Found 4 results
Open Access
Review
10 April 2026Hepatic Stellate Cells Interact with the Immune System: A Bidirectional Crosstalk Network Driving Liver Fibrosis
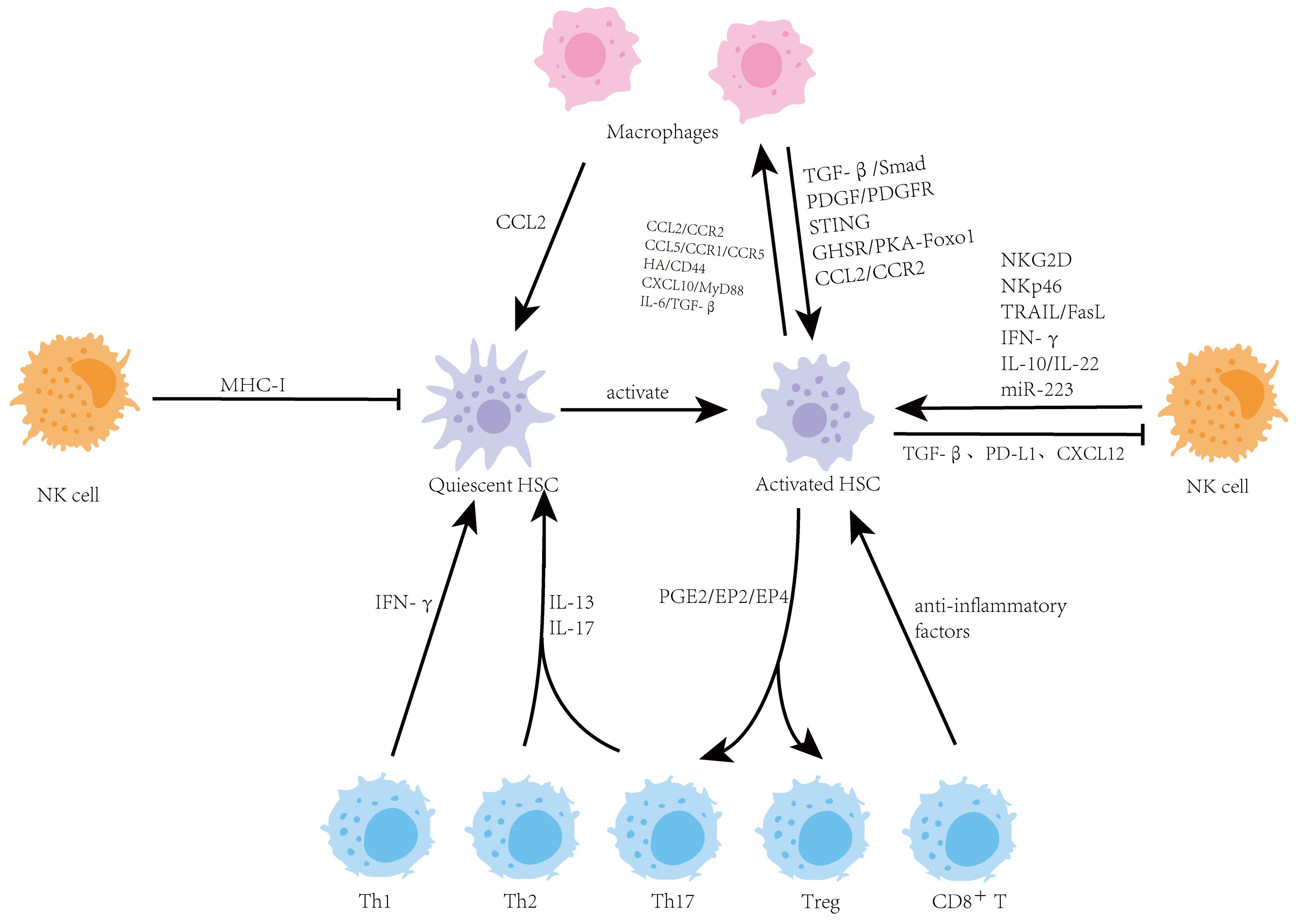
As a central metabolic and immune organ, the liver maintains a unique immune microenvironment which is crucial for sustaining health. When the immune balance in the liver is disrupted, it can drive the occurrence and progression of various chronic liver diseases, including liver fibrosis. Hepatic stellate cells (HSCs) are the key effector cells responsible for producing extracellular matrix (ECM) during liver fibrosis, and the hepatic immune microenvironment precisely regulates their activation. This review focuses on the complex bidirectional interaction network between HSCs and major immune cells in the liver, including macrophages, natural killer (NK) cells, and T cells. It systematically elucidates the central role of these interactions in maintaining hepatic homeostasis, mediating inflammatory responses, and driving the progression of fibrosis. A deeper understanding of the interaction between HSCs and immune cells is essential for elucidating the pathological mechanisms of liver fibrosis and will provide a theoretical basis for developing innovative therapeutic strategies targeting the immune microenvironment.
Open Access
Article
10 October 2025Immunoprofiling of Alcohol-Activated Hepatic Stellate Cells Reveals Mechanisms of Immune Evasion through NK/T Lymphocyte Checkpoint Signaling
Chronic alcohol consumption induces the pathogenic activation of hepatic stellate cells (HSC) and their conversion into proliferative myofibroblasts (Myo), which together constitute a disease hub in alcohol-associated liver disease (AALD). While natural killer (NK) lymphocytes efficiently target early activated HSC and ameliorate liver fibrosis in mouse models of diet- and alcohol-induced liver disease, late-activated HSC evade immune surveillance. To gain insight into evasive resistance mechanisms, we profiled the expression of immunoregulatory ligands by HSC and showed that HSC dynamically express CD80, a B7-family ligand that suppresses NK and T cell responses. Using a mouse model of acute-on-chronic alcohol consumption, we show that combined blockade of the CTLA-4//TIGIT/PD-1 inhibitory checkpoints overcomes this resistance mechanism, promoting the selective elimination of activated HSC (aHSC)/Myo, yet fails to diminish fibrosis or ameliorate liver function. Single-cell transcriptome profiling of liver non-parenchymal cells revealed that checkpoint blockade promotes hepatic infiltration of pro-fibrotic Th1 and Th17 T cell subpopulations, while decreasing immunosuppressive Treg. Strikingly, antibody-directed engagement of the PD-1 and TIGIT checkpoints also fails to reduce fibrosis or improve liver function. Thus, selective targeting of aHSC/Myo may be necessary to achieve significant therapeutic benefit.
Open Access
Letter
07 April 2025
Open Access
Communication
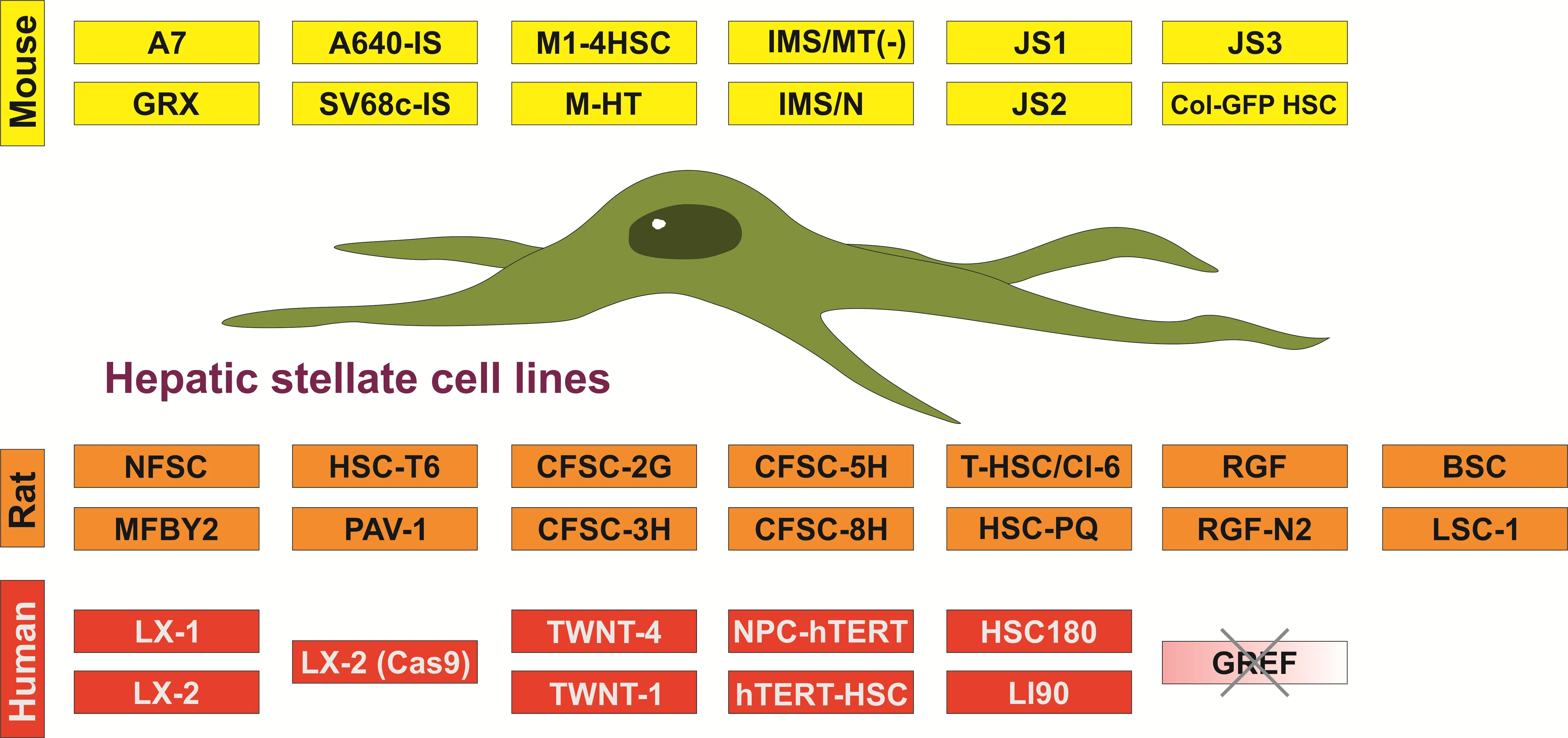
21 March 2023Established Hepatic Stellate Cell Lines in Hepatology Research
Hepatic stellate cells comprise a minor cell population in the liver, playing a key role in the pathogenesis of hepatic fibrosis. In chronic liver damage, these cells undergo a transition from a quiescent to a highly proliferative phenotype with the capacity to synthesize large quantities of extracellular matrix compounds such as collagens. Because of their pivotal role in liver disease pathogenesis, this hepatic cell population has become the focus of liver research for many years. However, the isolation of these cells is time consuming and requires the trained laboratory personnel. In addition, working with primary cells requires the following of ethical and legal standards and potentially needs the approval from respective authorities. Therefore, continuous growing hepatic stellate cells have become very popular in research laboratories because they are widely available and easy to handle, and allow a continuous supply of materials, and further reduction of lab animal use in biomedical research. This communication provides some general information about immortalized hepatic stellate cell lines from mouse, rats and humans.