effects, and the number and details of cases where cognitive therapy medications were initiated and evaluated through general examinations and blood tests.
2.8. Statistical Analysis
Background information was compared between the placebo and test food groups to confirm no significant differences in baseline characteristics. Regarding outcome measures, the change from baseline to 24 weeks was analyzed using an unpaired Student’s t-test. The significance level was set at 0.05, and SPSS Statistics Base Ver. 25 was used for statistical analyses. Intergroup comparisons between the placebo and test food groups were performed using unpaired Student’s t-tests, with changes in scores between baseline and 24 weeks as the outcome measure. The significance level was set at 0.05, and SPSS Statistics Base Ver. 25 was used.
3. Results
3.1. Population Analyses
Figure 1 shows a flow chart of study participants. The study was conducted as planned without any changes to the study protocol. A total of 113 participants enrolled in the study. Among randomized participants, one who had never consumed the test food and seven who withdrew their consent were excluded, resulting in a Full Analysis Set of 105 participants. The Safety Analysis Set consisted of 105 participants, excluding 3 who deviated from the selection criteria and 9 who violated the exclusion criteria. The Per Protocol Set, which was the population compliant with the clinical trial protocol, consisted of 93 participants.
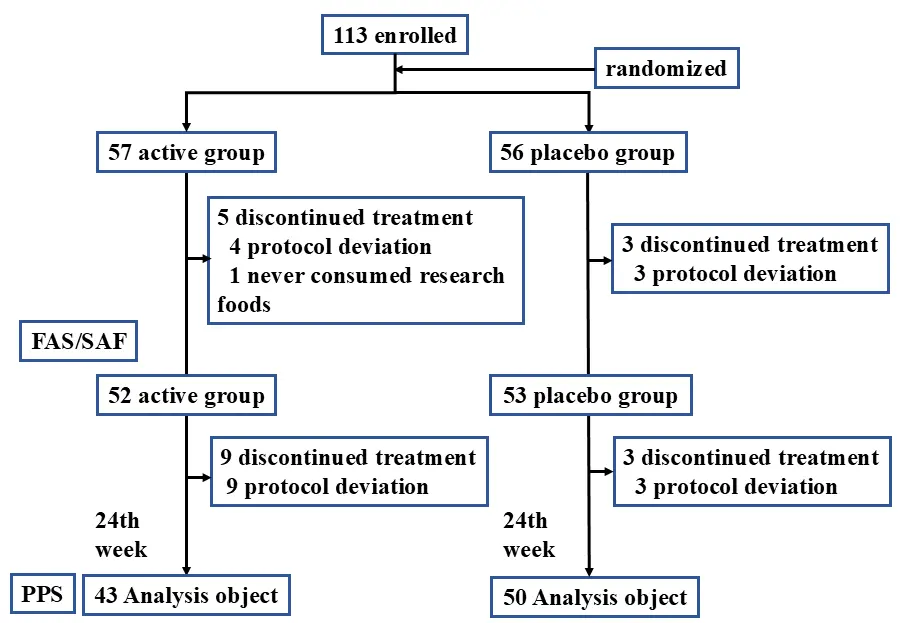
Figure 1. Flow diagram of participants in the test food and placebo groups throughout the study. FAS: Full Analysis Set; SAF: Safety Analysis Set; PPS: Per Protocol Set.
The breakdown by group was as follows: by age, the test food group included 65–69 years old (8 subjects), 70–74 years old (7 subjects), 75–79 years old (15 subjects), and 80 years or older (13 subjects), and the placebo group included 65–69 years old (11 subjects), 70–74 years old (13 subjects), 75–79 years old (14 subjects), and 80 years or older (12 subjects). No significant differences were observed in baseline characteristics between the test food and placebo groups (Table 1).
Table 1. Comparison of baseline characteristics of subjects in the test food and placebo groups.
|
Variables |
Test Food Group (n = 52) |
Placebo Group (n = 53) |
p Value |
||
|---|---|---|---|---|---|
|
Mean |
SD |
Mean |
SD |
||
|
Sex (male/female) |
18/34 |
17/36 |
0.78 |
||
|
Age (years) |
76.8 |
6.0 |
75.2 |
6.6 |
0.22 |
|
MMSE-J |
25.8 |
1.1 |
25.9 |
1.1 |
0.70 |
|
CFT |
15.0 |
4.3 |
13.9 |
3.4 |
0.17 |
|
ROCF-Copy |
34.9 |
1.5 |
34.6 |
3.4 |
0.61 |
|
ROCF-Immediate recall (3-min) |
16.9 |
6.5 |
16.9 |
7.0 |
0.99 |
|
SDMT |
33.5 |
9.4 |
33.6 |
9.5 |
0.96 |
|
AST (GOT) (U/L) |
23.5 |
5.2 |
24.3 |
10.0 |
0.59 |
|
ALT (GPT) (U/L) |
17.2 |
5.9 |
18.4 |
12.7 |
0.53 |
|
γ-GTP (IU/L) |
24.2 |
13.9 |
35.4 |
48.2 |
0.11 |
|
TP (Total protein) (g/dL) |
7.1 |
0.5 |
7.1 |
0.5 |
0.58 |
|
Alb (albumin) (g/dL) |
4.1 |
0.3 |
4.2 |
0.3 |
0.13 |
|
Total cholesterol (mg/dL) |
196.5 |
34.8 |
198.2 |
42.9 |
0.83 |
|
HDL cholesterol (mg/dL) |
56.9 |
12.9 |
57.2 |
14.9 |
0.91 |
|
LDL cholesterol (mg/dL) |
112.5 |
28.3 |
106.8 |
33.6 |
0.36 |
|
Triglycerides (mg/dL) |
145.1 |
73.4 |
180.6 |
231.4 |
0.29 |
|
HbA1c (NGSP) (%) |
5.8 |
0.7 |
5.8 |
0.4 |
0.76 |
|
Na (mmol/L) |
141.8 |
2.3 |
142.2 |
2.4 |
0.46 |
|
Cl (mmol/L) |
105.4 |
2.5 |
105.1 |
2.3 |
0.53 |
|
K (mmol/L) |
4.2 |
0.4 |
4.2 |
0.4 |
0.80 |
|
Fe (μg/dL) |
91.6 |
30.7 |
89.1 |
29.2 |
0.67 |
Mean test values in the test food and placebo groups were analyzed using a paired t-test.
3.2. Efficacy Evaluation
No significant difference was noted in the primary endpoint, the ROCF test result, between the test food and placebo groups (Table 2). On the other hand, since the ROCF test is a 36-point test requiring a high drawing ability, a subgroup analysis was conducted using a ROCF-Copy score of 33 points or higher, which is the average value for those aged 70 years or older (32.9 ± 2.7) [20], to remove the drawing ability bias. As a result, the mean change in the total score of ROCF-Copy was significantly higher in the test food (containing LNDP) group than in the placebo group (test food group: 0.2 ± 1.3, placebo group: −0.7 ± 1.5, p = 0.01) (Table 3). Furthermore, in the secondary evaluation items, the test food group showed significantly higher scores than the placebo group in the SDMT (test food group: 2.8 ± 4.2, placebo group: 1.1 ± 3.6, p = 0.04) (Table 2). Additionally, in subscale Q3 “Forgetfulness-what you tried to do” of the self-made VAS method memory loss questionnaire, the test food group showed significantly higher scores than the placebo group (test food group: −0.5 ± 2.0, placebo group: 0.4 ± 2.0, p = 0.03) (Table 2).
Table 2. Comparison of changes in evaluation items between the test food and placebo groups.
|
Variables |
Test Food Group (n = 43) |
Placebo Group (n = 50) |
p Value |
||
|---|---|---|---|---|---|
|
Mean |
SD |
Mean |
SD |
||
|
MMSE-J |
1.3 |
2.2 |
1.7 |
2.1 |
0.38 |
|
CFT |
0.5 |
3.8 |
1.3 |
2.6 |
0.22 |
|
ROCF-Copy # |
0.0 |
1.8 |
−0.4 |
1.9 |
0.29 |
|
ROCF-Immediate recall (3-min) # |
1.1 |
4.9 |
2.4 |
5.7 |
0.24 |
|
SDMT |
2.8 |
4.2 |
1.1 |
3.6 |
0.04 * |
|
Q1 Forgetfulness-names of people |
−0.8 |
2.8 |
−0.2 |
2.6 |
0.27 |
|
Q2 Forgetfulness-names of things |
−0.5 |
2.5 |
−0.2 |
2.3 |
0.57 |
|
Q3 Forgetfulness-what you tried to do |
−0.5 |
2.0 |
0.4 |
2.0 |
0.03 * |
|
Q4 Forgetfulness-what you tried to say |
−0.4 |
2.4 |
0.3 |
1.9 |
0.11 |
|
Q5 Memory of hours ago |
−0.8 |
2.6 |
0.1 |
2.0 |
0.06 |
|
Q6 Promises |
−0.1 |
1.8 |
0.0 |
2.2 |
0.66 |
|
Q7 Things you forgot |
−0.4 |
2.0 |
−0.1 |
2.1 |
0.44 |
|
Q8 Where you put things |
−0.5 |
1.9 |
0.0 |
2.1 |
0.17 |
|
Survey on forgetfulness of self-made VAS test-Total |
−4.0 |
12.5 |
0.5 |
11.7 |
0.07 |
Differences between the placebo and test food groups were analyzed using an unpaired t-test for means. # n = 91 (test food 43, placebo 48, except for 2 subjects due to an inability to draw). * p < 0.05.
Table 3. Comparison of mean changes in ROCF scores in test food and placebo groups by a subgroup stratified analysis after removal of the drawing ability bias.
|
Variables |
Test Food Group (n = 38) |
Placebo Group (n = 45) |
p Value |
||
|---|---|---|---|---|---|
|
Mean |
SD |
Mean |
SD |
||
|
ROCF-Copy |
0.2 |
1.3 |
−0.7 |
1.5 |
0.01 * |
|
ROCF-Immediate recall (3-min) |
1.1 |
5.1 |
2.3 |
5.5 |
0.34 |
ROCF: Rey-Osterrieth complex figure. Differences between the placebo and test food groups were analyzed using an unpaired t-test for means. * p < 0.05.
3.3. Safety Evaluation
General examinations, blood tests, and urine tests conducted before and after test food/placebo intake showed that values were within the reference range for each parameter, with no significant differences between the two groups, except for significantly higher levels of total protein and lower levels of serum iron in the test food group than in the placebo group. No adverse effects were reported (Table 4). There was little significant difference between active and placebo groups in values of red blood cell, white blood cell, hemoglobin and hematocrit.
Table 4. Comparison of mean changes in basic characteristics and blood test results between the test food and placebo groups.
|
Variable |
Test Food Group (n = 52) |
Placebo Group (n = 53) |
p Value |
||
|---|---|---|---|---|---|
|
Mean |
SD |
Mean |
SD |
||
|
BMI (kg/m2) |
0.6 |
0.6 |
0.3 |
0.8 |
0.10 |
|
Blood pressure (mmHg) |
|||||
|
Systolic |
11.1 |
20.5 |
12.4 |
16.7 |
0.70 |
|
Diastolic |
2.6 |
14.4 |
3.0 |
12.8 |
0.90 |
|
AST (GOT) (U/L) |
1.8 |
4.3 |
2.3 |
14.1 |
0.80 |
|
ALT (GPT) (U/L) |
1.8 |
4.0 |
1.7 |
11.9 |
1.00 |
|
γ-GTP (IU/L) |
1.5 |
4.6 |
−3.3 |
25.2 |
0.20 |
|
TP (Total protein) (g/dL) |
0.2 |
0.3 |
0.0 |
0.4 |
0.00 ** |
|
Alb (albumin) (g/dL) |
0.1 |
0.2 |
0.0 |
0.2 |
0.10 |
|
Total cholesterol (mg/dL) |
8.9 |
19.6 |
4.4 |
21.1 |
0.30 |
|
HDL cholesterol (mg/dL) |
3.2 |
7.2 |
2.7 |
7.3 |
0.70 |
|
LDL cholesterol (mg/dL) |
2.2 |
16.3 |
2.4 |
15.8 |
0.90 |
|
Triglycerides (mg/dL) |
−6.5 |
57.4 |
−22.7 |
102.6 |
0.30 |
|
HbA1c (NGSP) (%) |
−0.1 |
0.4 |
0.1 |
0.2 |
0.10 |
|
Na (mmol/L) |
−0.5 |
2.1 |
0.1 |
2.8 |
0.20 |
|
Cl (mmol/L) |
−1.5 |
2.5 |
−0.7 |
2.2 |
0.10 |
|
K (mmol/L) |
0.2 |
0.4 |
0.2 |
0.5 |
1.00 |
|
Ca (mg/dL) |
0.1 |
0.4 |
0.0 |
0.3 |
0.10 |
|
Fe (μg/dL) |
−5.8 |
30.3 |
9.9 |
35.3 |
0.00 ** |
Differences between the placebo and test food groups were analyzed using an unpaired t-test for means. ** p < 0.01.
4. Discussion
The intake of dairy products and the regular consumption of low-fat dairy products have been suggested to exert positive effects on neurocognitive health during aging [21,22]. Increased awareness of the effects of nutrients on physical and mental health has prompted interest in using milk and other dairy products to prevent or ameliorate age-related cognitive decline and dementia [23]. Ozawa et al. [5] found that increased intake of milk and dairy products attenuated increases in dementia incidence, particularly AD, in an elderly Japanese population. Ohsawa et al. [8] also examined the effects of a L. helveticus-fermented milk product, the whey of Calpis™ sour milk (Asahi Group Holdings, Ltd., Tokyo, Japan), on short-term memory impairment. They demonstrated that Calpis™ sour milk whey improved scopolamine-induced memory impairment in mice, and β-casein-derived LNDP was identified as the pharmacologically active ingredient in Calpis™ sour milk whey [9]. Furthermore, the intake of L. helveticus-fermented food containing LNDP improved the cognitive function of Japanese adults [10]. This preliminary human intervention study examined the effects of consuming the test food for 8 weeks in healthy middle-aged subjects, and cognitive function was assessed using a neuropsychological status (RBANS) test. The supplementation with LNDP in tablet form appeared to improve cognitive functions, including memory, attention, concentration, and information processing, in daily life, in healthy Japanese subjects aged 30–59 years as assessed by the Cognitrax [24], or aged 60–79 years, as assessed by the Uchida-Kraepelin test [25].
In the present study, the total score of the ROCF test, specifically the Copy and Immediate Recall (3-min) subscale scores, was extensively compared in elderly Japanese subjects with mild cognitive impairment of MMSE of 24–27 who consumed the test food (containing LNDP) and placebo for 24 weeks. In the stratified subgroup analysis of subjects with a ROCF-Copy score of 33 points or higher, the mean change in the total score of ROCF-Copy was significantly higher in the test food group than in the placebo group (Table 3). Furthermore, in the secondary evaluation, the test food group had significantly higher scores than the placebo group in the SDMT (Table 2). Additionally, in subscale Q3, “Forgetfulness-what you tried to do”, of the self-made VAS method memory loss questionnaire, the test food group showed significantly higher scores than the placebo group (Table 2). These results have shown that the repeated intake of food containing LNDP may significantly improve memory function in elderly subjects based on the results of the ROCF test and SDMT. The ROCF test and SDMT are characteristic tests that assess visual memory and attention [11,12,17,18,19]. Therefore, the results obtained herein indicate that the repeated intake of food containing LNDP may exert beneficial effects on visual working memory and attention function with aging in elderly subjects. Yamada et al. [26,27] previously reported that a nobiletin-containing food and formulated supplements of plasmalogen and elastin may be beneficial for improving visual memory. Therefore, similar to these supplements, food containing LNDP has potential as a visual care support for improving cognitive function.
LNDP was previously shown to improve amyloid β peptide-induced memory impairment in mice [28]. The precise molecular mechanisms of action of LNDP on brain neurons remain unknown. A previous study showed that memory formation required the expression of a large number of genes related to memory and transcription [29]. Therefore, we investigated the effects of LNDP on learning and memory-related gene expression in human-induced pluripotent stem cell-derived neurons using real-time quantitative RT-PCR. LNDP significantly up-regulated the gene expression of the neurotrophic factors, brain-derived neurotrophic factor and nerve growth factor, a family of receptor tyrosine kinases (Trk receptors), the muscarinic M1 receptor subtype, choline acetyltransferase, and N-methyl-D-aspartate receptors (Kimura et al., unpublished results). Overall, the up-regulation of gene expression for these learning and memory-related molecules by LNDP may contribute, in part, to the improvement in cognitive function observed in elderly subjects following repeated intake of food containing LNDP. Brain aging is associated with prominent neurodegenerative changes and oxidative damage. Whey protein has been shown to possess antioxidant properties [30]. The nuclear factor erythrogen-2 associated factor 2 (Nrf2) signaling pathway is an antioxidant defense system, and Nfr2 activity declines with age. Whey protein has been shown to activate the Nrf2 pathway, reducing cell senescence and gliosis [30]. LNDP may also act on functional proteins and enhance the Nrf2 signaling pathway, thereby attenuating cognitive dysfunction.
5. Conclusions
The present study has shown that the intake of food containing LNDP for 24 weeks by Japanese elderly subjects with mild cognitive decline improved cognitive and memory function based on the results of the ROCF test.
Acknowledgments
The authors would like to thank Kazuhiko Matsumoto for his assistance with statistical analyses. We also thank Michiyo Ishida, Eriko Amano, and Naomi Kajiyama for their support throughout the project.
Author Contributions
Study design and concept, S.Y., M.S., F.N., K.O. and M.K. Data collection, M.S., M.W. and M.K. Data analysis, S.Y. and M.S. All authors contributed to preparing the draft and gave their final approval of the manuscript for publication. All authors have read and agreed to the published version of the manuscript.
Ethics Statement
This study was conducted according to the guidelines of the Declaration of Helsinki (2013). Ethical approval for the trial was obtained from the Research Review Committee of University of Shizuoka on 3 February 2017 (No. 28-45).
Informed Consent Statement
Informed consent was obtained from all subjects involved in this study.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Funding
This study was funded by Asahi Group Holdings, Ltd. (Tokyo, Japan).
Declaration of Competing Interest
F.N. and K.O. are employed by Asahi Quality & Innovations, Ltd., a group company under Asahi Group Holdings, Ltd. This affiliation may be considered a potential conflict of interest. The company has no role in data collection or data analysis.
References
- Fekete M, Lehoczki A, Tarantini S, Fazekas-Pongor V, Csípő T, Csizmadia Z, et al. Improving cognitive function with nutritional supplements in aging: A comprehensive narrative review of clinical studies investigating the effects of vitamins, minerals, antioxidants, and other dietary supplements. Nutrients. 2023, 15, 5116. DOI:10.3390/nu15245116 [Google Scholar]
- Kim B, Hong VM, Yang J, Hyun H, Im JJ, Hwang J, et al. A review of fermented foods with beneficial effects on brain and cognitive function. Prev. Nutr. Food Sci. 2016, 21, 297–309. DOI:10.3746/pnf.2016.21.4.297 [Google Scholar]
- Luque-Uría Á, Calvo MV, Visioli F, Fontecha J. Milk fat globule membrane and its polar lipids: Reviewing preclinical and clinical trials on cognition. Food Funct. 2024, 15, 6783–6797. DOI:10.1039/D4FO00659C [Google Scholar]
- Liu J, Yu C, Li R, Liu K, Jin G, Ge R, et al. High-altitude Tibetan fermented milk ameliorated cognitive dysfunction by modified gut microbiota in Alzheimer’s disease transgenic mice. Food Funct. 2020, 11, 5308–5319. DOI:10.1039/C9FO03007G [Google Scholar]
- Ozawa M, Ohara T, Ninomiya T, Hata J, Yoshida D, Mukai N, et al. Milk and dairy consumption and risk of dementia in an elderly Japanese population: The Hisayama Study. J. Am. Geriatr. Soc. 2014, 62, 1224–1230. DOI:10.1111/jgs.12887 [Google Scholar]
- Clare DA, Swaisgood HE. Bioactive milk peptides: A prospectus. J. Dairy Sci. 2000, 83, 1187–1195. DOI:10.3168/jds.S0022-0302(00)74983-6 [Google Scholar]
- Leszek JE, Inglot AD, Janusz MA, Lisowski JÓ, Krukowska KA, Georgiades JA. Colostrinin: A proline-rich polypeptide (PRP) complex isolated from ovine colostrum for treatment of Alzheimer’s disease. A double-blind, placebo-controlled study. Clin. Trial Arch. Immunol. Ther. Exp. 1999, 47, 377–385. [Google Scholar]
- Ohsawa K, Uchida N, Ohki K, Nakamura Y, Yokogoshi H. Lactobacillus helveticus-fermented milk improves learning and memory in mice. Nutr. Neurosci. 2015, 18, 232–240. DOI:10.1179/1476830514Y.0000000122 [Google Scholar]
- Ohsawa K, Uchida N, Ohki K, Yokogoshi H. Identification of peptides present in sour milk whey that ameliorate scopolamine-induced memory impairment in mice. Int. J. Food Sci. Nutr. 2018, 69, 33–45. DOI:10.1080/09637486.2017.1324564 [Google Scholar]
- Ohsawa K, Nakamura F, Uchida N, Mizuno S, Yokogoshi H. Lactobacillus helveticus-fermented milk containing lactononadecapeptide (NIPPLTQTPVVVPPFLQPE) improves cognitive function in healthy middle-aged adults: A randomised, double-blind, placebo-controlled trial. Int. J. Food Sci. Nutr. 2018, 69, 369–376. DOI:10.1080/09637486.2017.1365824 [Google Scholar]
- Tombaugh TN, Hubley AM. Four studies comparing the Rey-Osterrieth and Taylor complex figures. J. Clin. Exp. Neuropsychol. 1991, 13, 587–599. DOI:10.1080/01688639108401073 [Google Scholar]
- Tremblay MP, Potvin O, Callahan BL, Belleville S, Gagnon JF, Caza N, et al. Normative data for the Rey-Osterrieth and the Taylor complex figure tests in Quebec-French people. Arch. Clin. Neuropsychol. 2015, 30, 78–87. DOI:10.1093/arclin/acu069 [Google Scholar]
- Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. DOI:10.1016/0022-3956(75)90026-6 [Google Scholar]
- Lee CH, Kim JI, Lee KM, Park JH, Bae K. Neurocognitive assessment in relation to hearing impairment and retinal neurodegeneration. Neurol. Sci. 2025, 46, 4309–4320. DOI:10.1007/s10072-025-08305-5 [Google Scholar]
- Kawano N, Umegaki H, Suzuki Y, Yamamoto S, Mogi N, Iguchi A. Effects of educational background on verbal fluency task performance in older adults with Alzheimer’s disease and mild cognitive impairment. Int. Psychogeriatr. 2010, 22, 995–1002. DOI:10.1017/S1041610210000839 [Google Scholar]
- McDonnell M, Dill L, Panos S, Amano S, Brown W, Giurgius S, et al. Verbal fluency as a screening tool for mild cognitive impairment. Int. Psychogeriatr. 2020, 32, 1055–1062. DOI:10.1017/S1041610219000644 [Google Scholar]
- Forn C, Belenguer A, Belloch V, Sanjuan A, Parcet MA, Ávila C. Anatomical and functional differences between the Paced Auditory Serial Addition Test and the Symbol Digit Modalities Test. J. Clin. Exp. Neuropsychol. 2011, 33, 42–50. DOI:10.1080/13803395.2010.481620 [Google Scholar]
- Benedict RH, DeLuca J, Phillips G, LaRocca N, Hudson LD, Rudick R. Validity of the Symbol Digit Modalities Test as a cognition performance outcome measure for multiple sclerosis. Mult. Scler. J. 2017, 23, 721–733. DOI:10.1177/1352458517690821 [Google Scholar]
- Sheridan L, Fitzgerald H, Adams K, Nigg J, Martel M, Puttler L, et al. Normative Symbol Digit Modalities Test performance in a community-based sample. Arch. Clin. Neuropsychol. 2006, 21, 23–28. DOI:10.1016/j.acn.2005.07.003 [Google Scholar]
- Golden CJ, Espe-Pfeifer P, Wachsler-Felder J. Neuropsychological Interpretation of Objective Psychological Tests; Critical Issues in Neuropsychology (CINP); Springer: New York, NY, USA, 2002. [Google Scholar]
- Crichton GE, Murphy KJ, Bryan J. Dairy intake and cognitive health in middle-aged South Australians. Asia Pac. J. Clin. Nutr. 2010, 19, 161–171. [Google Scholar]
- Crichton GE, Elias MF, Dore GA, Robbins MA. Relation between dairy food intake and cognitive function: The Maine-Syracuse Longitudinal Study. Int. Dairy J. 2012, 22, 15–23. DOI:10.1016/j.idairyj.2011.08.001 [Google Scholar]
- Camfield DA, Owen L, Scholey AB, Pipingas A, Stough C. Dairy constituents and neurocognitive health in ageing. Br. J. Nutr. 2011, 106, 159–174. DOI:10.1017/S0007114511000158 [Google Scholar]
- Sasai M, Kato M, Ohsawa K, Sashihara K, Nakamura Y, Kaneko T. Effects of a single dose of tablets containing lactononadecapeptide on cognitive function in healthy adults: A randomized, double-blind, crossover, placebo-controlled trial. Biosci. Biotechnol. Biochem. 2021, 85, 948–956. DOI:10.1093/bbb/zbaa117 [Google Scholar]
- Nakamura F, Ohsawa K, Nakamura Y, Sawabe A, Miura N, Okuda J. Effect of tablets containing lactononadecapeptide (NIPPLTQTPVVVPPFLQPE) on cognitive function in healthy middle-aged and senior adults. Jpn. Pharmacol. Ther. 2017, 45, 1303–1314. [Google Scholar]
- Yamada S, Shirai M, Ono K, Teruya T, Yamano A, Tae Woo J. Beneficial effects of a nobiletin-rich formulated supplement of Siikuwha (C. depressa) peel on cognitive function in elderly Japanese subjects; A multicenter, randomized, double-blind, placebo-controlled study. Food Sci. Nutr. 2021, 9, 6844–6853. DOI:10.1002/fsn3.2640 [Google Scholar]
- Yamada S, Shirai M, Katsumata M, Kurono M, Matahira Y, Ono K, et al. Beneficial effects of a formulated supplement of Ascidiacea (Halocynthia-roretzi)-derived plasmalogen and Tuna-derived elastin on memory function in elderly Japanese subjects; A randomized, double-blind, placebo-controlled study. J. Oleo Sci. 2024, 73, 1319–1328. DOI:10.5650/jos.ess24128 [Google Scholar]
- Nakamura F, Ohsawa K, Sato K, Nakamura Y. Lactononadecapeptide (NIPPLTQTPVVVPPFLQPE) improves amyloid β peptide-induced memory impairment in mice. Pharmacometrics 2018, 95, 9–18. Available online: https://jglobal.jst.go.jp/en/detail?JGLOBAL_ID=201802228617002424 (accessed on 8 December 2025)
- Kimura J, Shimizu K, Takito J, Nemoto K, Degawa M, Yokosuka A, et al. Upregulatory effects of nobiletin, a citrus flavonoid with anti-dementia activity, on the gene expression of mAChR, ChAt, and CBP. Planta Med. Lett. 2015, 2, e12–e14. DOI:10.1055/s-0035-1545937 [Google Scholar]
- Sarhan NR, El Nashar EM, Hamza E, El-Beah SM, Alghamdi MA, Al-Khater KM, et al. Nuclear factor erythrogen-2 associated factor 2 (Nrf2) signaling is an essential molecular pathway for the anti-aging effect of whey protein in the prefrontal cortex of aging rat model (Histological and Biochemical Study). Tissue Cell. 2023, 84, 102192. DOI:10.1016/j.tice.2023.102192 [Google Scholar]
