Technological Innovation in Syngas Fermentation and Prospects for Industrial Application
Received: 12 January 2026 Revised: 23 January 2026 Accepted: 04 February 2026 Published: 24 February 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Global climate change is a significant challenge facing humanity, driven primarily by greenhouse gas emissions from human activities. Among these, CO2 accounts for 76.7% of total global greenhouse gas emissions, making it the most dominant greenhouse gas. CO2 is not only a major contributor to global climate change but also poses a serious threat to ecosystem stability and human sustainable development [1,2]. The CO2 concentration in 2024 reached 422.3 ppm, continuing the upward trend and setting another all-time high [3]. In addition, CO is also an important component of industrial exhaust gas. Incomplete combustion of fossil fuels, steelmaking, and other industrial processes emits large amounts of CO, CO2, and H2 [4,5,6]. Traditional combustion for power generation generates substantial pollutants and has low energy utilization efficiency.
In response to the challenges, the Paris Agreement explicitly sets the goal of limiting global temperature rise to well below 2 °C above pre-industrial levels, and the United Nations further advocates that all countries achieve carbon neutrality by 2050 [7]. Achieving this goal requires transitioning to a bio-based economy and renewable energy to reduce emissions from sources, and it also relies on breakthroughs in key technologies such as carbon capture, utilization, and storage (CCUS) [8,9,10,11]. CCUS technology captures CO2 from industrial emissions for geological sequestration or resource utilization, while bio-CCUS technology sequesters CO2 using microorganisms, plants, and algae [12,13]. Despite its broad prospects, CCUS technology still faces challenges, including potential leakage risks from geological sequestration (annual leakage rates of 0.00001–1%) and high costs [14,15].
Among numerous technologies for reducing carbon emissions and improving resource utilization, syngas fermentation has attracted widespread attention [5,16]. Syngas is a mixed gas containing CO, CO2, and H2, and syngas fermentation converts gaseous feedstock into high-value-added chemicals and biofuels in a single step through microbial fermentation [17,18,19,20]. Using syngas as the carbon and energy source, this technology employs acetogens to convert CO2 and CO into acetyl-CoA via the Wood-Ljungdahl pathway, which is then converted into various high-value chemicals and biofuels, such as ethanol, acetic acid, butanol, 2,3-butanediol, and butyric acid [21,22,23,24,25,26,27]. Compared with traditional chemical conversion technologies, syngas fermentation offers distinct advantages, including operation at low temperature and pressure and the ability to handle flexible gas compositions. It is particularly suitable for the resource utilization of industrial exhaust gas from steel plants and other industrial facilities [28,29,30,31,32,33]. However, this technology still faces bottlenecks, such as limited gas-liquid mass transfer efficiency and difficulty in scale-up, which restrict its large-scale industrial application [31].
In summary, syngas fermentation technology has significant strategic value for carbon neutrality and the utilization of industrial exhaust gas resources. However, numerous challenges remain, including the in-depth exploration of breakthroughs in key technologies and the implementation of industrialization. Existing studies have not yet formed a systematic technical system or industrialization solutions. Based on this, this review focuses on strain construction, technology optimization, process integration, and breakthroughs in bottlenecks, systematically analyzing and discussing core technologies, key process technologies, full-process integration, and industrialization challenges. It aims to provide comprehensive theoretical and technical support for the sustainable development of syngas fermentation technology, facilitating its greater role in energy transition practices.
2. Syngas Fermentation Strains
Syngas fermentation is a highly promising biotechnological route that relies on acetogens to convert syngas into biofuels and bio-based chemicals. Industrial strains directly determine the range of feedstocks that can be used, the types of target products, and industrial production costs. Acetogens possess the most extensive and flexible substrate utilization system among known microorganisms; they can not only utilize simple C1 compounds such as CO and CO2 but also metabolize complex carbohydrates [34,35,36,37].
2.1. Key Enzymes of Acetogens
Acetogens are strictly anaerobic, Gram-positive bacteria, with typical representatives including Clostridium autoethanogenum, Clostridium ljungdahlii, Acetobacterium woodii, and Moorella thermoacetica [24,38]. As shown in Figure 1, the Wood-Ljungdahl pathway (WLP) is the core pathway for acetogens to metabolize syngas, in which Acetyl-CoA is generated through the synergistic cooperation of the methyl and carbonyl branches [39]. In the methyl branch, CO2 is reduced by a sequence of metalloenzymes using tetrahydrofolate as the cofactor, namely formate dehydrogenase (FDH), formyltetrahydrofolate synthetase (FHS), methenyltetrahydrofolate cyclohydrolase/dehydrogenase (FCH/MDH), methylenetetrahydrofolate reductase (MTHFR), and methyltransferase (MT). Ultimately, the methylcobamide iron-sulfur protein (Methyl-CoFeS) serves as the methyl donor. In the carbonyl branch, the carbon monoxide dehydrogenase/acetyl-CoA synthase (CODH/ACS) complex catalyzes the reduction of another molecule of CO2 to CO [40]. Subsequently, the methyl donor, coenzyme A, and CO generated from the carbonyl branch condense, mediated by CODH/ACS, to form acetyl-CoA [40,41,42]. If CO is present in the substrate, it can be directly utilized by the CODH/ACS complex.
WLP cannot generate net ATP via substrate-level phosphorylation, and insufficient energy supply is a core bottleneck in its metabolism. In response to this bottleneck, acetogens have evolved two key energy conservation mechanisms: first, establishing a proton-motive force via membrane-bound ferredoxin: NAD⁺ oxidoreductase (Rnf) or energy-converting hydrogenase complexes to drive ATP synthesis through chemiosmotic phosphorylation; second, enhancing energy utilization efficiency by coupling exergonic reactions with endergonic reactions through the flavin-based electron bifurcation mechanism [43,44].
In Clostridium autoethanogenum, there are three carbon monoxide dehydrogenase (CODH) isozymes, namely AcsA, CooS1, and CooS2. Among them, AcsA is the CODH subunit of the CODH/ACS complex, which converts CO2 to CO as its core function. It provides the critical CO precursor for the complex to synthesize acetyl-CoA, and the CO generated by AcsA is transferred to the ACS active site through a molecular tunnel to participate in the condensation reaction [45]. Inactivation of this enzyme results in the inability of the strain to grow under CO conditions, making it an indispensable core unit of the WLP. Additionally, CooS1 and CooS2 are monofunctional CODHs that primarily catalyze the reversible conversion between CO2 and CO, thereby exhibiting functional redundancy. CooS1 oxidizes CO for energy production and provides electrons for hydrogen production (reaction: CO + H2O → CO2 + 2H⁺ + 2e⁻), and is responsible for extracting and transferring electrons from CO [46]. Similarly, CooS2 is a monofunctional CODH that usually coexists with CooS1, and its function is more biased toward CO2 reduction under anaerobic conditions (reaction: CO + H2O ⇌ CO2 + 2H⁺ + 2e⁻), providing CO precursors and reducing power [46]. In C. autoethanogenum, acsA is indispensable for autotrophic growth, while deletion of cooS1 and cooS2 exerts only a minor impact [47].
During CO conversion in Carboxydothermus hydrogenoformans, carbon monoxide dehydrogenase (CODH) serves as the core catalytic component, and ChCODH-Ⅱ is a key functional unit of this pathway by virtue of its high catalytic activity [48]. However, ChCODH-Ⅱ exhibits extreme sensitivity to oxygen and is prone to conformational disruption of the active site, leading to catalytic inactivation. This inherent characteristic severely restricts its large-scale application in complex industrial gas environments. To address this bottleneck, Kim adopted a comparative structural biology approach to systematically elucidate the structural differences between the oxygen-sensitive ChCODH-Ⅱ and its oxygen-tolerant homolog, ChCODH-Ⅳ, ultimately identifying the key amino acid residue A559 in the substrate channel [48]. The research team then conducted site-directed mutagenesis on ChCODH-Ⅱ, which significantly reduced its sensitivity to oxygen while retaining almost all of its original high CO conversion activity.
Hydrogenases and formate dehydrogenases (FDHs) also play crucial roles in electron transfer and carbon fixation, and their functional efficiency determines the syngas utilization rate and product yield. In autotrophic growth, hydrogenases primarily provide reducing power and energy to cells by oxidizing H2 and transferring electrons to carriers, such as ferredoxins, to drive energy-consuming reactions, including CO2 reduction. Based on the metals in their active sites, hydrogenases are classified into [NiFe]-, [FeFe]-, and [Fe]-types. These types differ in catalytic activity and oxygen sensitivity, and most acetogens contain multiple hydrogenases to meet diverse metabolic demands [49]. Formate dehydrogenases, on the other hand, are responsible for CO2 fixation in the methyl branch of the WLP, catalyzing the conversion of CO2 to formate and fixing the carbon atoms of the methyl group into acetyl-CoA.
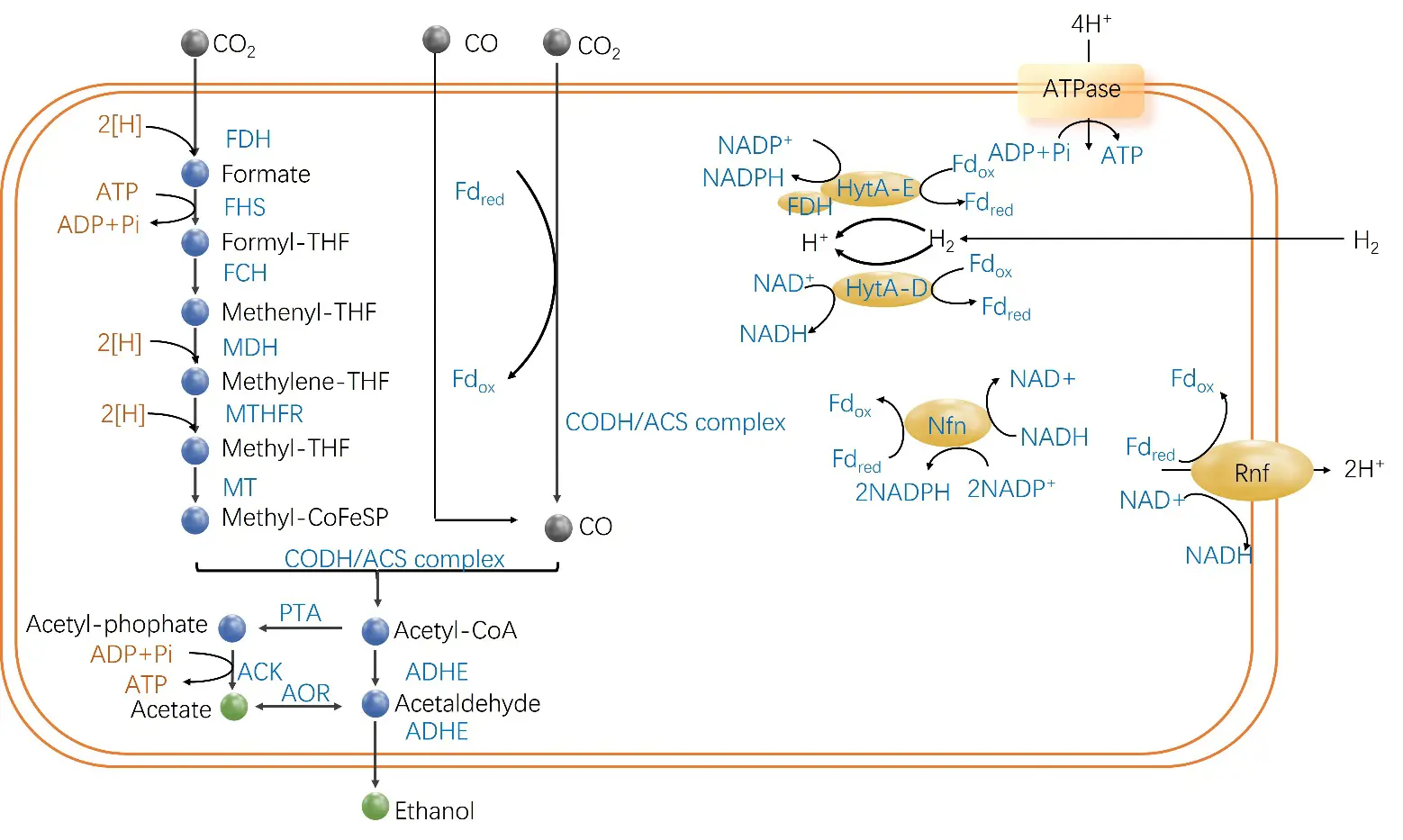
Figure 1. Key metabolic pathways and key enzymes of acetogens [50]. Adapted from reference [50]. THF: Tetrahydrofolate; FDH: Formate dehydrogenase; FHS: Formyl-tetrahydrofolate synthase; FCH: Formyl-cyclohydrolase; MDH: Methylene-tetrahydrofolate dehydrogenase; MTHFR: Methylene-tetrahydrofolate reductase; MT: Methyltransferase; CoFeSP: Corrinoid iron-sulfur protein; CODH: CO dehydrogenase; ACS: Acetyl-CoA synthase; PTA: Phosphotransacetylase; ACK: Acetate kinase; Fdox: Oxidized ferredoxin; Fdred: Reduced ferredoxin.
The synthesis of ethanol from acetyl-CoA as the precursor is tightly regulated in C. autoethanogenum, relying primarily on two core metabolic pathways: the AOR (aldehyde: ferredoxin oxidoreductase) pathway, which first reduces acetate to acetaldehyde and then reduces acetaldehyde to ethanol via alcohol dehydrogenase (Adh); and the ADHE (bifunctional aldehyde/alcohol dehydrogenase) pathway, in which AdhE directly reduces acetyl-CoA to ethanol. The genome of C. autoethanogenum encodes two AOR isoenzymes (Aor1 and Aor2) and two AdhE isoenzymes (AdhE1 and AdhE2). When using syngas, the AOR pathway exhibits tighter coupling with ATP metabolism, thereby conferring a competitive advantage in energy-limited environments. Furthermore, previous studies have shown that aor2 knockout enhances ethanol synthesis, suggesting functional differences between Aor1 and Aor2; aor2 deletion likely reduces metabolic flux diversion or substrate competition. Similarly, knockout of adhE1 or adhE2 also increases ethanol production, presumably by blocking the AdhE-mediated reversible reaction that oxidizes ethanol back to acetate, thereby diverting more reducing power and carbon flux toward the AOR pathway [51]. In addition, both the AOR and ADHE pathways possess bidirectional catalytic activity: they not only reduce acetate to ethanol during the exponential growth phase of the strain but also oxidize ethanol to acetate during the stationary growth phase or under specific conditions.
2.2. Metabolic Regulation Strategies of Acetogens
The construction and optimization of strains represent a core step in enhancing the commercial feasibility of syngas fermentation processes. Adaptive laboratory evolution (ALE) has been successfully applied to the performance improvement of acetogens. For instance, the C. autoethanogenum strain LAbrini, obtained via ALE, exhibits superior growth performance, product synthesis efficiency, and environmental robustness compared to the wild-type strain [52]. The development of specialized molecular tools is the fundamental basis for the engineering application of acetogens [53]. Given that most acetogens are strictly anaerobic Gram-positive bacteria, researchers have established shuttle plasmid systems that can be stably replicated and expressed in hosts. These systems provide a platform for the efficient expression of heterologous genes and genetic manipulation. Gene editing technologies have evolved from early traditional methods based on homologous recombination to efficient editing strategies based on the CRISPR-Cas system, enabling precision manipulations such as gene knockout, knock-in, and base editing. This advancement provides critical technical support for precise, multi-site genome modifications [54,55,56,57]. In recent years, three anaerobically compatible fluorescent systems—FAST (fluorescent-activating and absorption-shifting tag), HaloTag, and SNAP-tag—have been reported, laying the foundation for high-sensitivity, fluorescent-based, high-throughput, rapid screening of acetogens [58,59].
In terms of high-value and diversified product modification, the expansion and regulation of product profiles have been successfully achieved through strategies such as gene deletion, modification, and heterologous pathway expression: knockout of the bifunctional aldehyde/alcohol dehydrogenase in C. ljungdahlii can regulate the yield ratio of acetate to ethanol [60,61]; heterologous expression of acetone and butanol synthesis pathways, as well as construction of non-natural product synthesis pathways, have enabled the synthesis of various high-value target products including acetone, butanol, biodiesel precursors, and polyhydroxybutyrate (PHB), thereby enhancing the industrial value of syngas fermentation [25,62,63,64]. In addition, researchers have further improved the fermentation performance of strains through auxiliary engineering strategies: by regulating factors related to extracellular polysaccharide synthesis and biofilm formation, combined with the development of hollow fiber membrane bioreactors, the reaction stability of strains has been improved [65]; by precisely regulating the expression of Spo0A, a key regulatory factor for sporulation, or targeting downstream regulators to construct asporogenous strains, the negative impact of sporulation on fermentation efficiency has been reduced [66].
2.3. High-Throughput Construction of Syngas-Fermenting Strains
High-throughput construction of syngas-fermenting strains is a key step toward overcoming the inherent drawbacks of acetogens and promoting their industrial application. Relying on gene editing, automation, and screening technologies, it enables rapid, large-scale construction and screening of genetically modified strains, thereby optimizing gas utilization efficiency, product yield, tolerance, and strain robustness. Compatible with automated strain construction, high-throughput screening technologies have developed rapidly, enabling rapid screening of mutants with application value from genetically diverse strain libraries and enhancing the industrial application capacity of the strains [67,68,69].
Currently, microplate, microdroplet, and single-cell screening are widely used high-throughput screening systems [70]. For the high-throughput screening under anaerobic conditions, there are presently successful cases focusing on key areas such as strain viability detection and metabolite screening. For metabolite screening, researchers have developed a high-throughput spectrophotometric detection method based on the alcohol dehydrogenase of the thermophilic anaerobic bacterium Thermotoga hypogea. Using microplates, this method can quickly and efficiently screen high-butanol-producing mutant libraries under anaerobic conditions [71]. In evaluating the growth characteristics of anaerobic production strains, researchers have combined high-throughput anaerobic growth curve determination technology with a systematic data analysis framework, and this strategy has been successfully applied to screening research on Clostridium kluyveri [72]. This integrated technology can simultaneously detect and analyze the concentrations of multiple substrates and products, providing strong technical support for bioprocess modeling and optimization as well as the screening of high-efficiency strains. The core design concept of anaerobic high-throughput screening platforms is to adapt and integrate high-throughput detection and culture systems for use in an anaerobic environment.
However, reports on high-throughput screening systems targeting syngas-fermenting microorganisms are relatively rare. The toxicity, flammability, and explosiveness of CO and H2 in syngas have led to a lag in the research and development of high-throughput screening technologies for strictly anaerobic acetogens, and there is a lack of mature commercial solutions. In addition, the system requires extensive customization to ensure stable operation, further increasing the use cost [73]. In 2019, LanzaTech established the world's first fully automated, high-throughput Clostridium workstation, achieving whole anaerobic operation in a toxic, flammable gaseous environment [74]. The construction and screening of thousands of strains were completed based on microplate fermentation technology, providing a paradigm for technological breakthroughs in this field. Regarding the workstation-specific design principles and operational mechanisms, no detailed elaboration has been provided in the relevant reports. Nevertheless, this microplate platform has drawbacks, including limited gas-liquid mass transfer, inability to fully simulate the real fermentation environment, cumbersome isolation and purification of dominant strains, and limited iterative throughput. Therefore, there is an urgent need to develop high-throughput screening systems with smaller systems and higher mass transfer efficiency. Microdroplet culture systems have advantages such as microliter-scale culture, efficient gas-liquid mass transfer, online culture, and in-situ detection and sorting [75]. No relevant research reports on the application of microdroplet systems in syngas fermentation have been published so far. Although this strategy has significant application potential, syngas fermentation strains generally grow slowly and require long-term incubation. Therefore, two key issues urgently need to be addressed for the engineering application of this technology. One is maintaining the physical and chemical stability of microdroplets for several days; the other is efficiently recovering strain cells from target droplets after screening to support downstream culture and sequencing analysis [75].
Currently, the high-throughput construction and screening of syngas-fermenting strains remain challenging. At the host level, the low transformation efficiency and homologous recombination efficiency of some acetogens limit the construction of large-scale libraries. Moreover, wild-type strains exhibit slow growth and poor tolerance, requiring optimization of chassis cells through adaptive evolution or rational design, which is a time-consuming process. At the screening technology level, high-throughput methods for gas fermentation-related phenotypes remain immature. The most essential traits require verification in small-scale bioreactors, which is time-consuming and labor-intensive. At the automation level, it is necessary to address challenges such as equipment stability and reliability, the balance between standardization and flexibility, and the development and compatibility of information automation protocols [76,77]. Furthermore, due to workflow variability, customized solutions need to be designed for different scenarios [76].
3. Scale-Up of Syngas Fermentation
The large-scale production of syngas fermentation is a highly integrated biomanufacturing platform. The full release of its industrial potential relies on the collaborative optimization and in-depth coupling of the upstream, midstream, and downstream integrated industrial chain technology systems. Upstream technology focuses on syngas purification as the core; its technical level directly determines the purity, composition stability, and supply cost of the feed gas, thereby affecting the microbial activity and product selectivity in the subsequent fermentation process. Midstream technology focuses on the construction and regulation of the fermentation system, which determines product yield, production efficiency, and process stability. Downstream technology involves the separation and purification of target products, the utilization of by-products as resources, and heat recovery. The application of efficient separation technology can significantly reduce energy consumption in product purification, while recycling fermentation wastewater can achieve the dual goals of efficient resource utilization and environmental friendliness.
3.1. Feed Gas Pretreatment
Feed gas pretreatment is a critical link to ensuring stable fermentation operation. Its main objectives are to remove or reduce toxic impurities harmful to fermentative microorganisms, while adjusting the physicochemical parameters of the gas to better match fermentation conditions. Syngas is mainly derived from coal or biomass gasification or from industrial exhaust gas, and it contains various impurities, such as particulates, ash, tar, sulfides, and nitrides [64]. As shown in Table 1, particulates and ash can cause equipment abrasion and pipeline blockages; tar can cover microbial cell surfaces, hinder mass transfer, or damage membrane integrity; sulfides and other substances can destroy enzyme activity and inhibit metabolism. Untreated syngas severely inhibits cell growth and can even terminate fermentation, whereas appropriately pretreated syngas can significantly promote the synthesis of target products, such as ethanol [39,78,79,80]. As shown in Figure 2, as a multi-step integrated system, the core processes of pretreatment include: first, cooling and particulate removal, which removes particulates and ash from high-temperature gasification gas (>800 ℃) through equipment such as cyclone separators and ceramic filters [81]; second, tar removal, which can adopt technologies such as scrubbing, catalytic cracking and adsorption; third, desulfurization treatment, which achieves deep removal of sulfides through amine absorption or adsorption by zinc oxide and activated carbon [82,83]; fourth, gas conditioning, which adjusts the gas composition ratio by pressure swing adsorption (PSA) or membrane separation, combined with compression regulation to match fermentation requirements [84]. The pretreated feed gas must meet the cleanliness requirement, with impurity content below the microbial tolerance threshold, and have an appropriate composition that meets the metabolic needs of the strains.
Table 1. Common impurities in syngas.
|
Impurity Categories |
Typical Representatives |
Main Sources |
Tolerance Threshold |
|---|---|---|---|
|
Particulates and Ash |
Dust, carbon particles |
Entrained by gasification feedstock, generated during reactions [64] |
No mentioned |
|
Tar and Aromatic Hydrocarbons |
Benzene, toluene, naphthalene, phenols |
Incomplete pyrolysis products of biomass/coal [78] |
Benzene, toluene, and xylenes (BTX) < 0.19mM [85] |
|
Sulfides |
Carbonyl sulfide (COS) |
Converted from sulfur elements in the feedstock during gasification [80] |
No mentioned |
|
Nitrides |
Hydrogen cyanide (HCN) |
Converted from nitrogen elements in feedstock [79] |
<0.1 mM [86] |
|
Other Impurities |
O2, acetylene |
Feedstock impurities or incomplete reactions [79] |
<1900 ppm O2 |
In practical applications, multiple factors need to be considered when selecting purification technologies. The pretreatment process of feed gas is a core challenge for its industrial application. This process is not merely a collection of purification steps, but a dynamic, integrated systems engineering approach that must cope with upstream gas-source fluctuations and connect to downstream bioconversion [79]. Its implementation approaches can be divided into the following aspects. At the process design level, flexible modular configuration, multi-stage enhanced purification combination, online monitoring with adaptive control, and reliability design are adopted. These measures enable the system to adapt to fluctuations in the composition, impurity content, and flow rate of complex industrial gas sources, ensuring that the outlet gas quality remains within the safe tolerance range for microorganisms. Among them, the adoption of high-efficiency technologies, such as catalytic cracking, further expands the scope of treatable gas sources. At the strain level, targeted measures are implemented to enhance strain resistance to residual impurities: exploring the intrinsic tolerance of industrial strains and reinforcing their detoxification mechanisms via metabolic engineering modification. This not only relaxes the requirements for pretreatment depth to cut costs, but also serves as an essential supplementary approach for improving the overall tolerance of the system [84,87]. Economy is the core inspection criterion for the pretreatment process, and a balance must be achieved between purification depth and investment-energy consumption. Through energy integration strategies such as heat and pressure energy recovery and a long-term, stable operation design that ensures catalyst lifespan and adsorbent regeneration efficiency, life-cycle cost optimization is realized.
3.2. Syngas Fermentation
3.2.1. Large-Scale Reactors
Gases supply both the carbon source and energy for syngas fermentation. Among them, the low solubility of CO and H2 impairs mass transfer efficiency, thereby limiting the metabolic rate of strains [89,90,91]. Gas-liquid mass transfer refers to the process of mass exchange between the gas and liquid phases. It occurs as gas molecules diffuse across the interfacial boundary layer at the phase interface, thereby accomplishing the transfer from the gas phase into the liquid phase. A three-phase system consisting of a mixture of gaseous substrates, a liquid nutrient solution, and cells suspended in the broth is present in the reactor. For gases with poor solubility, the primary mass transfer resistance lies in the liquid film at the gas-liquid interface [92]. The volumetric mass transfer coefficient, KLa, is commonly used to evaluate the mass transfer characteristics of the reaction system. KLa is specific to each system and depends on various parameters, such as agitation speed, gas flow rate, bubble size, liquid circulation rate, reactor geometry, internal structure, or packing material [93,94]. Gas-liquid mass transfer is a vital reactor characteristic that can be improved by selecting appropriate parameters or designs. Since systems with efficient gas-liquid mass transfer usually require increased energy consumption, which reduces efficiency, the KLa value must be correlated with the required volumetric power input (P/V) for a more reliable comparison [95,96]. While increasing power input enhances mass transfer, the optimization objective is to minimize energy consumption while meeting process requirements. This necessitates a careful consideration of reactor geometry, operating conditions, and biological system characteristics. In gas fermentation, this balance is critical, as it directly affects the economic feasibility of the process. Therefore, optimizing bioreactor design is crucial for improving cell growth and unit productivity in gas fermentation [31,97,98].
Currently, improvements in KLa are primarily accomplished by increasing the gas-liquid contact area, reducing mass transfer resistance, and enhancing the mass transfer driving force. Hollow fiber membrane reactors (HFMRs) utilize a unique membrane structure in which micropores in the membrane wall serve as gas-distribution units. HFMRs leverage the high specific surface area of membrane materials to intensify gas-liquid mass transfer. The membrane also serves as a carrier for microbial biofilm attachment [99,100]. As shown in Table 2, Yasin constructed a membrane reactor using hydrophobic polyvinylidene fluoride (PVDF) material, achieving a KLa of 155.16 h−1 [99]. In the membrane reaction system established by Shen et al., the KLa for CO reached a peak of 1096.2 h−1, and efficient mass transfer was maintained even at low gas flow rates [101,102].
Table 2. Reactors applied to syngas fermentation and their fermentation performance.
|
Reactor Configuration |
Microorganisms |
Gas Composition |
KLa (h−1) |
Product |
Produtivity/Titer |
P/V |
|---|---|---|---|---|---|---|
|
CSTR |
C. ljungdahlii |
Syngas consisted of 70% CO |
135 |
Ethanol, acetate |
Total concentration |
Not mentioned |
|
Airlift reactor |
C. carboxidivorans |
CO, CO2 |
23.8–153.6 (CO) |
Acetate |
Acetate 7.2 mmol/L, |
3–23 W/m3 [104] |
|
HFMBR |
Eubacterium limosum |
CO |
63.72–135.72 (CO) |
Acetate |
20.92 mmol/L [99] |
Not mentioned |
|
HFMBR |
C. carboxidivorans |
CO, CO2, H2 |
400–1096.2 (CO) |
Ethanol |
3.12 mmol/L/h [101] |
Not mentioned |
|
Trickle bed |
Not applied |
Air |
421 [105] |
Not applied |
Not applied |
Not mentioned |
|
Trickle bed |
Clostridium ragsdalei |
CO, N2, CO2 and H2 |
Not mentioned |
Ethanol |
3.43 mmol/L/h [106] |
Not mentioned |
Airlift reactors drive liquid circulation relying on density differences, eliminating the need for mechanical agitation. When optimized with microporous spargers, the maximum KLa can reach 91.1 h−1, featuring uniform flow fields and mild shear stress [107]. Biofilm-packed trickle bed reactors form thin liquid-film structures within the packing materials, which shorten the substrate diffusion path; their mass transfer rates are significantly higher than those of traditional continuous stirred-tank reactors (CSTRs) [105,108,109]. CSTRs can improve flow field distribution and intensify bubble breakup by optimizing the agitation structure, thereby further enhancing KLa [110]. The bulk-gas-to-atomized-liquid (BGAL) contactors disperse liquids into micron-scale droplets via atomization, reconstructing the gas-liquid contact interface. When oxygen is used as the model gas, the KLa can reach approximately 8208 h−1, and the mass transfer efficiency is improved by more than one order of magnitude compared with traditional reactors [88,107].
Gas distributors can enhance KLa by regulating the initial bubble size and distribution; their performance determines the initial level of gas-liquid contact area [111]. Optimizing the structure, pore size, and material can achieve bubble miniaturization and homogenization, thereby significantly improving mass transfer efficiency. Among them, microbubble dispersers are the most widely used and effective, capable of generating microbubbles with diameters less than 100 μm. Compared with traditional large bubbles, these microbubbles have a much larger specific surface area, a slower rising speed, and a longer liquid-phase residence time, which can significantly increase gas-liquid contact opportunities and reduce liquid film thickness [111].
Without changing the reactor hardware configuration and components, optimizing operating conditions and process parameters is the preferred approach to improving mass transfer efficiency of existing devices. According to Henry's law, increasing the reactor operating pressure can directly increase the solubility of gaseous substrates in the fermentation broth, enhance the mass transfer driving force, and thus improve KLa; increasing gas and liquid flow rates can promote bubble breakup and dispersion, strengthen liquid phase turbulence, and reduce liquid film thickness and boundary layer thickness. Shen et al. have confirmed that increasing gas-liquid flow rates in hollow fiber membrane reactors can significantly improve the KLa for CO, and higher liquid flow rates can also avoid bubble coalescence and maintain the renewal frequency of the gas-liquid interface; for stirred-tank reactors, agitation speed is a key control parameter [101,102]. Increasing the rotation speed can enhance impeller shear force, refine bubble size, and improve the uniformity of liquid-phase mixing. However, excessively high rotation speed may damage microbial cells and inhibit metabolic activity, and this balance needs to be considered in combination with strain characteristics.
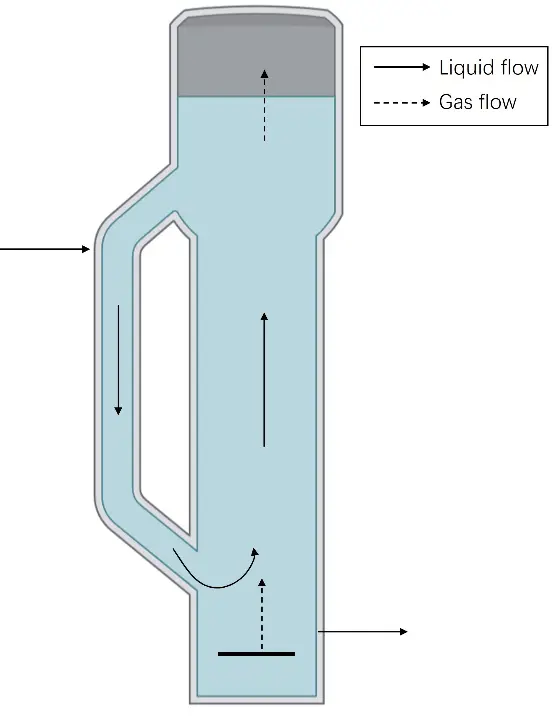
The design objective of commercial reactors is to enhance feed gas conversion efficiency with low energy consumption, thereby improving the yield of target products. Therefore, this objective should serve as a benchmark for balancing the relationships among various operating parameters. Airlift reactors offer advantages such as no mechanical agitation required, low maintenance costs, energy conservation, low shear forces, and a simple mechanical structure. They can improve gas conversion efficiency by extending gas residence time [104,112,113]. As shown in Figure 3, the reactor consists of two interconnected compartments: a riser section for receiving gas flow and a downcomer section for liquid reflux. The density difference between the riser and downcomer sections drives liquid circulation [108]. In addition, due to their unique design, airlift reactors offer advantages for handling high-viscosity or high-density liquids, thereby effectively enhancing gas-liquid mass transfer and promoting microbial growth. Peter conducted syngas fermentation using Clostridium carboxidivorans in an airlift reactor and a CSTR, with product concentrations of 1.5 g/L ethanol and 0.7 g/L butanol, and 1.2 g/L ethanol and 0.4 g/L butanol, respectively [104]. Under the same operating conditions, compared with CSTRs, the volumetric power input, mass transfer coefficient, and mixing time of the airlift bioreactor are 0.05%, 5%, and up to 2806% of those of the CSTRs, respectively [104]. Therefore, compared with CSTRs, airlift reactors have lower operating costs.
For syngas fermentation to achieve economic feasibility on a commercial scale, it must rely on a simplified process design and cost control as prerequisites [114]. During scale-up, if productivity increases solely by replicating the number of reactors, production costs will increase linearly. According to the estimates by Takors et al., the volume of a single bioreactor for syngas fermentation must exceed 500 m3, and the specific power consumption must be within 0.3 kW/m3 for the entire process to be economically viable [115].
The economics of reactor selection and scale-up are a crucial factor limiting the commercialization of syngas fermentation. Although CSTRs are widely used in laboratory research, they face high operating costs during scale-up. The gas-liquid mass transfer limitation in the syngas fermentation process must be overcome by high-speed agitation, but this high volumetric agitation power results in a drastic increase in energy consumption. In addition, although membrane reactors have the advantage of a large specific surface area for improving mass transfer efficiency, they also face prominent economic bottlenecks in scale-up: on one hand, membrane reactors have inherent problems such as membrane fouling and high investment costs, with membrane modules accounting for up to 73% of the equipment procurement cost; on the other hand, the modular nature of membrane systems determines that capacity expansion relies on increasing membrane area or adding module quantities, and scale economies cannot be achieved by increasing the size of a single unit [5]. This results in investment costs that are difficult to decrease linearly with scale expansion, constituting a significant barrier to scale-up. The economic limitations of the above two types of reactors fully indicate that the scale-up of syngas fermentation must overcome the problem of economic adaptability at the reactor level, which is supported by commercial cases represented by Shougang LanzaTech, which have selected airlift reactors, a type of reactor that achieves a better balance between mass transfer efficiency and operating costs.
3.2.2. Fermentation Strategy
Single-stage fermentation is a process completed in a single reactor, in which syngas is directly converted into target products. Its advantages lie in a simple process flow and low equipment requirements. At the same time, its disadvantages include the potential for microbial strains to be subject to product inhibition, leading to reduced product yield. Consequently, single-stage fermentation is suitable for simple, rapid production, but its efficiency and yield may be constrained. In single-stage syngas fermentation, ethanol inhibits microbial metabolic activity, limits ethanol concentration in the fermentation broth, and thereby affects product yield [107].
Two-stage fermentation divides the syngas fermentation process into two stages for treatment. In the first stage, syngas is converted into intermediate products, and in the second stage, these intermediates are further transformed into the final target products. Different operating conditions or microorganisms can be adopted in each stage to optimize fermentation efficiency [116]. Butyrivibrio methylotrophicus converts syngas into acetic acid and butyric acid, and then Clostridium acetobutylicum is used to convert the products of the first stage into ethanol and butanol [117]. By separating the growth and production stages and optimizing the process parameters for each, the production efficiency of syngas fermentation can be further improved. C. ljungdahlii conducts syngas fermentation, which is divided into growth and production stages through different reactors and operating conditions. The operating conditions of both stages are optimized to adapt to their respective objectives, and the final ethanol production rate can reach 0.37 g/L/h [118]. Meanwhile, this system recovers and reuses the incompletely converted gas to improve substrate conversion rate and reduce production costs. Multi-stage fermentation can not only increase product yield but also enhance product diversity. C. carboxidivorans is first used to convert syngas into acetic acid and butyric acid, and then Yarrowia lipolytica utilizes acetic acid and butyric acid to synthesize β-carotene [119].
The feed gas for syngas fermentation is mainly derived from industrial off-gases, suggesting that a continuous fermentation process is suitable. Upstream industrial processes, such as blast furnace ironmaking and coal gasification, operate continuously 24 h a day, and off-gases are continuously produced [88]. The CO-containing feed gas is toxic, flammable, and has a low energy density. Large-scale storage is not only technically complex and costly but also poses significant safety hazards; the optimal approach is on-site production and immediate use. Continuous fermentation can stably and continuously consume the continuously supplied gas source, matching the upstream production [120]. They avoid the shutdown issues caused by batch switching, equipment preparation, and sterilization in batch fermentation, thereby preventing resource waste and carbon emissions resulting from feed gas venting [121]. Dynamic balance is achieved through continuous material flow, improving the material utilization rate [122]. From an operational perspective, because the growth of syngas-fermenting microorganisms is slower than in batch fermentation, continuous fermentation reduces batch intervals, shortens the fermentation cycle, improves equipment utilization, and lowers operating costs.
3.2.3. Fermentation Process Control
In the process control of syngas fermentation, in addition to routine monitoring of temperature, pH, ORP (oxidation-reduction potential), cell density, and other parameters, gas composition analysis is also essential. Mass spectrometry can detect almost all gases and enable simultaneous multi-component analysis in a single measurement, making it suitable for the accurate detection of complex gas mixtures [123].
During syngas fermentation, real-time monitoring of dissolved CO, CO2, and H2 concentrations in the fermentation broth is crucial for process control. However, commercial electrodes capable of directly detecting these dissolved gases have not yet been developed. Current technologies mostly rely on offline analysis methods, which still have limitations in practical applications [124]. Although the CO-myoglobin optical absorption method can detect dissolved CO, it is complex to operate, and the protein has a limited lifespan, making long-term online monitoring difficult [124]. The low solubility of CO and H2 results in extremely low concentrations in the liquid phase, requiring sensors with extremely high sensitivity [39]. Complex components in the fermentation broth may interfere with sensor signals, so it is necessary to develop anti-contamination materials or specific recognition technologies [124]. In industrial-scale reactors, the concentration of dissolved gases fluctuates on a second-by-second scale, requiring sensors with a response time of less than 1 s, which is far beyond current technical capabilities [125].
3.2.4. Process Simulation and Reactor Scale-Up Design
Modeling of the syngas fermentation process can be classified into three closely related systems, ranging from microscale to macroscale, based on the research methods and scales used. Cell-scale metabolic and kinetic modeling focuses on intracellular metabolism and quantifies the kinetic behaviors of microbial growth, substrate consumption, and product synthesis. Based on analyses of the metabolic pathways of specific acetogens, it uses classical kinetic equations, such as the Monod and Haldane equations, or genome-scale metabolic network models to clarify growth rates, substrate inhibition effects, and product distribution rules. It serves as the foundation for revealing the intrinsic mechanisms of biotransformation and optimizing strain performance [126,127]. Interphase mass transfer modeling targets the rate-limiting step in the three-phase gas-liquid-solid system of syngas fermentation, describing the transfer of gas from bubbles to the liquid phase and then to the cell surface [112]. By calculating the KLa and constructing mass transfer equations based on the two-film theory, it quantifies the limiting effect of mass transfer rate on the overall reaction rate, linking gas-phase substrate supply with cellular metabolism. Hydrodynamic and system modeling at the reactor scale enables the coupling of microscale kinetics, mass transfer processes, and macroscale reactor behavior, and is primarily applied to process scale-up design and optimization. Specifically, this modeling method establishes mass and momentum balance equations for different reactor types. It also simulates the flow field, bubble distribution, and concentration field inside the reactor by integrating computational fluid dynamics (CFD) techniques [128].
In addition, the core bottleneck in the industrial-scale up of syngas fermentation lies in the heterogeneity of large-scale reactors. In laboratory-scale small-volume reactor systems, approximate ideal mixing conditions can be achieved through intensified stirring, thereby maintaining uniformity in key reaction parameters such as substrate concentration, pH, and pressure, providing a stable environment for microbial metabolism. However, fundamental changes occur in the hydrodynamic characteristics of the system as the reactor scale is scaled up from several liters to hundreds of cubic meters, and these changes trigger a series of chain reactions. On the one hand, macro mixing and circulation time are prolonged, with the time required for the liquid to complete a full cycle in the reactor increasing significantly [129]. On the other hand, the characteristic time constants (e.g., microbial substrate consumption rate and gaseous substrate dissolution rate) may be shorter than the fluid circulation time. This further results in the formation of significant gradients in concentration, pressure, and gas composition along the fluid flow direction inside the reactor [130]. During fluid circulation, microorganisms experience periodic drastic environmental fluctuations between “nutrient-rich zones” and “nutrient-depleted zones”, which directly affect their metabolic activity and stability [131].
To address heterogeneity challenges in large-scale reactors, optimizing reactor structures and operational modes is essential. Airlift reactors, driven by bubble ascent, offer lower energy consumption and larger gas-liquid contact areas, making them the preferred option for large-scale production [127]. Meanwhile, mass transfer efficiency can be enhanced and concentration gradients mitigated through gas recirculation (recovering unreacted off-gas) and microbubble generation [108]. Furthermore, mixing performance can be optimized by adjusting circulation parameters and gas delivery strategies, thereby reducing reactor heterogeneity [132].
In addition, the CRD-CFD coupled model can facilitate the accurate design and regulation of industrial scale-up [133]. CFD is employed to simulate the flow field, mixing time, shear stress, and concentration fields of key substances within the reactor, identifying dead zones and gradient regions [134]. The fluid environment information is then coupled with the CRD cell metabolism and growth model, and the Euler-Lagrange method is used to track virtual cell trajectories, predicting their physiological responses and differences in population metabolic activity [133]. To reduce computational costs, the CFD flow field can be divided into several perfectly mixed flow regions; flow rates derived from CFD are used to connect these regions, and the CRD model is run within each region, balancing computational efficiency with spatial heterogeneity. Based on scale-down model experiments, reaction kinetic parameters and microbial environmental response characteristics are determined. The CFD-CRD coupled model is established and calibrated using these parameters to achieve an integrated simulation of flow, mass transfer, and reaction. The model is iteratively calibrated using pilot-scale data, and the validated model serves as a "digital twin" tool for virtual screening of optimal configurations and operational parameters for industrial reactors, minimizing scale-up risks [132,135,136].
The CFD-CRD coupled model also faces several challenges. Full-scale, long-term, high-resolution simulations entail high computational costs; therefore, intelligent coupling strategies, such as surrogate modeling or machine learning, are used to improve computational efficiency while ensuring accuracy. Existing CRD models are relatively simplistic and fail to adequately reflect dynamic cellular stress responses. It is necessary to couple dynamic Metabolic Flux Analysis (dMFA) with CFD, and integrate real-time calibration using online sensor data to enhance prediction reliability [137]. Cells in the reactor exhibit heterogeneity in physiological states and other aspects; thus, population balance models (PBM) or individual-based models (IBM) should be incorporated into the CFD-CRD framework to comprehensively simulate changes in the distribution of cell population characteristics induced by environmental gradients and evaluate process stability [138,139].
Industrial pilot-scale studies have been conducted to tackle the heterogeneity challenge. For example, during syngas fermentation for acetone and isopropanol production at scales exceeding 120,000 L, LanzaTech achieved stable microbial production under fluctuating concentration conditions by deepening understanding of microbial metabolic robustness and optimizing gas delivery and mixing strategies [132]. In conclusion, addressing heterogeneity in large-scale syngas fermentation reactors is a systematic engineering task that requires integrating innovative reactor design, an optimized operational plan, and advanced simulation tools.
3.3. Separation and Purification of Products
In syngas fermentation, downstream product separation and purification are complex processes that aim to remove impurities and recover high-purity target products [16]. The concentration of products in the broth directly affects the cost and energy consumption of separation and extraction, and determines the economic benefits of the entire process. In the practical operation of syngas fermentation, the concentration of target products is typically low, leading to high energy consumption during extraction. In addition, the target products contain numerous impurities, including microbial cell debris and metabolic by-products. The presence of these impurities further complicates separation and extraction. Ethanol is currently the main commercial product of syngas fermentation, which is mainly recovered by distillation. In the ethanol industry, distillation has emerged as the preferred option, as it delivers high ethanol recovery rates and can be paired with professional software to enable process simulation [140]. However, it still has shortcomings in practical applications: it requires additional separation steps to handle azeotropes, consumes high energy, and high-temperature operation may inactivate microorganisms and proteins [141].
Pervaporation is a membrane separation technology in which liquid permeates (such as ethanol) are vaporized downstream of the membrane under vacuum conditions and then recovered through condensation [16]. Pervaporation performance depends on the separation membrane characteristics, but in practice, membrane fouling and scaling require frequent cleaning, which, in turn, affects production efficiency. Previous studies have shown that combining membrane separation, extraction, and vacuum distillation can achieve efficient ethanol separation [142].
Given that each separation technology has its unique advantages and limitations, integrating multiple separation unit modules and implementing stage-wise separation would be an effective strategy to improve efficiency and compensate for the shortcomings of each method. However, many separation technologies are still in the early stages of development, and further studies are needed to confirm their industrial applicability. The future development of downstream separation in syngas fermentation will focus on the application of in situ product-removal technology. This technology can simultaneously separate products during fermentation, effectively reducing product inhibition and increasing cell density and yield. Meanwhile, the co-development of by-products into high-value products such as chemical raw materials or bio-feed can expand diversified revenue channels to improve process economics.
In the industrial field of syngas fermentation, distillation is a validated and widely adopted separation technology (exemplified by Shougang LanzaTech). Although technologies such as pervaporation offer significant energy-saving potential, they have not yet become mainstream industrial options, and their application remains at the pilot-scale-up or evaluation phase for specific projects.
3.4. Process Integration
In discussions of the stability and efficiency of syngas fermentation technology, the efficient treatment of upstream feed gas and the separation and recovery of downstream products are undeniably crucial. Meanwhile, the stable operation of auxiliary systems, such as water circulation and heat recovery, also significantly affects them. To utilize these feedstocks efficiently and sustainably, stable auxiliary facilities for water, electricity, gas, heat, and other resources must be equipped. The high integration of these links is a key factor in ensuring continuous, stable fermentation product output and high-efficiency production. Shougang LangzaTech has integrated six sections—raw gas pretreatment, biological fermentation, distillation dehydration, protein separation, waste heat recovery, and sewage treatment—into an integrated process technology. The high-temperature steam generated during the fermentation process can be used as a heat source for the process. Through waste heat recovery technology, it not only improves energy utilization efficiency but also reduces carbon emissions. By-products produced during fermentation need to be effectively separated and recovered; otherwise, they will adversely affect the stable operation of the system. Through protein separation, microbial proteins are removed from the fermentation broth, thereby avoiding interference with system operation and enabling further utilization of these resources. This highly integrated process design not only improves resource utilization efficiency but also reduces production costs and environmental impacts.
4. Economics and Policy Support of Syngas Fermentation
The main products derived from syngas fermentation are bulk chemicals, which exhibit significant price volatility and low profit margins. Although this technology features high resource utilization efficiency and environmental friendliness, its economics and profitability are constrained by multiple factors, requiring a comprehensive techno-economic analysis of specific projects. The construction of syngas fermentation facilities entails substantial capital expenditure (Capex), including reactors, gas pretreatment systems, sewage treatment systems, and other equipment. Operating expenditure (OpEx) is equally essential and covers feedstock costs, energy consumption, equipment maintenance, and more. Syngas can be derived from diverse sources, such as biomass gasification, coal gasification, and iron and steel plant off-gases. Syngas from different sources exhibits significant cost differences: the costs of syngas derived from natural gas and corn stover are 1724 USD/ton and 2878 USD/ton, respectively [143]. Many studies on the techno-economic analysis of syngas fermentation for fuel ethanol and microbial protein production overlook these factors, focusing primarily on environmental impacts [144]. Vlaeminck developed a comprehensive model covering the fermentation process of acetic acid and microbial protein production via gas fermentation, as well as gas pretreatment and downstream processing technologies. Techno-economic analysis using this model demonstrated the economic feasibility of the process. The study showed that increasing the acetic acid concentration to 45 g/L and the productivity to 4 g/L/h could reduce the cost from 4.15 USD/kg to 2.78 USD/kg. However, this model also overlooked many other details; the impact of ambient temperature on reactor temperature control cannot be ignored. In cost-sensitive regions, the accumulation of these secondary factors can also affect the final profit margin.
The feedstock price for syngas fermentation is subject to fluctuations in the coal or biomass markets; for instance, rising coal prices may drive up syngas costs. Meanwhile, process energy consumption accounts for 20–30% of total costs, and fluctuations in energy prices directly affect ethanol costs [145,146]. Fuel ethanol projects are price-sensitive and highly influenced by ethanol selling prices, which directly determine their economic viability. The price of fuel ethanol is closely correlated with that of gasoline. Changes in government subsidy can also lead to periodic price fluctuations [147]. Issues such as regional supply disruptions or demand changes in international trade and supply chains may disrupt the balance of the global ethanol market, thereby affecting ethanol prices [148]. Currently, only a small number of demonstration projects for syngas fermentation have achieved commercialization, and the scale effect and economic benefits have not yet been fully realized. Due to the dynamic changes and interconnections among the aforementioned factors, short-term operational data from a small number of projects cannot fully reflect the complex situation across the entire industry, nor can a clear conclusion be drawn about the economic benefits of syngas fermentation.
The United States regards bioconversion technology as an essential technical support for achieving carbon neutrality goals, with multi-departmental collaboration in conducting research and development (R&D) and industrial layout. Strategic documents issued by the United States, such as Biomanufacturing to Advance the Bioeconomy, released in 2022, and Bold Goals for U.S. Biotechnology and Biomanufacturing, issued in 2023, explicitly identify the conversion of renewable feedstocks, the development of low-carbon product preparation pathways, and the circular utilization of carbon sources such as CO2 as core R&D directions. In October 2024, the U.S. Bioindustrial Manufacturing and Design Ecosystem (BioMADE) explicitly identified gas fermentation as a key technical pathway for producing fuels and chemicals using waste carbon sources such as industrial exhaust gas and syngas. The annual tender theme and the project scale determine the project funding amount. In January 2025, the Bioenergy Technologies Office (BETO) of the U.S. Department of Energy (DOE) and the Environmental Protection Agency (EPA) jointly announced the provision of a total of 6 million US dollars in funding for three biofuel promotion projects to support the leading position in energy and emission innovation. Among them, Air Company Holdings, based in Brooklyn, New York, received 2 million US dollars in funding for its project “Conversion of Biobased Carbon Dioxide to Sustainable Aviation Fuel”. On 16 January 2025, the U.S. Department of Energy (DOE) announced an investment of approximately 13.7 million US dollars in four carbon conversion technology R&D projects, aiming to promote the conversion of carbon dioxide into environmentally friendly, high-value-added products and ensure U.S. energy and critical mineral security. Among them, the project “Large-Scale Electrolysis of Carbon Dioxide to Produce Carbon Monoxide and Chemicals” received approximately 5.9397 million US dollars in funding; another project converts carbon dioxide generated from starch fermentation in biorefining into carbon monoxide through electrocatalysis, which is then transported to a gas fermenter to produce ethanol.
The European Union (EU) takes the development of the circular bioeconomy and the transition to industrial decarbonization as its core orientation. By formulating a series of strategic plans and providing exceptional funding support, it promotes the innovative development of bioconversion technologies. Policy documents such as the 2023 Strategic Research and Innovation Agenda for Bioenergy identify CO2 bioconversion and synthetic fuel production as key technical directions, among which the PyroCO2 innovation project focuses on industrial CO2 conversion. The Industrial Carbon Management Strategy clearly defines phased goals for CCUS from 2030 to 2040. In 2021, the European Union (EU) invested 44 million euros to launch the PyroCO2 innovation project, focusing on converting industrial carbon dioxide into chemical building blocks via biotechnology, and constructing facilities capable of capturing 10,000 tons of industrial carbon dioxide annually. On 16 November 2021, the EU announced an investment of 1.1 billion euros in 7 large-scale cross-border innovation projects across multiple European countries under the “Innovation Fund” framework, aimed at promoting the commercialization of breakthrough decarbonization technologies for energy-intensive industries. This funding covers technologies such as hydrogen energy, CCUS, and so on. In 2022, under the “Innovation Fund” framework, the EU launched a 150-million-euro large-scale climate transition innovation project to construct the first large-scale commercial bioenergy with carbon capture and storage (BECCS) facility.
Other developed countries have also deployed targeted policy frameworks to promote the development of relevant technologies based on their respective energy and environmental strategic needs. The United Kingdom has identified technologies such as bioenergy with carbon capture and storage (BECCS) and CO2 conversion for sustainable aviation fuel production as core supporting technologies for achieving net-zero carbon emission goals, through measures including annual special funding and project carriers such as the “Direct Air Capture Technology Competition”. Within the framework of the Green Growth Strategy for Carbon Neutrality 2050, Japan focuses on supporting core technical areas, such as the development of CO2-fixing microbial platforms and CO/CO2 feedstock-oriented manufacturing technologies.
China’s goals for carbon neutrality provide strategic guidance for the biomanufacturing industry, and a series of policy documents have established a multi-level policy support system covering technological R&D, industrial application, and financial incentives. Policy documents, such as the 14th Five-Year Plan for Bioeconomy Development, explicitly identify biomanufacturing, CCUS, and non-grain-based biobased materials as key development areas. The Guiding Opinions on Accelerating the Green Development of Manufacturing Industry further focus on key technical fields such as industrial process coupling with CCUS and CO2 bioconversion and utilization. The technology of producing ethanol from industrial off-gases has been successively included in the list of green and low-carbon technology demonstration projects and the Fifth Batch of National Key Promoted Low-Carbon Technology Catalog. C. autoethanogenum protein has been selected into the list of landmark biomanufacturing products, forming a full-chain policy incentive system covering technological R&D, pilot-scale amplification, and industrial application.
Overall, the United States emphasizes overall bioeconomy competitiveness, the European Union focuses on climate goals and circular economy, while China prioritizes integration with industrial system upgrading and comprehensive resource utilization. However, their commonality lies in providing extensive support for cutting-edge green technologies from technological breakthroughs to market introduction through systematic policy design, thus accelerating their progression from the laboratory to industrialization.
Global policies universally endorse low-carbon technologies, yet specific provisions, including fiscal and tax incentives and integration with carbon markets that target syngas fermentation, have not been clarified. The primary barriers to the current promotion of syngas fermentation are the lack of detailed policy guidelines, high technological costs, and inadequate collaboration along the industrial chain. To address these challenges, efforts should focus on special incentives, standard formulation, demonstration projects, and international cooperation.
5. Challenges and Outlook
Although syngas fermentation technology offers advantages across multiple areas, such as carbon neutrality and process stability, it still needs breakthroughs in the following areas to support its commercialization.
The future development of industrial strains will center on the in-depth integration of the Design-Build-Test-Learn (DBTL) cycle: genome-scale metabolic models and machine learning are leveraged to predict design targets, with automated platforms combined to enable rapid construction of strain libraries; microreactor arrays coupled with gas consumption and product generation detection are developed to realize high-throughput phenotypic screening under actual fermentation conditions; and multi-omics analyses including transcriptomics, proteomics and metabolomics are applied to analyze microbial physiological responses, thereby guiding the next round of precise strain design. With the maturation of high-throughput screening technology for gaseous substrates, the automation and standardization of strain construction and testing will be gradually realized, thereby generating high-quality and large-scale datasets. These datasets support complex experimental design, facilitate close, iterative feedback between machine learning models and strain testing, and accelerate the research and development of syngas fermentation strains.
At the core technical level, low gas mass transfer efficiency constitutes a technical bottleneck that severely restricts production intensity. There is an urgent need to develop novel and high-efficiency reactors while reducing energy consumption associated with agitation, mixing, and other processes. Additionally, the issues of feed gas quality and high purification costs cannot be overlooked. Syngas derived from industrial exhaust gas or biomass gasification exhibits significant compositional fluctuations and contains impurities. Although deep purification can mitigate this issue, it substantially increases capital and operating costs. In the future, priority should be given to developing more tolerant engineered strains to reduce gas-purity requirements, while optimizing low-cost, high-efficiency online gas purification technologies to lower energy consumption and pretreatment costs.
In terms of engineering and operations, high energy consumption in downstream separation is a core challenge. The ethanol concentration in fermentation broth is usually low, resulting in high energy consumption during product separation, concentration, and purification. The development of in-situ product separation technology to continuously remove ethanol during fermentation can not only alleviate product inhibition but also reduce downstream separation load. Furthermore, the difficulties in scale-up and integration optimization urgently need to be addressed. Moreover, integrating and matching the energy requirements of multiple units, including purification, fermentation, and separation, is complex. It is necessary to develop more accurate scale-up models and process simulation tools, and to promote modular, standardized plant design to reduce investment risks and shorten the construction cycle.
In the economic and market domain, weak cost competitiveness constitutes the primary obstacle to commercialization. The large initial investment scale and persistently high operating costs render syngas-to-ethanol less competitive under the current price structure for fossil fuels and traditional bioethanol. It is imperative to reduce whole-process costs through technological breakthroughs while exploring business models for the co-production of high-value products, such as long-chain alcohols and microbial proteins, to enhance overall economic viability. Additionally, inadequate policy and market mechanisms restrict industrial development, and the carbon-emission-reduction value of syngas-based ethanol has not been fully realized through carbon markets. There is a need to establish a carbon credit mechanism to monetize environmental benefits.
At the industrial chain and infrastructure construction level, the instability of the raw material supply chain is particularly prominent. Reliance on steel plant exhaust gas is constrained by geography and industry cycles, while biomass raw materials face high collection, transportation, and storage costs, as well as scattered supply. In the future, it is necessary to develop small- to medium-sized, modular production models adapted to distributed raw materials such as agricultural waste and municipal solid waste, while establishing long-term, stable cooperative relationships with large-scale industrial emission sources.
Under the guidance of the “dual carbon” goals, carbon capture and utilization technology serves as a core pillar for building a low-carbon energy and material system. Syngas fermentation, as a unique biocatalytic pathway, acts as a “flexible and efficient carbon molecular hub”. It forms strong complementary relationships with diverse carbon utilization technologies, jointly building a resilient carbon-neutrality solution.
Syngas fermentation exhibits significant complementary and synergistic relationships with other CCUS carbon utilization technologies. Traditional CCUS focuses on CO2 geological storage and emission reduction. Syngas fermentation can share its capture and transportation infrastructure, create value through carbon utilization, and reduce storage costs, forming economic complementarity. CO2 thermocatalytic/electrocatalytic conversion requires harsh conditions and has high reaction rates but limited product types. It complements syngas fermentation in technical routes, as the latter offers mild conditions, strong impurity tolerance, and a diverse product range. Biomethanation specifically produces CH4 suitable for natural gas pipelines. It can flexibly adjust the output of energy and chemical products through syngas fermentation to meet end-user needs, thereby achieving product-matrix complementarity. Microalgal carbon sequestration relies on light, covers large areas, and has a long growth cycle. It complements syngas fermentation in terms of temporal and spatial efficiency, as syngas fermentation enables all-weather industrial production with higher volumetric productivity and adapts to different application scenarios.
Syngas fermentation is not intended to replace other carbon utilization pathways; instead, it serves as a key, flexible node in the carbon cycle network. Leveraging its bioconversion advantages, it converts waste carbon sources into green products. It forms a synergistic pattern in which storage secures the bottom line, and utilization generates economic value with storage, thermocatalysis, and other technologies. Its core positioning lies in enhancing the economic viability and industrial attractiveness of the CCUS system, serving as an essential engine for deep industrial decarbonization and the cultivation of emerging green industries.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this manuscript, the authors used DeepSeek to improve the grammar and syntax. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the published article.
Author Contributions
Investigation, X.L.; Writing—Original Draft Preparation, N.L.; Writing—Review & Editing, X.B.; Project Administration, M.Y. and R.W.; Funding Acquisition, W.C.
Ethics Statement
Not applicable for studies not involving humans or animals.
Informed Consent Statement
Not applicable for studies not involving humans.
Data Availability Statement
As this article is a review paper, no original research data were generated. All information and data cited in this study were obtained from the published literature.
Funding
This work was supported by the National Key Research and Development Program of China (2024YFA0918100).
Declaration of Competing Interest
The authors declare no conflicts of interest. The authors are employees of Beijing Shougang LangzaTech Technology Co., Ltd; however, the company had no role in the study design, data collection, data analysis, decision to publish, or preparation of the manuscript.
References
-
Razak AAA, Saaid IM, Yusof MAMd, Husein N, Zaidin MF, Sabil KM. Physical and chemical effect of impurities in carbon capture, utilisation and storage. J. Pet. Explor. Prod. Technol. 2023, 13, 1235–1246. DOI:10.1007/s13202-023-01616-3 [Google Scholar]
-
Mathew MD. Nuclear energy: A pathway towards mitigation of global warming. Prog. Nucl. Energy 2022, 143, 104080. DOI:10.1016/j.pnucene.2021.104080 [Google Scholar]
-
Shi S, Hu YH. 2024, A landmark year for climate change and global carbon capture, utilization, and storage: Annual progress review. Energy Sci. Eng. 2025, 13, 3448–3458. DOI:10.1002/ese3.70091 [Google Scholar]
-
Sun X, Atiyeh HK, Huhnke RL, Tanner RS. Syngas fermentation process development for production of biofuels and chemicals: A review. Bioresour. Technol. Rep. 2019, 7, 100279. DOI:10.1016/j.biteb.2019.100279 [Google Scholar]
-
Gunes B. A critical review on biofilm-based reactor systems for enhanced syngas fermentation processes. Renew Sustain. Energy Rev. 2021, 143, 110950. DOI:10.1016/j.rser.2021.110950 [Google Scholar]
-
Benevenuti C, Amaral P, Ferreira T, Seidl P. Impacts of syngas composition on anaerobic fermentation. Reactions 2021, 2, 391–407. DOI:10.3390/reactions2040025 [Google Scholar]
-
Gao Y, Gao X, Zhang X. The 2 °C global temperature target and the evolution of the long-term goal of addressing climate change—from the United Nations Framework Convention on Climate Change to the Paris Agreement. Engineering 2017, 3, 272–278. DOI:10.1016/j.eng.2017.01.022 [Google Scholar]
-
Liu X, Wang X, Meng X. Carbon emission scenario prediction and peak path selection in China. Energies 2023, 16, 2276. DOI:10.3390/en16052276 [Google Scholar]
-
Chu H, Huang Z, Zhang Z, Yan X, Qiu B, Xu N. Integration of carbon emission reduction policies and technologies: Research progress on carbon capture, utilization and storage technologies. Sep. Purif. Technol. 2024, 343, 127153. DOI:10.1016/j.seppur.2024.127153 [Google Scholar]
-
Hanson E, Nwakile C, Hammed VO. Carbon capture, utilization, and storage (CCUS) technologies: Evaluating the effectiveness of advanced CCUS solutions for reducing CO2 emissions. Results Surf. Interfaces 2025, 18, 100381. DOI:10.1016/j.rsurfi.2024.100381 [Google Scholar]
-
Mollahosseini A, Dafchahi MN, Salestan SK, Chew JW, Mozafari M, Soroush M, et al. Polymeric membranes in carbon capture, utilization, and storage: current trends and future directions in decarbonization of industrial flue gas and climate change mitigation. Energy Environ. Sci. 2025, 18, 5025–5092. DOI:10.1039/d4ee05328a [Google Scholar]
-
Davoodi S, Al-Shargabi M, Wood DA, Rukavishnikov VS, Minaev KM. Review of technological progress in carbon dioxide capture, storage, and utilization. Gas Sci. Eng. 2023, 117, 205070. DOI:10.1016/j.jgsce.2023.205070 [Google Scholar]
-
Liang B, Chen C, Jia C, Wang C, Wang X, Zha Y, et al. Carbon capture, utilization and storage (CCUS) in oil and gas reservoirs in China: Status, opportunities and challenges. Fuel 2024, 375, 132353. DOI:10.1016/j.fuel.2024.132353 [Google Scholar]
-
Yaïci W, Entchev E, Longo M. Recent advances in small-scale carbon capture systems for micro-combined heat and power applications. Energies 2022, 15, 2938. DOI:10.3390/en15082938 [Google Scholar]
-
Plaza MG, Ribeiro RPPL. Special issue “CO2 capture and renewable energy.” Energies 2022, 15, 5187. DOI:10.3390/en15145187 [Google Scholar]
-
Devi NB, Pugazhenthi G, Pakshirajan K. Synthetic biology approaches and bioseparations in syngas fermentation. Trends Biotechnol. 2025, 43, 111–130. DOI:10.1016/j.tibtech.2024.07.008 [Google Scholar]
-
Comley JG, Scott JA, Laamanen CA. Utilizing CO2 in industrial off-gas for microalgae cultivation: considerations and solutions. Crit. Rev. Biotechnol. 2024, 44, 910–923. DOI:10.1080/07388551.2023.2233692 [Google Scholar]
-
Fackler N, Heijstra BD, Rasor BJ, Brown H, Martin J, Ni Z, et al. Stepping on the gas to a circular economy: accelerating development of carbon-negative chemical production from gas Fermentation. Annu. Rev. Chem. Biomol. 2021, 12, 1–32. DOI:10.1146/annurev-chembioeng-120120-021122 [Google Scholar]
-
Dowaidar M. Synthetic biology of metabolic cycles for enhanced CO2 capture and sequestration. Bioorg. Chem. 2024, 153, 107774. DOI:10.1016/j.bioorg.2024.107774 [Google Scholar]
-
Calvo DC, Luna HJ, Arango JA, Torres CI, Rittmann BE. Determining global trends in syngas fermentation research through a bibliometric analysis. J. Environ. Manag. 2022, 307, 114522. DOI:10.1016/j.jenvman.2022.114522 [Google Scholar]
-
Udemezue V, Shaikh KM, Vorontsova M, Valgepea K. Optimization of plasmid curing from genetically engineered Clostridium autoethanogenum. ACS Synth. Biol. 2025, 14, 4967–4972. DOI:10.1021/acssynbio.5c00456 [Google Scholar]
-
Lemaire ON, Belhamri M, Shevchenko A, Wagner T. Carbon-monoxide-driven bioethanol production operates through a tungsten-dependent catalyst. Nat. Chem. Biol. 2026, 22, 28–36. DOI:10.1038/s41589-025-02055-3 [Google Scholar]
-
Ragsdale SW, Pierce E. Acetogenesis and the Wood-Ljungdahl pathway of CO(2) fixation. Biochim. Biophys. Acta 2008, 1784, 1873–1898. DOI:10.1016/j.bbapap.2008.08.012 [Google Scholar]
-
Zhang J-Z, Li Y-Z, Xi Z-N, Gao H-P, Zhang Q, Liu L-C, et al. Engineered acetogenic bacteria as microbial cell factory for diversified biochemicals. Front. Bioeng. Biotechnol. 2024, 12, 1395540. DOI:10.3389/fbioe.2024.1395540 [Google Scholar]
-
Lauer I, Philipps G, Jennewein S. Metabolic engineering of Clostridium ljungdahlii for the production of hexanol and butanol from CO2 and H2. Microb. Cell Factories 2022, 21, 85. DOI:10.1186/s12934-022-01802-8 [Google Scholar]
-
Ghadermazi P, Re A, Ricci L, Chan SHJ. Metabolic engineering interventions for sustainable 2,3-butanediol production in gas-fermenting Clostridium autoethanogenum. mSystems 2022, 7, e01111-21. DOI:10.1128/msystems.01111-21 [Google Scholar]
-
Lanzillo F, Ruggiero G, Raganati F, Russo ME, Marzocchella A. Batch syngas fermentation by Clostridium carboxidivorans for production of acids and alcohols. Processes 2020, 8, 1075. DOI:10.3390/pr8091075 [Google Scholar]
-
Beneroso D, Bermúdez JM, Arenillas A, Menéndez JA. Comparing the composition of the synthesis-gas obtained from the pyrolysis of different organic residues for a potential use in the synthesis of bioplastics. J. Anal. Appl. Pyrolysis 2015, 111, 55–63. DOI:10.1016/j.jaap.2014.12.011 [Google Scholar]
-
Stoll IK, Boukis N, Sauer J. Syngas fermentation to alcohols: reactor technology and application perspective. Chem. Ing. Tech. 2020, 92, 125–136. DOI:10.1002/cite.201900118 [Google Scholar]
-
Latif H, Zeidan AA, Nielsen AT, Zengler K. Trash to treasure: Production of biofuels and commodity chemicals via syngas fermenting microorganisms. Curr. Opin. Biotechnol. 2014, 27, 79–87. DOI:10.1016/j.copbio.2013.12.001 [Google Scholar]
-
Munasinghe PC, Khanal SK. Biomass-derived syngas fermentation into biofuels: Opportunities and challenges. Bioresour. Technol. 2010, 101, 5013–5022. DOI:10.1016/j.biortech.2009.12.098 [Google Scholar]
-
Kumar T, J SE. Review and perspectives of emerging green technology for the sequestration of carbon dioxide into value-added Products: An intensifying development. Energy Fuels 2023, 37, 3570–3589. DOI:10.1021/acs.energyfuels.2c04122 [Google Scholar]
-
Owoade A, Alshami AS, Levin D, Onaizi S, Malaibari ZO. Progress and development of syngas fermentation processes toward commercial bioethanol production. Biofuels Bioprod. Biorefin. 2023, 17, 1328–1342. DOI:10.1002/bbb.2481 [Google Scholar]
-
You M, Zhao Q, Liu Y, Zhang W, Shen Z, Ren Z, et al. Insights into lignocellulose degradation: comparative genomics of anaerobic and cellulolytic Ruminiclostridium-type species. Front. Microbiol. 2023, 14, 1288286. DOI:10.3389/fmicb.2023.1288286 [Google Scholar]
-
Stark C, Münßinger S, Rosenau F, Eikmanns BJ, Schwentner A. The potential of sequential fermentations in converting C1 substrates to higher-value products. Front. Microbiol. 2022, 13, 907577. DOI:10.3389/fmicb.2022.907577 [Google Scholar]
-
Tamaru Y, Miyake H, Kuroda K, Nakanishi A, Matsushima C, Doi RH, et al. Comparison of the mesophilic cellulosome‐producing Clostridium cellulovorans genome with other cellulosome‐related clostridial genomes. Microb. Biotechnol. 2011, 4, 64–73. DOI:10.1111/j.1751-7915.2010.00210.x [Google Scholar]
-
Fernández‐Blanco C, Robles‐Iglesias R, Naveira‐Pazos C, Veiga MC, Kennes C. Production of biofuels from C1‐gases with Clostridium and related bacteria—Recent advances. Microb. Biotechnol. 2023, 16, 726–741. DOI:10.1111/1751-7915.14220 [Google Scholar]
-
Piatek P, Humphreys C, Raut MP, Wright PC, Simpson S, Köpke M, et al. Agr quorum sensing influences the Wood-Ljungdahl pathway in Clostridium autoethanogenum. Sci. Rep. 2022, 12, 411. DOI:10.1038/s41598-021-03999-x [Google Scholar]
-
Abubackar HN, Veiga MC, Kennes C. Biological conversion of carbon monoxide: rich syngas or waste gases to bioethanol. Biofuels Bioprod. Biorefin. 2011, 5, 93–114. DOI:10.1002/bbb.256 [Google Scholar]
-
Harahap BM, Ahring BK. Acetate production from syngas produced from lignocellulosic biomass materials along with gaseous fermentation of the syngas: A review. Microorganisms 2023, 11, 995. DOI:10.3390/microorganisms11040995 [Google Scholar]
-
Schwarz FM, Ciurus S, Jain S, Baum C, Wiechmann A, Basen M, et al. Revealing formate production from carbon monoxide in wild type and mutants of Rnf‐ and Ech‐containing acetogens, Acetobacterium woodii and Thermoanaerobacter kivui. Microb. Biotechnol. 2020, 13, 2044–2056. DOI:10.1111/1751-7915.13663 [Google Scholar]
-
Okolie JA, Sajeev E, Shekher S, Desongu KS, Ogbaga CC, Gunes B. Application of Magnetic Nanoparticles in Bioreactors to Enhance Mass Transfer during Syngas Fermentation. 2022. Available online:https://www.preprints.org/manuscript/202208.0415 (accessed on 3 February 2026).
-
Müller B, Sun L, Schnürer A. First insights into the syntrophic acetate‐oxidizing bacteria – a genetic study. MicrobiologyOpen 2013, 2, 35–53. DOI:10.1002/mbo3.50 [Google Scholar]
-
Adam PS, Borrel G, Gribaldo S. An archaeal origin of the Wood–Ljungdahl H4MPT branch and the emergence of bacterial methylotrophy. Nat. Microbiol. 2019, 4, 2155–2163. DOI:10.1038/s41564-019-0534-2 [Google Scholar]
-
Maynard EL, Lindahl PA. Evidence of a molecular tunnel connecting the active sites for CO2 reduction and acetyl-coa synthesis in acetyl-coa synthase from Clostridium thermoaceticum. J. Am. Chem. Soc. 1999, 121, 9221–9222. DOI:10.1021/ja992120g [Google Scholar]
-
Toshchakov SV, Lebedinsky AV, Sokolova TG, Zavarzina DG, Korzhenkov AA, Teplyuk AV, et al. Genomic insights into energy metabolism of Carboxydocella thermautotrophica coupling hydrogenogenic CO oxidation with the reduction of Fe(III) minerals. Front. Microbiol. 2018, 9, 1759. DOI:10.3389/fmicb.2018.01759 [Google Scholar]
-
Liew F, Henstra AM, Winzer K, Köpke M, Simpson SD, Minton NP. Insights into CO2 fixation pathway of Clostridium autoethanogenum by targeted mutagenesis. Mbio 2016, 7, e00427-16. DOI:10.1128/mbio.00427-16 [Google Scholar]
-
Kim SM, Lee J, Kang SH, Heo Y, Yoon H-J, Hahn J-S, et al. O2-tolerant CO dehydrogenase via tunnel redesign for the removal of CO from industrial flue gas. Nat. Catal. 2022, 5, 807–817. DOI:10.1038/s41929-022-00834-y [Google Scholar]
-
Liu W, Zhang K, Liu J, Wang Y, Zhang M, Cui H, et al. Bioelectrocatalytic carbon dioxide reduction by an engineered formate dehydrogenase from Thermoanaerobacter kivui. Nat. Commun. 2024, 15, 9962. DOI:10.1038/s41467-024-53946-3 [Google Scholar]
-
Wan S, Wang H, Ma X, Tan Y, Liu L, Li F. Genetic modification of acetogens and optimization of fermentation process in C1-gas bioconversion. Sheng Wu Gong Cheng Xue Bao Chin. J. Biotechnol. 2023, 39, 2410–2429. DOI:10.13345/j.cjb.221004 [Google Scholar]
-
Liew F, Henstra AM, Kӧpke M, Winzer K, Simpson SD, Minton NP. Metabolic engineering of Clostridium autoethanogenum for selective alcohol production. Metab. Eng. 2017, 40, 104–114. DOI:10.1016/j.ymben.2017.01.007 [Google Scholar]
-
Ingelman H, Heffernan JK, Harris A, Brown SD, Shaikh KM, Saqib AY, et al. Autotrophic adaptive laboratory evolution of the acetogen Clostridium autoethanogenum delivers the gas-fermenting strain LAbrini with superior growth, products, and robustness. N. Biotechnol. 2024, 83, 1–15. DOI:10.1016/j.nbt.2024.06.002 [Google Scholar]
-
Humphreys CM, Minton NP. Advances in metabolic engineering in the microbial production of fuels and chemicals from C1 gas. Curr. Opin. Biotech. 2018, 50, 174–181. DOI:10.1016/j.copbio.2017.12.023 [Google Scholar]
-
Jin S, Bae J, Song Y, Pearcy N, Shin J, Kang S, et al. Synthetic biology on acetogenic bacteria for highly efficient conversion of C1 gases to biochemicals. Int. J. Mol. Sci. 2020, 21, 7639. DOI:10.3390/ijms21207639 [Google Scholar]
-
Nagaraju S, Davies NK, Walker DJF, Köpke M, Simpson SD. Genome editing of Clostridium autoethanogenum using CRISPR/Cas9. Biotechnol. Biofuels 2016, 9, 219. DOI:10.1186/s13068-016-0638-3 [Google Scholar]
-
Fackler N, Heffernan J, Juminaga A, Doser D, Nagaraju S, Gonzalez-Garcia RA, et al. Transcriptional control of Clostridium autoethanogenum using CRISPRi. Synth. Biol. 2021, 6, ysab008. DOI:10.1093/synbio/ysab008 [Google Scholar]
-
Poulalier-Delavelle M, Baker JP, Millard J, Winzer K, Minton NP. Endogenous CRISPR/Cas systems for genome engineering in the acetogens Acetobacterium woodii and Clostridium autoethanogenum. Front. Bioeng. Biotechnol. 2023, 11, 1213236. DOI:10.3389/fbioe.2023.1213236 [Google Scholar]
-
Charubin K, Streett H, Papoutsakis ET. Development of strong anaerobic fluorescent reporters for Clostridium acetobutylicum and Clostridium ljungdahlii using HaloTag and SNAP-tag Proteins. Appl. Environ. Microbiol. 2020, 86, e01271-20. DOI:10.1128/aem.01271-20 [Google Scholar]
-
Streett HE, Kalis KM, Papoutsakis ET. A strongly fluorescing anaerobic reporter and protein-tagging system for Clostridium organisms based on the Fluorescence-Activating and Absorption-Shifting Tag Protein (FAST). Appl. Environ. Microbiol. 2019, 85, e00622-19. DOI:10.1128/aem.00622-19 [Google Scholar]
-
Lo J, Humphreys JR, Jack J, Urban C, Magnusson L, Xiong W, et al. The metabolism of Clostridium ljungdahlii in phosphotransacetylase negative strains and development of an ethanologenic strain. Front. Bioeng. Biotechnol. 2020, 8, 560726. DOI:10.3389/fbioe.2020.560726 [Google Scholar]
-
Liu Y, Zhang Z, Jiang W, Gu Y. Protein acetylation-mediated cross regulation of acetic acid and ethanol synthesis in the gas-fermenting Clostridium ljungdahlii. J. Biol. Chem. 2022, 298, 101538. DOI:10.1016/j.jbc.2021.101538 [Google Scholar]
-
Nogle R, Nagaraju S, Utturkar SM, Giannone RJ, Reynoso V, Leang C, et al. Clostridium autoethanogenum isopropanol production via native plasmid pCA replicon. Front. Bioeng. Biotechnol. 2022, 10, 932363. DOI:10.3389/fbioe.2022.932363 [Google Scholar]
-
Dykstra JC, Oort J van, Yazdi AT, Vossen E, Patinios C, Oost J van der, et al. Metabolic engineering of Clostridium autoethanogenum for ethyl acetate production from CO. Microb. Cell Factories 2022, 21, 243. DOI:10.1186/s12934-022-01964-5 [Google Scholar]
-
Liew F, Martin ME, Tappel RC, Heijstra BD, Mihalcea C, Köpke M. Gas fermentation—A flexible platform for commercial scale production of low-carbon-fuels and chemicals from waste and renewable feedstocks. Front. Microbiol. 2016, 7, 694. DOI:10.3389/fmicb.2016.00694 [Google Scholar]
-
Zhang H, Yang P, Wang Z, Li M, Zhang J, Liu D, et al. Clostridium acetobutylicum biofilm: Advances in understanding the basis. Front. Bioeng. Biotechnol. 2021, 9, 658568. DOI:10.3389/fbioe.2021.658568 [Google Scholar]
-
Harris LM, Welker NE, Papoutsakis ET. Northern, morphological, and fermentation analysis of spo0a inactivation and overexpression in Clostridium acetobutylicum ATCC 824. J. Bacteriol. 2002, 184, 3586–3597. DOI:10.1128/jb.184.13.3586-3597.2002 [Google Scholar]
-
Li Z-H, Li S, Chen H-B, Ji Y-Z, Zhang C. The advances of strategies and technologies in high-throughput microbial culturomics. Biotechnol. Adv. 2026, 87, 108783. DOI:10.1016/j.biotechadv.2025.108783 [Google Scholar]
-
Yan W, Li X, Zhao D, Xie M, Li T, Qian L, et al. Advanced strategies in high-throughput droplet screening for enzyme engineering. Biosens. Bioelectron. 2024, 248, 115972. DOI:10.1016/j.bios.2023.115972 [Google Scholar]
-
Longwell CK, Labanieh L, Cochran JR. High-throughput screening technologies for enzyme engineering. Curr. Opin. Biotechnol. 2017, 48, 196–202. DOI:10.1016/j.copbio.2017.05.012 [Google Scholar]
-
Kwon KK, Lee J, Kim H, Lee D-H, Lee S-G. Advancing high-throughput screening systems for synthetic biology and biofoundry. Curr. Opin. Syst. Biol. 2024, 37, 100487. DOI:10.1016/j.coisb.2023.100487 [Google Scholar]
-
Agu CV, Lai SM, Ujor V, Biswas PK, Jones A, Gopalan V, et al. Development of a high-throughput assay for rapid screening of butanologenic strains. Sci. Rep. 2018, 8, 3379. DOI:10.1038/s41598-017-18074-7 [Google Scholar]
-
Candry P, Daele TV, Denis K, Amerlinck Y, Andersen SJ, Ganigué R, et al. A novel high-throughput method for kinetic characterisation of anaerobic bioproduction strains, applied to Clostridium kluyveri. Sci. Rep. 2018, 8, 9724. DOI:10.1038/s41598-018-27594-9 [Google Scholar]
-
Sundstrom ER, Criddle CS. Optimization of methanotrophic growth and production of Poly(3-Hydroxybutyrate) in a high-throughput microbioreactor system. Appl. Environ. Microbiol. 2015, 81, 4767–4773. DOI:10.1128/aem.00025-15 [Google Scholar]
-
Köpke M, Simpson SD. Pollution to products: recycling of ‘above ground’ carbon by gas fermentation. Curr. Opin. Biotech. 2020, 65, 180–189. DOI:10.1016/j.copbio.2020.02.017 [Google Scholar]
-
Payne EM, Holland-Moritz DA, Sun S, Kennedy RT. High-throughput screening by droplet microfluidics: perspective into key challenges and future prospects. Lab a Chip 2020, 20, 2247–2262. DOI:10.1039/d0lc00347f [Google Scholar]
-
Chao R, Mishra S, Si T, Zhao H. Engineering biological systems using automated biofoundries. Metab. Eng. 2017, 42, 98–108. DOI:10.1016/j.ymben.2017.06.003 [Google Scholar]
-
Hecht A, Filliben J, Munro SA, Salit M. A minimum information standard for reproducing bench-scale bacterial cell growth and productivity. Commun. Biol. 2018, 1, 219. DOI:10.1038/s42003-018-0220-6 [Google Scholar]
-
Figueras J, Benbelkacem H, Dumas C, Buffiere P. Inhibitions from syngas impurities: Impact of common tar components on a consortium adapted for syngas biomethanation. Waste Biomass Valorization 2024, 15, 2427–2437. DOI:10.1007/s12649-023-02237-x [Google Scholar]
-
Ellacuriaga M, Gil MV, Gómez X. Syngas fermentation: Cleaning of syngas as a critical stage in fermentation performance. Fermentation 2023, 9, 898. DOI:10.3390/fermentation9100898 [Google Scholar]
-
Ramachandriya KD, Kundiyana DK, Sharma AM, Kumar A, Atiyeh HK, Huhnke RL, et al. Critical factors affecting the integration of biomass gasification and syngas fermentation technology. AIMS Bioeng. 2016, 3, 188–210. DOI:10.3934/bioeng.2016.2.188 [Google Scholar]
-
Laurence LC, Ashenafi D. Syngas treatment unit for small scale gasification—Application to IC engine gas quality requirement. J. Appl. Fluid Mech. 2012, 5, 95–103. DOI:10.36884/jafm.5.01.11963 [Google Scholar]
-
Xiao Z, Liu Y, Pan J, Wang Y. Review of the adsorption of sulfur-containing gaseous pollutants by biochar: Progress, challenges, and perspectives. Energy Fuels 2025, 39, 4151–4176. DOI:10.1021/acs.energyfuels.4c06274 [Google Scholar]
-
Jian H, Haobo G, Liyu G, Hao S, Yuling L, Lei Z. Formulated absorbent for fine desulfurization and carbon capture of blast furnace gas based on alcohol amine solution. Environ. Eng. Res. 2025, 30, 240648. DOI:10.4491/eer.2024.648 [Google Scholar]
-
Safarian S, Unnthorsson R, Richter C. Bioethanol production via herbaceous and agricultural biomass gasification integrated with syngas fermentation. Fermentation 2021, 7, 139. DOI:10.3390/fermentation7030139 [Google Scholar]
-
Piatek P, Olsson L, Nygård Y. Adaptation during propagation improves Clostridium autoethanogenum tolerance towards benzene, toluene and xylenes during gas fermentation. Bioresour. Technol. Rep. 2020, 12, 100564. DOI:10.1016/j.biteb.2020.100564 [Google Scholar]
-
Oswald F, Zwick M, Omar O, Hotz EN, Neumann A. Growth and product formation of Clostridium ljungdahlii in presence of cyanide. Front. Microbiol. 2018, 9, 1213. DOI:10.3389/fmicb.2018.01213 [Google Scholar]
-
Gao S, Liao Y, He H, Yang H, Yang X, Xu S, et al. Advance of tolerance engineering on microbes for industrial production. Synth. Syst. Biotechnol. 2023, 8, 697–707. DOI:10.1016/j.synbio.2023.10.004 [Google Scholar]
-
Ahuja V, Bhatt AK, Ravindran B, Yang Y-H, Bhatia SK. A mini-review on syngas fermentation to bio-alcohols: Current status and challenges. Sustainability 2023, 15, 3765. DOI:10.3390/su15043765 [Google Scholar]
-
Enriquez FA, Ahring BK. Strategies to overcome mass transfer limitations of hydrogen during anaerobic gaseous fermentations: A comprehensive review. Bioresour. Technol. 2023, 377, 128948. DOI:10.1016/j.biortech.2023.128948 [Google Scholar]
-
Puiman L, Elisiário MP, Crasborn LML, Wagenaar LECH, Straathof AJJ, Haringa C. Gas mass transfer in syngas fermentation broths is enhanced by ethanol. Biochem. Eng. J. 2022, 185, 108505. DOI:10.1016/j.bej.2022.108505 [Google Scholar]
-
Rittmann SK-MR, Lee HS, Lim JK, Kim TW, Lee J-H, Kang SG. One-carbon substrate-based biohydrogen production: Microbes, mechanism, and productivity. Biotechnol. Adv. 2015, 33, 165–177. DOI:10.1016/j.biotechadv.2014.11.004 [Google Scholar]
-
Klasson KT, Ackerson MD, Clausen EC, Gaddy JL. Bioreactors for synthesis gas fermentations. Resour. Conserv. Recycl. 1991, 5, 145–165. DOI:10.1016/0921-3449(91)90022-g [Google Scholar]
-
Fadavi A, Chisti Y. Gas–liquid mass transfer in a novel forced circulation loop reactor. Chem. Eng. J. 2005, 112, 73–80. DOI:10.1016/j.cej.2005.06.009 [Google Scholar]
-
Drzyzga O, Revelles O, Durante‐Rodríguez G, Díaz E, García JL, Prieto A. New challenges for syngas fermentation: towards production of biopolymers. J. Chem. Technol. Biotechnol. 2015, 90, 1735–1751. DOI:10.1002/jctb.4721 [Google Scholar]
-
Daniell J, Köpke M, Simpson S. Commercial biomass syngas fermentation. Energies 2012, 5, 5372–5417. DOI:10.3390/en5125372 [Google Scholar]
-
Ungerman AJ, Heindel TJ. Carbon monoxide mass transfer for syngas fermentation in a stirred tank reactor with dual impeller configurations. Biotechnol. Prog. 2007, 23, 613–620. DOI:10.1021/bp060311z [Google Scholar]
-
Shen Y, Brown R, Wen Z. Enhancing mass transfer and ethanol production in syngas fermentation of Clostridium carboxidivorans P7 through a monolithic biofilm reactor. Appl. Energy 2014, 136, 68–76. DOI:10.1016/j.apenergy.2014.08.117 [Google Scholar]
-
Jang N, Lee M, Yasin M, Chang IS. Behavior of CO-water mass transfer coefficient in membrane sparger-integrated bubble column for synthesis gas fermentation. Bioresour. Technol. 2020, 311, 123594. DOI:10.1016/j.biortech.2020.123594 [Google Scholar]
-
Yasin M, Park S, Jeong Y, Lee EY, Lee J, Chang IS. Effect of internal pressure and gas/liquid interface area on the CO mass transfer coefficient using hollow fibre membranes as a high mass transfer gas diffusing system for microbial syngas fermentation. Bioresour. Technol. 2014, 169, 637–643. DOI:10.1016/j.biortech.2014.07.026 [Google Scholar]
-
Lee P-H, Ni S-Q, Chang S-Y, Sung S, Kim S-H. Enhancement of carbon monoxide mass transfer using an innovative external hollow fiber membrane (HFM) diffuser for syngas fermentation: Experimental studies and model development. Chem. Eng. J. 2012, 184, 268–277. DOI:10.1016/j.cej.2011.11.103 [Google Scholar]
-
Shen Y, Brown R, Wen Z. Syngas fermentation of Clostridium carboxidivoran P7 in a hollow fiber membrane biofilm reactor: Evaluating the mass transfer coefficient and ethanol production performance. Biochem. Eng. J. 2014, 85, 21–29. DOI:10.1016/j.bej.2014.01.010 [Google Scholar]
-
Ahmed T, Semmens MJ. Use of sealed end hollow fibers for bubbleless membrane aeration: Experimental studies. J. Membr. Sci. 1992, 69, 1–10. DOI:10.1016/0376-7388(92)80162-d [Google Scholar]
-
Asimakopoulos K, Gavala HN, Skiadas IV. Reactor systems for syngas fermentation processes: A review. Chem. Eng. J. 2018, 348, 732–44. DOI:10.1016/j.cej.2018.05.003 [Google Scholar]
-
Riegler P, Chrusciel T, Mayer A, Doll K, Weuster-Botz D. Reversible retrofitting of a stirred-tank bioreactor for gas-lift operation to perform synthesis gas fermentation studies. Biochem. Eng. J. 2019, 141, :89–101. DOI:10.1016/j.bej.2018.09.021 [Google Scholar]
-
Orgill JJ, Atiyeh HK, Devarapalli M, Phillips JR, Lewis RS, Huhnke RL. A comparison of mass transfer coefficients between trickle-bed, hollow fiber membrane and stirred tank reactors. Bioresour. Technol. 2013, 133, 340–346. DOI:10.1016/j.biortech.2013.01.124 [Google Scholar]
-
Devarapalli M, Lewis RS, Atiyeh HK. Continuous ethanol production from synthesis gas by Clostridium ragsdalei in a trickle-bed reactor. Fermentation 2017, 3, 23. DOI:10.3390/fermentation3020023 [Google Scholar]
-
Dhakal N, Acharya B. Syngas fermentation for the production of bio-based polymers: A review. Polymers 2021, 13, 3917. DOI:10.3390/polym13223917 [Google Scholar]
-
Ayol A, Peixoto L, Keskin T, Abubackar HN. Reactor designs and configurations for biological and bioelectrochemical C1 gas conversion: A Review. Int. J. Environ. Res. Public Heal. 2021, 18, 11683. DOI:10.3390/ijerph182111683 [Google Scholar]
-
Asimakopoulos K, Kaufmann-Elfang M, Lundholm-Høffner C, Rasmussen NBK, Grimalt-Alemany A, Gavala HN, et al. Scale up study of a thermophilic trickle bed reactor performing syngas biomethanation. Appl. Energy 2021, 290, 116771. DOI:10.1016/j.apenergy.2021.116771 [Google Scholar]
-
Bredwell MD, Worden RM. Mass‐Transfer properties of microbubbles. 1. Experimental studies. Biotechnol. Prog. 1998, 14, 31–38. DOI:10.1021/bp970133x [Google Scholar]
-
Bredwell MD, Srivastava P, Worden RM. Reactor design issues for synthesis‐gas fermentations. Biotechnol. Prog. 1999, 15, 834–844. DOI:10.1021/bp990108m [Google Scholar]
-
Munasinghe PC, Khanal SK. Syngas fermentation to biofuel: Evaluation of carbon monoxide mass transfer coefficient (KLa) in different reactor configurations. Biotechnol. Prog. 2010, 26, 1616–1621. DOI:10.1002/btpr.473 [Google Scholar]
-
Besagni G, Inzoli F, Ziegenhein T. Two-phase bubble columns: A comprehensive review. ChemEngineering 2018, 2, 13. DOI:10.3390/chemengineering2020013 [Google Scholar]
-
Medeiros EM de, Noorman H, Filho RM, Posada JA. Production of ethanol fuel via syngas fermentation: Optimization of economic performance and energy efficiency. Chem. Eng. Sci. X 2020, 5, 100056. DOI:10.1016/j.cesx.2020.100056 [Google Scholar]
-
Takors R, Kopf M, Mampel J, Bluemke W, Blombach B, Eikmanns B, et al. Using gas mixtures of CO, CO2 and H2 as microbial substrates: the do’s and don’ts of successful technology transfer from laboratory to production scale. Microb. Biotechnol. 2018, 11, 606–625. DOI:10.1111/1751-7915.13270 [Google Scholar]
-
Henriques TM, Pereira SR, Serafim LS, Xavier AMRB. Two-stage aeration fermentation strategy to improve bioethanol production by Scheffersomyces stipitis. Fermentation 2018, 4, 97. DOI:10.3390/fermentation4040097 [Google Scholar]
-
Klasson KT, Ackerson MD, Clausen EC, Gaddy JL. Biological conversion of coal and coal-derived synthesis gas. Fuel 1993, 72, 1673–1678. DOI:10.1016/0016-2361(93)90354-5 [Google Scholar]
-
Richter H, Martin M, Angenent L. A two-stage continuous fermentation system for conversion of syngas into ethanol. Energies 2013, 6, 3987–4000. DOI:10.3390/en6083987 [Google Scholar]
-
Robles-Iglesias R, Naveira-Pazos C, Nicaud J-M, Veiga MC, Kennes C. Two-stage syngas fermentation into microbial oils and β-carotene with Clostridium carboxidivorans and engineered Yarrowia lipolytica. J CO2 Util. 2023, 76, 102593. DOI:10.1016/j.jcou.2023.102593 [Google Scholar]
-
Dong Y, Zhang Y, Liu D, Chen Z. Strain and process engineering toward continuous industrial fermentation. Front. Chem. Sci. Eng. 2023, 17, 1336–1353. DOI:10.1007/s11705-022-2284-6 [Google Scholar]
-
Bankar SB, Survase SA, Ojamo H, Granström T. Biobutanol: the outlook of an academic and industrialist. RSC Adv. 2013, 3, 24734–24757. DOI:10.1039/c3ra43011a [Google Scholar]
-
Diba F, Mokhtarani B, Panahi R. Dilution rate control for overproduction of rhamnolipid in continuous fermentation. Int. J. Environ. Sci. Technol. 2024, 21, 371–386. DOI:10.1007/s13762-023-05253-5 [Google Scholar]
-
Jochum T, Michalzik B, Bachmann A, Popp J, Frosch T. Microbial respiration and natural attenuation of benzene contaminated soils investigated by cavity enhanced Raman multi-gas spectroscopy. Analyst 2015, 140, 3143–3149. DOI:10.1039/c5an00091b [Google Scholar]
-
Dang J, Wang N, Atiyeh HK. Review of Dissolved CO and H2 measurement methods for syngas fermentation. Sensors 2021, 21, 2165. DOI:10.3390/s21062165 [Google Scholar]
-
Puiman L, Benalcázar EA, Picioreanu C, Noorman HJ, Haringa C. Downscaling industrial-scale syngas fermentation to simulate frequent and irregular dissolved gas concentration shocks. Bioengineering 2023, 10, 518. DOI:10.3390/bioengineering10050518 [Google Scholar]
-
Mohammadi M, Mohamed AR, Najafpour GD, Younesi H, Uzir MH. Kinetic Studies on fermentative production of biofuel from synthesis gas using Clostridium ljungdahlii. Sci. World J. 2014, 2014, 910590. DOI:10.1155/2014/910590 [Google Scholar]
-
Chen J, Gomez JA, Höffner K, Barton PI, Henson MA. Metabolic modeling of synthesis gas fermentation in bubble column reactors. Biotechnol. Biofuels 2015, 8, 89. DOI:10.1186/s13068-015-0272-5 [Google Scholar]
-
Eskandari M, Puiman L, Zeitler J. Multi-fidelity Bayesian optimisation of syngas fermentation simulators. arXiv 2023. DOI:10.48550/arxiv.2311.05776 [Google Scholar]
-
Nadal-Rey G, McClure DD, Kavanagh JM, Cornelissen S, Fletcher DF, Gernaey KV. Understanding gradients in industrial bioreactors. Biotechnol. Adv. 2021, 46, 107660. DOI:10.1016/j.biotechadv.2020.107660 [Google Scholar]
-
Lara AR, Galindo E, Ramírez OT, Palomares LA. Living with heterogeneities in bioreactors. Mol. Biotechnol. 2006, 34, 355–381. DOI:10.1385/mb:34:3:355 [Google Scholar]
-
Pigou M, Morchain J. Investigating the interactions between physical and biological heterogeneities in bioreactors using compartment, population balance and metabolic models. Chem. Eng. Sci. 2015, 126, 267–282. DOI:10.1016/j.ces.2014.11.035 [Google Scholar]
-
Liew FE, Nogle R, Abdalla T, Rasor BJ, Canter C, Jensen RO, et al. Carbon-negative production of acetone and isopropanol by gas fermentation at industrial pilot scale. Nat. Biotechnol. 2022, 40, 335–344. DOI:10.1038/s41587-021-01195-w [Google Scholar]
-
Delvigne F, Noorman H. Scale‐up/Scale‐down of microbial bioprocesses: a modern light on an old issue. Microb. Biotechnol. 2017, 10, 685–687. DOI:10.1111/1751-7915.12732 [Google Scholar]
-
Benalcázar EFA. Modeling the anaerobic fermentation of CO, H2 and CO2 mixtures at large and micro-scales. Doctoral Dissertation, Delft University of Technology, Faculty of Science in Life Science & Technology, Delft, The Netherlands, 2023. [Google Scholar]
-
Haringa C, Deshmukh AT, Mudde RF, Noorman HJ. Euler-Lagrange analysis towards representative down-scaling of a 22 m3 aerobic S. cerevisiae fermentation. Chem. Eng. Sci. 2017, 170, 653–669. DOI:10.1016/j.ces.2017.01.014 [Google Scholar]
-
Lapin A, Müller D, Reuss M. Dynamic behavior of microbial populations in stirred bioreactors simulated with Euler−Lagrange methods: Traveling along the lifelines of single cells. Ind. Eng. Chem. Res. 2004, 43, 4647–4656. DOI:10.1021/ie030786k [Google Scholar]
-
Lee H-J, Shen F, Eames A, Jedrychowski MP, Chandrasekaran S. Dynamic metabolic network modeling of a mammalian cell cycle using time-course multi-omics data. bioRxiv 2021, 2021.10.11.464012. DOI:10.1101/2021.10.11.464012 [Google Scholar]
-
Camphuijsen J, Rivera FJC, Potter G, Clark C, Chen J, Kahan S, et al. Modeling the biomechanics of cells on microcarriers in a stirred-tank bioreactor. bioRxiv 2022, 2022.08.31.505282. DOI:10.1101/2022.08.31.505282 [Google Scholar]
-
Wang X, Mohsin A, Sun Y, Li C, Zhuang Y, Wang G. From spatial-temporal multiscale modeling to application: Bridging the valley of death in industrial biotechnology. Bioengineering 2023, 10, 744. DOI:10.3390/bioengineering10060744 [Google Scholar]
-
Janković T, Straathof AJJ, McGregor IR, Kiss AA. Bioethanol separation by a new pass-through distillation process. Sep. Purif. Technol. 2024, 336, 126292. DOI:10.1016/j.seppur.2024.126292 [Google Scholar]
-
Zentou H, Abidin ZZ, Yunus R, Biak DRA, Korelskiy D. Overview of alternative ethanol removal techniques for enhancing bioethanol recovery from fermentation broth. Processes 2019, 7, 458. DOI:10.3390/pr7070458 [Google Scholar]
-
Huang T-Y, Ho JS, Goh S, Chong TH. Ethanol recovery from dilute aqueous solution by perstraction using supported ionic liquid membrane (SILM). J. Clean Prod. 2021, 298, 126811. DOI:10.1016/j.jclepro.2021.126811 [Google Scholar]
-
Redl S, Sukumara S, Ploeger T, Wu L, Jensen TØ, Nielsen AT, et al. Thermodynamics and economic feasibility of acetone production from syngas using the thermophilic production host Moorella thermoacetica. Biotechnol. Biofuels 2017, 10, 150. DOI:10.1186/s13068-017-0827-8 [Google Scholar]
-
Jean AB, Brown RC. Techno-economic analysis of gas fermentation for the production of single cell protein. Environ. Sci. Technol. 2024, 58, 3823–3829. DOI:10.1021/acs.est.3c10312 [Google Scholar]
-
Cheng M-H, Wang Z, Dien BS, Slininger PJW, Singh V. Economic analysis of cellulosic ethanol production from sugarcane bagasse using a sequential deacetylation, Hot Water and Disk-Refining Pretreatment. Processes 2019, 7, 642. DOI:10.3390/pr7100642 [Google Scholar]
-
Giwa AS, Sheng M, Maurice NJ, Liu X, Wang Z, Chang F, et al. Biofuel recovery from plantain and banana plant wastes: Integration of biochemical and thermochemical approach. J. Renew Mater. 2023, 11, 2593–2629. DOI:10.32604/jrm.2023.026314 [Google Scholar]
-
Anekwe IMS, Nyembe N, Nqakala LC, Madikizela M, Isa YM. Sustainable fuels: Lower alcohols perspective. Environ Prog. Sustain. Energy 2023, 42, e14175. DOI:10.1002/ep.14175 [Google Scholar]
-
Knittel C, Meiselman B, Stock J. The pass-through of RIN prices to wholesale and retail fuels under the renewable fuel standard. J. Assoc. Environ. Resour. Econ. 2015. DOI:10.3386/w21343 [Google Scholar]