Synthesis and Properties of Fully Biobased Plastics from Biuret and Diamines
Yuezong Chen 1 Bochao Pan 1 Donglin Tang 1,2,*
Received: 04 January 2026 Revised: 05 February 2026 Accepted: 09 March 2026 Published: 19 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Researchers nowadays pay attention to the environmental and resource issues on polymer materials which are applied broadly in our daily life [1]. These materials rely substantially on petroleum oil resources. To deplete the dependence on the limited petroleum oil and find environmentally friendly materials, biobased polymers are currently in high demand [2]. Biobased polymers are those synthesized from natural resources biologically, chemically, or physically [3]. Polyureas (PUrs) are the polymers bearing urea groups in the backbone [4]. The existence of urea groups contributes to the formation of bidentate hydrogen bonds, which brings the polymers with diverse, remarkable, and excellent properties, such as waterproof, wear-resistant, and anticorrosive [5]. PUrs are therefore widely applied in many fields, such as construction, industrial, and military [6]. Traditional PUrs are synthesized from isocyanates and amines [7]. The reactivity of isocyanates is so high that amines are able to react quickly with isocyanates at room temperature, however, the choice of isocyanates is so limited that retards the expanding use of PUrs, especially fully biobased PUrs [8,9]. The high reactivity of isocyanates increases transportation and storage costs. Another issue for isocyanates is that they are toxic and harmful to physical health [10]. Therefore, alternative synthetic routes, namely non-isocyanate routes, are becoming more attractive, especially in the current underground of green chemistry. There are several environmentally friendly synthetic routes that have been proposed after numerous years of development and research, for instance, the dicarbamate route, the carbon dioxide (CO2) route, and the urea route [11,12,13].
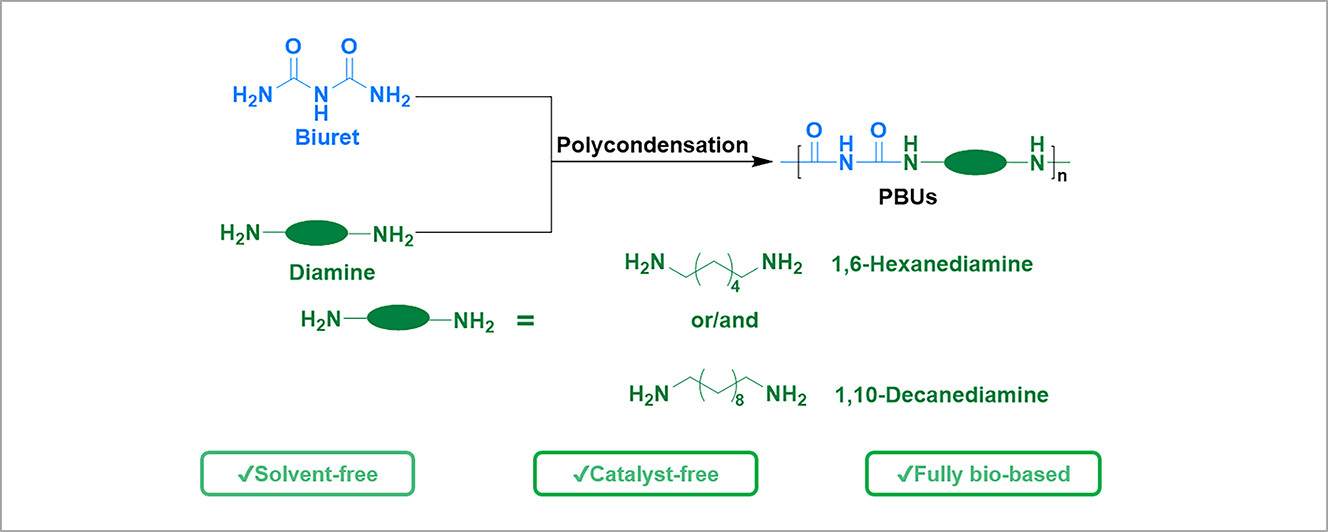
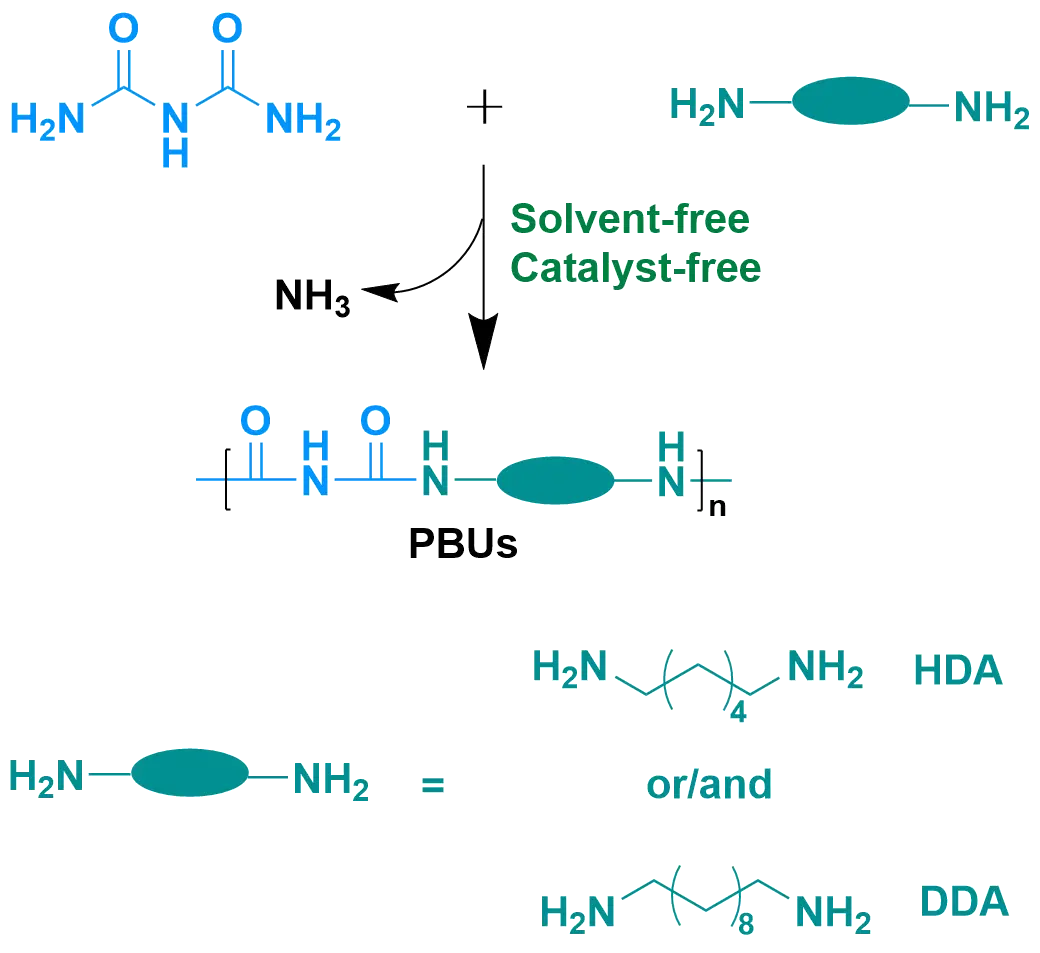
Urea (NH2CONH2) exists naturally or can be manufactured from CO2, so it is considered a cost-efficient and environmentally friendly chemical [14,15]. The high reactivity of urea with amines makes it more attractive and feasible in the synthesis of PUrs [16]. Lin et al. [11] applied urea and two fully biobased diamines, namely 1,6-hexanediamine (HDA) and 1,10-decanediamine (DDA), to synthesize fully biobased high-performance PUrs without using any catalyst and solvent. Considering that the choice of fully biobased diamines is still limited nowadays, another strategy can be applied: utilizing biuret, an environmentally friendly compound, in the construction of polymers to enhance their properties. The simplest biuret (NH2CONHCONH2) is the dimer of urea, and it shows similar reactivity to urea when reacting with amines. The biuret is commercially available and cost-efficient. The polymers synthesized from biuret and diamines are named as polybiurets (PBUs). Polymers with higher hydrogen bonding density brings them enhanced thermal and excellent mechanical properties [17,18,19]. The introduction of the biuret function has been proved by Pan et al. [20] that it is able to considerably improve the mechanical properties of polyureas based on priamine. Compared to other existing solutions, biuret is proposed to be utilized to react with two diamines (HDA or/and DDA), achieving the formation of PBU plastics with enhanced performance in this work. Due to the existence of biuret groups, PBUs, compared with PUrs, are expected to possess higher hydrogen bonding density, investing those polymers more extraordinary and outstanding thermal and mechanical properties.
2. Materials and Methods
2.1. Materials
Biuret was purchased from Senrise Technology Co., Ltd. (Anqing, China). HDA (99.5%) was bought from Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). DDA (97%) was bought from Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). All chemicals were used as received without further treatment.
2.2. Preparation of PBUs
The synthetic procedures and reaction conditions for the five polymers are the same, with different feedings of the two diamines. Taking the synthesis of PBU-6501050 as an example, biurets (100 mmol, 10.31 g), HDA (50 mmol, 5.81 g), and DDA (50 mmol, 8.62 g) were added into a 250 mL four-neck spherical flange reactor equipped with a gas inlet, a PTFE mechanical stirrer, and a gas outlet. The reaction was first performed at 180 °C under argon until the mixture became turbid. Then, the temperature was increased to 280 °C, and the reaction was run for another 2 h. Vacuum (0.08 MPa) was applied, and the reaction was kept for another 1 h to allow further chain extension. The products were collected when the reaction finished.
2.3. Characterizations
Solid-state 13C nuclear magnetic resonance (NMR) spectra were recorded by a JEOL-ECZ400R/M1 (400 MHz) spectrometer (JEOL RESONANCE Inc., Japan) with an HXY 3.2 mm probe. Samples were spun at 15,000 Hz, and the spectra were recorded at 25 °C. Fourier transform infrared (FTIR) was carried out on a SHIMADZU IR Tracer 10 (SHIMADZU, Japan). The measurements were taken out with an accumulation of 32 scans at room temperature. The spectra were obtained in the range of 400–4000 cm−1 with a resolution of 4 cm−1. Thermogravimetric analysis (TGA) was carried out by a NETZSCH TG209 F3 analyzer (NETZSCH, Germany) from 100 to 600 °C with a heating rate of 20 °C/min, under N2 atmosphere. Differential scanning calorimetry (DSC) measurements were carried out under N2 atmosphere at a heating rate of 20 °C/min and a cooling rate of 10 °C/min using a NETZSCH DSC 204 F1 calorimeter (NETZSCH, Germany), from 20 to 280 °C. Dynamic mechanical thermal analysis (DMA) was conducted on a DMA 242E dynamic mechanical analyzer (NETZSCH, Germany). For each experiment, a sample cut into a rectangular shape (10 × 5 × 0.5 mm3) was loaded on a tension-type fixture with a frequency of 1 Hz. The temperature was set ranging from 15 °C to 220 °C with a heating rate of 5 °C/min. The samples for both DMA and tensile tests were prepared by compression molding. Compression molding was performed at 195 °C for 10 min, with a clamping pressure of 20 MPa. The obtained film samples were cut into dumbbell shape according to the ISO 37 Type 4 standard (the size of the test section is 10 × 2 × 0.5 mm3). Tensile tests were performed on an INSTRON 5960 (INSTRON, USA) universal tensile tester equipped with a load cell of 10 kN, and a 50 mm/min tensile speed was applied. For each sample, at least five specimens were tested, and the results were given by taking the average value.
3. Results and Discussion
3.1. Synthesis and Characterization
PBUs are proposed in this work to be synthesized from biurets and diamines via melting polycondensation, without using any catalyst or solvent (see Scheme 1). The two diamines utilized are fully biobased, namely HDA and DDA. The synthesized PBUs are nominated as PBU-6x10100−x, in which x is the molar fraction of HDA (x = 0, 33, 67, 100). The synthetic process is designed to proceed in three steps. In the first step, the polymerization was carried at lower temperature at 180 °C to facilitate the consumption of the volatile diamines. The temperature was increased in the second step to afford a faster polymerization rate and a higher conversion of the functional groups. A vacuum was then applied in the final step to allow further chain extension and to achieve higher molecular weight for the products.
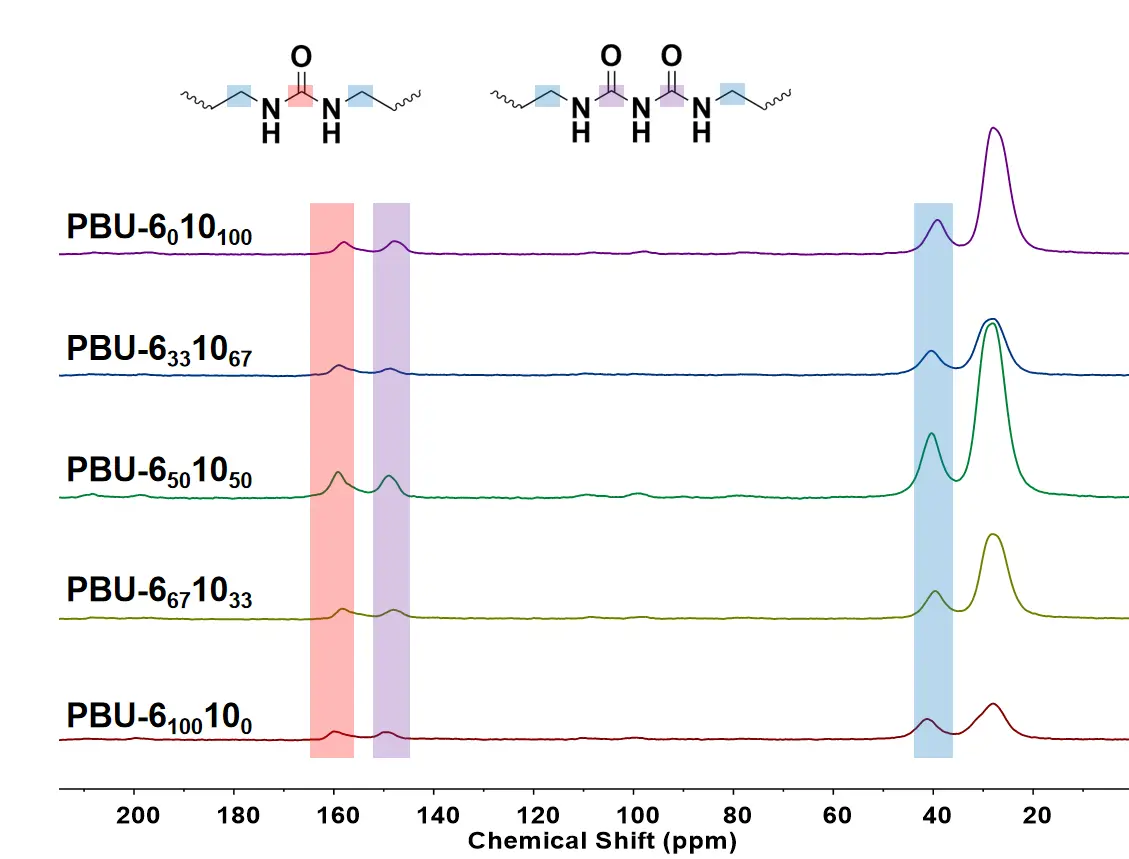
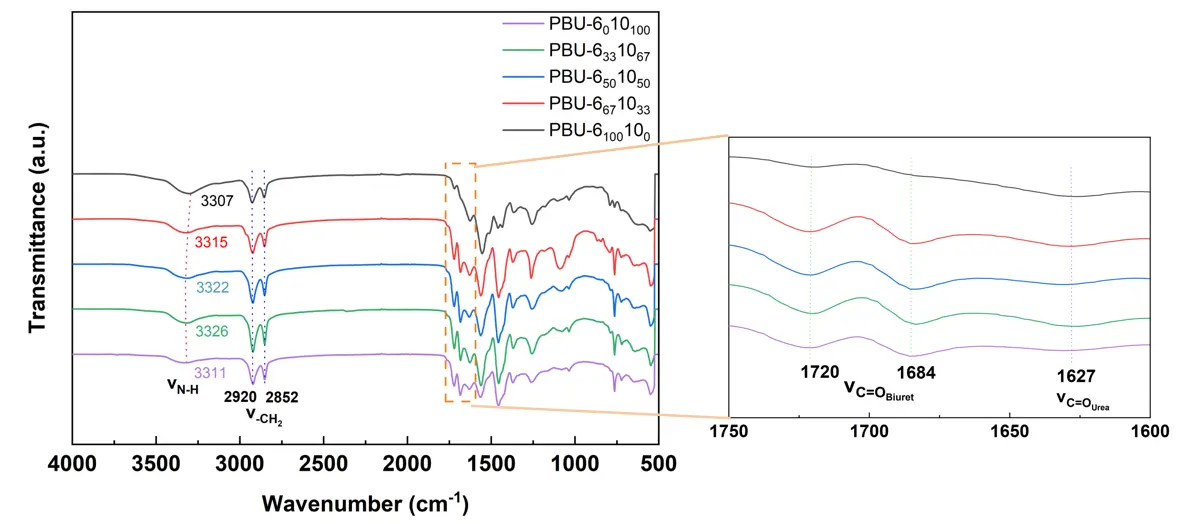
PBUs are resistant to most organic solvents, including acetone, methanol, dichloromethane, tetrahydrofuran, acetic acid, dimethylformamide, dimethylacetamide, N-methylpyrrolidone, dimethylsulfoxide, hexafluoroisopropanol, and even trifluoroacetic acid. Therefore, solid state 13C NMR was chosen to verify the chemical structure of PBUs, which are shown in Figure 1. The carbon signals next to the urea (CH2-NHCONH-) and biuret groups (CH2-NHCONH-CONH-) can be observed at 41 ppm. The signals of carbonyl carbon in both urea (-NHCONH-) and biuret functions (-NHCONHCONH-) can be observed at 160 and 150 ppm, respectively, which implies the formation of PUrs and PBUs [20]. The formation of urea groups can be explained by the attack of both carbonyl functions by the amino group. The structure of PBUs was detected by FTIR as well, and the results are shown in Figure 2. The peaks at 1720 and 1684 cm−1 are the signals of the stretching vibrations of C=O in biuret groups with disordered and ordered hydrogen bonds, respectively. The peak at about 1627 cm−1 corresponds to the stretching vibrations of C=O in the urea group. The stretching vibrations of N–H in biuret and urea can be observed at about 3320 cm−1. Red shift in the N–H stretching vibrations is present because of the denser hydrogen bonding of PBU-6100100 [21]. A red shift of N–H stretching vibration appears for PBU-6100100 and PBU-6010100, comparing with PBU-6331067, PBU-6501050, and PBU-6671033, due to the less hydrogen bonding in the copolymers [11]. As the proportion of DDA increases to 67%, the stretching vibration of N–H shifts from 3307 cm−1 to 3326 cm−1. The peaks at about 2920 and 2852 cm−1 are the signals of the stretching vibrations of -CH2 in the main chains.
3.2. Thermal Properties
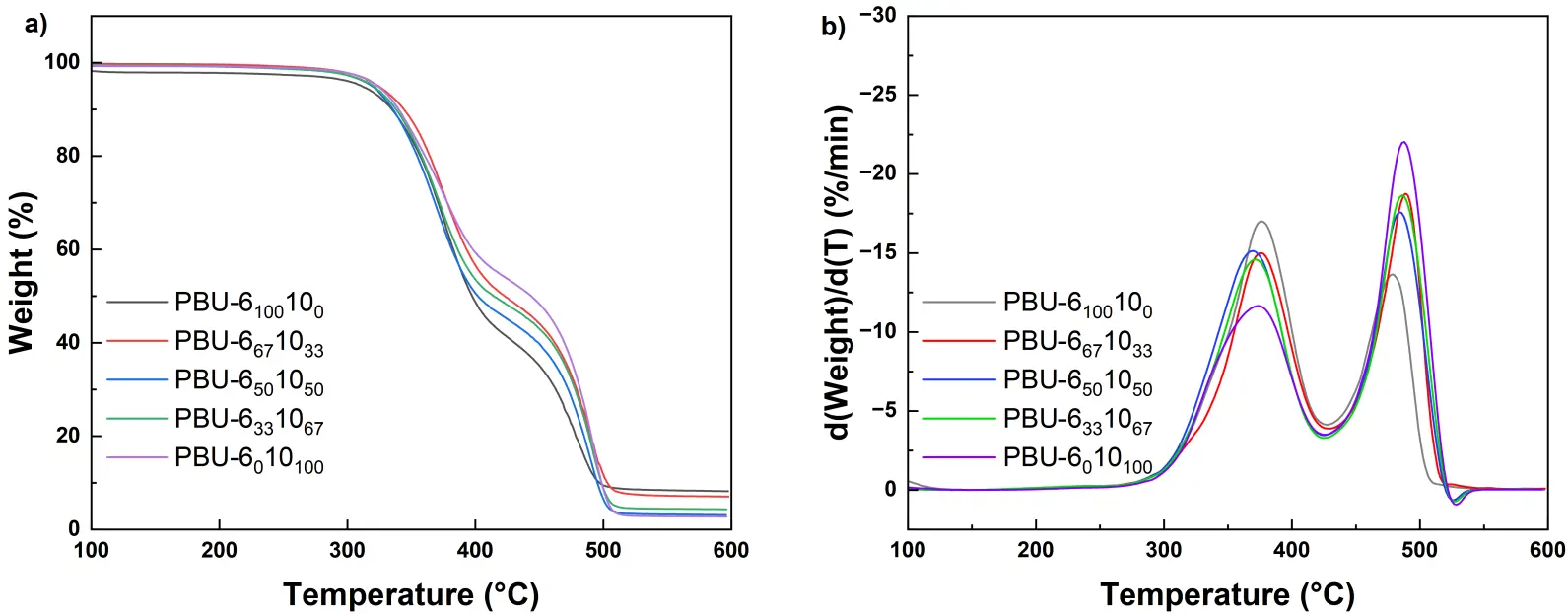
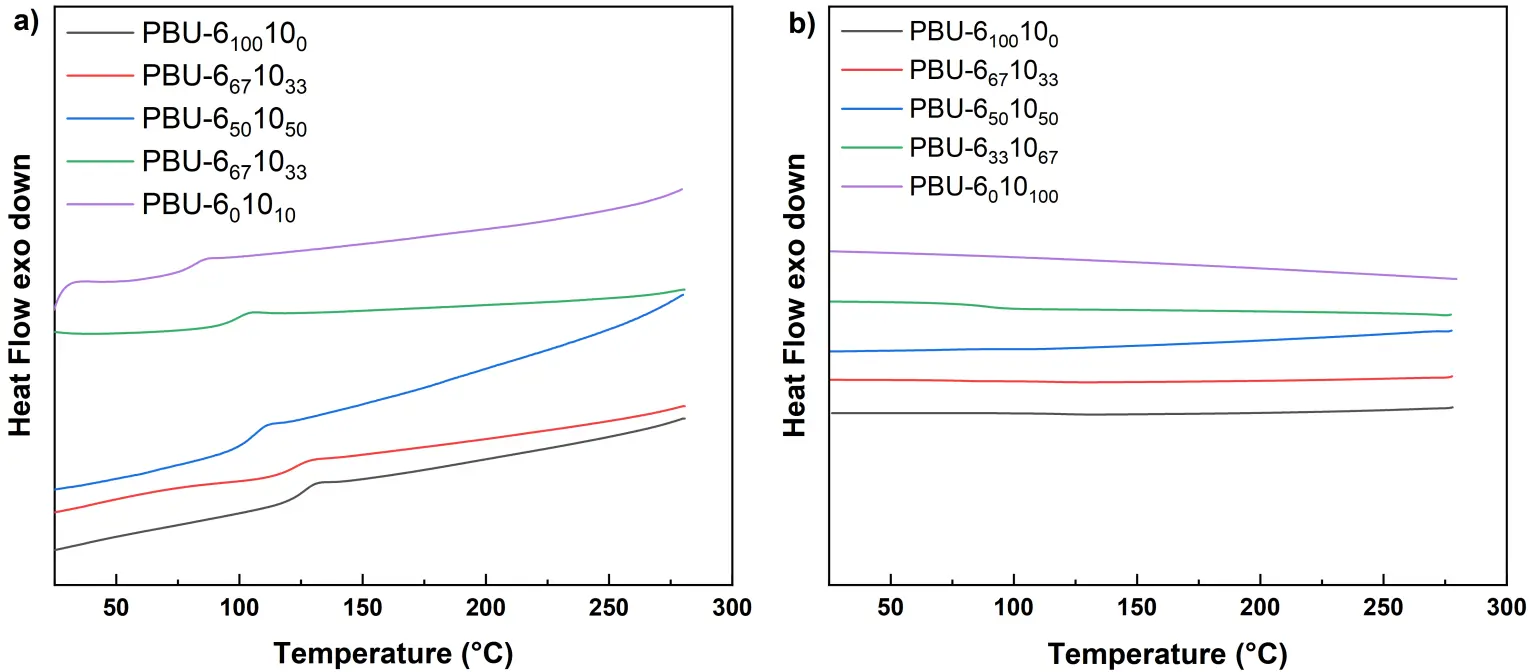
The thermostability of PBUs was tested by thermogravimetric analysis (TGA), and the curves are shown in Figure 3. For all the PBU samples, the initial decomposition temperature (Td,5%) is found to be above 310 °C. There is a slight increase in Td,5% with the increase of DDA content. The degradation behavior of these samples seems similar, which includes two steps: the breakage of biuret or urea functions and the degradation of alkylene functions. Thermal transition behavior of the samples was also probed by differential scanning calorimetry (DSC) (see Figure 4), and the results are listed in Table 1. There is no melting peak observed in the second heating curves for all PBUs samples. In addition, no evident changes are observed in the DSC cooling curves. It is interesting that those PBUs have a higher glass transition temperature (Tg) than PUrs with similar structures [11]. For instance, the Tg of PUr-6 was detected to be 90.4 °C, but that of PBU-6100100 increases to 131.2 °C. This should be attributed to the strong hydrogen bonding effects [22] provided by the biuret groups, which would restrict the flexibility of hexamethylene segments in PBUs. The Tg of PBU decreases gradually as the increase of DDA content. It is understandable since the introduction of DDA portion would be able to enhance the flexibility of PBU chains.
Table 1. Thermal properties of PBUs.
|
Sample |
Td,5% (°C) |
Td,1 a (°C) |
Td,2 a (°C) |
Tg b (°C) |
Tg,1 c (°C) |
Tg,2 c (°C) |
|---|---|---|---|---|---|---|
|
PBU-6100100 |
311.1 |
376.3 |
477.3 |
131.2 |
138.4 |
- |
|
PBU-6671033 |
324.3 |
373.9 |
488.2 |
130.0 |
93.1 |
123.0 |
|
PBU-6501050 |
319.1 |
370.0 |
489.3 |
107.4 |
102.1 |
122.7 |
|
PBU-6331067 |
320.7 |
373.0 |
491.0 |
106.4 |
81.5 |
104.2 |
|
PBU-6010100 |
323.6 |
378.9 |
492.0 |
89.8 |
91.9 |
- |
a Td,1 is the maximum decomposition rate of temperature of the first stage; Td,2 is the maximum decomposition rate of temperature of the second stage. b Tg was measured by the second heating curves of DSC. c Tg,1 and Tg,2 were measured by Tan δ curves of DMA.
3.3. Mechanical Properties
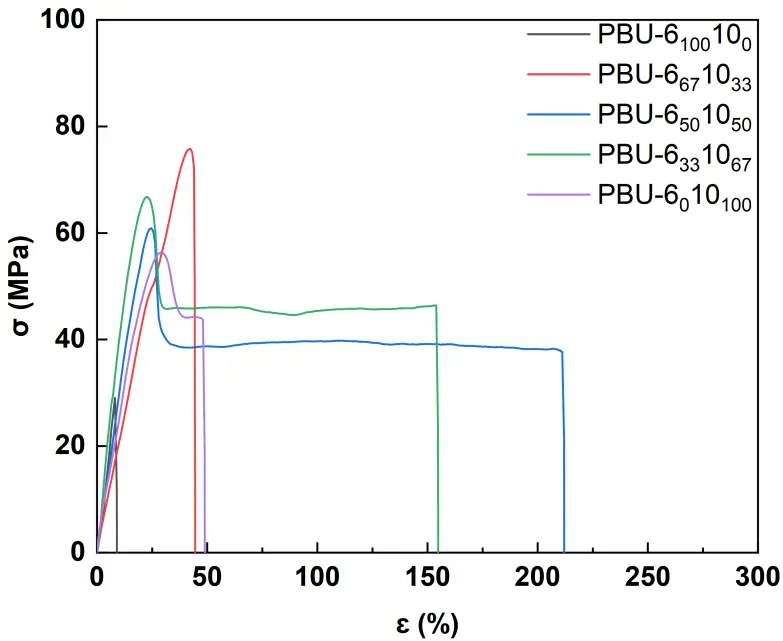
The curves of dynamic mechanical thermal analysis (DMA) and Tg was measured by the Tan δ curves of DMA (see Figure 5 and Table 1). On the storage modulus curves, a rubbery plateau is observed above 150 °C, indicating that all polymers possess a micro-crosslinked structure. For copolymers, two peaks can be observed in the tan δ curves, which dedicates to the different flexibility of hexamethylene and decamethylene moieties [23]. Tensile tests were conducted to explore the mechanical properties of PBUs (see Figure 6), and the data are listed in Table 2. As we can see from the typical stress-strain curves, PBU-6100100 seems rigid and brittle, which may be due to the rigid structure with high hydrogen bonding density. When introducing the part of DDA, for instance, PBU-6671033, the product is also rigid but less brittle and the elongation at break (εb) increases from 8% to 44%. The value of strength at break (σb) achieves the highest level at 77 MPa. Further increasing the feeding ratio of DDA, the materials become tougher, the toughness increases from 1.3 (PBU-6100100) to 85.7 J·cm−3 (PBU-6501050). When compared to the corresponding PUr, PBU-6671033 shows higher σb (54.6 vs. 77.4 MPa) [11]. The values of σb for other PBUs are quite similar to those of the corresponding PUrs.
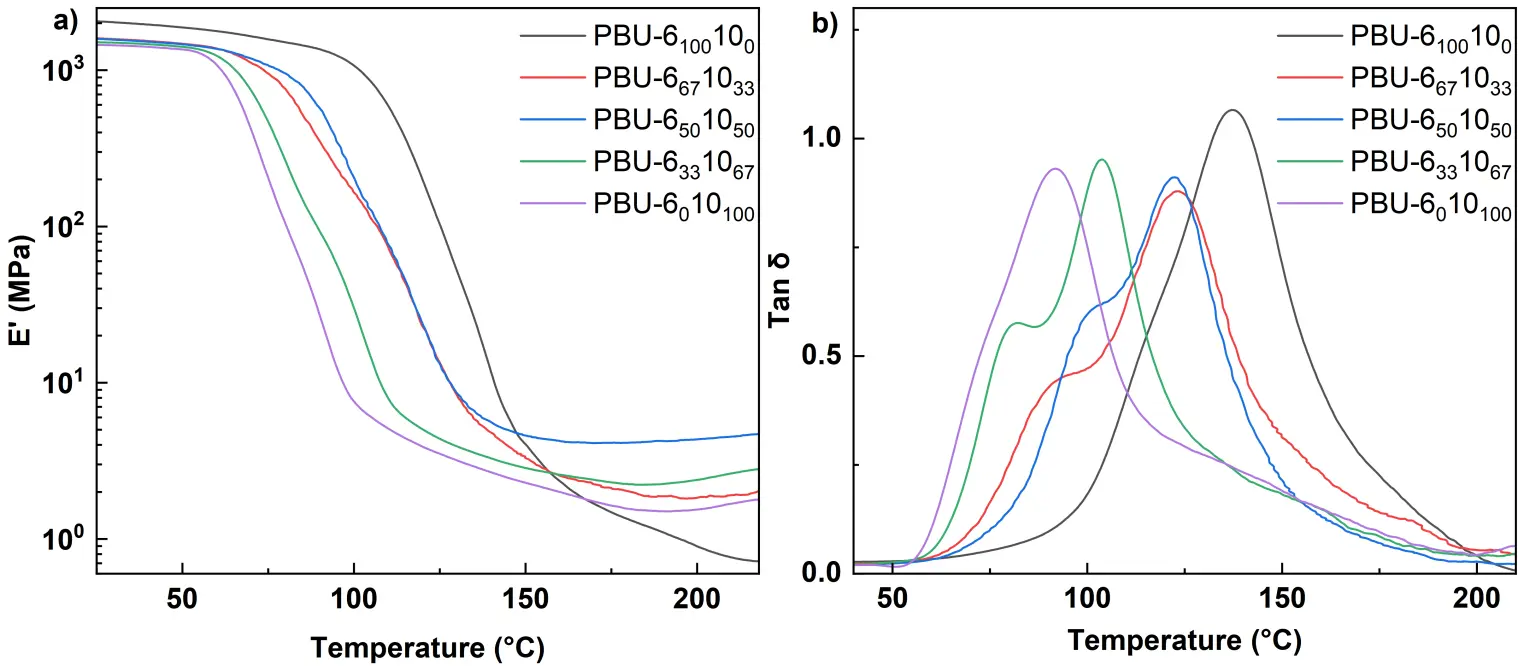
Figure 5. (a) Storage modulus and (b) Tan δ curves of PBUs in DMA testings at 5 °C/min with a frequency of 1 Hz.
Table 2. Mechanical properties of PBUs.
|
Sample |
E a (MPa) |
σy a (MPa) |
σb a (MPa) |
ɛb a (%) |
Toughness b (J·cm−3) |
|---|---|---|---|---|---|
|
PBU-6100100 |
465.7 ± 159.2 |
- |
30.4 ± 7.7 |
8 ± 0.1 |
1.3 ± 0.3 |
|
PBU-6671033 |
315.5 ± 71.8 |
- |
77.4 ± 1.5 |
44 ± 6 |
19.3 ± 3.7 |
|
PBU-6501050 |
386.1 ± 16.0 |
61.2 ± 0.3 |
42.0 ± 5.1 |
220 ± 14 |
85.7 ± 7.6 |
|
PBU-6331067 |
277.1 ± 46.5 |
60.0 ± 6.0 |
47.8 ± 7.2 |
167 ± 92 |
70.4 ± 40.0 |
|
PBU-6010100 |
256.3 ± 109.2 |
55.1 ± 2.7 |
55.3 ± 17.8 |
57 ± 18 |
21.0 ± 5.9 |
a The values of Young’ s modulus (E), yielding strength (σy), strength at break (σb), and elongation at break (ɛb) were determined by tensile test with a tensile speed at 50 mm/min. b Data were calculated by integrating the stress-strain curves.
4. Conclusions
In this work, fully biobased PBUs were synthesized from biuret with two fully biobased diamines (HDA and DDA) via solvent-free and catalyst-free melting polycondensation. In the PBUs, not only the biuret group but also the urea group can be observed, and they are lightly cross-linked. Compared with traditional PUrs, the introduction of biuret groups results in a rise of hydrogen bonding density and affords polymers with enhanced thermal and mechanical properties. The glass transition temperature is about 40 °C higher and the strength at break achieves as high as 77.4 MPa. The mechanical properties of PBUs can be feasibly tuned by adjusting the fraction of these two fully biobased diamines. The strength becomes higher when the feeding ratio of HDA increases. The toughness reaches at the highest level when the feeding ratio of HDA and DDA is at 1/1 (PBU-6501050). This work provides a novel solution in developing fully biobased plastics by simply introducing biuret functions in the backbone to improve the thermal and mechanical properties.
Acknowledgments
The authors are grateful for the supports of the Fund of Guangdong Provincial Key Laboratory of Luminescence from Molecular Aggregates (South China University of Technology, No. 2023B1212060003).
Author Contributions
Conceptualization, D.T. and B.P.; Methodology, D.T., B.P. and Y.C.; Validation, D.T., B.P. and Y.C.; Formal Analysis, B.P. and Y.C.; Investigation, D.T.; Resources, D.T.; Data Curation, B.P. and Y.C.; Writing—Original Draft Preparation, Y.C.; Writing—Review & Editing, D.T. and Y.C.; Supervision, D.T.; Project Administration, D.T.; Funding Acquisition, D.T.
Ethics Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Funding
This research was funded by the Fund of Guangdong Provincial Key Laboratory of Luminescence from Molecular Aggregates (South China University of Technology, No. 2023B1212060003).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Hessefort LZ, Long TE. Urea as a monomer: Synthesis and characterization of semi-aromatic thermoplastic non-isocyanate polyureas. Polym. Chem. 2025, 16, 3319–3328. DOI:10.1039/D5PY00445D [Google Scholar]
- Calle M, Lligadas G, Ronda JC, Galià M, Cádiz V. Non-isocyanate route to biobased polyurethanes and polyureas via AB-type self-polycondensation. Eur. Polym. J. 2016, 84, 837–848. DOI:10.1016/j.eurpolymj.2016.04.022 [Google Scholar]
- Ban JL, Li SQ, Yi CF, Zhao JB, Zhang ZY, Zhang JY. Amorphous and Crystallizable Thermoplastic Polyureas Synthesized through a One-pot Non-isocyanate Route. Chin. J. Polym. Sci. 2019, 37, 43–51. DOI:10.1007/s10118-018-2165-0 [Google Scholar]
- Kébir N, Benoit M, Legrand C, Burel F. Non-isocyanate thermoplastic polyureas (NIPUreas) through a methyl carbamate metathesis polymerization. Eur. Polym. J. 2017, 96, 87–96. DOI:10.1016/j.eurpolymj.2017.08.046 [Google Scholar]
- Post C, Maniar D, Folkersma R, Voet VSD, Loos K. Panoramic view of biobased BHMTHF-based polymers. Polym. Chem. 2025, 16, 3587–3596. DOI:10.1039/D5PY00544B [Google Scholar]
- Martin A, Lecamp L, Labib H, Aloui F, Kébir N, Burel F. Synthesis and properties of allyl terminated renewable non-isocyanate polyurethanes (NIPUs) and polyureas (NIPUreas) and study of their photo-crosslinking. Eur. Polym. J. 2016, 84, 828–836. DOI:10.1016/j.eurpolymj.2016.06.008 [Google Scholar]
- Li S, Zhao J, Zhang Z, Zhang J, Yang W. Aliphatic thermoplastic polyurethane-ureas and polyureas synthesized through a non-isocyanate route. RSC Adv. 2015, 5, 6843–6852. DOI:10.1039/C4RA12195C [Google Scholar]
- Lv X, Wang L, Li S, Sheng C, Liu Y, Zhang Y, et al. Molecular engineering of transparent non-isocyanate polyurea elastomers: Multi-scale hydrogen-bond design for simultaneous mechanical robustness, self-healing and recyclability. Chem. Eng. J. 2025, 523, 168758. DOI:10.1016/j.cej.2025.168758 [Google Scholar]
- Kébir N, Benoit M, Burel F. Elaboration of AA-BB and AB-type Non-Isocyanate Polyurethanes (NIPUs) using a cross metathesis polymerization between methyl carbamate and methyl carbonate groups. Eur. Polym. J. 2018, 107, 155–163. DOI:10.1016/j.eurpolymj.2018.07.045 [Google Scholar]
- Dennis JM, Steinberg LI, Pekkanen AM, Maiz J, Hegde M, Müller AJ, et al. Synthesis and characterization of isocyanate-free polyureas. Green Chem. 2018, 20, 243–249. DOI:10.1039/C7GC02996A [Google Scholar]
- Lin C, Xie K, Tang D. High-performance thermoplastic polyureas via a non-isocyanate route from urea and aliphatic diamines. J. Appl. Polym. Sci. 2022, 139, e52513. DOI:10.1002/app.52513 [Google Scholar]
- Li H, Cheng H, Zhao F. A Review on CO2-Based Polyureas and Polyurea Hybrids. Asian J. Org. Chem. 2022, 11, e202200338. DOI:10.1002/ajoc.202200338 [Google Scholar]
- Ding L, Wang Y, Lin J, Ma M, Hu J, Qiu X, et al. Recent advances in polyurea elastomers and their applications in blast protection: A review. J. Mater. Sci. 2024, 59, 14893–14923. DOI:10.1007/s10853-024-10050-7 [Google Scholar]
- Zhang H, Meng J, Cai L, Wang Z, Tong G. Polyurea: Evolution, synthesis, performance, modification, and future directions. Prog. Org. Coat. 2025, 201, 109127. DOI:10.1016/j.porgcoat.2025.109127 [Google Scholar]
- Kumar A, Luk J. Catalytic Hydrogenation of Urea Derivatives and Polyureas. Eur. J. Org. Chem. 2021, 2021, 4546–4550. DOI:10.1002/ejoc.202100775 [Google Scholar]
- Luk J, Goodfellow AS, More ND, Bühl M, Kumar A. Exploiting decarbonylation and dehydrogenation of formamides for the synthesis of ureas, polyureas, and poly(urea-urethanes). Chem. Sci. 2024, 15, 16594–16604. DOI:10.1039/D4SC03948C [Google Scholar]
- Yu K, Xin A, Feng Z, Lee KH, Wang Q. Mechanics of self-healing thermoplastic elastomers. J. Mech. Phys. Solids 2020, 137, 103831. DOI:10.1016/j.jmps.2019.103831 [Google Scholar]
- Guo K, Qu J, Chen R, Liu Q, Yu J, Liu J, et al. A hierarchical energy dissipation strategy for impact-resistant polyurea driven by multiple hydrogen bonds. Constr. Build. Mater. 2025, 487, 142056. DOI:10.1016/j.conbuildmat.2025.142056 [Google Scholar]
- Holzworth K, Jia Z, Amirkhizi AV, Qiao J, Nemat-Nasser S. Effect of isocyanate content on thermal and mechanical properties of polyurea. Polymer 2013, 54, 3079–3085. DOI:10.1016/j.polymer.2013.03.067 [Google Scholar]
- Pan B, Tang D. Synthesis and Properties of Fully Biobased Thermoplastic Elastomers from Priamine. ACS Macro Lett. 2025, 14, 1661–1667. DOI:10.1021/acsmacrolett.5c00614 [Google Scholar]
- Coleman MM, Sobkowiak M, Pehlert GJ, Painter PC, Iqbal T. Infrared temperature studies of a simple polyurea. Macromol. Chem. Phys. 1997, 198, 117–136. DOI:10.1002/macp.1997.021980110 [Google Scholar]
- Jiang S, Shi R, Cheng H, Zhang C, Zhao F. Synthesis of polyurea from 1,6-hexanediamine with CO2 through a two-step polymerization. Green Energy Environ. 2017, 2, 370–376. DOI:10.1016/j.gee.2017.05.001 [Google Scholar]
- Bao C, Miao R, Yin Y, Xu Y, Ding Y, Liu J, et al. Mechanically Robust yet Body-Temperature Self-Healable Polyurethane Elastomer via the Cross-Linking of Dynamic Boroxines. ACS Appl. Polym. Mater. 2024, 6, 797−805. DOI:10.1021/acsapm.3c02418 [Google Scholar]