Full-Spectrum Heart Failure Management by Tracking Performance of Multiple Cardiac Chambers via Cardiac Time Intervals
Received: 13 February 2026 Revised: 16 March 2026 Accepted: 23 March 2026 Published: 07 April 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Heart failure (HF) is a global epidemic affecting over 60 million people and imposing high healthcare burdens. Early detection of cardiac decompensation and tailored therapy adjustments are crucial to improving outcomes. Traditional monitoring, such as symptom onset, weight gain, and blood pressure changes, has limitations in sensitivity and timeliness of intervention, and is generally a late-stage fluid marker. Invasive hemodynamic sensors like the CardioMEMS® pulmonary artery (PA) pressure monitor have shown reduced hospitalizations in both HFrEF and HFpEF by detecting pressure rises weeks before overt symptoms. However, these require implantation and are primarily geared towards fluid management/congestion monitoring, but do not directly provide data on other aspects of HF, such as contractility or possible HF-related etiologies.
Recent advances in non-invasive wearables allow measurement of cardiac time intervals (CTIs), the timings of key cardiac events, as novel markers of cardiac chamber performance. Changes in CTIs have been linked to systolic and diastolic dysfunction. This narrative review examines the physiological relevance of CTIs (mitral valve closure/opening, aortic valve closure/opening, isovolumetric contraction/relaxation times), how they reflect chamber performance, and how they can be monitored remotely. Articles in this review deemed pertinent regarding CTI’s were included, as well as landmark studies evaluating other adjunctive detection modalities assessing early cardiac decompensation. CTIs enable the evaluation of multiple cardiac chamber performance, including contractility, relaxation, and ejection timing, providing real-time insight that enables earlier detection of deterioration, better therapy guidance, and proactive care without the need for invasive procedures. Additionally, this review compares current commercially available products with their use in the real world of heart failure to a novel device, the HEMOTAG system, currently completing an NHLBI-sponsored multi-site prospective trial evaluating how the utility of CTI’s compares to currently used hemodynamic monitoring systems [1,2,3,4].
2. Cardiac Time Intervals: Definitions and Physiological Significance
Cardiac time intervals (CTIs) are the durations between specific events in the cardiac cycle, typically the time differences between valve openings and closures. They encode information about systolic contraction, ejection, and diastolic relaxation of the heart, offering indirect but precise markers of contractility, preload, afterload, and relaxation (Table 1).
Table 1. Summary of cardiac time interval events.
|
Event/Interval |
Timing in Cardiac Cycle |
Physiological Significance [5]. |
|---|---|---|
|
Mitral Valve Closure (MVC) |
Closure of mitral valve at end-diastole |
Onset of ventricular systole; end of LV filling [6]. Abnormal delay or advance in closure may indicate impaired diastolic function or elevated filling pressures [7]. |
|
Isovolumetric Contraction Time (IVCT) |
From MVC to aortic valve opening (AVO) |
Duration of LV pressure build-up before ejection. Short IVCT implies efficient pressure generation by the ventricle. Prolongation indicates impaired contractility or increased afterload [8]. |
|
Aortic Valve Opening (AVO) |
Opening of aortic valve when LV pressure > aortic pressure |
Start of LV ejection; end of isovolumetric contraction. Delayed aortic opening can signal reduced contractility, Correlates with afterload [6]. |
|
Left Ventricular Ejection Time (LVET) |
From AVO to aortic valve closure (AVC) |
Duration of systolic ejection. Shortens with reduced stroke volume or tachycardia [9,10]. |
|
Aortic Valve Closure (AVC) |
Closure of aortic valve at end-systole (S2 sound) |
End of systole; onset of diastole [5,11]. Delayed AVC indicates high afterload, abnormal systolic unloading or altered aortic compliance |
|
Isovolumetric Relaxation Time (IVRT) |
From AVC to mitral valve opening (MVO) |
Duration of LV relaxation before filling. Prolongation indicates impaired relaxation, while a short IVRT is sign of elevated filling pressures (i.e., congestion) [12,13]. |
|
Mitral Valve Opening (MVO) |
Opening of mitral valve when LV pressure falls below LA pressure |
Start of diastolic filling; end of isovolumetric relaxation. Delayed or shortened MVO often suggests elevated left atrial pressure and reduced ventricular compliance [6]. |
|
Diastolic Filling Time (DFT) |
MVO to next MVC (diastole duration) |
Available time for ventricular filling; can be shortened at high heart rates, reduced preload, and elevated atrial pressure [14,15]. |
LV—left ventricle.
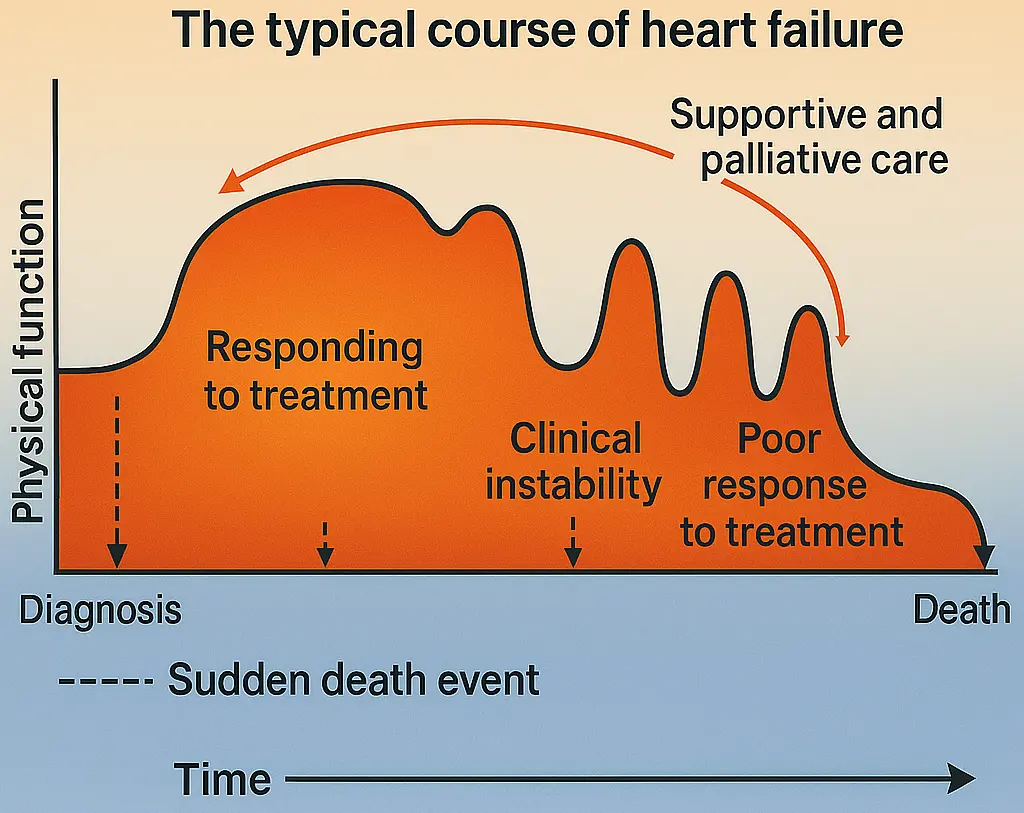
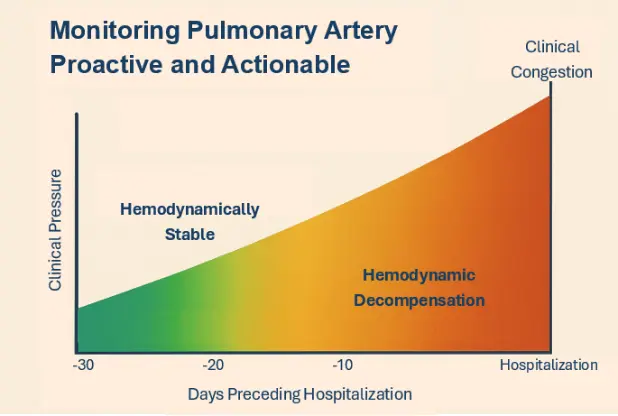
Effective surveillance of heart failure progression is central to preventing avoidable deterioration. When early physiologic worsening is not detected, patients may present later with symptomatic congestion and require hospitalization (Figure 1A). Heart failure is a chronic syndrome in which the heart cannot fill and/or eject blood adequately to meet the body’s needs. The resulting rise in cardiac filling pressures and reduced forward flow can trigger fluid accumulation in the lungs and peripheral tissues, leading to dyspnea, exercise intolerance, fatigue, edema, and weight gain. Episodes of “decompensation” often evolve over days to weeks and may be preceded by measurable shifts in intracardiac hemodynamics, changes in preload (venous return and ventricular filling), afterload (arterial load the ventricle must pump against), and contractility (intrinsic pumping strength) (Figure 1B). Tracking these parameters either directly or via validated physiologic surrogates may support earlier recognition of deterioration, more individualized titration of diuretics and guideline-directed therapies, and timelier intervention before severe congestion and hospitalization occur.
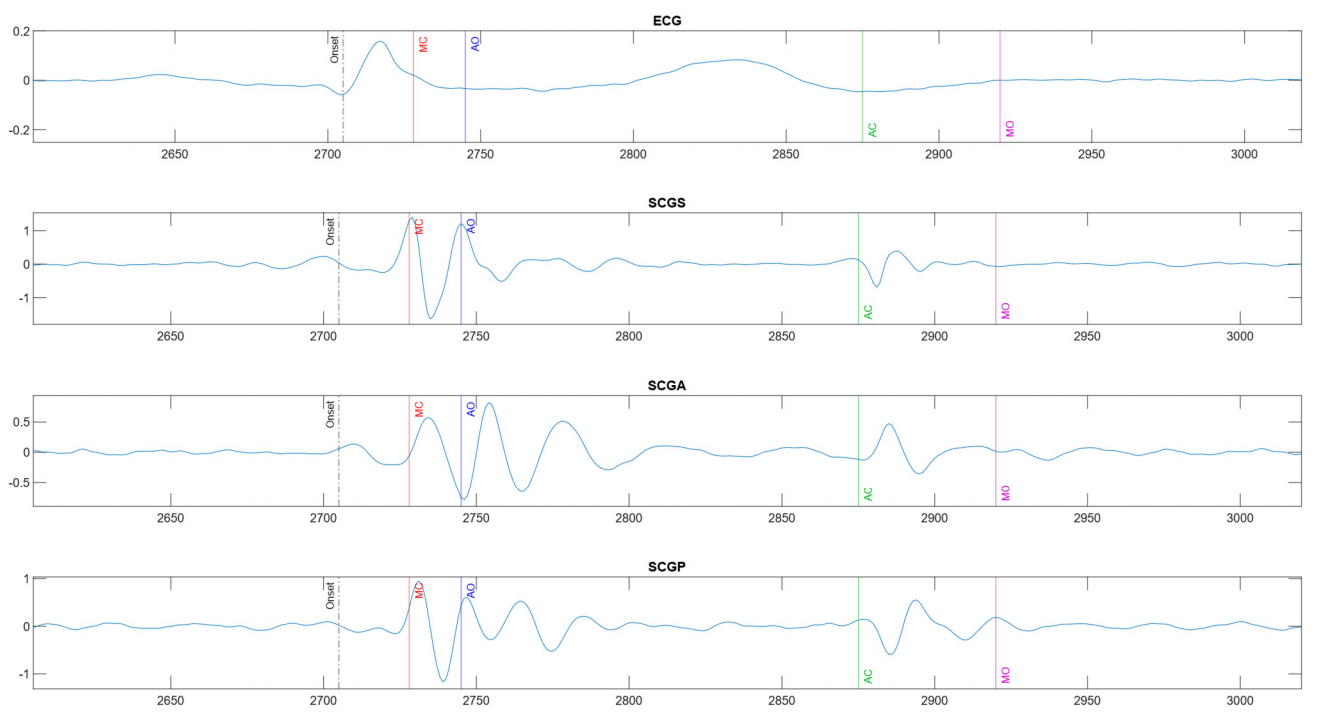
Figure 1C illustrates a normal sequence of these events on an electromechanical timeline. The mitral valve closure (MVC) coincides with the first heart sound (S1) and marks the beginning of systole. The interval from MVC to AVO is the isovolumetric contraction time (IVCT), during which the ventricles contract with all valves closed, rapidly raising intraventricular pressure. A longer IVCT suggests that the ventricle is taking extra time to generate sufficient pressure to open the aortic valve, often due to weakened contractility or increased afterload [8]. Aortic valve opening (AVO) initiates the ejection phase; the duration from AVO to aortic valve closure (AVC) is the left ventricular ejection time (LVET). Shortened LVET can reflect reduced stroke volume (as in severe systolic dysfunction) or excessive tachycardia limiting ejection time. Aortic valve closure (AVC), corresponding to the second heart sound (S2), marks the end of systole and the start of diastole. The interval from AVC to mitral valve opening (MVO) is the isovolumetric relaxation time (IVRT), representing the time for active myocardial relaxation and pressure drop before the mitral valve reopens. Prolonged IVRT is a hallmark of diastolic dysfunction, indicating a stiff ventricle slow to relax (often seen in HF with preserved EF). Short IVRT is a sign of elevated filling pressures (i.e., congestion). Post-diuretic results in IVRT normalization, providing a marker of decongestion and improved diastolic dynamics [16]. This makes IVRT a useful non-invasive parameter for monitoring response to diuretic therapy, especially in HFpEF, where traditional markers (like weight or jugular venous pressure) can be less sensitive. Finally, mitral valve opening (MVO) begins ventricular filling; the length of diastole (from MVO to the next MVC), Diastolic Filling Time (DFT), determines left vertical filling time and can be compromised in HF by high heart rates or poor relaxation.
 |
 |
|
(A) |
(B) |
 |
|
|
(C) |
|
Figure 1. (A). Typical Patient Course in Congestive Heart Failure; (B). Pulmonary artery pressures leading up to symptomatic heart failure decompensation; (C). Multi-channel phonocardiography (PCG) and electrical activity traces with annotated cardiac time intervals. Key fiducial points include Q wave, mitral valve closure (MC on PCG), aortic valve opening (AO), aortic valve closure (AC), aortic valve closing (AC), mitral valve opening (MO), and are indicated.
Physiologically, these CTIs provide quantitative metrics of cardiac performance. Systolic time intervals have long been used as proxies for contractile function. For example, the pre-ejection period (PEP), often defined as the interval from electrical depolarization (Q wave) to AVO, prolongs as left ventricular (LV) contractility declines or as afterload increases. Meanwhile, LVET tends to shorten in severe systolic dysfunction or when stroke volume is reduced. The systolic time ratio (STR), PEP/LVET, is a classic composite index of systolic performance: a higher PEP/LVET (indicating a disproportionately long isovolumic phase relative to ejection duration) correlates with worse LV ejection fraction. Weissler et al. termed this ratio the Contractility Coefficient, finding that it inversely correlates with LVEF. A normal PEP/LVET is roughly <0.3, whereas values > 0.3 suggest markedly depressed systolic function [17].
Diastolic intervals are equally informative. A prolonged IVRT denotes slow LV pressure decay and delayed relaxation, often corresponding to elevated filling pressures and a noncompliant ventricle (e.g., in HFpEF). Indeed, increased late-systolic load (as from arterial stiffening in HFpEF) can prolong overall systolic duration and delay the onset of ventricular relaxation. A shortened diastolic filling time, especially at higher heart rates, can impair preload—a particular concern in HFpEF, where the ventricle requires ample filling time due to reduced compliance. Thus, CTIs collectively reflect both systolic performance (through MVC, AVO, IVCT, LVET) and diastolic function (through AVC, MVO, IVRT, DFT), as well as the coordination of cardiac cycle events.
Traditionally, CTIs were measured via imaging or specialized physiology recordings. Echocardiography (M-mode or Doppler) is considered a gold standard for capturing valve event timings [18]. Classic analog methods combined phonocardiography (heart sounds) and carotid pulse tracings with ECG to derive systolic time intervals noninvasively. These methods established normal ranges (e.g., normal IVCT on echo ~20–40 ms, IVRT ~60–100 ms, etc.) and demonstrated that prolongation of CTIs is a marker of ventricular dysfunction. However, serially measuring CTIs outside the clinic was impractical until the development of modern sensor technology. Recent advances in high-fidelity wearable sensors, signal processing, and AI now enable CTI measurement at home, providing a potential early-warning tool for HF management.
3. Clinical Applications Across the HF Spectrum
A notable strength of CTI monitoring is its applicability to both major HF phenotypes: heart failure with reduced ejection fraction (HFrEF) and heart failure with preserved ejection fraction (HFpEF). In HFrEF (reduced EF), with impairment of systolic function, one expects prolonged IVCT and PEP, reduced LVET, and often the presence of a third heart sound or other acoustic markers of dilated ventricles. These changes can be tracked. For instance, as a patient’s EF improves with therapy such as reverse remodeling with GDMT, the Systolic Time Ratio should decrease toward normal, and IVCT may shorten. Conversely, an acute drop in EF would prolong these intervals. In HFpEF, EF is normal, but filling is impaired, and CTIs can reveal the diastolic etiology [19]. A patient with HFpEF might have a relatively normal IVCT and ejection time, but an IVRT that is prolonged (relaxation abnormalities), and an S3 sound from abrupt deceleration of blood is present in early diastole. Mitral Valve Closure (MVC) helps determine if the filling phase is prematurely terminated, which may occur in stiff ventricles as seen in patients with HFpEF. Altered timing may suggest elevated LV end-diastolic pressure or impaired compliance. Delayed AVO may indicate systolic dysfunction or increased afterload, which is common in HFrEF.
As HFpEF-related congestion worsens, IVRT may paradoxically shorten (when left atrial pressure rises to the degree in which the mitral valve opens sooner), thus trends in IVRT alongside heart sounds could signal transitions from pre-clinical stiffness to overt pulmonary edema, useful for monitoring diuretic response or evaluating diastolic dysfunction severity. Monitoring DFT allows optimization of heart rate and the timing of diuretics and beta-blockers. CTI monitoring could help assess chronotropic responses by examining how diastolic time shortens with heart rate. A recent study noted that exercise in HFpEF can drastically shorten diastolic filling time and raise filling pressures, explaining exercise intolerance.
Remote CTI monitoring can assist in therapy optimization. For example, when initiating a beta-blocker or titrating a vasodilator, one might expect improvements in CTIs (beta blockade might slightly prolong diastole but improve filling; vasodilators might shorten IVCT by reducing afterload). If CTIs worsen with prolongation of the IVCT, despite up-titration, it could indicate an inadequate response or need for alternative therapy. In device-managed patients (e.g., CRT for LBBB), improvements in dyssynchrony would shorten IVCT and may even produce a crisper S1. If a persistently prolonged IVCT is present in a CRT recipient, it might prompt an echocardiographic reassessment of lead timing or represent an inadequacy of resynchronization therapy. Below is a comprehensive breakdown of CTIs for Chamber Performance, and how each plays a critical role in the management of heart failure, especially in distinguishing and tracking both HFpEF (preserved EF) and HFrEF (reduced EF). It is important to be aware of conduction system abnormalities affecting CTI’s. Among patients with conduction system disease, CTI’s and, particularly, IVCT can be affected. These include left bundle branch blocks, in which ventricular dyssynchrony, resulting from discordant contractions of the septum and lateral wall, can prolong IVCT as coordinated pressure generation within the cardiac chambers is impaired. Additionally, loss of atrial kick among patients in atrial fibrillation can alter IVCT beat to beat as a result of decreased preload. Heart rate variability tends to have minimal effects. Key Systolic and Diastolic Events and Intervals in Cardiac Chamber Performance (Table 2).
Table 2. Clinical relevance of CTIs in HFrEF and HFpEF.
|
Interval |
Definition |
Chamber Performance Insight |
HFrEF Implication |
HFpEF Implication |
|---|---|---|---|---|
|
Mitral Valve Closure (MVC) |
From Q wave to MVC |
Marks the end of diastolic filling and the beginning of systolic contraction |
Prolonged timing may suggest elevated LV end-diastolic pressure or impaired compliance |
Prolonged if elevated LV end-diastolic pressure |
|
Aortic Valve Opening (AVO), Pre-Ejection Period (PEP) |
From Q wave to AVO |
Reflects electrical-to-mechanical delay |
Prolonged in electromechanical delay, poor contractility. Delayed opening indicates systolic dysfunction or increased afterload [10]. |
May be normal or slightly prolonged. |
|
Isovolumetric Contraction Time (IVCT) |
Time from mitral valve closure to aortic valve opening |
Measures ventricular contractile readiness before ejection |
Prolonged in low contractility. Reduced contractility or increased afterload [10,20]. |
May be normal or prolonged if afterload is elevated. |
|
Left Ventricular Ejection Time (LVET or ET) |
Time from aortic valve opening to closing |
Duration of active blood ejection |
Shortened due to reduced stroke volume. ET is directly correlated with cardiac output [10]. |
May be normal or prolonged if compensation occurs |
|
Systolic Time (ST) |
IVCT + ET or MVC to AVC |
Total systolic mechanical duration |
Reduced ST in decompensated HF [21]. |
Usually normal [20]. |
|
Aortic Valve Closure (AVC) |
From Q wave to AVC |
End of systole/start of diastole. Anchors diastolic timing |
Delayed AVC may indicate abnormal systolic unloading or altered aortic compliance, common in HF with high afterload (e.g., hypertension, aortic stiffening) |
Abnormal AVC timing may reflect impaired relaxation despite preserved ejection fraction |
|
Mitral Valve Opening (MVO) |
From Q wave to MVO |
When LV pressure falls below LA pressure. Indicates readiness for diastolic filling |
Delayed or shortened MVO often suggests elevated left atrial pressure and reduced ventricular compliance |
Prolonged filling times with low velocity reflect poor ventricular suction and often correlate with worse prognosis |
|
Isovolumetric Relaxation Time (IVRT) |
Time from AVC to MVO |
Assesses ventricular relaxation and pressure decay |
Shortened if LA pressure is high. Sign of elevated filling pressures |
Prolonged in impaired relaxation; shortened if filling pressures rise. Post-diuretic IVRT normalization is a marker of decongestion and improved diastolic dynamic [22]. |
|
Diastolic Filling Time (DFT) |
Time from MVO to MVC of next heartbeat |
Total period of ventricular filling, Reflects diastolic suction and compliance |
Often shortened due to tachycardia or poor relaxation. Shortened DFT during tachycardia impaired stroke volume and increased filling pressures [20,23]. |
May be shortened, especially in fast HR. Short DFT → high LA pressure, diastolic congestion [20,23]. |
|
Diastolic Time (DT) |
IVRT + DFT or AVC to MVC of the next heartbeat |
Total duration of ventricular filling |
Reduced in decompensated state |
Reduced in tachycardia and stiff ventricles |
LV—left ventricle, HFrEF—heart failure with reduced ejection fraction, HFpEF—heart failure with preserved ejection fraction.
The following table (Table 3) was included to provide a clear reference for established normal values of cardiac time intervals stratified by sex. Presenting these normative ranges provides context for interpreting CTIs, supports accurate comparisons across patient groups, and ensures that sex-specific physiological differences are appropriately accounted for in the analysis.
Table 3. normal values of the cardiac time intervals stratified by sex [24].
|
Cardiac Time Intervals |
All Genders |
Females |
Males |
p Value for Sex Difference |
|---|---|---|---|---|
|
IVCT, ms |
40 ± 10 |
41 ± 9 |
38 ± 10 |
<0.001 |
|
LVET, ms |
292 ± 23 |
296 ± 22 |
286 ± 23 |
<0.001 |
|
IVRT, ms |
96 ± 19 |
94 ± 19 |
100 ± 19 |
<0.001 |
|
MPI |
0.47 ± 0.09 |
0.46 ± 0.09 |
0.49 ± 0.09 |
<0.001 |
IVCT—isovolumetric contraction time, LVET—left ventricular ejection time, IVRT—isovolumetric relaxation time, MPI—myocardial performance index.
Isovolumic contraction time (IVCT) is an important measure for predicting heart failure. Normal IVCT is approximately 40 ms. When IVCT is longer than this, it indicates that the heart requires more time to generate sufficient pressure to open the aortic valve, which can reflect early impairment of systolic function. Evidence from the Copenhagen City Heart Study shows that for every 10 ms increase in IVCT, the risk of developing heart failure rises by about 24–29%, even after adjusting for traditional risk factors and left ventricular ejection fraction. These findings highlight that even modest prolongation beyond 40 ms may serve as an early warning sign of future heart failure [24].
4. HEMOTAG, for Full-Spectrum Heart Failure Management
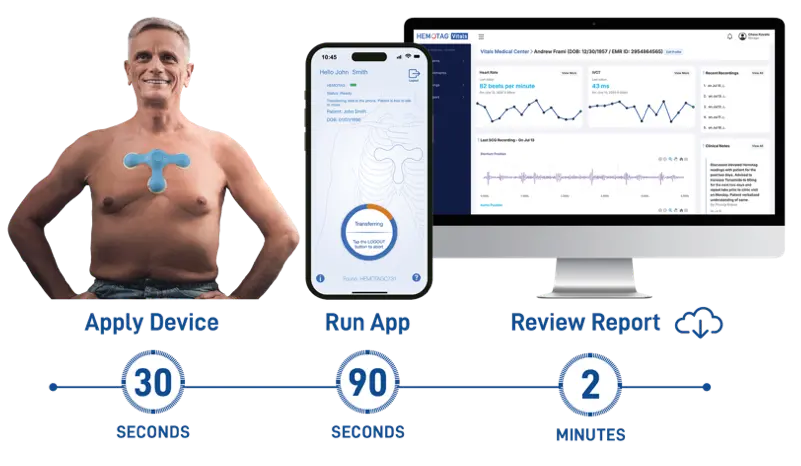
HEMOTAG is a novel system designed for hemodynamic management in patients across the HF spectrum, both HFpEF (preserved EF) and HFrEF (reduced EF). It is a small, non-invasive device placed on the chest that records time-synchronized cardiac vibrations and heart rate in a 30-s measurement (Figure 2). HEMOTAG introduces a new sensing class with a spatial-electrical-synchronized hardware in precise anatomical geometry to detect multi-chamber cardiac mechanics using a proprietary quad-sensor array. HEMOTAG captures subtle mechanical signals from multiple chest locations (including near the aortic and pulmonic areas and the sternum) simultaneously with the electrical activity signal. In essence, it operates as a sophisticated multi-channel digital stethoscope/Phonocardiogram (PCG): it detects heart sounds (S1, S2) and mechanical micro-accelerations corresponding to valve movements, all aligned with the electrical cycle. This allows the device to pinpoint the moments of mitral valve closure/opening and aortic valve opening/closure, from which the key CTIs (MVC, AVO, AVC, MVC, IVCT, LVET, IVRT, MPI, STR, STRi, HR) are computed. HEMOTAG’s multi-sensor approach is critical by recording vibrations at multiple sites simultaneously. It can identify valve events with excellent precision, including across the full spectrum of heart failure severity, BMI, gender, valve disease, and variation in placement on the chest. HEMOTAG tracks key systolic and diastolic events, offering a window into cardiac chamber hemodynamics that helps clinicians personalize heart failure therapy in real time before symptoms worsen.
The device has integrated electrical and acoustic/vibratory sensors that the patient positions on the chest. In one described configuration, its flexible “arms” contact the high left and right sternal borders (over aortic and pulmonic areas) and the lower sternum/apical region. A single button press initiates a recording: within ~30 s, the system records a segment of electrical activity and three synchronized vibrations. The raw data is transmitted wirelessly to a smartphone app, which then sends it to a cloud platform for reporting. The reporting identifies the fiducial points for each heartbeat onset, MVC, AVO, AVC, MVO, and calculates the CTIs beat-by-beat. The result is a set of hemodynamic intracardiac “vitals.” The processed results are available on a secure clinician dashboard. HEMOTAG is a non-invasive heart failure monitoring platform that delivers real-time insights into cardiac chamber performance. While others rely on implants in the pulmonary artery, left atrium, or inferior vena cava, HEMOTAG takes a smarter path, capturing real intracardiac markers directly from heart valves without an invasive procedure. By analyzing cardiac time intervals, it detects subtle changes in contractility, relaxation, and pressure dynamics, enabling earlier intervention and better outcomes without relying on invasive tools or late-stage fluid markers.
Validation and Clinical Studies
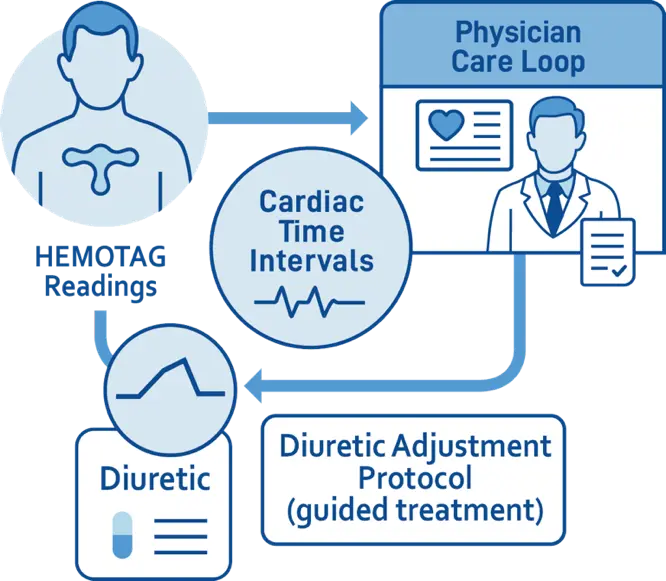
Clinical evaluations have successfully demonstrated that HEMOTAG’s CTI measurements correlate with congestion and ventricular dysfunction. In the HATS OFF (Hemotag Assessment for Short-term Outcomes of Heart Failure), In-hospital study [1], 94 hospitalized patients were evaluated to determine if HEMOTAG could distinguish acute decompensated HF (ADHF) from other conditions. HEMOTAG derived IVCT was markedly prolonged in ADHF patients (mean ~53 ms) compared to controls (~32 ms). Using a threshold IVCT ≥40 ms, the device identified ADHF (defined by clinical presentation and NT-proBNP ≥1800 pg/mL) with high sensitivity (95%) and specificity (84%). The HEMOTAG recording device was used to track daily IVCTs (normal <40 ms) in all patients. For ADHF patients, the mean IVCT decreased from 59 ± 9 ms (first day of hospitalization) to 48 ± 7 ms (final day of hospitalization). Conversely, the control arm did not have any significant changes in their daily IVCTs, demonstrated by a mean of 32 ± 9 ms (first day of hospitalization) to a mean of 35 ± 9 ms (final day of hospitalization). This demonstrates that a prolonged isovolumic contraction time on this 30-s, noninvasive test strongly indicated elevated LV filling pressure and acute HF exacerbation. Notably, these CTI markers corresponded to natriuretic peptide levels and congestion status. No adverse events occurred with device use. This study concluded that HEMOTAG could accurately detect plus track response to treatment in acutely decompensated HF (Figure 2b).
 |
 |
|
(a) |
(b) |
Figure 2. (a). HEMOTAG device kit and module application; (b). HEMOTAG clinical management flow.
Beyond the acute setting, HEMOTAG has shown promise in remote monitoring post-discharge and in chronic HF. In a home monitoring context, patients perform daily or weekly measurements with HEMOTAG. In the context of home monitoring, patients can perform regular measurements using HEMOTAG, with results securely transmitted to clinicians for review. The system’s ability to track cardiac timing intervals (CTIs) noninvasively offers a promising approach for identifying early signs of hemodynamic changes. While formal studies are ongoing, the potential for CTI trends such as changes in IVCT, IVRT, or PEP/LVET ratio to inform clinical decision-making is actively being explored. Patient involvement in their own monitoring may also enhance engagement and adherence to care plans. Compared to invasive alternatives like the CardioMEMS implant, HEMOTAG offers a non-invasive, user-friendly solution that could support proactive heart failure management once further validation is available.
While these findings are encouraging, several limitations should be considered when interpreting the current evidence. The HATS-OFF study represents an early validation cohort with a relatively small sample size and was conducted in a controlled inpatient setting. As such, the generalizability of these results to broader and more heterogeneous heart failure populations remains uncertain. In addition, although CTI parameters such as IVCT demonstrated strong associations with clinical congestion and natriuretic peptide levels, the extent to which these measurements directly reflect left ventricular filling pressures or intracardiac hemodynamics requires further validation against invasive gold-standard measurements.
Ongoing and future studies are needed to better define the role of CTI-guided monitoring in clinical practice, particularly in outpatient and remote management settings. Prospective, multicenter trials evaluating clinical endpoints such as hospitalization rates, symptom burden, and mortality will be essential to determine whether integration of HEMOTAG into routine care improves outcomes. Furthermore, comparative effectiveness studies against established monitoring strategies, including implantable hemodynamic systems such as the CardioMEMS HF System, will help clarify its relative clinical value and optimal use case.
HEMOTAG’s innovation combines multiple sensors and advanced analytics to extract clinical meaning from cardiac micro-vibrations. The concept of using chest vibration signals (PCG) for cardiac timing is not entirely new, research in the 2010s demonstrated that PCG fiducial points correspond to valve movements on echo [25]. However, earlier single-sensor PCG approaches struggled in patients with abnormal cardiac motion or obesity, where waveforms can be distorted. HEMOTAG circumvents some of these issues by using four synchronized signals to effectively capture a composite 3D vibration pattern. The multiple synchronous signals help to identify the distinct “sources” of sounds corresponding to each valve event. By distinguishing these, HEMOTAG can more reliably timestamp MVC, AVO, AVC, and MVO even in the presence of murmurs or co-morbidities.
5. Comparison with Other Cardiac Monitoring Systems
CTI-based assessment represents a new class in non-invasive hemodynamic telemonitoring. It is instructive to compare its capabilities with other commercial and clinical cardiac monitoring systems used in HF care (Table 4). These systems vary in invasiveness, parameters measured, and evidence base.
Table 4. Capabilities of commercial cardiac monitoring systems.
|
System |
Type |
Metrics Monitored |
Invasive? |
Clinical Evidence |
|---|---|---|---|---|
|
HEMOTAG [1] |
Wearable chest sensor and heart rhythm |
Cardiac time intervals: HR, MVC, AVO, AVC, MVO, IVCT, LVET, IVRT, heart sounds |
No (external) |
Detects acute HF decompensation via prolonged CTIs; HATS-OFF RCT demonstrated guided therapy reduced 30, 60 and 90-day readmissions [1]. |
|
Implanted PA pressure sensor |
Direct pressure trends in the pulmonary artery |
Yes (implanted via cath) |
Reduced HF hospitalizations ~30–40% in trials (CHAMPION for HFrEF/HFpEF) [28]; improved QoL. FDA-approved for NYHA III; high clinician adoption for high-risk HF [11]. |
|
|
External dielectric sensor |
Lung fluid/thoracic fluid index (%) congestion level assessment congestion level assessment lung measurements |
No |
Shown to detect pulmonary edema; guided therapy reduced 3-month readmissions. Used post-discharge for fluid management with some success in small studies [31,32]. |
|
|
Implantable device with algorithm (in ICD/CRT) |
Multi-sensor index: third heart sound (S3), thoracic impedance, respiratory rate, heart rate, activity, etc. |
Yes (requires ICD) |
70% sensitivity for predicting HF events ~1 month in advance (MultiSENSE study); alerts associated with fewer worsening HF events. Requires implanted defibrillator [34]. |
|
|
Telemonitoring (BP/Weight) [35] |
Home monitoring & symptom tracking |
Weight, blood pressure, symptoms, heart rate |
No |
Mixed results in trials [36]; some meta-analyses show reduced mortality/hospitalization, but others (e.g., Tele-HF) were neutral. Compliance and timely response are challenges [37]. |
|
Digital stethoscope + ECG with AI analysis |
Heart sounds, single-lead ECG; AI predicts low EF or abnormal hemodynamics |
No |
Recent study (JACC 2024) showed ~77% sensitivity, 78% specificity for detecting EF ≤40% [40]. Promising for screening in primary care, but not continuous monitoring [41]. |
|
|
Bodyport smart scale [42] |
Weight, pulse wave velocity, impedance |
Congestion index |
No |
2024 data (SCALE-HF 1 study) detected ~70% of impending HF events vs. 35% by weight gain [37,43]. |
|
Zoll Heart Failure Management System [44] |
Impedance path for continuous wear |
Thoracic Fluid Index, heart rate, activity, posture, and heart rhythm |
No |
Reduced 90-day hospital re-admission by 38% [45]. |
ReDS—remote dielectric sensing, ICD—implantable cardioverter-defibrillator, AI—artificial intelligence, CRT—cardiac resynchronization therapy, ECG—electrocardiogram, HR—heart rate, MVC—mitral valve closure, AVO—aortic valve opening, AVC—aortic valve closure, MVO—mitral valve opening, IVCT—isovolumetric contraction time, LVET—left ventricular ejection time, IVRT—isovolumetric relaxation time, EF—ejection fraction, CTI—cardiac time interval, RCT—randomized controlled trial, HFrEF—heart failure with reduced ejection fraction, HFpEF—heart failure with preserved ejection fraction.
5.1. HEMOTAG vs. Invasive Hemodynamic Monitors
An established invasive option for proactive heart failure (HF) management is the CardioMEMS HF System, an implantable pulmonary artery (PA) pressure sensor used with patient-initiated (typically daily) home transmissions. In published clinical studies, management strategies that incorporate PA pressure trends have been reported to reduce HF hospitalizations in selected patient populations, and PA diastolic pressure elevations can precede overt clinical decompensation. HEMOTAG is being developed as a non-invasive approach that focuses on cardiac mechanical timing rather than direct intracardiac or PA pressure measurement. The underlying hypothesis is that changes in filling pressures and ventricular performance may be reflected in cardiac time intervals (CTIs) derived from synchronized acoustic/mechanical signals and ECG obtained during a brief chest recording. For example, impaired relaxation and increased filling pressures may be associated with changes in diastolic timing (e.g., IVRT), while altered contractility/afterload may be associated with systolic timing changes (e.g., IVCT) [13]. Compared with implantable pressure monitors, a non-invasive CTI-based approach could be applicable to a broader range of HF phenotypes (including HFpEF and HFrEF) [46] and to patients who are not candidates for, or prefer to avoid, implantation. However, CTIs are indirect physiologic markers and require clinical validation to determine how reliably they track hemodynamic states, how they perform across diverse HF phenotypes and comorbidities, and how they should be incorporated into clinical decision-making. Both approaches can be limited by sampling frequency and adherence: CardioMEMS data are typically obtained via scheduled patient transmissions rather than continuous streaming, and HEMOTAG requires patient-initiated recordings. In both cases, missed measurements or inconsistent use could reduce the ability to detect meaningful trends.
5.2. HEMOTAG vs. Other Non-Invasive Wearables
Several non-invasive technologies support HF monitoring using different physiological proxies: (A) ReDS™ uses low-power radiofrequency signals to estimate lung fluid content. Some studies have reported fewer early readmissions when ReDS measurements are used to guide post-discharge diuretic management; however, lung fluid measurements are an indirect marker of decompensation and can be influenced by pulmonary comorbidities and other confounders. (B) Bioimpedance-based systems (e.g., ZOLL μCor HFMS; Bodyport’s Cardiac Scale) aim to estimate thoracic fluid status and/or hemodynamic change using impedance and algorithmic features. These approaches primarily emphasize congestion- and fluid-related metrics rather than direct measures of cardiac mechanical timing. (C)Wearable adhesive patches (e.g., VitalConnect VitalPatch; ZOLL VitalPatch) measure ECG and activity-related signals (and, in some configurations, respiratory parameters and/or heart sounds). These devices may support the detection of physiologic deterioration (e.g., rhythm changes, activity decline) and, in some cases, attempt to infer congestion. In contrast, HEMOTAG’s stated intent is to derive CTIs (e.g., IVCT, IVRT, PEP, LVET) from synchronized acoustic/mechanical and ECG signals, with the goal of capturing timing features related to contraction, relaxation, and valve events. This approach may provide information that is complementary to fluid-focused monitoring, but head-to-head comparative performance and clinical decision utility require prospective validation. Implantable sensors under investigation (e.g., Cordella, V-LAP, FIRE1) target direct hemodynamic measurements (pulmonary or atrial pressures) and may offer different tradeoffs (procedural risk, patient selection constraints, signal specificity). AI-enhanced stethoscope systems (e.g., Eko DUO) combine ECG and phonocardiography for point-of-care screening applications (e.g., reduced EF detection reported with AUROC metrics in published studies). These tools are typically used during clinical encounters rather than for serial home monitoring, so comparisons should distinguish between use cases (screening vs. longitudinal monitoring), measurement types (pressure vs. timing vs. fluid proxy), and workflow.
5.3. Usability and Patient Experience
HEMOTAG is designed to support a brief, user-initiated measurement (e.g., ~30 s) with one-button operation and wireless upload to a secure cloud environment, enabling integration into remote monitoring workflows [1,2]. Unlike implantable systems (e.g., CardioMEMS), HEMOTAG does not require implantation and therefore avoids the procedural risks associated with invasive device placement [3]. Continuous wearables can provide high-frequency data but may also increase data volume and the need for signal quality controls, alert management, and clinical workflow integration to reduce false or non-actionable notifications [4]. HEMOTAG’s approach emphasizes targeted CTI metrics intended to support trend review; however, thresholds (e.g., IVCT cutoffs explored in inpatient/acute settings) should be described as exploratory unless validated for home monitoring and linked to clinically meaningful endpoints [47]. As with other home monitoring tools, adherence, correct positioning, and adequate signal quality are necessary for interpretable data. Early feasibility/usability assessments may inform training requirements and learning curves, but conclusions about long-term adherence and patient experience should be limited to what has been directly measured and reported [26].
6. Benefits and Limitations of Remote CTI Monitoring
Remote monitoring of CTIs offers several potential benefits for HF management:
-
-
Early Warning of Decompensation: By tracking subtle changes in cardiac function (before overt weight gain or symptoms), CTI monitoring can alert clinicians about impending decompensation. As noted, prolonged CTIs correlated strongly with high BNP in acute HF, and anecdotal use showed detection of abnormalities before symptom onset. This could translate to timely intervention (e.g., adjust diuretics) and prevention of hospitalizations.
-
-
Objective Therapy Guidance: CTIs provide quantitative, beat-to-beat information on how the heart is responding. This could guide the titration of medications. For instance, an ACE inhibitor might be increased until no further improvement in PEP or IVRT is observed, indicating an optimal hemodynamic effect. In contrast, patient-reported symptoms or weights can be quite variable and subjective.
-
-
Applicability to Diverse HF Populations: CTI monitoring is useful in HFrEF, HFpEF, and even other cardiac conditions (valve disease, post-myocardial infarction dysfunction, etc.) because it measures fundamental aspects of cardiac cycle timing. This is unlike some telemonitors that focus solely on fluid (more relevant to congestion) or arrhythmia (relevant to certain subsets).
-
-
Non-Invasive and Patient-Centric: With systems like HEMOTAG, patients can perform daily 30-s readings at home, without blood draws or imaging. Empowering patients to be active participants in their care. It also reduces the need for frequent surveillance clinic visits, which is important given the burden of HF clinic follow-ups.
However, there are important limitations and challenges:
-
-
Signal Quality and Patient Factors: Not every patient’s cardiac signals are easy to interpret via chest wall sensors. SCG/PCG signals can be attenuated by obesity, COPD (hyperinflated lungs), or simply poor positioning, inadequate patient and medical staff training. HEMOTAG’s multi-sensor design significantly improves the odds, from the HATS-OFF study, in about 1% of patients, the vibrational waveform was “unacceptable”, and 3% of patients, the electrical signal fiducial points were “Unacceptable”. Arrhythmias, such as atrial fibrillation, also complicate CTI analysis, as beat-to-beat variability is high and averaging may be required. In such cases, trends might be obscured.
-
-
Data Interpretation and Alerts: Just as with any monitoring, false positives/negatives are a risk. A spurious prolongation of IVCT (due to a premature beat or user error in one measurement) could trigger an unnecessary alert. Conversely, a gradual decline in function might be missed if changes fall within normal variability or if measurements are infrequent. Setting the right thresholds for alerting is key. The HATS-OFF study’s cutoff of IVCT ≥ 40 ms for acute HF was useful diagnostically, but for home monitoring, each patient may need an individualized baseline. AI could eventually establish each patient’s “dry” baseline CTIs and flag significant deviations.
-
-
Integration into Care Workflow: Remote monitoring only improves outcomes if coupled to a robust clinical response system. This was seen with CardioMEMS: the sensor itself does not treat HF; it is the clinical team’s timely intervention on pressure rise that prevents hospitalization. Similarly, CTI data must be continuously reviewed (or automatically triaged by algorithms) and acted upon. This demands infrastructure (nurses, telehealth communication, etc.) that not all practices have readily available. Until this cutting-edge CTI monitoring technology is integrated into standard care pathways or HF disease management programs, its full benefit may not be realized. Current guidelines have been cautious, offering limited endorsement of remote monitoring technologies due to mixed trial results and implementation challenges. Demonstrating outcome improvements in rigorous trials will be key to broader adoption.
-
-
Specificity of CTI Changes: CTIs can be influenced by many factors beyond HF status. For example, IVCT and IVRT vary with heart rate. If a patient’s beta-blocker dose is increased, heart rate drops; this alone lengthens diastolic intervals (more filling time, which is beneficial), but one must distinguish this from an intrinsic relaxation improvement. Dehydration could shorten IVRT (less filling pressure), but that might be misconstrued as improved diastolic function. Thus, context is needed; ideally, CTI data is interpreted alongside other data (heart rate, blood pressure, perhaps patient symptoms). HEMOTAG’s ecosystem could incorporate some of this context (the device also measures heart rate and perhaps can correlate with the timing of medication dosing or weight changes if those are logged in the app).
-
-
Patient adherence: A key practical limitation of remote monitoring strategies in heart failure is variable patient adherence, which can substantially influence both data reliability and clinical effectiveness. Sustained engagement with home-based monitoring requires patients to perform regular measurements, correctly use the device, and remain responsive to care team feedback—tasks that may be challenging in a population often characterized by advanced age, multimorbidity, cognitive impairment, and fluctuating functional status. Digital literacy and access to reliable technology (e.g., smartphones, internet connectivity) further contribute to disparities in adherence, particularly among socioeconomically disadvantaged groups. Even among motivated patients, adherence may decline over time due to monitoring fatigue, perceived lack of immediate benefit, or competing health priorities. Inconsistent data acquisition can lead to gaps in longitudinal trends, potentially limiting the ability of clinicians to detect early physiologic changes or intervene in a timely manner. Additionally, reliance on patient-generated data introduces variability related to measurement technique and environmental factors, which may affect signal quality and interpretation. From a systems perspective, suboptimal adherence may reduce the overall effectiveness and cost-efficiency of remote monitoring programs, particularly if actionable alerts are missed or if additional resources are required to re-engage patients. These challenges underscore the importance of designing user-centered monitoring platforms with simplified workflows, automated data capture where possible, and integrated patient education and support mechanisms. Future studies should incorporate adherence metrics as key endpoints and evaluate strategies—such as behavioral interventions, caregiver involvement, and adaptive monitoring protocols—to enhance sustained engagement and ensure that the potential benefits of non-invasive remote monitoring are realized in real-world heart failure populations.
7. Conclusions
Measuring cardiac chamber performance via cardiac time intervals offers a window into the heart’s mechanical performance that is highly relevant for managing heart failure. CTIs like MVC, AVO, IVCT, and IVRT distill complex hemodynamics into quantifiable markers that reflect contractility, afterload, and relaxation. The advent of home-based CTI monitoring, exemplified by the HEMOTAG system, represents a significant innovation in HF care. HEMOTAG’s non-invasive sensor technology can detect the same valvular timing intervals that were previously measurable only in echo labs or cath labs, bringing sophisticated hemodynamic assessment to the patient’s living room. Early studies have shown that CTI changes correlate with acute HF decompensation and may forecast clinical deterioration, enabling interventions that improve outcomes. While challenges like signal quality, data management, and clinical integration need to be addressed, the potential upside is substantial: fewer hospitalizations, more personalized therapy titration, improved quality of life, and lower out-of-pocket costs for patients who can age gracefully and live longer, happier, and healthier lives together at home.
In the evolving landscape of HF management, remote monitoring is increasingly recognized as a key component, complementing pharmacotherapy and device therapy. CTI monitoring via HEMOTAG or similar systems could fill a niche that current telemonitoring approaches struggle to provide: direct, real-time insight into cardiac function without invasive sensors. As evidence grows (from ongoing trials and real-world use) and as algorithms become more refined, we can expect CTI-guided care to earn a place in guidelines alongside PA pressure monitoring and other modalities. For cardiologists, being familiar with these time interval metrics and their interpretation will be important, as they provide insights that were not available before and, daily, represent another set of vital signs for the failing heart. Ultimately, harnessing CTIs non-surgically within a home monitoring strategy aligns with the broader goal in HF management: anticipating and preventing decompensation rather than reacting to it. The HEMOTAG system, with its robust capture of mitral and aortic valve events, exemplifies how advanced engineering can augment clinical acumen, translating the subtle sounds and motions of the heart into actionable data for better patient care.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this manuscript, the authors used ChatGPT in order to review language, clarity and grammar of the text. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the published article.
Acknowledgments
The authors acknowledge Aventusoft LLC for providing administrative and technical support, as well as access to relevant technical and clinical information related to the HEMOTAG system that informed portions of this manuscript.
Author Contributions
R.C., F.R.T., J.E.P. and D.S. contributed to the conceptualization, review, and editing of the manuscript. Authors provided critical input, approved the final version, and agree to be accountable for all aspects of the work.
Ethics Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data are protected under SBIR data protection guidelines for a period of up to 20 years and are therefore not publicly available. Access may be granted by the corresponding author upon reasonable request and with permission from the funding agency.
Funding
This research was funded by the National Heart, Lung, And Blood Institute grant number R44HL145941 and R44HL149561.
Declaration of Competing Interest
None of the authors have any conflicts of interest, financial or otherwise, to declare in regard to this study.
References
- Chait R, Ramos Tuarez F, Pino JE, Uppal D, Snipelisky D. Non-Invasive Evaluation by the HEMOTAGTM Recording Device to Tailor Treatment of Acutely Decompensated Heart Failure. Cardiovasc. Sci. 2025, 2, 10007. DOI:10.70322/cvs.2025.10007 [Google Scholar]
- Nudelman BG, Tejeda APT, Patsias I. HEMOTAG: A Novel Non-Invasive Device for Reducing Hospital Readmissions in Acute Decompensated Heart Failure Patients. J. Card. Fail. 2026, 32, 291. DOI:10.1016/j.cardfail.2025.11.297 [Google Scholar]
- Nudelman BG, Patsias I. Remote monitoring with HEMOTAG: Preventing heart failure readmissions through early detection of atrial flutter. JACC 2025, 85 (Suppl. S12), 3895. DOI:10.1016/S0735-1097(25)04379-7 [Google Scholar]
- Uppal D, Sanchez A, Lewis A, Juan C, Alonso M, Estep JD, et al. Use of HEMOTAG cardiopulmonary assessment system for predicting clinical decompensation in heart failure in the inpatient setting. J. Am. College Cardiol. 2024, 83, 3569. DOI:10.1016/s0735-1097(24)05559-1 [Google Scholar]
- Robinson S, Ring L, Oxborough D, Harkness A, Bennett S, Rana B, et al. The assessment of left ventricular diastolic function: guidance and recommendations from the British Society of Echocardiography. Echo Res. Pract. 2024, 11, 16. DOI:10.1186/s44156-024-00051-2 [Google Scholar]
- Pollock JD, Makaryus AN. Physiology, Cardiac Cycle. National Library of Medicine. Published 3 October 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459327/ (accessed on 5 December 2025).
- Tsutsui H, Urabe Y, Mann DL, Tagawa H, Carabello BA, Cooper G, et al. Effects of chronic mitral regurgitation on diastolic function in isolated cardiocytes. Circ. Res. 1993, 72, 1110–1123. DOI:10.1161/01.RES.72.5.1110 [Google Scholar]
- Biering-Sørensen T. Cardiac Time Intervals by Tissue Doppler Imaging M-mode echocardiography: Reproducibility, Reference values, Association with Clinical Characteristics and Prognostic Implications. Dan. Med. J. 2016, 63, B5279. Available online: https://www.researchgate.net/profile/Tor-Biering-Sorensen/publication/305903418_Cardiac_time_intervals_by_tissue_Doppler_imaging_M-mode_echocardiography_reproducibility_reference_values_association_with_clinical_characteristics_and_prognostic_implications/links/57a6177508aefe6167b69740/Cardiac-time-intervals-by-tissue-Doppler-imaging-M-mode-echocardiography-reproducibility-reference-values-association-with-clinical-characteristics-and-prognostic-implications.pdf (accessed on 5 December 2025).
- Alhakak AS, Sengeløv M, Jørgensen PG, Bruun NE, Johnsen C, Abildgaard U, et al. Left ventricular systolic ejection time is an independent predictor of all-cause mortality in heart failure with reduced ejection fraction. Eur. J. Heart Fail. 2022, 23, 240–249. DOI:10.1002/ejhf.2022 [Google Scholar]
- Alhakak AS, Teerlink JR, Lindenfeld J, Böhm M, Rosano GMC, Biering-Sørensen T. The Significance of Left Ventricular Ejection Time in Heart Failure with Reduced Ejection Fraction. Eur. J. Heart Fail. 2021, 23, 541–551. DOI:10.1002/ejhf.2125 [Google Scholar]
- Napoli N. CardioMEMS Sensor Reliably Safe, Cuts Hospitalizations by More than Half. American College of Cardiology. Published 17 March 2019. Available online: https://www.acc.org/about-acc/press-releases/2019/03/17/09/05/cardiomems-sensor-reliably-safe (accessed on 5 December 2025).
- CardioMEMS HF System Clinical Evidence. Cardiovascular Abbott. Zamani. Available online: https://www.cardiovascular.abbott/us/en/hcp/products/heart-failure/pulmonary-pressure-monitors/cardiomems/clinical-evidence.html (accessed on 5 December 2025).
- Sohn DW. Heart Failure Due to Abnormal Filling Function of the Heart. J. Cardiol. 2011, 57, 148–159. DOI:10.1016/j.jjcc.2011.01.001 [Google Scholar]
- Lala A, Barghash MH, Giustino G, Alvarez-Garcia J, Konje S, Parikh A, et al. Early Use of Remote Dielectric Sensing After Hospitalization to Reduce Heart Failure Readmissions. ESC Heart Fail. 2021, 8, 1047–1054. DOI:10.1002/ehf2.13026 [Google Scholar]
- Nishimura RA, Tajik AJ. Evaluation of Diastolic Filling of Left Ventricle in Health and Disease: Doppler Echocardiography Is the Clinician’s Rosetta Stone. J. Am. Coll. Cardiol. 1997, 30, 8–18. DOI:10.1016/S0735-1097(97)00144-7 [Google Scholar]
- Aziz F, Luqman-Arafath TK, Enweluzo C, Dutta S, Zaeem M. Diastolic Heart Failure: A Concise Review. J. Clin. Med. Res. 2013, 5, 327. DOI:10.4021/jocmr1532w [Google Scholar]
- Weissler AM, Stack RS, Lee CC, Reddy BP, Taylor ML. Left ventricular performance in coronary artery disease by systolic time intervals and echocardiography. Trans. Am. Clin. Climatol. Assoc. 1976, 87, 36–47. [Google Scholar]
- Reant P, Dijos M, Donal E, Mignot A, Ritter P, Bordachar P, et al. Systolic time intervals as simple echocardiographic parameters of left ventricular systolic performance: correlation with ejection fraction and longitudinal two-dimensional strain. Eur. J. Echocardiogr. 2010, 11, 834–844. DOI:10.1093/ejechocard/jeq084 [Google Scholar]
- Oki T, Miyoshi H, Oishi Y, Iuchi A, Kusunose K, Yamada H, et al. Heart Failure With Preserved Ejection Fraction―Time for a Paradigm Shift Beyond Diastolic Function ―. Circ. Rep. 2019, 1, 8–16. DOI:10.1253/circrep.CR-18-0017 [Google Scholar]
- Mayo Clinic. Heart Failure with Preserved Ejection Fraction (HFpEF): More Than Diastolic Dysfunction. Published 20 May 2015. Available online: https://www.mayoclinic.org/medical-professionals/cardiovascular-diseases/news/heart-failure-with-preserved-ejection-fraction-hfpef-more-than-diastolic-dysfunction/mac-20430055 (accessed on 5 December 2025).
- Patel PA, Ambrosy AP, Phelan M, Alenezi F, Chiswell K, Van Dyke MK, et al. Association Between Systolic Ejection Time and Outcomes in Heart Failure by Ejection Fraction. Eur. J. Heart Fail. 2020, 22, 1174–1182. DOI:10.1002/ejhf.1659 [Google Scholar]
- Isovolumic Relaxation Time—An Overview. Available online: https://www.sciencedirect.com/topics/medicine-and-dentistry/isovolumic-relaxation-time (accessed on 5 December 2025).
- Kagami K, Obokata M, Harada T, Kato T, Wada N, Adachi T, et al. Diastolic Filling Time, Chronotropic Response, and Exercise Capacity in Heart Failure and Preserved Ejection Fraction With Sinus Rhythm. J. Am. Heart Assoc. 2022, 11. DOI:10.1161/JAHA.121.026009 [Google Scholar]
- Alhakak AS, Olsen FJ, Skaarup KG, Lassen MCH, Johansen ND, Jørgensen PG, et al. Age- and sex-based normal reference ranges of the cardiac time intervals: the Copenhagen City Heart Study. Clin. Res. Cardiol. 2023, 114, 430–442. DOI:10.1007/s00392-023-02269-2 [Google Scholar]
- Tavakolian K. Systolic Time Intervals and New Measurement Methods. Cardiovasc. Eng. Technol. 2016, 7, 118–125. DOI:10.1007/s13239-016-0262-1 [Google Scholar]
- Abraham WT, Adamson PB, Hasan A, Bourge RC, Pamboukian SV, Aaron MF, et al. Safety and accuracy of a wireless pulmonary artery pressure monitoring system in patients with heart failure. Am. Heart J. 2011, 161, 558–566. DOI:10.1016/j.ahj.2010.10.041 [Google Scholar]
- Rakita V. CardioMEMSTM Device for Monitoring Advanced Heart Failure: 5 Things to Know. Temple Health. Published 16 December 2021. Available online: https://www.templehealth.org/about/blog/cardiomems-device-monitoring-advanced-heart-failure-5-things-to-know (accessed on 5 December 2025).
- Patel AH, Natarajan B, Pai RG. Current Management of Heart Failure with Preserved Ejection Fraction. Int. J. Angiol. 2022, 31, 166–178. DOI:10.1055/s-0042-1756173 [Google Scholar]
- Amir O, Ben-Gal T, Weinstein JM, Schliamser J, Burkhoff D, Abbo A, et al. Evaluation of remote dielectric sensing (ReDS) technology-guided therapy for decreasing heart failure re-hospitalizations. Int. J. Cardiol. 2017, 240, 279–284. DOI:10.1016/j.ijcard.2017.02.120 [Google Scholar]
- ReDSTM Pro System Remote Dielectric Sensing. Sensible Medical. Published 15 January 2025. Available online: https://sensible-medical.com/solution/ (accessed on 5 December 2025).
- Adamson PB, Abraham WT, Bourge RC, Costanzo MR, Hasan A, Yadav C, et al. Wireless Pulmonary Artery Pressure Monitoring Guides Management to Reduce Decompensation in Heart Failure With Preserved Ejection Fraction.Circ. Heart Fail. 2014, 7, 935–944. DOI:10.1161/CIRCHEARTFAILURE.113.001229 [Google Scholar]
- Alvarez-Garcia J, Lala A, Rivas-Lasarte M, De Rueda C, Brunjes D, Lozano-Jimenez S, et al. Remote Dielectric Sensing Before and After Discharge in Patients With ADHF. JACC Heart Fail. 2024, 12, 695–706. DOI:10.1016/j.jchf.2024.01.002 [Google Scholar]
- Boehmer JP, Hariharan R, Devecchi FG, Smith AL, Molon G, Capucci A, et al. A Multisensor Algorithm Predicts Heart Failure Events in Patients With Implanted Devices. JACC Heart Fail. 2017, 5, 216–225. DOI:10.1016/j.jchf.2016.12.011 [Google Scholar]
- Singh JP, Wariar R, Ruble S, Kwan B, Averina V, Stolen CM, et al. Prediction of Heart Failure Events With the HeartLogic Algorithm: Real-World Validation. J. Card. Fail. 2024, 30, 509–512. DOI:10.1016/j.cardfail.2023.10.478 [Google Scholar]
- Mohebali D, Kittleson MM. Remote monitoring in heart failure: current and emerging technologies in the context of the pandemic. Heart 2021, 107, 366–372. DOI:10.1136/heartjnl-2020-318062 [Google Scholar]
- Chaudhry SI, Mattera JA, Curtis JP, Spertus JA, Herrin J, Lin Z, et al. Telemonitoring in Patients with Heart Failure. N. Engl. J. Med. 2010, 363, 2301–2309. DOI:10.1056/NEJMoa1010029 [Google Scholar]
- O’Riordan M. Smart Scale Better at Predicting HF Events Than Weight-Based Monitoring. Published 11 March 2024. Available online: https://www.tctmd.com/news/smart-scale-better-predicting-hf-events-weight-based-monitoring (accessed on 5 December 2025).
- Littrell A. AI-Powered Stethoscope Enhances Early Heart Failure Detection in Primary Care. Available online: https://www.medicaleconomics.com/view/ai-powered-stethoscope-enhances-early-heart-failure-detection-in-primary-care (accessed on 25 November 2025).
- Peters NS. Triple Cardiovascular Disease Detection with an Artificial Intelligence-Enabled Stethoscope (TRICORDER). Clinicaltrials.gov. Published 11 July 2024. Available online: https://clinicaltrials.gov/study/NCT05987670 (accessed on 5 December 2025).
- Guo L, Pressman GS, Kieu SN, Marrus SB, Mathew G, Prince J, et al. Automated Detection of Reduced Ejection Fraction Using an ECG-Enabled Digital Stethoscope. JACC Adv. 2025, 4, 101619. DOI:10.1016/j.jacadv.2025.101619 [Google Scholar]
- Heart Failure Management System (HFMS) . Zoll. Published 2024. Available online: https://cardiac.zoll.com/patients/heart-failure-management-system (accessed on 5 December 2025).
- Prithul Bom B. K211585—Bodyport Cardiac Scale . FDA; 2022. Available online: https://www.accessdata.fda.gov/cdrh_docs/pdf21/K211585.pdf (accessed on 5 December 2025).
- Stevenson LW, Ross HJ, Rathman LD, Boehmer JP. Remote Monitoring for Heart Failure Management at Home. J. Am. College Cardiol. 2023, 81, 2272–2291. DOI:10.1016/j.jacc.2023.04.010 [Google Scholar]
- Heart Failure Management System (HFMS) Reduce Heart Failure Readmissions and Improve Patient Outcomes. Published 2024. Available online: https://cardiac.zoll.com/medical-professionals/heart-failure-management-system (accessed on 5 December 2025).
- Boehmer JP, Cremer S, Abo-Auda WS, Stokes DR, Hadi A, McCann PJ, et al. Impact of a Novel Wearable Sensor on Heart Failure Rehospitalization. JACC Heart Fail. 2024, 12, 2011–2022. DOI:10.1016/j.jchf.2024.07.022 [Google Scholar]
- Simmonds SJ, Cuijpers I, Heymans S, Jones EAV. Cellular and Molecular Differences between HFpEF and HFrEF: A Step Ahead in an Improved Pathological Understanding. Cells 2020, 9, 242. DOI:10.3390/cells9010242 [Google Scholar]
- Işilay Zeybek ZM, Racca V, Pezzano A, Tavanelli M, Di Rienzo M. Can Seismocardiogram Fiducial Points Be Used for the Routine Estimation of Cardiac Time Intervals in Cardiac Patients? Front. Physiol. 2022, 13, 825918. DOI:10.3389/fphys.2022.825918 [Google Scholar]