A Novel Approach to Synthesis Alkyd Resin from Recycled Polyethylene Terephthalate (rPET)
Received: 07 January 2026 Revised: 02 February 2026 Accepted: 03 March 2026 Published: 12 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
1.1. Significant Environmental Issue: Global Warming
Global warming is recognized as a critical challenge worldwide due to its harmful impact on the environment. A goal has been established to reduce global greenhouse gas emissions by 2.6 percent by 2030, relative to 2019 levels, as per the UN Environment Programmer’s (UNEP) Emissions Gap Report 2024 [1]. This imposes a considerable obligation on every nation to tackle this issue. To meet this target, emissions must be cut by 43 percent by 2030, with the aim of achieving net zero by 2050 [2,3]. Various initiatives are being implemented across all sectors globally to minimize carbon footprints. In particular, the chemical industry is focusing on developing sustainable products to address environmental issues, including waste management and the depletion of fossil fuel resources.
1.2. Polyethylene Terephthalate (PET): Major Contributor to Plastic Waste and Environmental Issues
Plastic waste is one of the major contributors to carbon emissions, particularly polyethylene terephthalate (PET), which is widely used in commodity applications [4,5]. The global production of PET exceeds eighty-two million metric tons annually, making it a significant source of waste and environmental problems [6,7]. The waste PET is utilized across various sectors, including textiles, packaging, construction, chemicals, and coatings by chemical recycling. In the coatings industry, PET waste undergoes chemical recycling to produce alkyd and polyester resins [8].
1.3. Global Market of Alkyd Resin
Alkyd resin, a workhorse in the paint industry, offers various properties, such as flexibility, gloss, and air-drying capability, making it a popular choice for coatings [9]. The worldwide market for alkyd resins is projected to increase from USD 23 billion to approximately USD 39 billion by the year 2030, reflecting a compound annual growth rate of about 7.5% [10,11]. Basically, alkyd is an oil-modified polyester resin produced through the condensation reaction of polyols with polybasic acids to form ester linkages [12]. rPET has been used in the production of as such polyester resin at commercial scale, but producing alkyd (oil-based polyester) faces a number of challenges [13,14]. As triglyceride (oil) is integrated into polyester reactions through monoglyceride formation via the transesterification reaction with polyol. Alkyd is categorized based on the percentage of oil content in an alkyd, like long oil alkyd (oil content exceeding 55%) utilized in air drying systems for decorative enamels, industrial enamels, and wood finishes. Medium oil alkyd (45–55% oil length) is employed in primer applications for decorative and industrial coatings. Short oil alkyd (less than 45% oil length) is suitable for quick-drying industrial paints, auto-refinish, wood coatings, and more [15]. The oil length formula is given in Equation (1) below. The best way of chemical recycling of rPET is the utilization of rPET into alkyd resin synthesis.
1.4. Utilization of Recycled Polyethylene Terephthalate in Alkyd Resin Synthesis
The conventional method for chemical recycling of PET involves glycolysis, in which ethylene glycol breaks the ester linkage to form bis(2-hydroxylethyl terephthalate) (BHET) [16]. This BHET is then used in the alkyd esterification reaction, as demonstrated by Nawal E. et al., who synthesized the medium oil alkyd resin from glycolyzed PET waste using propylene glycol and diethylene glycol [17,18,19]. Vandana Jamdar et al. also explored chemical recycling of PET waste through reaction with ethylene glycol [20]. Büyükyonga et al. investigated the glycolysis reactions of waste PET flakes using dipropylene glycol, diethylene glycol, and triethylene glycol, with the resultant products used in the formulation of medium-oil acrylic-modified water-reducible alkyd resins [21]. Spasojević et al. investigated the glycolysis of waste PET using multifunctional alcohols such as glycerol, trimethylolethane, trimethylolpropane, pentaerythritol, diethylene glycol, propylene glycol, and dipropylene glycol, which were then utilized in alkyd resin synthesis [22]. While the conventional approach has several limitations like two-step synthesis, inability to produce long oil alkyd, and long drying time. The new proposed synthesis route presented in this research work offers the advantage of a single step synthesis methodology that directly introduces rPET into the reactor during alkyd resin synthesis, allowing for simultaneous rPET depolymerization and resin formation, partially replacing the petroleum-based polyol or polyacid typically used in the resin composition. This innovative approach provides satisfactory drying time and improved performance properties. The main objective of this research work is to produce a sustainable product that aligns with the principles of the circular economy, aiming to replace the end-of-life of PET waste with a model focused on sharing, leasing, reusing, repairing, refurbishing, and recycling polyethylene terephthalate as long as possible [23].
This research focused on the creation of three long oil alkyd resins utilizing rPET, soybean oil, glycerin, pentaerythritol, phthalic anhydride, dibutyl tin-oxide (DBTO) as a catalyst and ortho xylene as azeotropic solvent. Incorporating different proportions of rPET, Alkyd A includes 8% rPET, Alkyd B has 12% rPET, and Alkyd C comprises 16% rPET. The performance of these resins was evaluated against the benchmark Alkyd D, which does not contain rPET.
2. Experimental
2.1. Materials
Alkali refined soybean oil industrial grade sourced from Adani Wilmer Pvt Ltd. (now often operating as AWL Agri Business Ltd., Ahmedabad, India). The recycled polyethylene terephthalate (rPET) material is acquired from Dalmia Polypro Industries Pvt Ltd., Mumbai, India, with its properties provided in Table 1. Dibutyl tin-oxide (DBTO) catalysts of 98% assay are obtained from Guldbrandsen Chemicals Private Ltd., Vadodara, India. Refined Grade Glycerin with Glycerol content of 98% supplied by Godrej Industries, Mumbai, India. While Phthalic Anhydride of 98% purity is purchased from Thirumalai Chemicals Limited, Chennai, India. Pentaerythritol of Penta Mono grade obtained from Perstorp Chemicals, Mumbai, India. Ortho xylene of 98% purity is utilized from Reliance Industries, Mumbai, India. Mineral turpentine oil (MTO) is obtained from Bharat Petroleum Corporation Limited, Mumbai, India. Cobalt octoate, with a 5% metal content, is used as a surface drier from Arum Pharmachem Pvt. Ltd., Mumbai, India. Zirconium octoate, containing 18% metal content, is employed as a through drier from Notional Specialties Products Company, Vapi, India. Calcium Octoate, with a 3% metal content, is utilized as an auxiliary drier from Bhagwati Allied Products, Chhatrapati Sambhajinagar, India.
Table 1. Properties of recycled PET used for the experimentation. (As per supplier COA).
|
Sr. No. |
Tests |
Units |
Results |
|---|---|---|---|
|
1 |
Visual Appearance |
- |
Flakes |
|
2 |
Intrinsic Viscosity |
dL/g |
0.698 |
|
3 |
Moisture |
Percentage |
0.47 |
|
4 |
Ash |
Percentage |
0.29 |
|
5 |
Bulk Density |
Kg/m3 |
310 |
|
6 |
Flakes Dimension |
mm |
8 to 13 (70%) 2 to 8 (30%) |
|
7 |
Water pH before and after washing flakes |
- |
7.5 |
2.2. Methods
2.2.1. Synthesis of Alkyd Resin Based on rPET
Alkyd A (8% rPET), Alkyd B (12% rPET), and Alkyd C (16% rPET) were synthesized based on the formula of Alkyd D as detailed in Table 2. This formula includes zero percentage of rPET. As we increase the amount of rPET in the formula, the raw materials such as soybean oil, glycerin, DBTO, and ortho xylene remain constant. However, as the quantity of rPET increases, the amounts of raw materials like pentaerythritol and phthalic anhydride decrease in proportion to the quantity of rPET. The synthesis of alkyd resin was conducted in 2-L 4 neck round bottom flask equipped with a stirrer, dean & stark, temperature sensor, water condenser and nitrogen purging tube. The flask was placed in a heating mantle to ensure uniform heating [24]. The stirring speed is maintained at 180 ± 10 rpm. The soybean oil, glycerin, pentaerythritol, and DBTO catalyst were added at room temperature. The heating rate was adjusted to reach the final temperature of 245–250 °C. After one hour of reaching the 245 °C temperature, a sample was taken to measure the extent of monoglyceride formation using alcohol tolerance [25]. Once the desired alcohol tolerance was achieved, the heating of the reaction was stopped. At a temperature of 190 °C, recycled PET flakes were added and heated up to 240 °C for 1 h. Within this hour, the recycled PET underwent depolymerization, resulting in a clear material in the system. The reaction scheme is shown in section 3.2. After 1 h, the material was cooled to 170 °C, and phthalic anhydride, along with ortho-xylene (azeotropic solvent), was charged [26]. The heating was then restarted, and the reaction temperature was raised to 230 °C. Acid value and viscosity of the batch were monitored after the removal of 80% water of the reaction based on the theoretical value [27]. Once the desired acid value and viscosity were achieved, the azeotropic solvent ortho-xylene was removed from the system by flushing with nitrogen gas. The batch was thinned to 60% solids at 170 °C using mineral turpentine oil.
Table 2. Formulation of Alkyd Resin.
|
Raw Materials |
Parts by Weight |
Molecular Weight (g/mol) |
|---|---|---|
|
Soyabean oil |
55.0 |
880 |
|
Pentaerythritol |
7.0 |
136 |
|
Glycerin |
8.0 |
92 |
|
DBTO |
0.05 |
248 |
|
rPET |
0 |
Not applicable |
|
Phthalic Anhydride |
26.80 |
148 |
|
Ortho Xylene |
3.15 |
106 |
|
Total |
100 |
2.2.2. Characterization
Physicochemical properties of the alkyd resins, such as acid value (ASTM D1639), viscosity (ASTM D1545), colour (ASTM D1544), non-volatile matter, and drying, were studied. Differential Scanning Calorimetry (DSC) analysis is used to determine the percentage crystallinity of the rPET sample. The DSC instrument used was TA Instrument Discovery DSC250 Auto by TA Waters, New Castle, DE, USA. The instrument conditions are equilibrate at 0 °C, 0 to 300 °C @ 10 °C/min. The FTIR spectroscopy was utilized to detect the presence of rPET in the alkyd resin. The FTIR analysis was conducted over a range of 4000–400 cm−1 with 16 scans on the FTIR-ATR device model Frontier by PerkinElmer India Private Limited, Thane city, India. The molecular weight of the alkyd resins was determined through Gel Permeation Chromatography, using the Agilent 1260 multi detector system by Agilent Technologies, Santa Clara, CA, USA. Dynamic mechanical analysis (DMA) was performed using the DMAQ 800 equipment by TA Waters, New Castle, DE, USA. With a temperature sweep test ranging from −30 to 150 °C at a rate of 3 °C/min, strain of 0.1%, and frequency of 1 Hz. Stress was applied to the system, and the resulting strain was measured. Thermogravimetric analysis (TGA) was carried out using the Mettler Toledo device TGA1 Star System by Mettler Toledo, Greifensee, Switzerland. In the temperature range of 30–600 °C at a ramp rate of 10 °C per minute under a nitrogen atmosphere with a N2 purging rate of 50 mL/min.
2.2.3. Film Preparation and Performance Evaluation Test Methods
Resin sample (100 g) was combined with 14 g of mineral turpentine oil (MTO) and driers, including 5% cobalt octoate (0.6 g), 18% zirconium octoate (1.67 g), and 3% calcium octoate (4 g) [28]. The quantity of driers used depended on the solid content of the binder. A uniform solution was prepared by stirring and mixing, it was left to sit overnight before being applied to metal panels. Dry film thickness was measured in accordance with ASTM D5796 using a Positector 6000 thickness gauge, while gloss was measured following ASTM D523 with a gloss meter. Scratch hardness was determined using an automatic scratch hardness tester REF 705, as per ASTM D5178. Pencil hardness was evaluated according to ASTM D3363, with hardness expressed as H, 2H, 3H, or 6H. The crosscut adhesion test was conducted following the ASTM D3359 standard. Image clarity was assessed based on the distinctness of the image (DOI) according to ASTM D5767. Impact resistance was tested using an Erichsen impact testing machine, as per ASTM D2794. The flexibility test was conducted using a conical mandrel tester in accordance with ASTM D4145. Weathering effect was studied by QUV (ASTM G53-96) by Q Lab solar eye, Westlake, OH, USA. The testing condition was an alternate cycle of 4 h UV and 4 h condensation. UV temperature 49–51 °C, Condensation Temperature 44–46 °C, Irradiance level 0.54–0.056 watts/m2/nm.
3. Results & Discussion
3.1. DSC Analysis of rPET
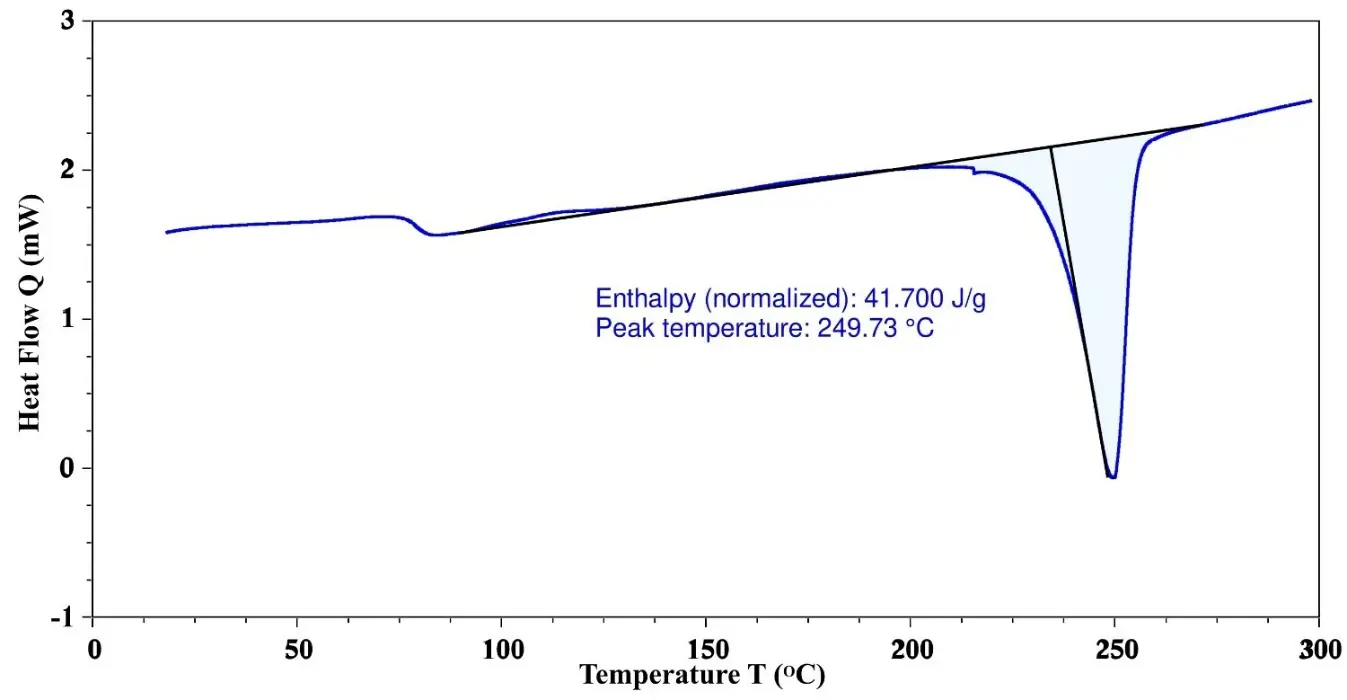
The depolymerization process of rPET is significantly influenced by its crystallinity. The crystalline regions are denser, more ordered, and chemically resistant, making ester bonds less accessible to depolymerization. This necessitates greater energy, time, and catalysts to break the ester bonds and achieve depolymerization. In contrast, the amorphous regions decompose at a much faster rate [29]. The percentage of crystallinity in the sample is assessed through DSC analysis. According to the literature, the enthalpy of 100% crystalline PET is 140 (J/g) [30]. The enthalpy of the rPET sample, as determined by DSC, is 41.7 (J/g), as given in Figure 1. Based on the formula, the percentage crystallinity of the rPET sample is calculated to be 29.78%. The results indicate that the rPET exhibits moderate crystallinity. This suggests that depolymerization is feasible and more efficient compared to 100% crystalline rPET. The depolymerization process is carried out at 240 °C in the presence of a tin-based catalyst, which can effectively depolymerize the rPET under these conditions.
3.2. Single Step rPET Based Alkyd Resin Synthesis and FTIR Spectroscopy
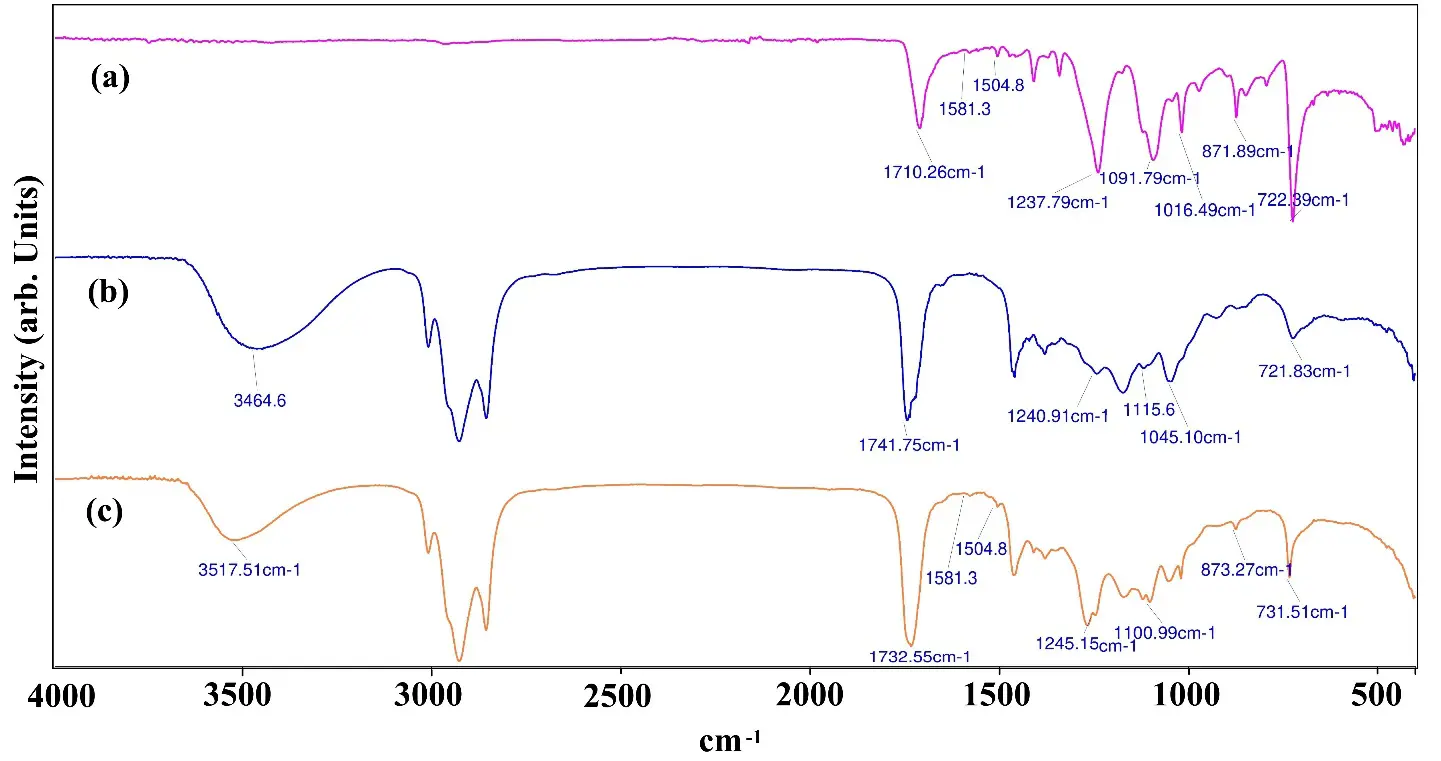
The alkyd resin derived from rPET was synthesized through a single-step process. As illustrated in Figure 2a, soybean oil underwent alcoholysis with polyols such as pentaerythritol and glycerin, facilitated by DBTO acting as a Lewis acid catalyst at a temperature of 245 °C [31]. The alcohol tolerance confirms the formation of monoglycerides. All reaction samples exhibited a minimum alcohol tolerance of 1:4, indicating that monoglyceride formation has indeed occurred [32]. The FTIR spectra of the reaction products, as shown in Figure 3 confirms the formation of monoglyceride and diglyceride due to the observation of a strong –OH peak around 3464 cm−1, which signifies the presence of free hydroxyl groups in the sample [33]. The carbonyl stretching at 1741 cm−1 further confirms the formation of esters, along with a peak at 1240–1045 cm−1, typical of ester linkages. The FTIR spectra of as such rPET exhibit a carbonyl stretching at 1710 cm−1, which is characteristic of the ester groups found in PET. The peaks at 1581 cm−1 and 1504 cm−1 correspond to the benzene ring present in the backbone of PET [34]. Additionally, the C–O stretching bands observed in the range of 1237–1091 cm−1 are associated with ester linkages [34]. Notably, there is no prominent –OH peak detected around 3400 cm−1, indicating that rPET contains a minimal amount of hydroxyl groups.
After completion of monoglyceride formation the rPET chips were introduced into the reaction mixture and heated for one hour at 240 °C. In this phase, DBTO, as a Lewis acid catalyst, activates the ester bonds of rPET at elevated temperatures. A nucleophilic attack occurs from the hydroxyl groups of the monoglyceride on the carbonyl carbon of the rPET ester group, as depicted in Figure 2b [35]. This results in partial cleavage of rPET chains, releasing small ethylene glycol fragments. A linkage forms between the hydroxyl group of the monoglyceride and the rPET terephthalate units, leading to the creation of hydroxyl-terminated oligomers [36]. At this point, the reaction mixture becomes completely clear, and the sample is withdrawn, showing clarity under cold conditions. This observation indicates that depolymerization of rPET has taken place. The FTIR spectra of this reaction sample show a strong band at 1732 cm−1, which is sharper than that observed in pure monoglyceride, indicating the formation of new esters [37]. The –OH band near 3464 cm−1 is diminished compared to that of the monoglyceride, suggesting the consumption of hydroxyl groups during the reaction. Bands at 1245–1100 cm−1 indicate the presence of new ester bonds. Peaks confirm the presence of an aromatic structure at 1504 cm−1 and 1581 cm−1 [38]. This confirms that the rPET has integrated into the reaction backbone.
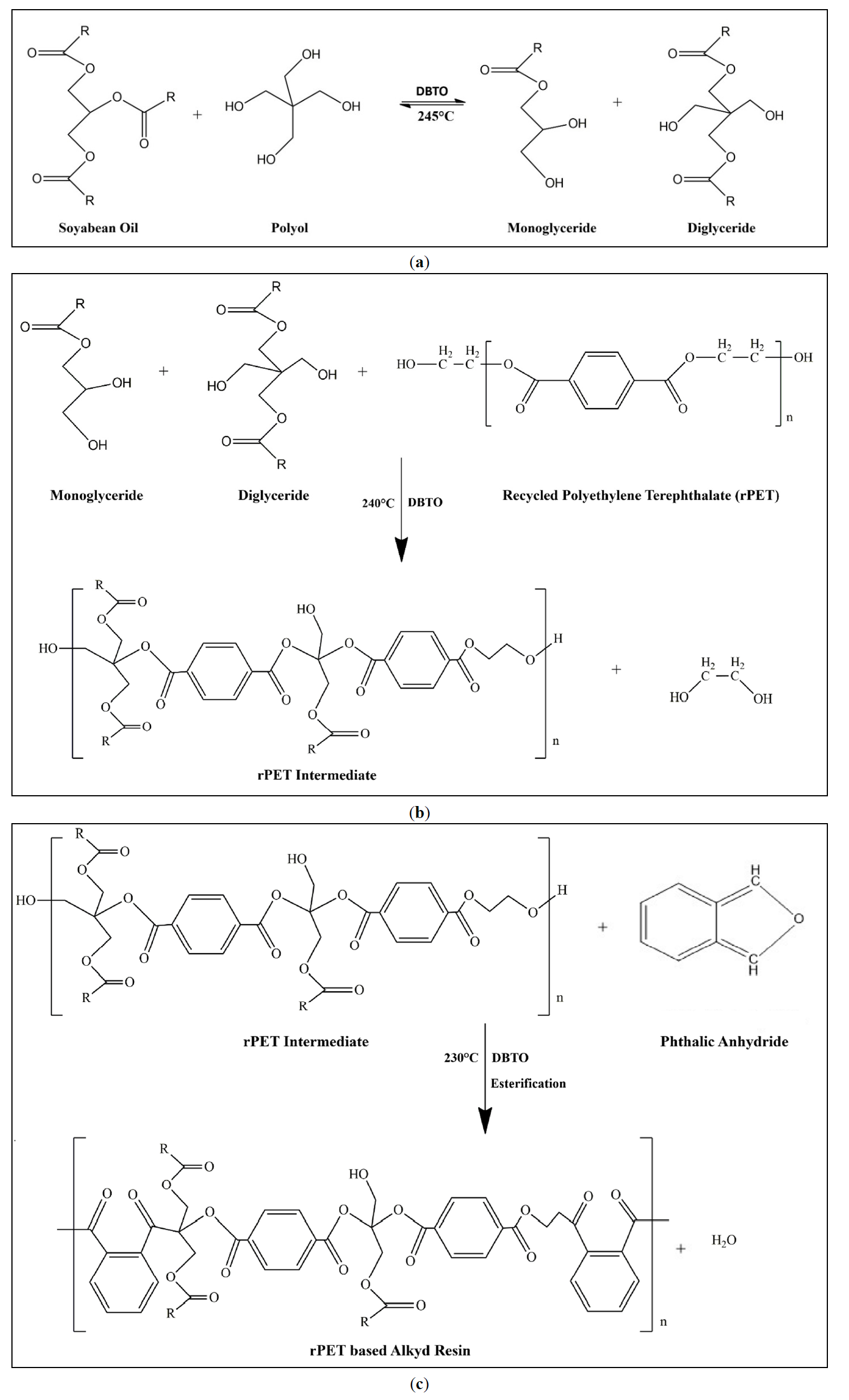
Figure 2. (a) Reaction Scheme: Synthesis of Monoglyceride; (b) Reaction Scheme: Synthesis of rPET intermediate; (c) Reaction Scheme: Synthesis of rPET based Alkyd.
After the depolymerization of rPET the reaction mass is cooled down to 170 °C. Further phthalic anhydride is added along with ortho-xylene (azeotropic solvent) and heated to 230 °C temperature for the esterification reaction. Phthalic anhydride reacts with the terminal hydroxyl of the rPET intermediate in the presence of a catalyst that will form the ester linkages, and water will be removed as a byproduct, as depicted in Figure 2c [39]. In Figure 3, if we compare the spectra of rPET based alkyd and without rPET based alkyd, the presence of a peak at 869 cm−1 shows the rPET integrated in the alkyd resin. This new synthesis route produced alkyd resin in one step by depolymerizing rPET in the same reactor and cooking the alkyd resin in the same reactor. Using this synthesis route long oil alkyd has been produced with shorter drying time.
 |
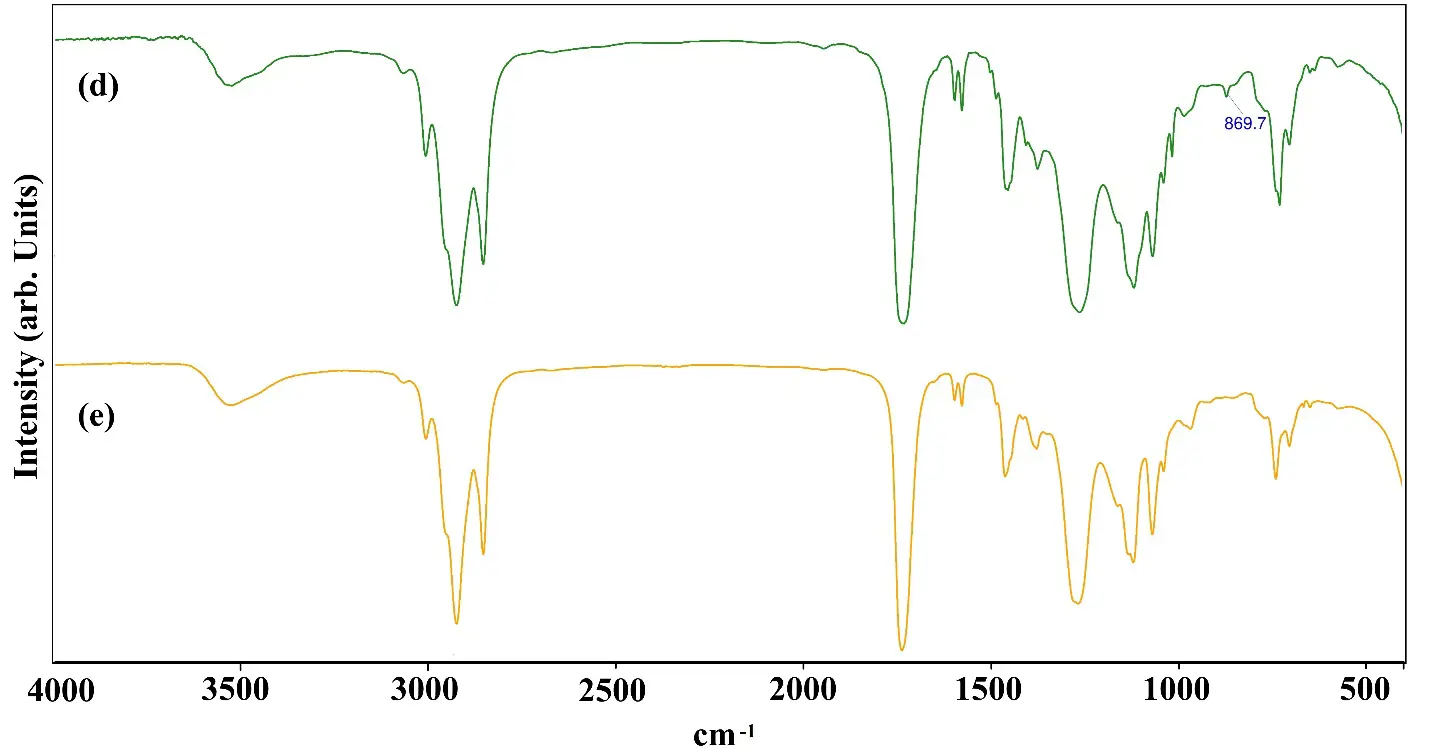 |
Figure 3. FTIR spectra: (a) rPET (b) Monoglyceride (c) rPET intermediate (d) rPET based Alkyd (e) Alkyd without rPET.
3.3. Physicochemical Properties
The physicochemical properties and GPC results of alkyds containing different proportions of rPET are detailed in the Table 3. All the resins underwent smooth processing and were thinned to 60% solids in MTO, resulting in completely transparent resins. The findings reveal that the color of the resins remains consistent regardless of the rPET content, falling within the 6–7 Gardner scale range. The acid value recorded was below 12 mg KOH/g. The viscosity for all resins remained consistent between W and Y on the Gardner scale at 25 °C. The drying results indicate that the alkyd resin containing 8% rPET becomes tack-free in 4.0 h, while the alkyd resin with 12% rPET achieves tack-free status in 4.30 h, and the alkyd resin with 16% rPET reaches tack-free in 8.15 h. The data suggest that as the rPET content in the alkyd increases, the drying properties are hindered. The drying process of alkyd resin is complex, occurring in two stages: the first is a physical process, and the second is a chemical process [40]. During the physical stage, the solvent evaporates, allowing the polymer chains to come together and initiate the chemical stage, which encompasses oxidation and crosslinking reactions. This chemical stage occurs in four distinct steps, oxygen induction, peroxide formation, free radical generation by peroxide decomposition, and crosslinking [41,42]. The molecular weight of the alkyd resin is critical to the drying process. The alkyd resin with 8% rPET has a reported molecular weight (Mw) of 19,727, the one with 12% rPET has an Mw of 24,197, and the alkyd resin with 16% rPET has an Mw of 31,301. The introduction of rPET in alkyd contributes to long oligomer chains by substituting smaller phthalic anhydride molecules which increase the molecular weight of alkyd [43]. The findings indicate that resins with a higher molecular weight exhibit extended drying times. This phenomenon occurs because the increase in molecular weight from rPET loading decreases the mobility of the polymer chains, thereby raising the glass transition temperature and subsequently reducing free volume. This decrease in free volume slows down the solvent diffusion rate through the film, leading to a longer tack-free time. Therefore, the alkyd containing the maximum amount of rPET has a larger molecular weight and a slower drying time, whereas the alkyd with the minimum rPET content has a lower molecular weight and a faster drying time [44,45].
Table 3. Physicochemical Properties of Alkyds.
|
Alkyd A |
Alkyd B |
Alkyd C |
Benchmark Alkyd D |
|
|---|---|---|---|---|
|
Percentage of rPET (%) |
8 |
12 |
16 |
0 |
|
Water of Reaction (%) |
3.98 |
4.41 |
4.79 |
3.20 |
|
Alcohol tolerance achieved |
1:4.0 |
1:Infinite |
1:Infinite |
1:4.5 |
|
Viscosity on Gardner scale @ 25 °C |
X |
W-X |
X-Y |
Y |
|
Acid value (mg KOH/g) |
10.1 |
9.25 |
10.92 |
9.02 |
|
Colour on Gardner scale |
6–7 |
6–7 |
6–7 |
6–7 |
|
Clarity |
Clear |
Clear |
Clear |
Clear |
|
NVM % (1 g/120 °C/1 h) |
60.89 |
60.65 |
60.35 |
61.03 |
|
Surface Dry (minutes) |
80 |
80 |
100 |
80 |
|
Tack Free (hours) |
4.0 |
4.30 |
8.15 |
4.0 |
|
Hard Dry (hours) |
14.0 |
14.0 |
14.0 |
14.0 |
|
GPC results |
||||
|
Mw |
19,727 |
24,197 |
31,301 |
26,128 |
|
Mn |
2745 |
2683 |
2659 |
3035 |
|
Mw/Mn |
7.18 |
9.02 |
11.77 |
8.61 |
3.4. Characterization of Alkyd Dried Films
The resin sample was combined with driers and thinner (MTO) as outlined in Section 2.2.3 and allowed to mature overnight. Following this maturation period, the lacquer was applied on releasing paper in accordance with the DFT specifications. Once applied, the film was permitted to cure for 7 days before undergoing testing.
3.4.1. TGA Analysis
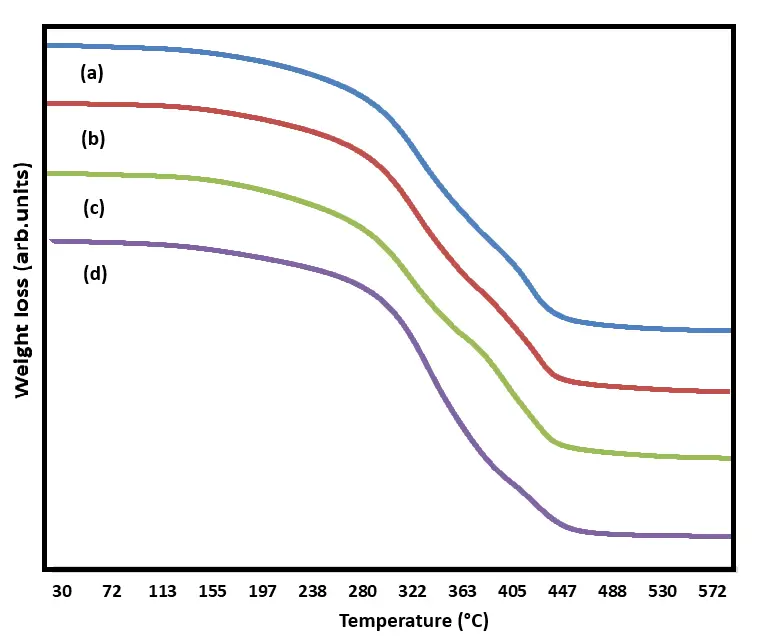
Using TGA, we gained insights into the thermal decomposition of the alkyd films. Table 4 illustrates the percentage weight loss, temperature, and residue at 600 °C after complete thermal decomposition, the thermogram presented in Figure 4. The findings indicated that benchmark alkyd D (without rPET) experiences weight loss at a lower temperature compared to the alkyd that contain rPET. Specifically, benchmark alkyd D exhibited 90% weight loss at 458 °C, whereas the rPET-containing alkyd showed 90% weight loss at temperatures exceeding 515 °C. This clearly demonstrates that incorporating rPET into the alkyd resin enhances its thermal stability, likely due to the introduction of long-chain and aromatic oligomers into the alkyd structure [46].
Table 4. TGA results of alkyd dried films.
|
Wt. Loss |
Alkyd A |
Alkyd B |
Alkyd C |
Benchmark Alkyd D |
|---|---|---|---|---|
|
50% |
369.50 °C |
369.16 °C |
373.00 °C |
360.16 °C |
|
60% |
394.00 °C |
395.00 °C |
400.66 °C |
376.83 °C |
|
70% |
420.16 °C |
419.16 °C |
419.66 °C |
397.00 °C |
|
80% |
439.16 °C |
440.16 °C |
440.33 °C |
426.50 °C |
|
90% |
553.16 °C |
515.50 °C |
569.50 °C |
458.66 °C |
3.4.2. DMA Analysis
The analysis conducted using DMA that provides insights into the viscoelastic properties exhibited by the films. These properties are closely linked to the crosslink density in the film. The crosslinked density, in turn, is influenced by factors such as molecular structure, processability, and drying characteristics of the polymer [47]. The findings are summarized in Table 5, which includes glass transition temperature and crosslinking density obtained from the DMA curve as given in Figure 5. By examining the storage modulus value in the rubbery plateau region, it is possible to determine the crosslinking density of the film, as this value correlates with the number of crosslinks in the polymer chain [48]. The crosslinking density is calculated using the equation as given below.
|
```latex\mathrm{q}=\frac{{\mathrm{E}}^{\mathrm{\prime}}}{3\mathrm{R}\mathrm{T}}``` |
where, q—Crosslinking Density; E′—Storage Modulus in the rubbery plateau Region; R—Universal Gas Constant; T—Absolute Temperature.
 |
 |
 |
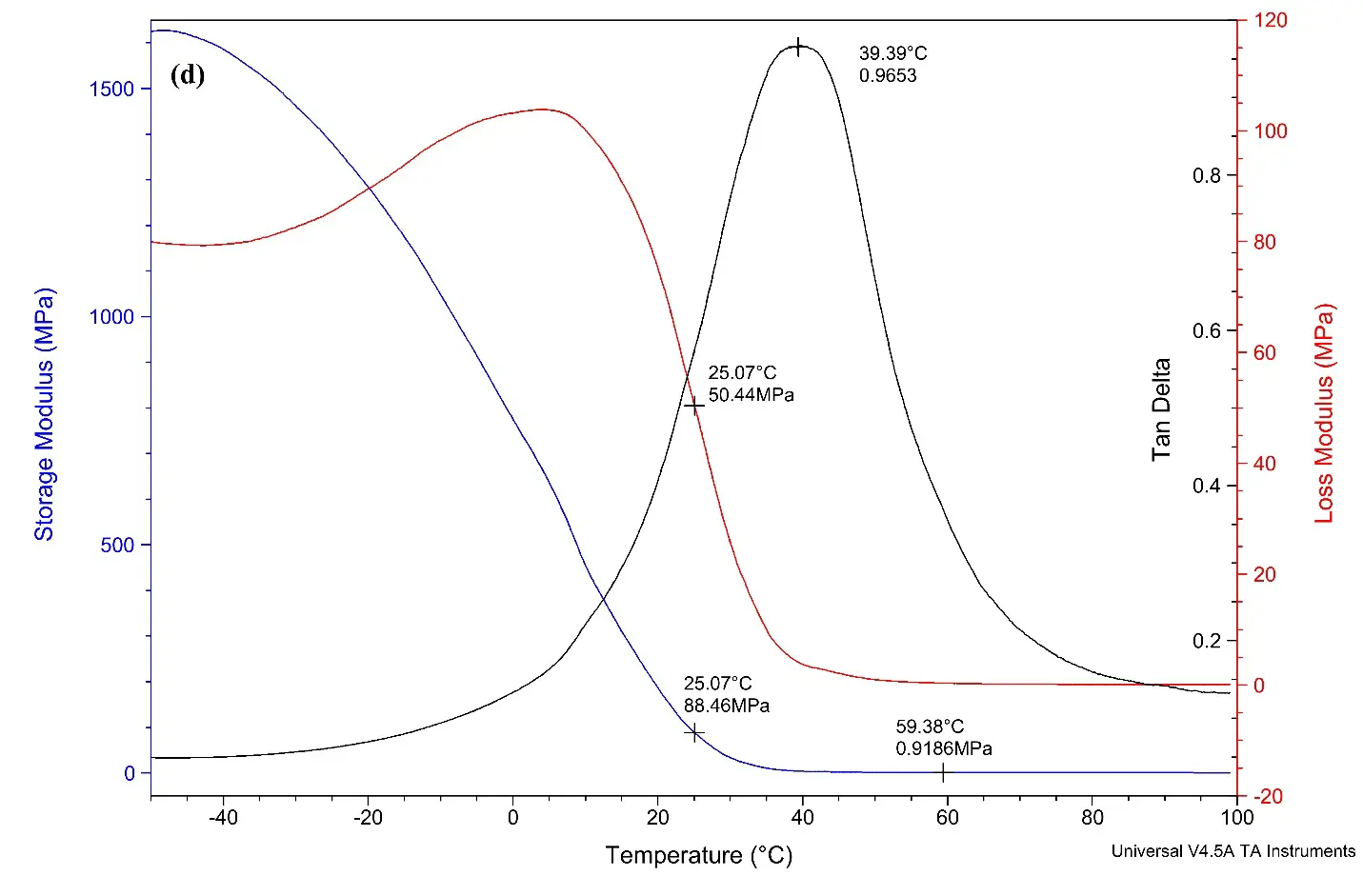 |
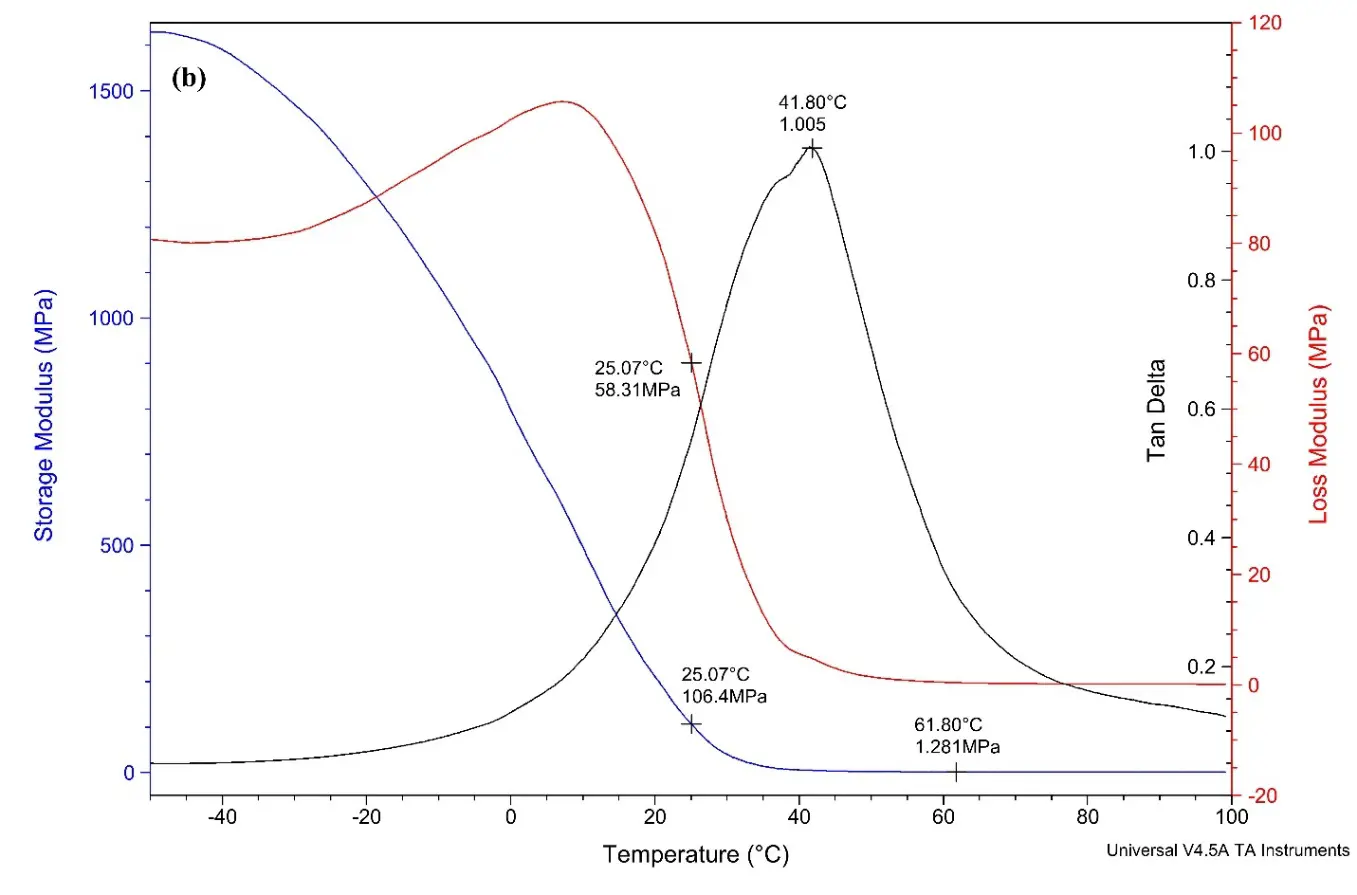
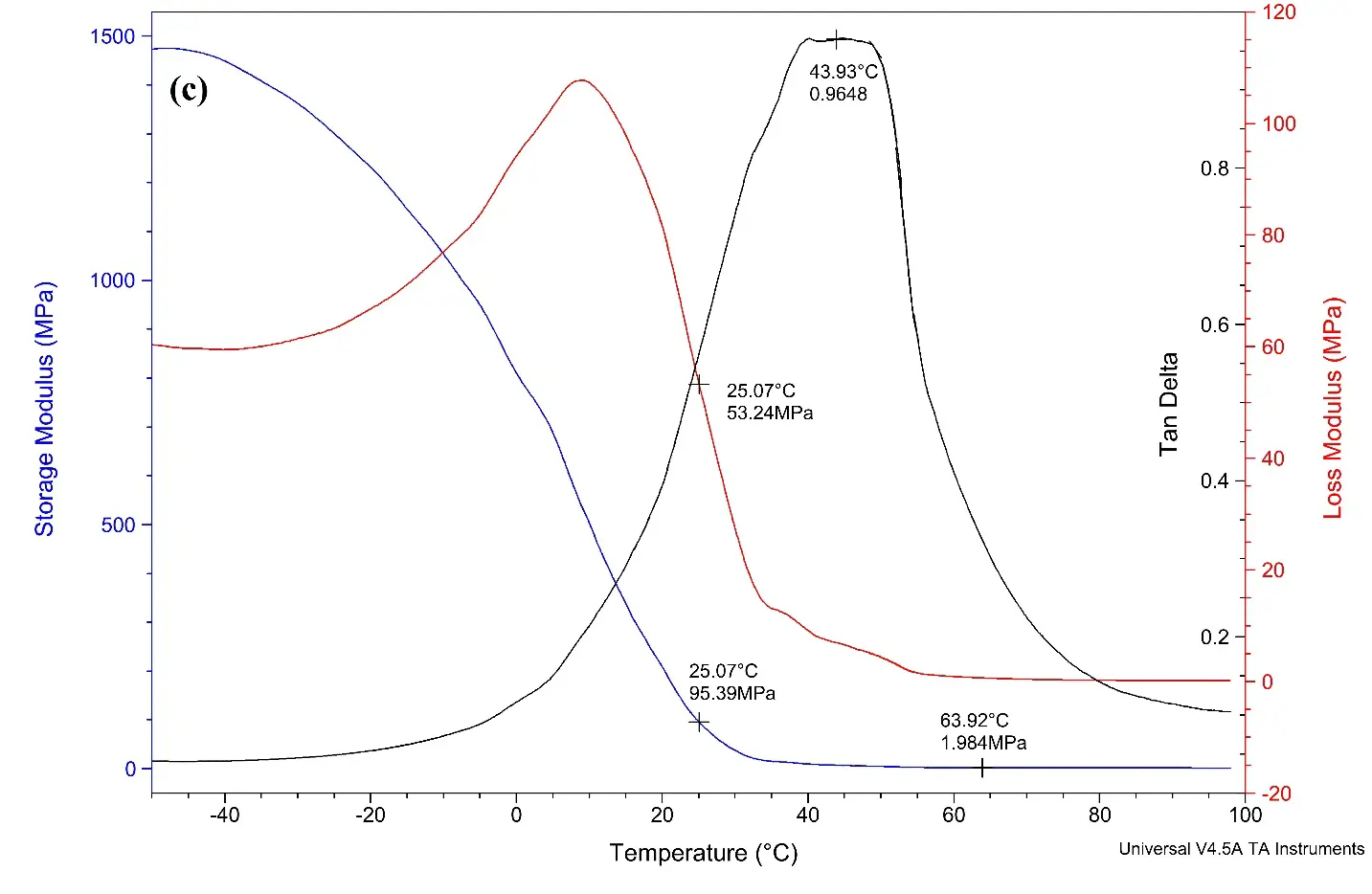
Figure 5. (a) DMA curve for alkyd A dried film; (b) DMA curve for alkyd B dried films; (c) DMA curve for alkyd C dried film; (d) DMA curve for alkyd D dried film.
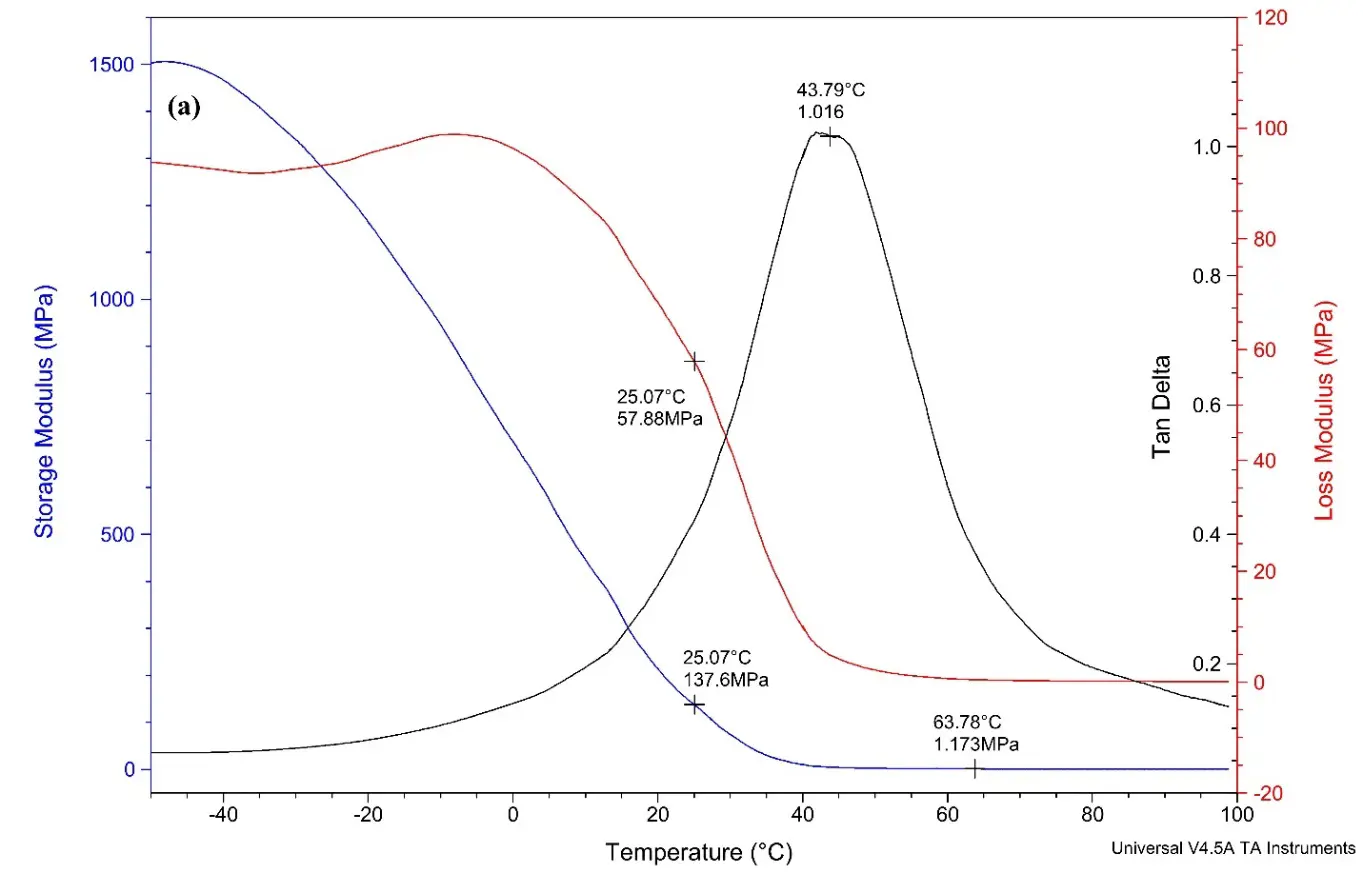
Figure 5a represents the DMA curve of alkyd A loaded with 8% of rPET. The curves report a damping factor that is the tan delta value of 1.016, with the Tg value calculated is 41.7 °C
Figure 5b shows the DMA curve of alkyd B loaded with 12% of rPET. The curves show the tan delta value of 1.005, with the Tg value calculated is 43.8 °C.
Figure 5c demonstrates the DMA curve of alkyd C loaded with 16% of rPET. The curves give the tan delta value of 0.9648, with the Tg value calculated is 44 °C.
Figure 5d displays the DMA curve of alkyd D loaded with 0% of rPET. The curves result the tan delta value of 0.9653, with the Tg value calculated is 39.4 °C.
The test findings reveal that the crosslink density increases when rPET is incorporated into the alkyd resin, in contrast to alkyd resins that do not contain rPET. This suggests that long oligomer chains contributed by rPET reduce the free volume within the alkyds. As a result of this reduction in free volume, the crosslinking density is enhanced [49]. According to the observed trend, an increase in rPET content correlates with an increase in both the crosslinking density and Tg value. However, it is important to note that exceeding a certain threshold of crosslinking density may negatively affect performance characteristics.
Table 5. DMA results for alkyd dried films.
|
Samples |
Tg (°C) |
Crosslinked Density (mol·cm−3) |
|---|---|---|
|
Alkyd A |
41.7 |
1.39 × 10−4 |
|
Alkyd B |
43.8 |
1.53 × 10−4 |
|
Alkyd C |
44 |
2.36 × 10−4 |
|
Alkyd D |
39.4 |
1.10 × 10−4 |
4. Performance Properties of Dried Films
The transparent lacquer was prepared according to the instructions provided in Section 2.2.3 and applied to Mild Steel (MS) and tin panels, which were then left overnight to dry. The dried panels underwent various performance tests in accordance with industrial standards, and the results have been compiled in Table 6. Based on the data obtained, it can be deduced that the distinctness of the image, flow & leveling of rPET containing alkyd are comparable to benchmark alkyd. The results verified that the film exhibits uniform drying and crosslinking. The compatibility of the rPET based oligomeric reactants contributed to effective film formation, optimal flow and leveling, as well as a desirable degree of optical clarity [50]. However, the gloss tends to deteriorate as the rPET content in the binder increases. This decrease in gloss value may be attributed by the aromatic oligomers; contributed by the rPET that may increase crosslinking density caused by the excessive loading of rPET [51]. While the gloss of Alkyd A is comparable to the benchmark alkyd, the results indicate that beyond 8% of rPET loading in the alkyd, the gloss diminishes. All the samples passed the crosscut adhesion test, indicating that the resin has sufficient amounts of terminal hydroxyl and carboxyl groups that anchor to the metal substrate [52]. In terms of scratch and pencil hardness, the alkyd with the highest rPET content exhibits superior hardness properties compared to other alkyds. This can be attributed to the increased crosslinking density resulting from higher rPET loading [51]. All the samples passed impact resistance, reverse impact and flexibility test, that specify the free volume that can effectively digest the external mechanical force [53]. One of the key tests conducted in the paint industry is the QUV test. In the QUV test, the alkyd with the lowest amount of rPET demonstrated 88.34% gloss retention after 240 h, while the benchmark alkyd showed 90.29% gloss retention after the same duration as shown in Figure 6. The trend indicates that the alkyd with the highest rPET content experienced a greater loss in gloss value. This may be due to excess crosslinking of the film, which becomes brittle under QUV exposure and causes gloss to decrease [54].
Table 6. Performance tests results of alkyd clear coat.
|
Test |
Alkyd A |
Alkyd B |
Alkyd C |
Alkyd D |
|---|---|---|---|---|
|
Dry Film Thickness (µm) |
24–29 |
23–28 |
22–27 |
23–28 |
|
Distinctness of Image |
Good |
Good |
Good |
Good |
|
Flow & leveling |
Satisfactory |
Satisfactory |
Satisfactory |
Satisfactory |
|
Gloss@ 20° angle |
86 |
85 |
79 |
86 |
|
Gloss@ 60° angle |
103 |
102 |
100 |
103 |
|
Cross-cut adhesion |
Pass |
Pass |
Pass |
Pass |
|
Scratch hardness (g) |
900 |
900 |
1100 |
1000 |
|
Pencil hardness |
H |
H |
2H |
H |
|
Impact resistance |
Pass |
Pass |
Pass |
Pass |
|
Reverse impact |
Pass |
Pass |
Pass |
Pass |
|
Flexibility |
Pass |
Pass |
Pass |
Pass |
|
QUV weathering test (240 h) |
Gloss retention 88.34% |
Gloss retention 84.31% |
Gloss retention 82% |
Gloss retention 90.29% |
5. Conclusions
The entire paint industry is exploring new approaches for sustainability. The approach of incorporating recycled polyethylene terephthalate in the alkyd resin will help in reducing the issue of PET waste and substituting the petroleum based raw material such as phthalic anhydride. Considering the production volume of alkyd resin, especially long oil alkyd, and the waste generated by PET, even a small amount of rPET incorporation into alkyd resin will have a massive impact on a larger scale.
In this research work, long oil alkyd was successfully produced by the novel synthesis route, that helped in overcoming the limitations of the conventional process of producing rPET based alkyd. Incorporating recycled PET into the alkyd resin enhances sustainability and environmental friendliness. FTIR analysis confirms the effective integration of recycled PET into the alkyd resin. GPC results reveal that increase in rPET content in the alkyd resin increases the molecular weight but affects the drying properties. This might be because of the reduction in free volume; it can be correlated with DMA results, as free volume decreases, the crosslinked density increases. The higher the rPET content in the alkyd resin higher the crosslinking density. TGA results show that the thermal stability of the alkyd film increases as the rPET content in the alkyd increases. alkyd A, containing 8% recycled PET, exhibits comparable performance to the benchmark alkyd D in terms of drying, gloss, and hardness. In exterior durability study, alkyd with highest loading of rPET showed less gloss retention. This is mainly due to the higher aromaticity of rPET, which increases the yellowing tendency of the alkyd film, whereas alkyd A, with a lower percentage of rPET, showed comparable performance to the benchmark alkyd D. This formulation (Alkyd A) is identified as the most robust and optimized for air-drying alkyd. These findings suggest that incorporating rPET into alkyd resin using this method is a promising approach for sustainability and the circular economy principle.
Acknowledgement
We express our sincere thanks towards the management of ‘Asian Paints limited’ to carried out this work. We are thankful to Ganesh Deokar, Subhajit Majhi and Ankit Joshi for carrying out the instrumental analysis. We are also thankful to Rajeevkumar Jain and Rajeevkumar Goel for giving us this opportunity.
Author Contributions
B.S.C.—Conceptualization, Methodology, Formal Analysis, validation, Writing Original Draft Preparation. V.S.—Supervision, Project Administration, Resources.
Ethics Statement
Not Applicable.
Informed Consent Statement
Not Applicable.
Data Availability Statement
Data will be made available on request.
Funding
This research received no external funding.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
-
UNEP—UN Environment Programme. Emission Gap Report 2024. Available online: https://www.unep.org/resources/emissions-gap-report-2024 (accessed on 17 April 2025).
-
Ritchie H, Rosado P, Roser M. CO2 and Greenhouse Gas Emissions. Our World in Data. Available online: https://ourworldindata.org/co2-and-greenhouse-gas-emissions?utm_source=pocket_shared (accessed on 17 April 2025).
-
Lamb WF, Wiedmann T, Pongratz J, Andrew R, Crippa M, Olivier JGJ, et al. A review of trends and drivers of greenhouse gas emissions by sector from 1990 to 2018. Environ. Res. Lett. 2021, 16, 073005. DOI:10.1088/1748-9326/abee4e [Google Scholar]
-
Kurdve M, Shahbazi S, Wendin M, Bengtsson C, Wiktorsson M. Waste flow mapping to improve sustainability of waste management: A case study approach. J. Clean. Prod. 2015, 98, 304–315. DOI:10.1016/j.jclepro.2014.06.076 [Google Scholar]
-
Zhou Z, Jiang H, Qin L. Life cycle sustainability assessment of fuels. Fuel 2007, 86, 256–263. DOI:10.1016/j.fuel.2006.06.004 [Google Scholar]
-
Techno-Economic, Life-Cycle, and Socioeconomic Impact Analysis of Enzymatic Recycling of Poly(Ethylene Terephthalate). Available online: https://www.nrel.gov/docs/fy21osti/79571.pdf (accessed on 22 April 2025).
-
Hamade R, Hadchiti R, Ammouri A. Making the environmental case for reusable PET bottles. Procedia Manuf. 2020, 43, 201–207. DOI:10.1016/j.promfg.2020.02.137 [Google Scholar]
-
Jankauskaitė V, Macijauskas G, Lygaitis R. Polyethylene terephthalate waste recycling and application possibilities: A review. Mater. Sci. 2008, 14, 119–127. Available online: https://epubl.ktu.edu/object/elaba:2924108/ (accessed on 23 April 2025).
-
Ifijen IH, Maliki M, Odiachi IJ, Aghedo ON, Ohiocheoya EB. Review on solvents based alkyd resins and water borne alkyd resins: Impacts of modification on their coating properties. Chem. Afr. 2022, 5, 211–225. DOI:10.1007/s42250-022-00318-3 [Google Scholar]
-
Alkyd Resin Market Size, Share, Industry Analysis Report, 2030. Available online: www.fnfresearch.com/alkyd-resin-market (accessed on 25 April 2025).
-
Heriyanto H, Suhendi E, Asyuni NF, Shahila IK. Effect of Bayah natural zeolite for purification of waste cooking oil as feedstock of alkyd resin. Tek. J. Sains Dan Teknol. 2022, 18, 49–55. DOI:10.36055/tjst.v18i1.15542 [Google Scholar]
-
Mukhtar A, Ullah H, Mukhtar H. Fatty acid composition of tobacco seed oil and synthesis of alkyd resin. Chin. J. Chem. 2007, 25, 705–708. DOI:10.1002/cjoc.200790132 [Google Scholar]
-
Ramli R, Ong HR, Hong CS, Khan MM, Yunus RM, Halim RM, et al. Investigation on the effect of monoglyceride concentration on palm oil based alkyd resin preparation. J. Oil Palm Res. 2021, 33, 299–306. DOI:10.21894/jopr.2020.0086 [Google Scholar]
-
Raheem AB, Uyigue L. The conversion of post-consumer polyethylene terephthalate (PET) into a thermosetting polyester resin. Arch. Appl. Sci. Res. 2010, 2, 240–254. Available online: https://www.researchgate.net/profile/Ademola-Raheem/publication/310748146_The_conversion_of_postconsumer_polyethylene_terephthalate_PET_into_a_thermosetting_polyester_resin/links/5bcd5ad2458515f7d9d026ba/The-conversion-of-postconsumer-polyethylene-terephthalate-PET-into-a-thermosetting-polyester-resin.pdf (accessed on 26 April 2025).
-
Lin KF. Alkyd resins. In Kirk-Othmer Encyclopedia of Chemical Technology; Wiley: Hoboken, NJ, USA, 2000. DOI:10.1002/0471238961.01121125120914.a01 [Google Scholar]
-
Hu Y, Wang Y, Zhang X, Qian J, Xing X, Wang X. Synthesis of poly(ethylene terephthalate) based on glycolysis of waste PET fiber. J. Macromol. Sci. Part A 2020, 57, 430–438. DOI:10.1080/10601325.2019.1709498 [Google Scholar]
-
Tuna Ö, Bal A, Güçlü G. Investigation of the effect of hydrolysis products of postconsumer polyethylene terephthalate bottles on the properties of alkyd resins. Polym. Eng. Sci. 2013, 53, 176–182. DOI:10.1002/pen.23247 [Google Scholar]
-
Bulak E, Acar I. The use of aminolysis, aminoglycolysis, and simultaneous aminolysis–hydrolysis products of waste PET for production of paint binder. Polym. Eng. Sci. 2014, 54, 2272–2281. DOI:10.1002/pen.23773 [Google Scholar]
-
Ikladious NE, Asaad JN, Emira HS, Mansour SH. Alkyd resins based on hyperbranched polyesters and PET waste for coating applications. Prog. Org. Coat. 2017, 102, 217–224. DOI:10.1016/j.porgcoat.2016.10.015 [Google Scholar]
-
Jamdar V, Kathalewar M, Dubey KA, Sabnis A. Recycling of PET wastes using Electron beam radiations and preparation of polyurethane coatings using recycled material. Prog. Org. Coat. 2017, 107, 54–63. DOI:10.1016/j.porgcoat.2017.02.007 [Google Scholar]
-
Büyükyonga ÖN, Akgün N, Acar I, Güçlü G. The usage of novel acrylic-modified water-reducible alkyd resin obtained from post-consumer PET bottles in water-based paint formulation. J. Mater. Cycles Waste Manag. 2019, 22, 187–196. DOI:10.1007/s10163-019-00929-y [Google Scholar]
-
Spasojević PM, Panić VV, Džunuzović JV, Marinković AD, Woortman AJ, Loos K, et al. High performance alkyd resins synthesized from postconsumer PET bottles. RSC Adv. 2015, 5, 62273–62283. DOI:10.1039/c5ra11777a [Google Scholar]
-
Kirchherr J, Reike D, Hekkert M. Conceptualizing the circular economy: An analysis of 114 definitions. Resour. Conserv. Recycl. 2017, 127, 221–232. DOI:10.1016/j.resconrec.2017.09.005 [Google Scholar]
-
Yin X, Duan H, Wang X, Sun L, Sun W, Qi H, et al. An investigation on synthesis of alkyd resin with sorbitol. Prog. Org. Coat. 2014, 77, 674–678. DOI:10.1016/j.porgcoat.2013.12.005 [Google Scholar]
-
Madiebo EM, Uzoh CF, Onukwuli OD, Ohale PE, Nweke CN, Igwegbe CA, et al. Synthesis and characterization of Cerbera odollam (sea mango) oil-based alkyd resin as binder for surface coating paint and matrix material for reinforced polymer composites. J. Coat. Technol. Res. 2024, 21, 1577–1589. DOI:10.1007/s11998-024-00917-4 [Google Scholar]
-
Donate FA, Timmers DA, Kappen JW. Solvent blends based on dipropylene glycol dimethyl ether for the production of alkyd and polyester resins by the azeotropic process. J. Coat. Technol. 2000, 72, 71–77. DOI:10.1007/BF02698007 [Google Scholar]
-
Misev TA. Calculating technique for formulating alkyd resins. Prog. Org. Coat. 1992, 21, 79–99. DOI:10.1016/0033-0655(92)80013-M [Google Scholar]
-
Dubrulle L, Lebeuf R, Thomas L, Fressancourt-Collinet M, Nardello-Rataj V. Catalytic activity of primary and secondary driers towards the oxidation and hydroperoxide decomposition steps for the chemical drying of alkyd resin. Prog. Org. Coat. 2017, 104, 141–151. DOI:10.1016/j.porgcoat.2016.12.018 [Google Scholar]
-
Liu G, Zuo W, Hao M, Zhu K, Wang F, Chen L. The influence of crystallinity on the depolymerization mechanism of PET fibers. Polym. Degrad. Stab. 2025, 13, 111670. DOI:10.1016/j.polymdegradstab.2025.111670 [Google Scholar]
-
Determination of Crystallinity of PET by DSC. Organic Material Science. Available online: https://setaramsolutions.com/app/uploads/sites/2/2020/09/ANS-002-Determination-of-cristallinity-of-PET-by-DSC.pdf (accessed on 12 April 2025).
-
Saad M, Wazarkar K, Singh V. Exploring the efficacy of tin-free catalysts in alkyd resin synthesis. J. Coat. Technol. Res. 2025, 23, 843–854. DOI:10.1007/s11998-025-01172-x [Google Scholar]
-
Arauz-Solís AB, Avalos-Belmontes F, Martínez-Cartagena ME, Banda-Villanueva A, Torres-Lubian JR, Ventura-Hunter C. Recent advances in hyperbranched alkyd resins. J. Coat. Technol. Res. 2025, 22, 149–169. DOI:10.1007/s11998-024-00995-4 [Google Scholar]
-
O’Connor RT, DuPre EF, Feuge RO. The infrared spectra of mono-, di-, and triglycerides. J. Am. Oil Chem. Soc. 1955, 32, 88–93. DOI:10.1007/BF02636511 [Google Scholar]
-
Brian C. Smith, Infrared Spectroscopy of Polymers, VIII: Polyesters and the Rule of Three. Spectroscopy 2022, 37, 25–28. DOI:10.56530/spectroscopy.ta9383e3 [Google Scholar]
-
Dullius J, Ruecker C, Ligabue R, Einloft S. Chemical recycling of post-consumer PET: Alkyd resins synthesis. Prog. Org. Coat. 2006, 57, 123–127. DOI:10.1016/j.porgcoat.2006.07.004 [Google Scholar]
-
Kawamura C, Ito K, Nishida R, Yoshihara I, Numa N. Coating resins synthesized from recycled PET. Prog. Org. Coat. 2002, 45, 185–191. DOI:10.1016/S0300-9440(01)00253-3 [Google Scholar]
-
Jumaah MA, Salih N, Salimon J. Optimization for esterification of saturated palm fatty acid distillate by D-optimal design response surface methodology for biolubricant production. Turk. J. Chem. 2021, 45, 1391–1407. DOI:10.3906/kim-2103-11 [Google Scholar]
-
Silverstein RM, Bassler GC. Spectrometric identification of organic compounds. J. Chem. Educ. 1962, 39, 546. DOI:10.1021/ed039p546 [Google Scholar]
-
Mazurek-Budzyńska MM, Rokicki G, Paśnik K. Utilization of poly(ethylene terephthalate) waste in the synthesis of air-drying polyester alkyd resins and aliphatic-aromatic poly(ester-carbonate)s. Polimery 2016, 61, 601–609. DOI:10.14314/polimery.2016.600 [Google Scholar]
-
Holmberg K. High Solids Alkyd Resins; CRC Press: Boca Raton, FL, USA, 2020. DOI:10.1201/9781003065814 [Google Scholar]
-
Atimuttigul V, Damrongsakkul S, Tanthapanichakoon W. Effects of oil type on the properties of short oil alkyd coating materials. Korean J. Chem. Eng. 2006, 23, 672–677. DOI:10.1007/BF02706813 [Google Scholar]
-
Mańczyk K, Szewczyk P. Highly branched high solids alkyd resins. Prog. Org. Coat. 2002, 44, 99–109. DOI:10.1016/S0300-9440(01)00249-1 [Google Scholar]
-
Atta AM, El-Ghazawy RA, El-Saeed AM. Corrosion protective coating based on alkyd resins derived from recycled poly (ethylene terephthalate) waste for carbon steel. Int. J. Electrochem. Sci. 2013, 8, 5136–5152. DOI:10.1016/S1452-3981(23)14668-2 [Google Scholar]
-
Van Gorkum R, Bouwman E. The oxidative drying of alkyd paint catalysed by metal complexes. Coord. Chem. Rev. 2005, 249, 1709–1728. DOI:10.1016/j.ccr.2005.02.002 [Google Scholar]
-
Erol T, Özaltun DH, Çavuşoğlu FC, Acar I, Güçlü G. The effect of linseed oil/canola oil blend on the coating and thermal properties of waste PET-based alkyd resins. Anais da Academia Brasileira de Ciências 2024, 96, 20230859. DOI:10.1590/0001-3765202420230859 [Google Scholar]
-
Adamu AA, Muhamad Sarih N, Gan SN. Thermal and anticorrosion properties of polyurethane coatings derived from recycled polyethylene terephthalate and palm olein-based polyols. R. Soc. Open Sci. 2021, 8, 201087. DOI:10.1098/rsos.201087 [Google Scholar]
-
Łabowska MB, Skrodzka M, Sicińska H, Michalak I, Detyna J. Influence of Crosslinking Conditions on Drying Kinetics of Alginate Hydrogel. Gels 2023, 9, 63. DOI:10.3390/gels9010063 [Google Scholar]
-
Chen TT. Quantifying Polymer Crosslinking Density Using Rheology and DMA; TA Instruments: New Castle, DE, USA, 2020. [Google Scholar]
-
Huang R, Zhao K, Cao P, Cao L, Liao H, Tang X. Molecular Simulation Study on the Impact of a Crosslinked Network Structure on the Tensile Mechanical Properties of PBT Substrates. Materials 2025, 18, 1675. DOI:10.3390/ma18071675 [Google Scholar]
-
Elba ME, Rehim EM, Ashery RE. Synthesis and Characterization Alkyd Resin Based on Soya Bean Oil and Gelycrin Using Zirconium Octoate as Catalyst. Int. J. Chem. Technol. 2018, 2, 34–43. Available online: https://dergipark.org.tr/en/download/article-file/471406 (accessed on 27 April 2025).
-
Güçlü G, Orbay M. Alkyd Resins Synthesized from Postconsumer PET Bottles. Prog. Org. Coat. 2009, 65, 362–365. DOI:10.1016/j.porgcoat.2009.02.004 [Google Scholar]
-
Lee LH. Molecular bonding and adhesion at polymer-metal interphases. J. Adhes. 1994, 46, 15–38. DOI:10.1080/00218469408026646 [Google Scholar]
-
Zhao Y, Huang R, Wu Z, Zhang H, Zhou Z, Li L, et al. Effect of free volume on cryogenic mechanical properties of epoxy resin reinforced by hyperbranched polymers. Mater. Des. 2021, 202, 109565. DOI:10.1016/j.matdes.2021.109565 [Google Scholar]
-
Erol T, Acar I. Ketone modification of alkyd synthesized from waste PET as a sustainable option: A comparative study of coating and thermal properties of alkyd–melamine–ketone resin systems. Polym. Eng. Sci. 2024, 64, 5259–5288. DOI:10.1002/pen.26935 [Google Scholar]