Contemporary Review on Multi-Modality Imaging Evaluation and Management of Functional Mitral Regurgitation
Received: 07 December 2025 Revised: 19 January 2026 Accepted: 05 March 2026 Published: 19 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Functional mitral regurgitation (FMR), or secondary mitral regurgitation, has traditionally been defined as valvular incompetence occurring in the setting of structurally normal mitral leaflets and chordae, where regurgitation results from adverse geometric remodeling of the left ventricle (LV) rather than from primary leaflet pathology [1]. In this classic, LV-centric paradigm, FMR is distinguished from primary (organic) mitral regurgitation by its etiology: global or regional LV remodeling displaces the papillary muscles and tethers the leaflets, thereby impairing systolic coaptation [1]. More recently, this concept has been refined, and FMR is now recognized to encompass at least two distinct pathophysiological phenotypes: ventricular FMR (VFMR) and atrial FMR (AFMR) [1]. VFMR represents the traditional LV-driven mechanism, whereas AFMR arises from left atrial (LA) enlargement with resultant annular dilatation, typically in the context of atrial fibrillation (AF) or heart failure with preserved ejection fraction (HFpEF) where left atrial pressure is chronically elevated, and this can occur despite preserved LV systolic function [1,2,3,4,5,6,7,8].
Irrespective of the underlying mechanism, FMR is a clinically important entity because of its rising prevalence, close association with heart failure symptoms, and adverse impact on prognosis [2,4,9,10,11]. Recognition of the distinct ventricular and atrial phenotypes is critical, as they differ in anatomy, clinical course, and response to therapy and may thus require tailored diagnostic and interventional strategies [1,2,3,4,5,6,7,8]. Multimodality imaging, including transthoracic and transesophageal echocardiography, cardiac magnetic resonance, and computed tomography, has become indispensable not only for accurate quantification of MR severity but also for distinguishing between AFMR and VFMR and for procedural planning [3,9,10]. In parallel, management has evolved from a foundation of guideline-directed medical therapy (GDMT) to a broad spectrum of surgical and, increasingly, transcatheter interventions targeted to the specific mechanism of regurgitation [10,12,13,14,15,16]. This review synthesizes contemporary understanding of FMR, emphasizing the pivotal role of multimodality imaging and outlining current and emerging management strategies across the spectrum of ventricular and atrial phenotypes.
2. Epidemiology and Clinical Impact
FMR is one of the most prevalent forms of moderate-to-severe mitral regurgitation in adults, accounting for approximately 65% of cases in a community-based cohort [17]. The estimated prevalence of moderate-to-severe mitral regurgitation in the general adult population ranges from 0.46% to 2.3% [2,17,18], with FMR occurring more frequently among older individuals and those with comorbid conditions, such as heart failure or atrial fibrillation [2,19,20,21].
The clinical burden of FMR is significant. Compared to the general population and to patients with primary mitral regurgitation of similar severity, individuals with FMR experience markedly increased morbidity and mortality [2,4,9,10,11]. The five-year heart failure incidence can be as high as 80% in VFMR and 59% in AFMR [22]. All-cause mortality is significantly elevated, with reported risk ratios ranging from 1.88 to 3.45 across FMR subtypes [17,23,24,25]. Notably, severe FMR confers a graded increase in mortality across all heart failure phenotypes, with hazard ratios reaching as high as 7.5 in the most advanced cases [24,25,26]. Despite its high prevalence and adverse prognosis, FMR remains a challenge to diagnose and treat. Fewer than 10% of affected patients undergo surgical or transcatheter mitral interventions [15], underscoring the need for improved recognition and management of this condition.
3. Pathophysiology & Management Considerations of FMR
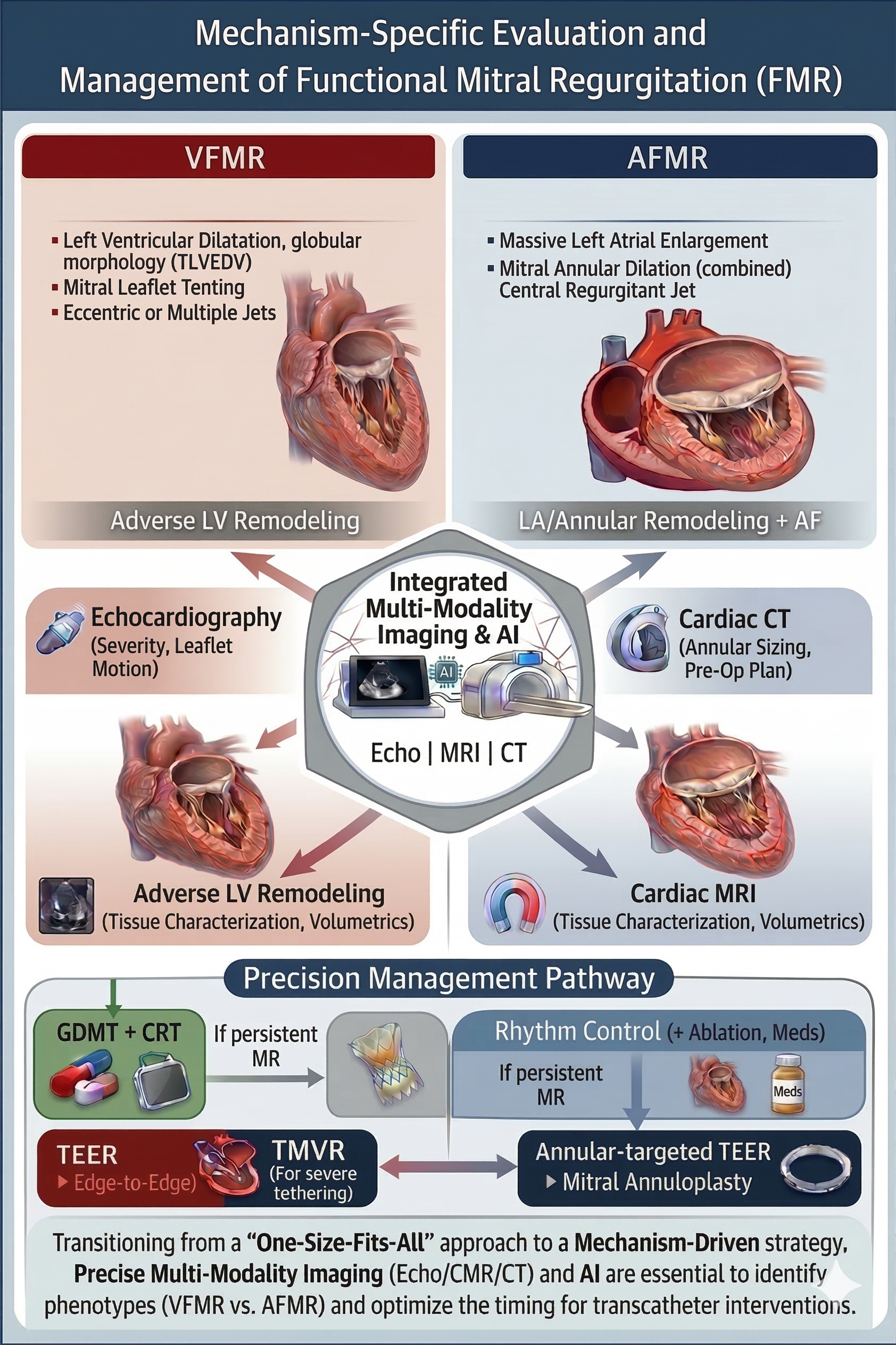
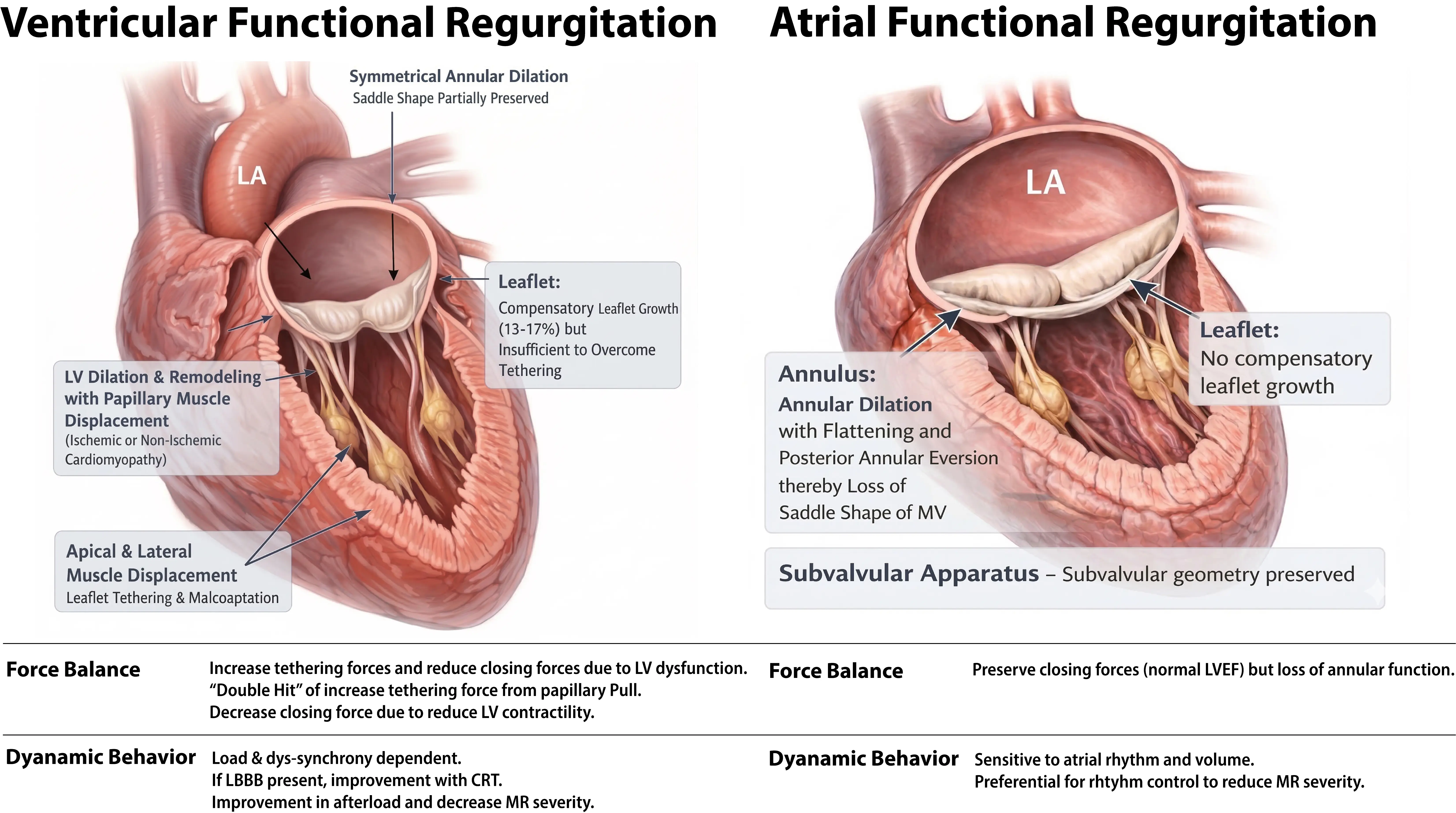
The mitral valve sits on a saddle-shaped annulus. There are 2 mitral valve leaflets, the anterior and posterior leaflets. There are chordae tendineae attached to each leaflet, which allow the mitral valve to perform its function during systole and diastole. FMR arises from perturbations in cardiac geometry of the saddle-shaped annulus and/or LV function that impair mitral valve competence despite structurally normal leaflets and sub-valvular apparatus [9]. On this basis, FMR can be further classified into three distinct phenotypes: VFMR, AFMR, and dual FMR [2,4,7,27,28,29]. VFMR is more common in males and is associated with left ventricular systolic dysfunction and ischemic heart disease [2,17,30,31,32], while AFMR is predominantly linked to AF and left atrial enlargement [19,27,33,34,35,36]. Dual FMR reflects the coexistence of atrial and ventricular mechanisms, in which both annular dilatation and leaflet tethering contribute to regurgitation, and may be encountered in advanced or long-standing cardiomyopathic and atrial remodeling [2,4,7,27,28,29]. Although their initiating mechanisms differ, all three phenotypes converge on a final common pathway: loss of effective leaflet coaptation due to an imbalance between tethering and closing forces [7,27,28,37]. Furthermore, FMR is a dynamic entity: its severity may fluctuate with loading conditions, heart rhythm, blood pressure, and ventricular synchrony [38,39,40]. Ultimately, a structurally normal mitral valve becomes incompetent due to its maladaptive mechanical environment [2], which clearly distinguishes FMR from primary (degenerative) mitral regurgitation, which is related to intrinsic issues with the mitral valve itself. The key mechanistic and clinical differences between VFMR and AFMR are detailed and summarized in Figure 1 and Table 1. Figure 2 details the differing management processes and considerations for VFMR and AFMR, which will be further elaborated in subsequent sections.
Table 1. Comparative Mechanistic and Clinical Characteristics of Ventricular vs. Atrial Functional Mitral Regurgitation.
|
VFMR |
AFMR |
|
|---|---|---|
|
Primary geometric driver |
LV dilation and remodeling due to ischemic or nonischemic cardiomyopathy with papillary muscle displacement |
Left atrial enlargement with mitral annular dilation in the setting of chronic AF or HFpEF and preserved LV size |
|
Imaging characteristics |
LV dilation, increased LVEDV, papillary muscle angles widened, increased tenting height/area (~10 mm in severe cases), eccentric jet. |
Marked LA dilation (>60 mL/m2), annular diameter ≥40 mm, height/width ratio <0.15, normal LV size/strain, central jet |
|
Clinical associations |
HFrEF, ischemic scar, LBBB |
Chronic AF, HFpEF, older age |
|
Prognosis |
Higher mortality and HF hospitalization risk |
Lower mortality and HF hospitalization risk; may progress to dual FMR |
|
First-line management |
GDMT for HFrEF; CRT if indicated |
Rhythm control for AF, HFpEF management, afterload reduction if hypertensive |
|
Device/interventional therapy |
TEER or surgical repair/replacement in select cases after GDMT optimization |
TEER or annuloplasty in select symptomatic patients |
|
Key RCTs |
COAPT, MITRA-FR, RESHAPE-HF2 |
No large RCTs specific to AFMR; most data from observational studies and subgroup analyses |
Abbreviations: VFMR = Ventricular Functional Mitral Regurgitation; AFMR = Atrial Functional Mitral Regurgitation; LV = Left Ventricle; LA = Left Atrium; HFpEF = Heart Failure with Preserved Ejection Fraction; HFrEF = Heart Failure with Reduced Ejection Fraction; CRT = Cardiac Resynchronization Therapy; LVEDV = Left Ventricular End-Diastolic Volume; TEER = Transcatheter Edge-to-Edge Repair; RCT = Randomized Controlled Trial; COAPT = Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients with Functional Mitral Regurgitation; MITRA-FR = Multicenter Study of Percutaneous Mitral Valve Repair MitraClip Device in Patients with Severe Secondary Mitral Regurgitation; RESHAPE-HF2 = Randomized Study of the MitraClip Device in Heart Failure Patients with Clinically Significant Functional Mitral Regurgitation.
3.1. VFMR
VFMR typically occurs in the context of ischemic or nonischemic cardiomyopathy and reflects the downstream consequences of LV dilation, papillary muscle displacement, and mitral annular enlargement [2,41]. These geometric alterations result in apical and lateral displacement of the papillary muscles, leading to increased leaflet tethering and preventing proper coaptation [29,41,42]. Impaired LV contractility reduces closing forces, further exacerbating regurgitation, even in the absence of structural valve abnormalities [3,43].
A contemporary conceptual framework, first articulated by Grayburn et al., categorizes VFMR into proportionate and disproportionate subtypes, depending on the relationship between the effective regurgitant orifice area (EROA) and the extent of LV remodeling [44]. In proportionate VFMR, MR severity aligns with the degree of LV dilation and dysfunction, implying that regurgitation is primarily a reflection of advanced myocardial disease [44]. Management in these patients should focus on reversing or stabilizing LV remodeling through GDMT, cardiac resynchronization therapy (CRT), or mechanical circulatory support, as valve intervention alone is unlikely to significantly alter outcomes [45,46,47]. Disproportionate VFMR is characterized by MR severity that is excessive relative to the degree of LV dilation, a scenario where the EROA is much larger than would be expected based on the LV end-diastolic volume (LVEDV) [44]. Mechanistically, this often results from regional wall motion abnormalities, desynchrony, or asymmetric leaflet tethering [44]. Here, MR itself is thought to drive symptoms and adverse events, so directly addressing the valve lesion surgically or via transcatheter edge-to-edge repair (TEER), offers substantial prognostic benefit [48,49]. This dichotomy has substantial clinical implications; it provides a potential explanation for divergent results in landmark trials: MITRA-FR (dominated by proportionate MR and large ventricles) found no benefit from TEER [16], whereas COAPT (which selected for those with disproportionate MR and smaller LV volumes) demonstrated significant improvements in heart failure hospitalization and mortality [49].
However, this framework has sparked considerable debate, and emerging evidence raises questions about its clinical utility. Hagendorff et al. provide a pointed critique, questioning the physiological validity of “disproportionate” MR and arguing that regurgitant flow cannot meaningfully exceed the limits set by ventricular remodeling [50]. They advocate for a comprehensive echocardiographic assessment over reliance on EROA/LVEDV ratios [50]. Similarly, Ooms et al., in a multicenter registry, observed that although patients classified as having disproportionate FMR initially experienced more robust MR reduction after TEER, this benefit was not sustained and did not translate into better long-term outcomes compared to those with proportionate MR [51]. Furthermore, Kałmucki et al. recently demonstrated that mitral annuloplasty with the Carillon device resulted in MR reduction and LV reverse remodeling, irrespective of “proportionality”, with outcomes not dependent on the EROA/LVEDV relationship [52]. These findings undermine the predictive value of the framework for durable clinical benefit [53].
Rather than viewing these perspectives as mutually exclusive, a more integrative interpretation is that proportionality may serve as a heuristic to frame competing ventricular versus valvular drivers of disease, but not as a binary determinant of treatment eligibility. When EROA and LVEDV appear discordant, a stepwise, physiology-informed approach may be more clinically useful as listed in Figure 3 which includes the following steps: (1) confirm MR severity using multiparametric echocardiographic assessment, including vena contracta width, regurgitant volume, and dynamic loading conditions; (2) interrogate mechanisms of tethering and annular deformation, with attention to regional wall motion abnormalities, dyssynchrony, and leaflet geometry; (3) assess the extent of myocardial disease and reversibility using LV volumes, strain, and cardiac magnetic resonance where available; and (4) evaluate clinical trajectory and response to optimized GDMT. In this framework, markedly dilated ventricles with only modest EROA should prompt caution that MR predominantly reflects advanced myocardial pathology, whereas severe MR in the setting of relatively preserved LV size and focal tethering may reasonably support consideration of valve intervention despite apparent “disproportionality”.
Taken together, the proportionate–disproportionate framework is best regarded as an interpretive lens rather than a prescriptive algorithm. Patient selection for surgical or transcatheter mitral intervention should ultimately rest on comprehensive multimodality imaging, longitudinal assessment of ventricular remodeling, clinical status, and heart team deliberation, rather than on simplified ratios alone. Future prospective studies integrating imaging-derived phenotypes with treatment response are needed to refine risk stratification beyond proportionality metrics and to identify patients most likely to derive sustained benefit from mitral valve intervention.
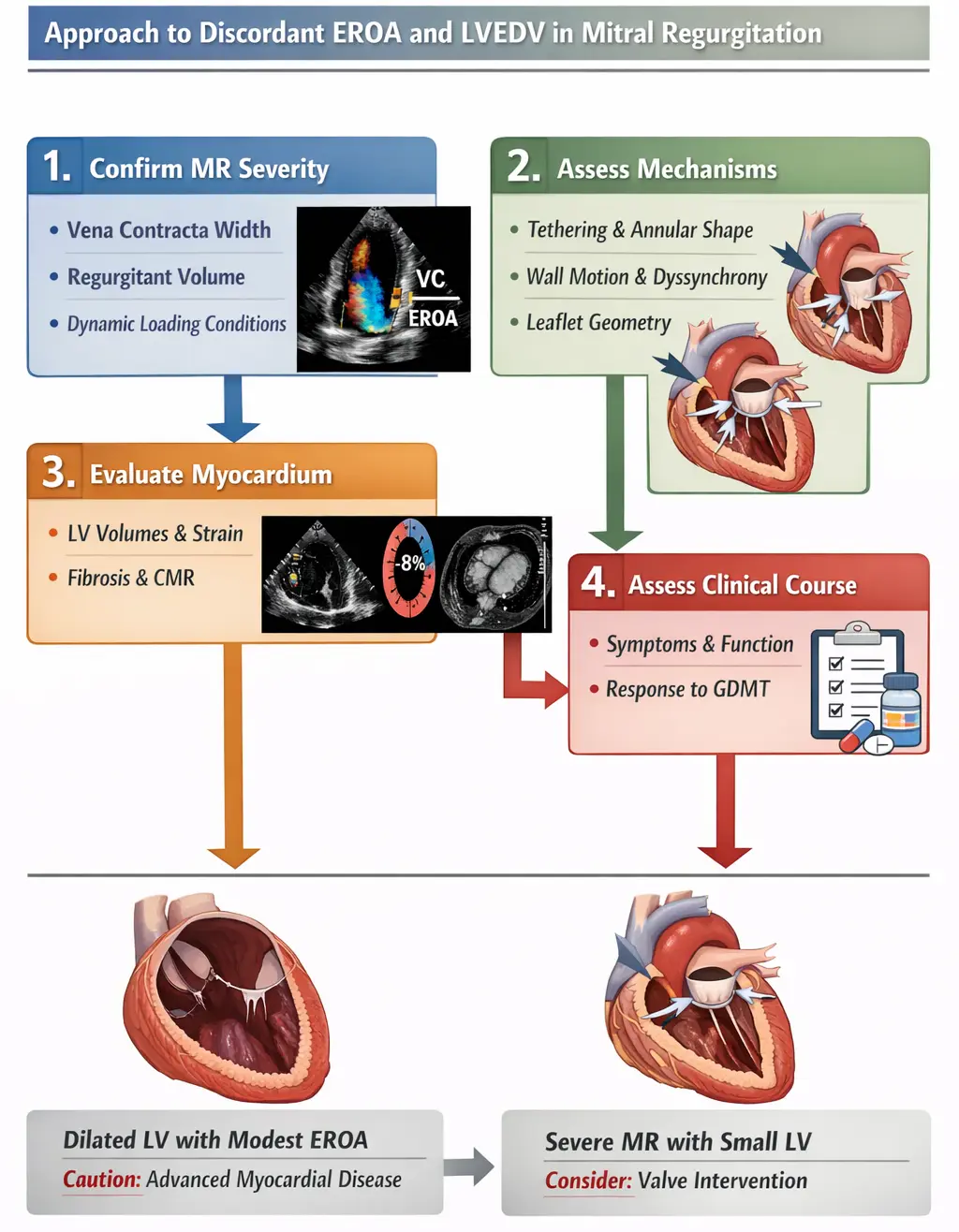
Figure 3. Approach to Ventricular Functional Mitral Regurgitation and Consideration of Management. Color Doppler: Red = flow toward the transducer, Blue = flow away from the transducer, Mosaic/mixed colors (yellow–green/orange) indicate turbulence and/or aliasing when velocities exceed the Nyquist limit (velocity scale). Global Longitudinal Strain from speckle-tracking echo: Red side = more myocardial shortening (i.e., more negative strain, typically closer to normal/better contraction; e.g., around –18% to –22% for GLS), Blue side = less shortening (i.e., less negative strain, closer to 0) or sometimes even positive strain (paradoxical lengthening/dyskinesis).
3.2. AFMR
AFMR occurs in the setting of preserved LV size and systolic function and is driven primarily by LA and mitral annular enlargement [4,28,35]. The most common etiologies are chronic AF and HFpEF [19,20]. Unlike VFMR, AFMR is not associated with papillary muscle displacement or LV dilation; rather, the pathological changes are atrial and annular in origin [4,22,28,35].
Chronic AF and/or elevated LA pressures lead to progressive LA dilation, which causes mitral annular enlargement and loss of its normal saddle shape [36,54,55,56]. The annulus becomes flattened and more planar, reducing leaflet coaptation and increasing the likelihood of central regurgitation [54,55,56]. Although the mitral leaflets are structurally normal, the annular dilation exceeds the compensatory capacity of the leaflets, resulting in a leaflet-to-annulus mismatch [57,58]. AF further compromises annular contractility, diminishing systolic annular shortening and thereby exacerbating regurgitation [59,60]. In some patients, posterior annular displacement may contribute to atriogenic tethering, compounding coaptation failure [34,54,56].
Not all FMR should be treated with the same algorithm. Given that the mechanistic of AFMR is due to atrial dilation, for the cohort with AFMR, treating the driver of atrial dilatation is of great importance. A proposed framework for the management of AFMR is listed as follows.
3.2.1. Step 1: Rhythm Control and HFpEF Optimization as First-Line Therapy
Given that AFMR is fundamentally driven by left atrial remodeling and dysfunction, therapeutic strategies that target the reduction of continuous adverse LA remodeling need to be considered at the forefront of management. Rhythm control, most commonly via catheter ablation, has emerged as a central therapeutic pillar in AFMR, with accumulating observational evidence demonstrating meaningful reductions in MR severity, left atrial volume, and annular dimensions, alongside improvements in diastolic function, natriuretic peptide levels, and functional status [7]. Importantly, MR improvement following successful rhythm control can reduce propensity for further atrial dilatation and loss of annular contraction in AFMR, rather than irreversible leaflet or ventricular pathology.
Although randomized data are lacking, several contemporary cohorts suggest that maintenance of sinus rhythm is associated with durable MR reduction and lower progression to dual FMR, particularly when intervention occurs before advanced atrial myopathy or severe annular dilation has developed [19]. These observations underscore the importance of early rhythm control in appropriately selected patients. Consistent with this concept, the Japanese Circulation Society assigns a Class IIa recommendation for catheter ablation in symptomatic patients with persistent atrial fibrillation and severe functional MR when sinus rhythm maintenance is considered achievable, highlighting atrial-directed therapy as a disease-modifying strategy rather than purely symptomatic management.
In cases of HFpEF that contribute to elevated LA pressure, guideline-directed therapy, including diuretics for volume optimization and aggressive blood pressure control, remains an essential adjunct.
3.2.2. Step 2: Indications for Escalation to Valve-Directed Intervention
Valve intervention should be considered in patients with persistent severe MR and NHYA class III–IV symptoms despite optimized rhythm control and medical therapy. Careful consideration of MR phenotype is critical to confirm atrial-predominant MR and to exclude advanced ventricular remodeling or mixed (dual) FMR, which is associated with worse outcomes and may not respond durably to atrial-focused strategies. Multimodality imaging is essential to identify patients in whom MR has become structurally entrenched and is less likely to regress with rhythm control alone.
3.2.3. Step 3: Selection of Interventional Strategy Based on Annular Pathophysiology and Atrial Substrate
For patients undergoing surgical intervention, such as mitral valve surgery, surgical mitral annuloplasty, when combined with a Cox-Maze procedure, is a viable interventional consideration for AFMR [61], as it simultaneously addresses annular dilatation and the underlying arrhythmic substrate. Observational data demonstrate substantially lower rates of recurrent MR when surgical annuloplasty is paired with atrial fibrillation ablation compared with annuloplasty alone, reinforcing the concept that isolated annular correction may be insufficient in the presence of ongoing atrial myopathy [61]. However, marked annular enlargement may necessitate aggressive ring under-sizing, increasing the risks of mitral stenosis or annular dehiscence, and mitral valve replacement may be required in select cases. Concomitant repair of atrial functional tricuspid regurgitation should be strongly considered when present.
Transcatheter edge-to-edge repair (TEER) represents an effective alternative in patients at elevated surgical risk and has demonstrated high procedural success and symptomatic improvement in AFMR cohorts. TEER appears to perform more favorably in AFMR than in VFMR, likely owing to preserved LV function and minimal leaflet tethering. Nonetheless, because TEER does not directly address annular dilatation or atrial remodeling, its durability may be limited in patients with progressive atrial myopathy, emphasizing the importance of careful patient selection. It is important to have a robust heart team meeting to discuss, as the success of TEER procedures often depends on the center’s success in treating mitral valve disease with TEER devices.
Direct and indirect transcatheter annuloplasty systems offer a more mechanistically aligned approach by targeting annular geometry and, in some cases, promoting left atrial reverse remodeling. While these strategies may achieve less immediate MR reduction than TEER, they may offer advantages in selected patients in whom atrial enlargement and annular dysfunction are the dominant drivers of regurgitation. Collectively, these considerations highlight that AFMR management should not default to annular intervention alone; rather, it should integrate rhythm control, atrial substrate modification, and valve therapy in a personalized, stepwise manner.
4. Clinical Features
The main symptoms of both VFMR and AFMR include exertional dyspnea, orthopnea, paroxysmal nocturnal dyspnea, fatigue, and decreased exercise tolerance [32,42]. These symptoms reflect elevated left-sided filling pressures and pulmonary congestion resulting from both primary ventricular dysfunction and the superimposed regurgitant volume load [9,20,62]. Palpitations and light-headedness can also be present, especially when AF is the underlying rhythm [37]. On physical examination, a holosystolic murmur is often appreciated at the cardiac apex and may radiate to the axilla [2,37,63]. Additional findings typically reflect the severity of heart failure and include signs of volume overload, such as elevated jugular venous pressure, pulmonary rales, hepatomegaly, and peripheral edema [63]. Evidence of underlying left ventricular systolic dysfunction, such as a displaced apical impulse or a soft S1, may also be present [2].
FMR is associated with a high risk of heart failure hospitalization and mortality, with outcomes closely tied to MR severity [17,22,24,25]. It is essential to note that the degree of regurgitation is often dynamic, fluctuating with changes in preload, afterload, or cardiac rhythm, which underscores the importance of serial assessment under physiologic conditions [4,37]. While both FMR subtypes carry increased risk, VFMR is generally associated with higher rates of mortality and heart failure hospitalization than AFMR [17,22,64]. However, in some patients, persistent atrial overload from AFMR can lead to progressive cardiac remodeling and eventual ventricular dysfunction, resulting in a transition to a mixed or “dual FMR” phenotype [22,37].
5. Diagnostic and Prognostic Evaluation
5.1. Electrocardiography, Laboratory Tests, and Chest Radiography
Electrocardiography (ECG) provides valuable clues to the underlying etiology of FMR, although findings are not specific. In VFMR, ECG may reveal features of structural heart disease, including Q waves or ST-T wave abnormalities indicative of prior myocardial infarction, left bundle branch block, or nonspecific intraventricular conduction delays [31,65]. These abnormalities reflect underlying ischemic or nonischemic cardiomyopathy and may contribute to dyssynchrony that exacerbates regurgitation [65,66]. In AFMR, AF is the most common rhythm disturbance, presenting with absent P waves and an irregularly irregular rhythm [4,28,35,58]. Alternatively, patients in sinus rhythm may exhibit P-wave changes consistent with left atrial enlargement, supporting the diagnosis of AFMR driven by atrial dilation [4,28,35,58].
Laboratory testing is largely non-specific in FMR. Elevated natriuretic peptides (BNP or NT-proBNP) are frequently observed in both VFMR and AFMR, reflecting elevated intracardiac pressures and neurohormonal activation associated with heart failure [67,68]. In more advanced cases, laboratory abnormalities related to end-organ dysfunction may emerge: impaired renal function due to cardiorenal syndrome and mild transaminase elevations due to hepatic congestion from right-sided heart failure [69,70]. However, these findings are not unique to FMR and should be interpreted in the broader clinical context.
Chest radiography offers supportive evidence of cardiac chamber enlargement and pulmonary congestion. Pleural effusions may be present in cases of volume overload [37,68]. Left ventricular enlargement is typically absent in AFMR, reflecting its preserved LV geometry [8,22,54,64]. These diagnostic tools, although not definitive, play a crucial role in the initial assessment and in guiding further imaging and risk stratification.
5.2. Echocardiography
Echocardiography serves as the cornerstone in the diagnosis and characterization of FMR, providing a direct view of the mitral valve apparatus and its interaction with surrounding cardiac structures [37,63]. A comprehensive echocardiographic examination, ideally combining 2D and 3D imaging, provides both hemodynamic grading and mechanistic insight [37,63]. Echocardiographic grading of MR severity requires a comprehensive, integrative approach, as no single parameter is definitive [37]. Figure 3 details the assessment of MR, both quantitative and semi-quantitative measures, as well as the guidelines of both ACC/AHA and ESC on MR severity thresholds.
As recommended by the ACC/AHA and ASE, this involves synthesizing qualitative parameters, such as valve morphology and color Doppler jet characteristics, with semiquantitative parameters like the vena contracta width (VCW; ≥0.7 cm for severe), pulmonary vein flow (systolic flow reversal is specific for severe MR), and a dense, triangular continuous-wave Doppler jet profile [37,63]. These are integrated with quantitative parameters, considered most specific, including regurgitant volume (RegVol; ≥60 mL for severe), regurgitant fraction (RF; ≥50% for severe), and effective regurgitant orifice area (EROA; ≥0.40 cm2 for severe) [37,63].
Despite these guidelines, accurately quantifying FMR remains challenging. The regurgitant orifice in FMR is often elliptical and dynamic, making the commonly used PISA method, which assumes a circular orifice, susceptible to underestimation [71]. Contemporary professional society guidelines generally agree on the importance of integrating quantitative and qualitative parameters when assessing FMR, differ in how rigidly EROA thresholds are applied, and how they inform TEER candidacy. The ACC/AHA guidelines define severe secondary MR using an EROA ≥ 0.40 cm2 (or regurgitant volume ≥ 60 mL) [37], consistent with primary MR, yet emphasize that intervention decisions, particularly TEER, should be guided by symptom burden, response to GDMT, and patient selection criteria derived from COAPT rather than by EROA alone [12]. In contrast, the ESC/EACTS guidelines adopt lower quantitative thresholds for severe FMR (EROA ≥ 0.20 cm2 or regurgitant volume ≥ 30 mL) [2]. The differences in EROA thresholds reflect the challenging aspects of MR assessment, given differences in mechanistic factors and the load-dependent nature of secondary MR, but both guidelines similarly stress comprehensive assessment and clinical context over isolated cutoffs. Recent studies suggest that an intermediate value of around 0.30 cm2 may better correlate with adverse outcomes and response to therapy [72,73]. The ASE/EACVI recommendations further reinforce this integrative approach, cautioning against reliance on a single parameter, such as EROA, and advocating multiparametric grading that incorporates ventricular size and function, regurgitant jet characteristics, and dynamic behavior [74]. Taken together, these documents are aligned in recognizing the limitations of rigid EROA thresholds in FMR and converge on the principle that TEER candidacy should be determined by a holistic evaluation of MR severity, ventricular remodeling, and clinical status rather than by any single quantitative metric. Figure 4 illustrates the multi-parametric transthoracic echocardiographic assessment used to grade severe AFMR, encompassing chamber enlargement, jet characteristics, and quantitative Doppler measurements.
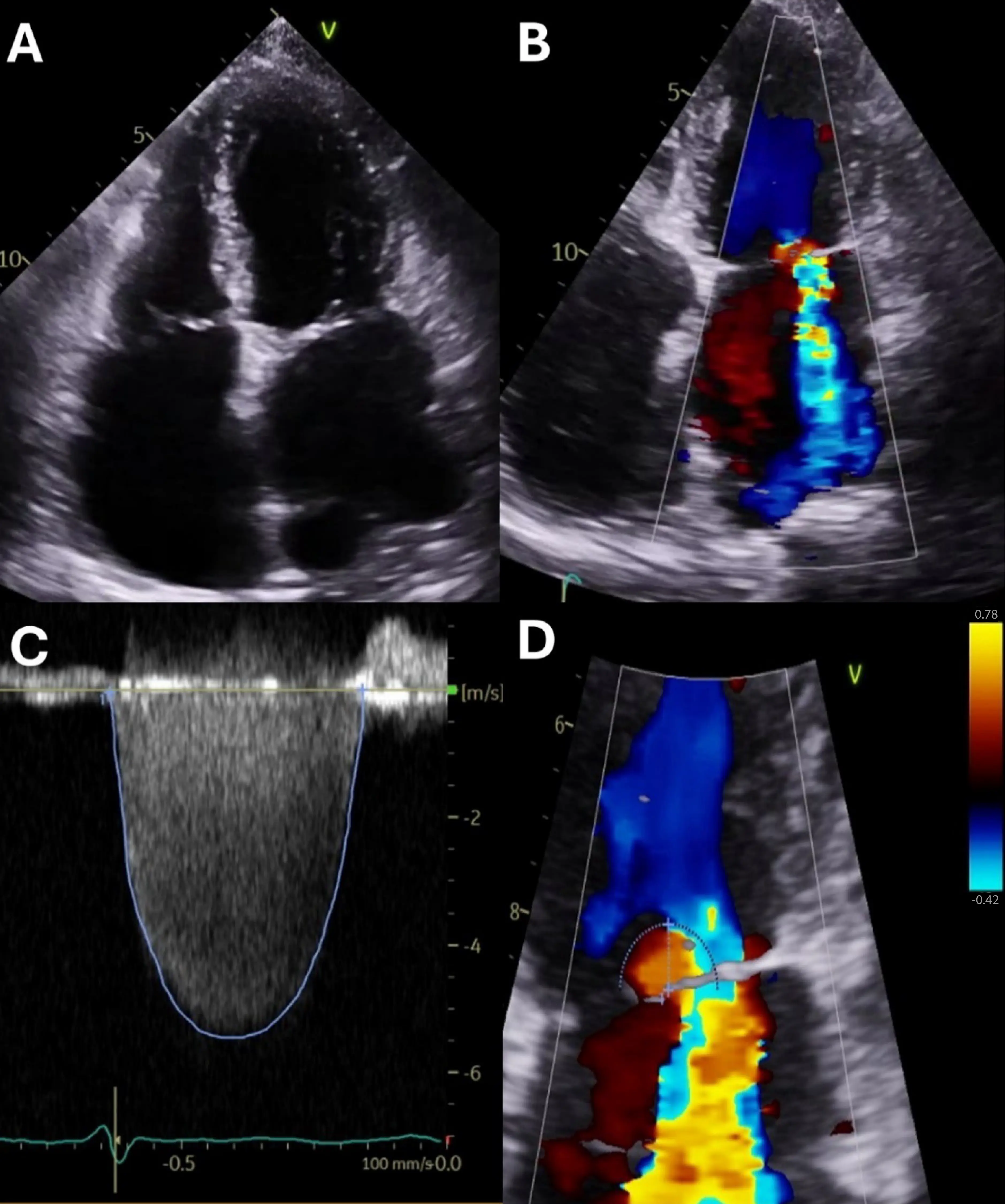
Figure 4. Transthoracic Echocardiographic Assessment of Severe Atrial Functional Mitral Regurgitation. (A) Apical four-chamber view demonstrating severe bi-atrial enlargement with marked dilation and flattening of the mitral annulus, consistent with atrial functional mitral regurgitation. The numbers along the side (e.g., 5, 10) are depth markers (in mm). (B) Color Doppler imaging in the same view reveals a broad, centrally directed jet of severe mitral regurgitation. Red vs. blue represent opposite flow directions relative to the transducer (conventionally: red = toward, blue = away, depending on the machine’s color map and orientation). Yellow/orange/mosaic colors indicate aliasing and turbulent/high-velocity flow (velocity exceeding the Nyquist limit). (C) Zoomed color Doppler view focused on the regurgitant orifice, showing a proximal isovelocity surface area (PISA) radius of 0.8 cm, supportive of significant regurgitant flow. The y-axis shows velocity (units shown as m/s). The horizontal baseline (0 m/s) separates flow directions (above vs. below baseline). The outlined envelope/contour (blue tracing) marks the spectral Doppler envelope used for measurement (e.g., peak velocity and/or velocity-time integral). The ECG trace at the bottom provides timing within the cardiac cycle. (D) Continuous-wave Doppler tracing across the mitral valve reveals a dense, holosystolic MR signal with a peak velocity of 5.5 m/s, consistent with elevated regurgitant volume and pressure gradient. This corresponds to an effective orifice regurgitant area (EROA) of 0.31 cm2. The dashed circular/semicircular outline highlights the proximal flow convergence/isovelocity contour. The straight line/arrow indicates the measured radius/distance from the flow origin to the aliasing boundary. The vertical color bar at the right is the velocity color scale, showing the range and direction of encoded velocities (i.e., the Nyquist limits and the color mapping).
Beyond grading, echocardiography is crucial for identifying the specific FMR mechanism [37,38]. In VFMR, the image typically shows a dilated or regionally remodeled left ventricle with apically and laterally displaced papillary muscles, leading to a tethered (Carpentier type IIIb) configuration [1,2,3,8,38]. Tenting parameters (height > 10 mm or area > 2.5 cm2) reflect this advanced distortion [1,3,38]. In contrast, AFMR is characterized by significant left atrial and annular enlargement (e.g., indexed LA volume > 34 mL/m2) in the setting of a preserved LV [7,22,28,38]. The annulus flattens and dilates, preventing the structurally normal leaflets from coapting, often creating a central regurgitant jet [4,7,41,75].
Assessment of LV and LA size, function, LV volumes, and LVEF and strain is central to this evaluation [37,38]. LA volume is a sensitive marker of chronic MR, as a normal LA size virtually excludes severe chronic MR [38,76]. Furthermore, LV global longitudinal strain (GLS) can detect early myocardial dysfunction before a decline in LVEF, while LA reservoir strain provides insight into atrial compliance; a reservoir strain below 18% indicates poor compliance, elevated pulmonary pressures, and a higher likelihood of failure with rhythm-control strategies [77,78,79,80,81,82]. This comprehensive chamber assessment is pivotal for applying the proportionate/disproportionate VFMR framework, which relies on the relationship between LVEDV and EROA to guide therapy [44,45].
Finally, specialized echocardiographic studies are used in specific clinical scenarios [44,63,83,84]. Stress echocardiography, preferably with exercise, is recommended when the severity of MR at rest does not correlate with clinical symptoms [40,63,85]. Key findings associated with adverse outcomes include a dynamic increase in MR severity (e.g., an EROA increase ≥13 mm2) or the development of pulmonary hypertension (PASP ≥ 60 mmHg) [40]. Transesophageal echocardiography (TEE) is essential when transthoracic echocardiography images are inconclusive and is mandatory for procedural planning and guidance [2,86]. TEE provides superior, high-resolution visualization of leaflet morphology, coaptation depth, and annular geometry (Figure 5) [2,86]. 3D TEE, in particular, offers a “surgical view” that is critical for assessing suitability for TEER and other transcatheter interventions [63,83,87]. During the MitraClip procedure, TEE is used for device navigation, guidance of transseptal puncture (e.g., measuring height from the annular plane), precise leaflet grasping, and assessment of the final post-deployment result [63,86]. This step-by-step guidance is critically important, as demonstrated in Figure 2.
To be clinically actionable, an echocardiographic report should document chamber dimensions, ejection fraction, annular size, leaflet tethering indices, and integrate multiple Doppler indices to estimate severity, explicitly stating which grading criteria were used. Most importantly, it should provide a mechanistic interpretation, concluding whether the regurgitation is predominantly ventricular, atrial, or mixed, as this narrative is essential for informing the management strategy.
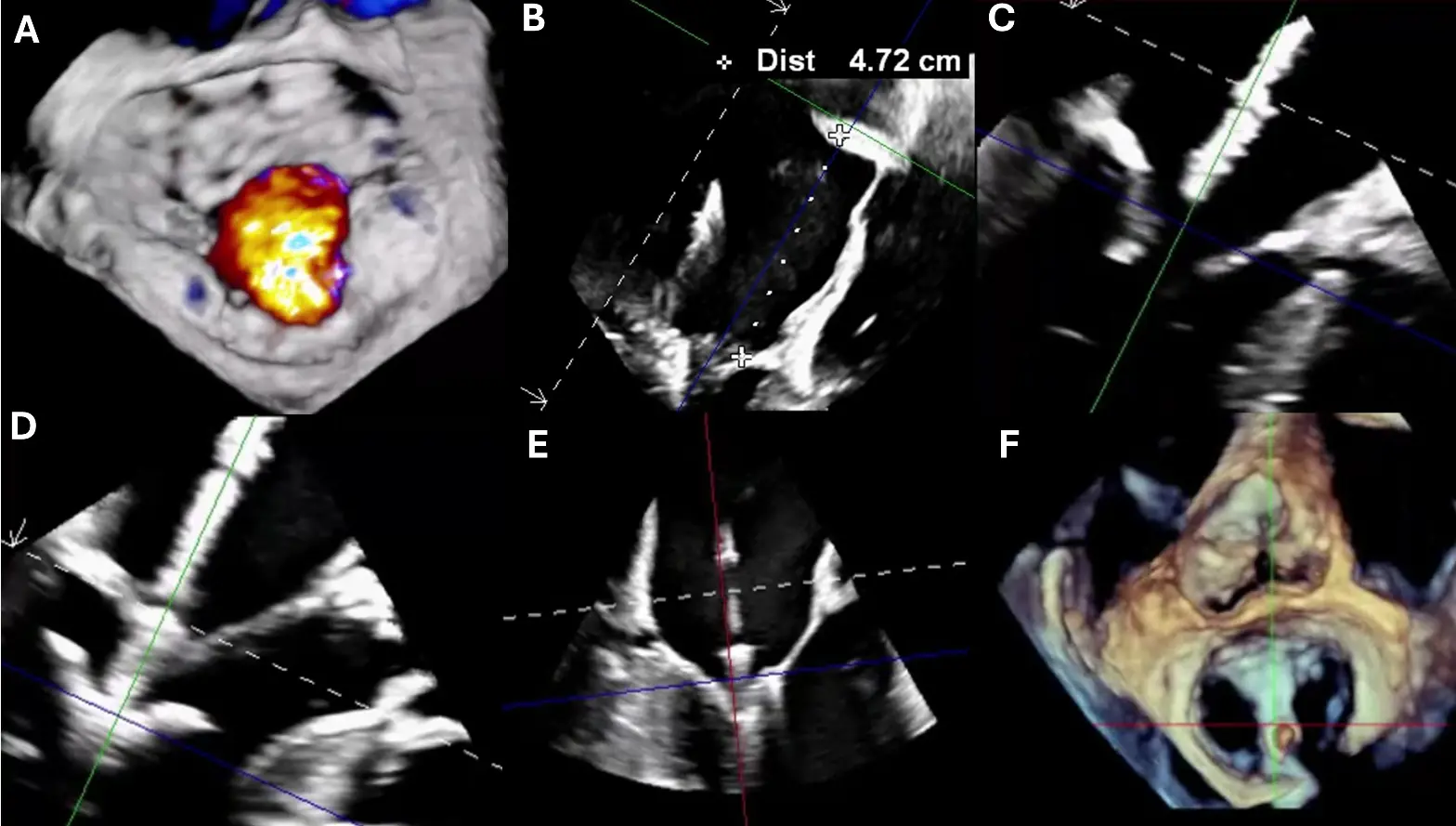
Figure 5. Transesophageal Echocardiographic Guidance During MitraClip Procedure for Severe Mitral Regurgitation. (A) Three-dimensional multiplanar reconstruction (MPR) of the mitral valve from the left atrial (surgeon’s) view demonstrating severe mitral regurgitation (aliasing/mosaic pattern consistent with high-velocity regurgitant flow). (B) Bi-caval TEE view showing successful transseptal puncture with a posterior and superior approach; height from the mitral annular plane measured at >4 cm, appropriate for clip delivery. (C) TEE long-axis view illustrating the MitraClip device crossing the mitral valve into the left ventricle. (D) Device arms fully opened within the left ventricle prior to leaflet capture. (E) Leaflet grasping with approximation of the anterior and posterior leaflets under real-time TEE guidance. (F) Final post-deployment result with stable clip position. Colored lines denote orthogonal 3D multiplanar reconstruction reference planes; white ‘+’ markers indicate caliper/cursor points; the ‘Dist’ line reports the measured transseptal height; dashed white lines outline the imaging sector/3D cropping or MPR slice limits; color Doppler shows the MR jet.
5.3. Cardiac Magnetic Resonance
Cardiac magnetic resonance (CMR) imaging offers a comprehensive, high-resolution, and reproducible assessment of both cardiac chambers and the mitral valve apparatus, making it an invaluable tool for evaluating FMR (Figure 6) [88,89,90,91]. As the reference standard for quantifying left ventricular and atrial volumes, systolic function, and remodeling, CMR plays an increasingly central role in the diagnosis, risk stratification, and longitudinal monitoring of patients with FMR [37,68,92,93].
One of the key advantages of CMR is its ability to quantify mitral regurgitant volume and fraction with high accuracy and low operator dependency [88,90]. This is achieved by subtracting aortic forward stroke volume, measured via phase-contrast velocity mapping, from the total left ventricular stroke volume derived from cine images [38,88,94,95]. The resulting regurgitant fraction (≥35% is often considered severe) is highly reproducible and particularly helpful when echocardiographic assessments are technically limited or yield discordant findings [78,96]. These volumetric parameters provide a robust estimate of MR severity and reflect its hemodynamic burden over time, thereby aiding in therapeutic decision-making [78,96].
Emerging 4D flow CMR techniques allow for direct visualization and quantification of complex regurgitant jets, including multiple or eccentric jets, and enable time-resolved assessment of regurgitant flow throughout systole [97]. While 4D flow offers theoretical advantages in capturing complex MR geometry, its use remains limited by longer acquisition times, post-processing complexity, and lack of standardized thresholds [98]; thus, it is currently best viewed as complementary to established indirect volumetric methods rather than a replacement in routine clinical practice.
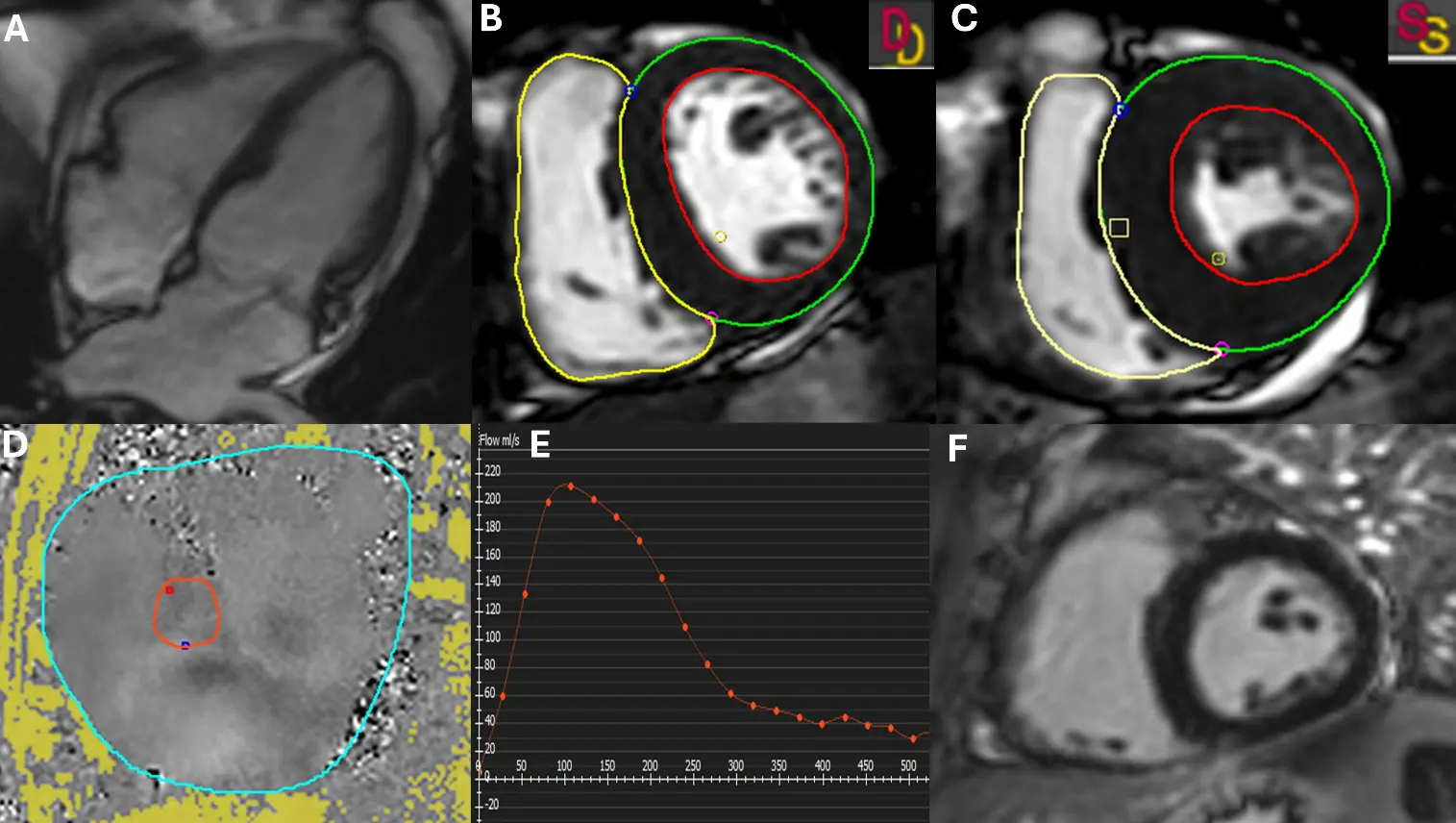
Figure 6. Cardiac Magnetic Resonance Evaluation of Mitral Regurgitation. (A) Cine four-chamber view demonstrating a systolic mitral regurgitant jet extending into the left atrium. (B,C) Short-axis stack at end-diastole (B) and end-systole (C) with manual contouring of the left ventricle to calculate end-diastolic volume (EDV), end-systolic volume (ESV), and stroke volume (SV). LV endocardial and epicardial contours are shown in red and green, respectively; the RV endocardial contour is shown in yellow. ED and ES frames are indicated (D = end-diastole; S = end-systole). (D) 2D phase-contrast flow imaging acquired at the sinotubular junction (STJ) for quantification of aortic forward flow. The aortic lumen ROI used for flow integration is outlined in red, and the reference/background (phase-offset correction) ROI is outlined in cyan. (E) Corresponding flow curve generated from (D) used to calculate regurgitant volume (RV = aortic flow − SV) and regurgitant fraction (RF = RV/SV). The orange curve/markers represent instantaneous aortic flow across the cardiac cycle (positive = forward flow). (F) Late gadolinium enhancement (LGE) imaging demonstrates no evidence of myocardial fibrosis or scar.
CMR is also exceptional at defining the underlying FMR mechanism [38,90,91]. In ventricular-driven FMR, it typically reveals left ventricular dilation, impaired systolic function, and regional wall motion abnormalities [1,41,99]. It can precisely visualize apical papillary muscle displacement and leaflet tethering, correlating well with echocardiographic findings [1,99]. In contrast, atrial-driven FMR exhibits a distinctly different pattern, characterized by significant left atrial and annular enlargement, while the left ventricle remains structurally and functionally preserved [4,7,58]. CMR may also visualize subtle forms of “atriogenic” leaflet tethering, in which posterior leaflet angulation arises from posterior annular displacement, reinforcing the mechanical nature of AFMR [38,90,91].
CMR’s most notable contribution is the characterization of myocardial tissue using late gadolinium enhancement (LGE) and T1 mapping techniques [68]. LGE detects replacement fibrosis (scar), which is typically focal and associated with prior infarction or advanced remodeling [100]. In FMR, the presence and extent of LGE are independently associated with adverse outcomes, including mortality and heart failure hospitalization [101]. For example, Wang et al. demonstrated that in both ischemic (ICM) and nonischemic (NICM) cardiomyopathy, LGE (≥5% in ICM or ≥2% in NICM) predicted worse event-free survival, with the combination of high MR fraction and LGE conferring an additive risk [102]. Similarly, Badau Riebel et al. found that replacement fibrosis detected by LGE was a strong independent predictor of adverse outcomes (HR 1.83) [103].
In addition, T1 mapping and extracellular volume (ECV) quantification enable the detection of diffuse interstitial fibrosis, which often precedes overt systolic dysfunction and is not visualized by LGE [104]. Elevated native T1 and ECV values are associated with adverse remodeling and worse prognosis in MR [105,106]. Badau Riebel et al. also demonstrated that interstitial fibrosis, as identified by T1 mapping, was an independent predictor of adverse outcomes (HR 1.61) [103]. The prognostic value of this tissue characterization is substantial: both replacement and interstitial fibrosis are associated with less improvement in LV function and symptoms after intervention [103,106]. These findings can thus inform the timing and selection of therapy, identifying high-risk patients who might benefit from earlier or alternative strategies [68,107].
Taken together, the strengths of CMR lie not only in its volumetric precision and reproducibility but also in its ability to integrate structural, functional, and tissue-based assessments in a single modality. As such, CMR serves as a valuable adjunct to echocardiography, particularly when discrepancies exist or when detailed anatomical and myocardial characterization is essential for guiding therapy. Its contributions are especially critical in distinguishing phenotypes of FMR, determining chronicity, and identifying candidates for valve-directed versus rhythm- or remodeling-focused therapies.
5.4. Computed Tomography
Cardiac computed tomography (CT) has emerged as a complementary modality for the structural assessment of FMR, providing high-resolution, three-dimensional visualization of the entire mitral apparatus—including the annulus, leaflets, chordae, and papillary muscles—with submillimeter spatial accuracy (Figure 7 and Figure 8) [108]. While three-dimensional echocardiography remains central to FMR evaluation, CT is widely regarded as the reference standard for mitral annular quantification and offers unique advantages in characterizing complex mitral geometry [109]. Multiphase CT reconstructions allow precise assessment of annular area, inter-commissural and septo-lateral diameters, annular nonplanarity, leaflet length, tenting height, and papillary muscle position throughout the cardiac cycle [110,111].
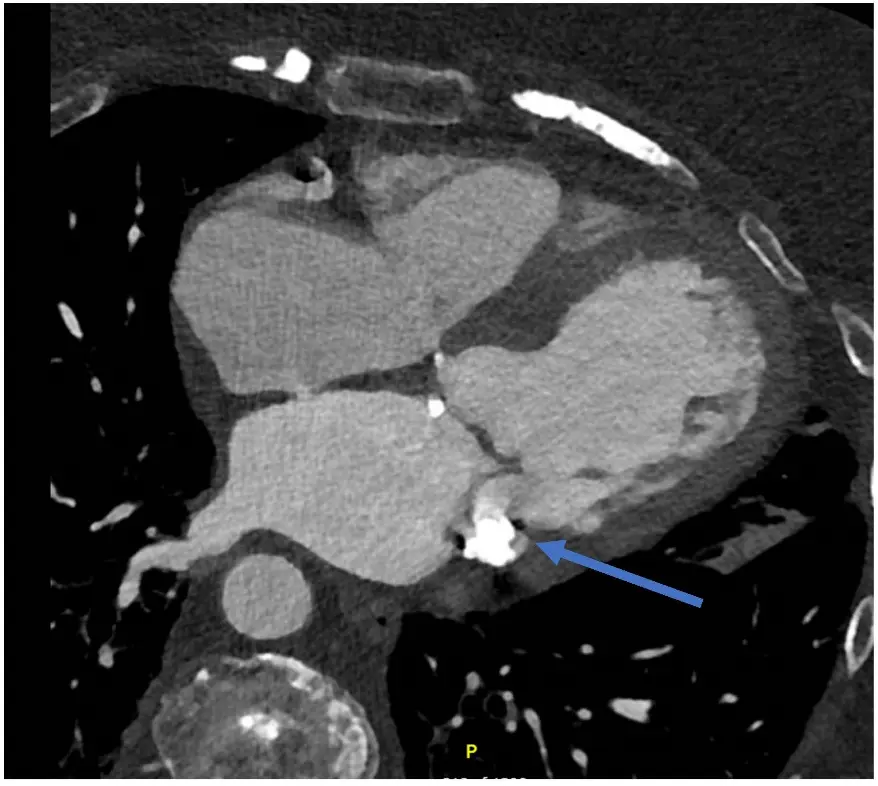
Figure 7. Cardiac Computed Tomography (CT) for Anatomy Evaluation. CT cardiac offers good temporal and spatial resolution for evaluation of cardiac structures and understanding the FMR mechanism and leaflet anatomy (including annulus, any mitral annular calcification, relation to LVOT). This image shows left atrial dilatation and LV dilatation. There is evidence of mitral annular calcification (indicated by the blue arrow).
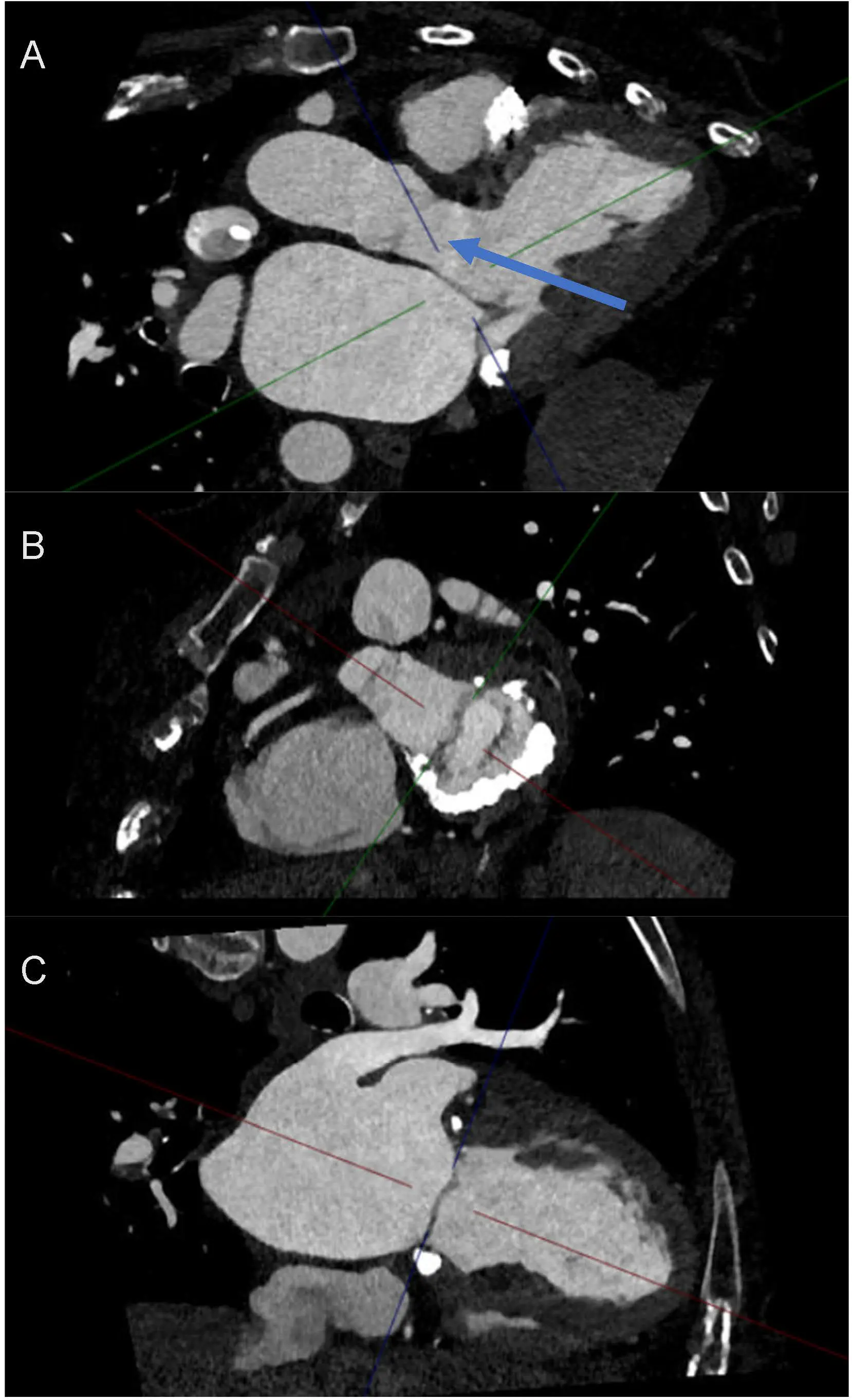
Figure 8. Use of Cardiac Computed Tomography in Functional Mitral Regurgitation Imaging. (A) 3 chamber view in relation to the mitral valve and the importance of evaluating LVOT (indicated by blue arrow) proximity to mitral valve intervention. (B) Short axis of mitral valve and appreciation of prohibitive anatomy for transcatheter or surgical intervention. This patient has significant mitral annular calcification, but the posterior leaflet still remains mobile. Loss of mitral valve saddle shape is predominantly due to the atrial functional mechanism. (C) 2-chamber view of the left atrium and left ventricle. Colored lines represent multiplanar reformation reference planes (crosshairs) displayed by the CT workstation.
Importantly, CT provides incremental value in phenotyping FMR mechanisms, particularly in differentiating ventricular from atrial functional MR. In VFMR, CT readily demonstrates LV dilation, papillary muscle displacement, leaflet tethering, and increased tenting geometry [22], whereas AFMR is characterized by marked annular dilatation, annular flattening, and preserved papillary muscle geometry in the setting of normal or near-normal LV size [4]. These distinctions are especially relevant in borderline or mixed phenotypes, where accurate mechanistic classification directly informs downstream management strategies.
CT is also indispensable for procedural planning of transcatheter mitral interventions [112,113]. In the context of transcatheter mitral valve replacement (TMVR), CT enables detailed prosthesis simulation, virtual valve deployment, and accurate calculation of the neo–left ventricular outflow tract (neo-LVOT). A projected neo-LVOT area <2.0 cm2 identifies patients at high risk for LVOT obstruction and strongly influences candidacy, device selection, and the need for adjunctive techniques such as intentional laceration of the anterior mitral leaflet (LAMPOON) [114,115]. The same datasets provide critical information on leaflet anatomy, annular dimensions, and the presence, distribution, and severity of mitral annular calcification (MAC), all of which impact the feasibility and durability of both TEER and TMVR [116,117]. In selected cases, CT planimetry of the anatomic regurgitant orifice area can further refine MR severity assessment, particularly when echocardiographic grading is limited by eccentric or multiple jets.
Finally, coronary computed tomography angiography (CCTA) plays an important adjunctive role in patients with suspected ischemic VFMR, where identification of significant coronary artery disease directly informs the need for concomitant revascularization as part of a combined surgical or hybrid strategy [32,100,118,119]. In contrast, broader assessment of extracardiac aortic pathology or general redo sternotomy planning, while valuable in selected surgical cases, is typically secondary to CT’s primary role in defining FMR mechanism and guiding mitral-specific intervention.
In summary, cardiac CT offers unparalleled anatomical detail and quantitative precision for FMR evaluation, with particular strength in mechanistic phenotyping, transcatheter procedural planning, and ischemic substrate assessment. When integrated with echocardiography and cardiac magnetic resonance imaging, CT provides essential complementary information that directly informs patient selection and procedural strategy in contemporary FMR management (Table 2).
Table 2. Diagnostic Imaging Modalities for Functional Mitral Regurgitation.
|
Diagnostic Tool |
Role in FMR Assessment |
MR Severity Quantification |
Strengths |
Limitations |
Best Clinical Use |
|---|---|---|---|---|---|
|
Echocardiography (TTE/TEE, 2D/3D) |
First-line modality for diagnosis, mechanistic classification (VFMR vs. AFMR), hemodynamic assessment, and longitudinal follow-up; essential for procedural guidance |
Integrative multiparametric grading using qualitative (jet morphology), semiquantitative (vena contracta width, pulmonary vein flow), and quantitative measures (EROA, regurgitant volume, regurgitant fraction) |
Widely available; real-time; assesses valve/ventricle/annulus; Doppler quantifies MR; 3D improves annular and leaflet geometry assessment |
Image quality dependent; limited by acoustic windows; Doppler parameters affected by loading; 2D may underestimate complex jets; TEE semi-invasive |
Initial diagnosis; serial follow-up; procedural guidance for TEER; dynamic assessment (stress echo) |
|
Cardiac Magnetic Resonance (CMR) |
Quantifies MR severity (regurgitant volume/fraction); precise LV/LA volumes; detects scar/fibrosis |
Indirect volumetric method: LV stroke volume (cine) minus aortic forward flow (phase-contrast); regurgitant fraction ≥35% often considered severe; 4D: direct time-resolved visualization and quantification of regurgitant jets |
Gold standard for MR quantification; geometry-independent quantification; reproducible; accurate in eccentric or multiple jets; comprehensive LV/LA assessment; tissue characterization; 4D captures complex, multiplanar, and eccentric jets; full volumetric flow mapping |
Limited availability; contraindications (e.g., devices); less spatial/temporal resolution for leaflets; not real-time for intervention guidance; longer acquisition time; 4D flow lacks standardized thresholds and widespread adoption |
Discordant or inconclusive echocardiography; prognostic stratification; assessment of remodeling and intervention candidacy; Selected cases or research settings with complex jet morphology |
|
Computed Tomography (CT) |
Anatomic assessment for intervention planning; annular sizing; access route |
Not a primary modality for functional MR severity; optional anatomic regurgitant orifice planimetry in selected cases with complex jets; Neo-LVOT prediction; annular sizing; MAC burden assessment |
Reference standard for annular geometry; excellent spatial resolution; distinguishes VFMR vs. AFMR anatomy; virtual valve deployment; accurate neo-LVOT calculation; device feasibility assessment |
Lacks hemodynamic assessment; radiation and contrast exposure; static assessment of a dynamic process; does not quantify functional severity; requires specialized software |
Phenotyping FMR mechanism; TMVR/TEER planning; annular and leaflet anatomy assessment |
Abbreviation: TTE = Transthoracic echocardiography; TEE = Transesophageal echocardiography; 2D = Two-dimensional; 3D = Three-dimensional; MR = Mitral regurgitation; FMR = Functional mitral regurgitation; VFMR = Ventricular functional mitral regurgitation; AFMR = Atrial functional mitral regurgitation; LV = Left ventricle/ventricular; LA = Left atrium/atrial; EROA = Effective regurgitant orifice area; CMR = Cardiac magnetic resonance; CT = Computed tomography; TMVR = Transcatheter mitral valve replacement; TEER = Transcatheter edge-to-edge repair; MAC = Mitral annular calcification; neo-LVOT = Neo–left ventricular outflow tract; 4D = Four-dimensional.
5.5. Standardization of Imaging Assessment
As listed above, there are many imaging modalities that prove useful for FMR assessment. Table 2 summarizes the different modalities described above, along with their strengths and limitations in FMR assessment.
FMR is inherently dynamic, with severity varying substantially according to loading conditions, heart rate, rhythm, and ventricular–atrial interactions [2]. Changes in preload, afterload, and contractility can meaningfully alter regurgitant volume and effective regurgitant orifice area over short time intervals, contributing to variability across imaging studies and potential misclassification of severity [5]. Given this marked load dependence, careful standardization of imaging conditions is essential to ensure that measured MR severity reflects the patient’s “true” clinical state rather than transient hemodynamic perturbations [38]. Whenever feasible, MR assessment should be performed under stable, near–euvolemic conditions, ideally after optimization of guideline-directed medical therapy [37]. Imaging performed during acute decompensation, excessive diuresis, dehydration, or uncontrolled hypertension may either exaggerate or underestimate regurgitation severity and should be interpreted with caution [37].
Practical steps to optimize and standardize imaging assessment include: (1) avoiding MR quantification during periods of rapid volume shifts (e.g., immediately after aggressive diuresis or intravenous fluid administration) [37]; (2) documenting blood pressure, heart rate, and rhythm at the time of imaging [37]; and (3) ensuring adequate blood pressure control, as acute elevations in afterload can disproportionately increase regurgitant severity [37]. In patients with atrial fibrillation, averaging measurements over multiple cardiac cycles is essential, and echocardiographic assessment should ideally be performed at a controlled ventricular rate [37]. Medication status should also be considered and reported. Imaging obtained before initiation or up-titration of vasodilators, diuretics, or rhythm-control therapies may not reflect steady-state conditions [32]. When serial imaging is used to guide management decisions, efforts should be made to perform studies under comparable hemodynamic and pharmacologic conditions to allow meaningful longitudinal comparison [38]. Stress echocardiography further highlights the dynamic nature of FMR by unmasking exertional increases in regurgitation severity and pulmonary pressures that may not be evident at rest [38]. However, resting MR severity used to guide interventional decisions should be grounded in standardized, clinically representative conditions rather than extreme loading states [38].
Taken together, recognition of FMR dynamism must be coupled with deliberate standardization of imaging acquisition and interpretation. Explicit documentation of loading conditions and thoughtful patient preparation reduces the risk of misclassification, improves reproducibility, and enhances the clinical relevance of imaging-based decision-making.
6. Therapeutic Modalities for Functional Mitral Regurgitation
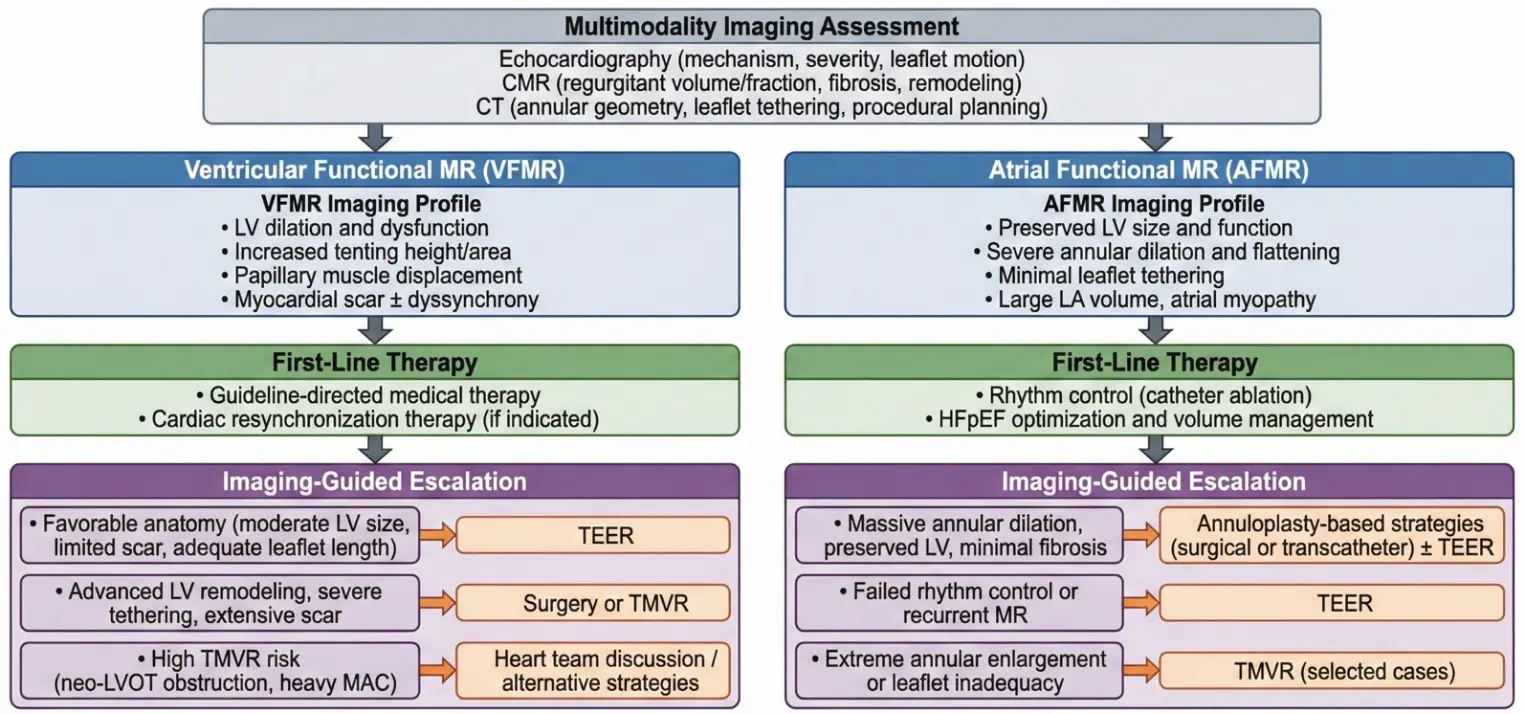
Treatment modalities for FMR vary significantly in patient selection, mechanism of action, supporting evidence, and clinical outcomes (Table 3). Figure 4 summarizes an imaging-guided management algorithm distinguishing VFMR from AFMR and illustrates how specific imaging phenotypes inform selection among medical therapy, TEER, surgery, and TMVR.
Table 3. Overview of Therapeutic Modalities for Functional Mitral Regurgitation.
|
Therapy |
Typical Candidates |
Mechanism |
Key Evidence/Trials |
Clinical Outcomes |
Limitations |
|---|---|---|---|---|---|
|
Medical and Device Optimization |
All with FMR; first-line for HFrEF; CRT for LBBB/QRS ≥ 150 ms; rhythm control for AFMR |
GDMT (beta-blocker, RAASi, ARNI, MRA, SGLT2i), CRT, AF ablation |
Multiple RCTs in HFrEF; observational in FMR |
Reduces MR severity by 40–45%, improves survival, and HF hospitalization |
Many remain symptomatic; MR often persists |
|
Surgical Repair or Replacement |
Severe, symptomatic FMR refractory to GDMT, or if other cardiac surgery is indicated |
Annuloplasty (repair) or chordal-sparing replacement |
CTSN SMR; observational studies |
No survival benefit over GDMT; repair: high recurrence; replacement: less MR recurrence |
High perioperative risk; no clear survival benefit in isolated FMR |
|
Transcatheter Edge-to-Edge Repair |
Severe, symptomatic FMR, LVEF 20–50%, persistent symptoms after GDMT, not surgical candidate |
MitraClip (edge-to-edge leaflet approximation) |
COAPT, MITRA-FR, RESHAPE-HF2 |
COAPT/RESHAPE-HF2: reduce HF hospitalization and mortality, improved QoL. MITRA-FR: neutral |
Benefit depends on MR/LV proportion, anatomy, operator experience |
|
Indirect Coronary Sinus Annuloplasty |
Symptomatic FMR, annular dilation, suitable coronary sinus anatomy |
Carillon device (annular cinching via coronary sinus) |
AMADEUS, TITAN, REDUCE-FMR |
Reduces MR, LV volumes, improves 6-min walk distance |
Anatomic variability, risk of circumflex impingement, less MR reduction than TEER |
|
Direct Annuloplasty (Cardioband, Millipede) |
Symptomatic FMR, annular dilation, high surgical risk, suitable anatomy |
Percutaneous direct annular reduction |
Cardioband feasibility, comparative studies |
Reduces annular size, MR, improves NYHA/6-min walk distance; similar or better functional outcomes vs. TEER in select patients |
Limited RCT data, device availability, learning curve |
|
Transcatheter Mitral Valve Replacement |
Severe, symptomatic FMR, high/prohibitive surgical risk, unsuitable for TEER/annuloplasty |
Transcatheter bioprosthesis (Tendyne, Intrepid) |
Tendyne, Intrepid, registries |
High device success, near-complete MR elimination, improved symptoms |
High procedural risk, LVOT obstruction risk; high early mortality in high-risk; limited long-term data |
Abbreviations: FMR = functional mitral regurgitation; HFrEF = heart failure with reduced ejection fraction; CRT = cardiac resynchronization therapy; LBBB = left bundle branch block; QRS = QRS duration; AFMR = atrial functional mitral regurgitation; GDMT = guideline-directed medical therapy; RAASi = renin-angiotensin-aldosterone system inhibitor; ARNI = angiotensin receptor-neprilysin inhibitor; MRA = mineralocorticoid receptor antagonist; SGLT2i = sodium-glucose cotransporter 2 inhibitor; MR = mitral regurgitation; HF = heart failure; TEER = transcatheter edge-to-edge repair; QoL = quality of life; LV = left ventricle; LVOT = left ventricular outflow tract.
6.1. Medical and Device Optimization
Medical and device-based optimization forms the foundation of management in FMR, with the overarching goals of relieving symptoms, reducing mitral regurgitant volume, and delaying or potentially avoiding invasive intervention [21,120]. Both the American College of Cardiology (ACC) and the Heart Failure Association of the European Society of Cardiology (ESC-HFA) emphasize that therapy should begin with comprehensive GDMT for heart failure, with escalation to device or interventional strategies reserved for patients who remain symptomatic despite optimal medical management [9,14,47].
In patients with FMR and reduced ejection fraction, GDMT represents the first-line approach [68]. According to joint recommendations from the ACC, American Heart Association (AHA), and Heart Failure Society of America (HFSA), all eligible patients should receive a combination of the following: a beta-blocker, a renin-angiotensin system inhibitor (ARNI preferred, or ACEi/ARB if ARNI is not tolerated), a mineralocorticoid receptor antagonist (spironolactone or eplerenone), and a sodium-glucose cotransporter 2 inhibitor (SGLT2i) [68]. These agents not only improve survival and reduce heart failure hospitalizations but have also been shown to directly impact the severity of FMR in many cases [63]. By promoting reverse remodeling, these medications reduce left ventricular volumes and loading conditions and improve geometry, often leading to a reduction in regurgitant severity [68].
Device-based therapies are crucial adjuncts in selected patients [2]. In particular, cardiac resynchronization therapy (CRT) is indicated for individuals with an LVEF ≤ 35%, sinus rhythm, left bundle branch block, and a QRS duration of ≥150 ms [121,122,123]. CRT improves left ventricular synchrony, facilitates reverse remodeling, and has been shown to reduce the degree of FMR [2,46,124,125,126]. For patients with AFMR complicated by AF, rhythm control strategies, including antiarrhythmic medications and catheter ablation, can reduce left atrial size and improve annular function, thereby mitigating the severity of regurgitation [7,127,128].
In ischemic FMR, coronary revascularization (either PCI or CABG) plays a crucial role [74]. Many patients with moderate ischemic MR experience MR improvement after revascularization alone, especially with CABG, which is associated with better event-free survival compared to PCI [129,130]. Concomitant mitral annuloplasty with CABG further improves outcomes in selected patients [130,131]. Decision-making should be multidisciplinary, considering comorbidities, operative risk, and individual patient factors [74,132].
6.2. Surgical Interventions
Surgical treatment for FMR is generally reserved for patients who remain symptomatic despite maximally optimized GDMT and, when appropriate, CRT [32,133]. According to current ACC/AHA recommendations, surgery is indicated in patients with severe FMR, whether of ventricular or atrial origin, primarily when concomitant cardiac surgery, such as coronary artery bypass grafting (CABG), is already planned [32]. Isolated mitral surgery for FMR is less commonly performed due to the lack of a proven survival benefit and the high rates of recurrence [32]. Therefore, patient selection is a nuanced process that requires a multidisciplinary approach [32,37,134].
The management of Ischemic FMR (VFMR) has been heavily influenced by two landmark randomized trials from the Cardiothoracic Surgical Trials Network (CTSN) published in The New England Journal of Medicine, which clarified the role of surgery in both moderate and severe disease (Table 4) [135,136]. For moderate ischemic MR, the trial by Smith et al. (2014) randomized 301 patients undergoing CABG to receive either CABG alone or CABG plus mitral valve repair (restrictive annuloplasty) [135]. At one year, the addition of mitral repair did not improve survival or left ventricular reverse remodeling [135]. While the combined group had less residual MR, this did not translate into improved clinical outcomes [135,136]. Furthermore, the addition of repair increased operative time, hospital length of stay, and the risk of neurologic events and arrhythmias [74,135,136]. Consequently, for patients with moderate ischemic MR, CABG alone is generally considered sufficient, as revascularization often reduces MR severity without the added morbidity of valve intervention [2,32,74,135,137]. For severe ischemic MR, the choice between repair and replacement was addressed by Acker et al. (2014) and Goldstein et al. (2016) [138,139]. This trial randomized 251 patients to either restrictive mitral annuloplasty (RMA), using a downsized rigid ring, or chordal-sparing mitral valve replacement [138]. The results highlighted a critical distinction between functional and primary MR [2]. Unlike primary MR, where repair is superior, in FMR, repair demonstrated poor durability [2,138,139]. At two years, the repair group had a significantly higher rate of recurrent moderate-to-severe MR (58.8% vs. 3.8%) and more heart failure-related adverse events compared to the replacement group [139]. However, there was no significant difference in survival between the two strategies [138,139]. These findings have shaped contemporary technical considerations. Repair remains an option for patients with mild tethering and less advanced ventricular remodeling, potentially favored in younger patients (<65 years) to avoid the risks of prosthetic valves [140]. However, because repair failure is driven by ongoing ventricular remodeling and leaflet tethering, chordal-sparing replacement is now generally preferred for patients with predictors of repair failure [2,32]. These predictors include a tenting height >10 mm, a posterior leaflet angle >45°, or significant infero-basal wall motion abnormalities [3,37]. Replacement in these high-risk anatomies provides a more durable correction of MR and fewer heart failure readmissions [2,139].
In contrast to the ventricular phenotype, AFMR requires a different surgical logic. Because the leaflets are typically structurally normal and regurgitation is driven by annular dilatation, isolated ring annuloplasty is highly effective and durable [27,141,142,143,144,145]. In the largest contemporary series, annuloplasty alone yielded a five-year survival rate of approximately 75% with low recurrence rates [141,142,144,145]. However, surgical success in AFMR often requires addressing the accompanying atrial pathology; standard practice includes concomitant Cox-Maze ablation and rhythm control strategies for atrial fibrillation, such as ablation or reducing afterload conditions that may contribute to elevated left ventricular/atrial filling pressures [111,142,143,146].
In summary, surgical management of FMR is mechanism specific. While AFMR is reliably treated with annuloplasty, moderate ischemic VFMR is often best managed with revascularization alone, and severe VFMR frequently necessitates valve replacement to ensure durability. Regardless of the technique, the decision to operate must weigh the limited survival benefit against the risks, prioritizing symptom relief and quality of life.
Table 4. Key Randomized Controlled Trials of Surgical Intervention for Functional Mitral Regurgitation.
|
Year |
Trial (N) |
Intervention |
Comparator |
Primary Outcome |
Result Summary |
Quality Considerations |
|---|---|---|---|---|---|---|
|
2014 |
CTSN Moderate IMR Trial (N = 301) |
CABG + mitral valve repair (restrictive annuloplasty) |
CABG alone |
LVESVI at 2 years |
No significant difference in LVESVI (41.2 vs. 43.2 mL/m2) or survival (10.0% vs. 10.6% mortality). Combined procedure reduced recurrent moderate-to-severe MR (11.2% vs. 32.3%, p < 0.001) but increased neurologic events and supraventricular arrhythmias. |
Multicenter RCT from CTSN; stopped early after interim analysis showed no benefit in the primary endpoint; patients had moderate IMR (effective regurgitant orifice area 0.2–0.4 cm2) |
|
2014 |
CTSN Severe IMR Trial (N = 251) |
Chordal-sparing mitral valve replacement |
Restrictive mitral annuloplasty (downsized rigid ring) |
LVESVI at 2 years |
No significant difference in LVESVI or survival (19.0% vs. 23.2% mortality, p = 0.39). Replacement had significantly lower recurrent moderate-to-severe MR (3.8% vs. 58.8%, p < 0.001), fewer heart failure-related adverse events (p = 0.05), and fewer cardiovascular readmissions (p = 0.01). |
Multicenter RCT from CTSN; patients had severe IMR (mean effective regurgitant orifice area 0.4 cm2); concomitant CABG was performed when indicated |
Abbreviations: CTSN = Cardiothoracic Surgical Trials Network; IMR = Ischemic mitral regurgitation; CABG = Coronary artery bypass grafting; LVESVI = Left ventricular end-systolic volume index; MR = Mitral regurgitation; RCT = Randomized controlled trial.
6.3. Transcatheter Interventions
6.3.1. Transcatheter Edge-to-Edge Repair (TEER)
TEER has become a cornerstone intervention for selected patients with FMR who remain symptomatic despite maximally optimized GDMT [12,32,37]. Applicable to both VFMR and AFMR subtypes, TEER offers a less invasive alternative to surgery for patients with prohibitive operative risk [32,37].
The American College of Cardiology recommends TEER in patients with severe, symptomatic FMR and a left ventricular ejection fraction (LVEF) between 20–50%, left ventricular end-systolic dimension <7.0 cm, pulmonary artery systolic pressure <70 mmHg, and effective regurgitant orifice area (EROA) ≥0.3 cm2, aligning with the inclusion criteria of the landmark COAPT trial [32,37]. These recommendations are made in the context of a multidisciplinary heart team decision, informed by comprehensive multimodality imaging to ensure anatomical suitability, specifically adequate leaflet length, coaptation depth, and a mitral valve area ≥4.0 cm2, using anatomical criteria originally derived from the EVEREST trials (though notably, EVEREST II primarily studied degenerative MR) [37].
The procedure is performed under general anesthesia with continuous transesophageal echocardiography (TEE) and fluoroscopic guidance [74,147]. Devices such as the MitraClip (Abbott) are advanced transseptally across the mitral valve, where the anterior and posterior leaflets are grasped at the origin of the regurgitant jet, creating a double-orifice valve that reduces regurgitation [2,147]. Multiple clips may be deployed as needed to optimize the result [74,147]. The clinical evidence base for TEER in FMR is defined by three pivotal randomized trials, MITRA-FR, COAPT, and RESHAPE-HF2, which yielded differing results driven by distinct patient selection strategies (Table 5) [2,12,13,15,37,68,123,148].
Table 5. Key Randomized Controlled Trials of TEER for Functional Mitral Regurgitation.
|
Year |
Trial (N) |
Intervention |
Comparator |
Primary Outcome |
Result Summary |
Quality Considerations |
|---|---|---|---|---|---|---|
|
2018 |
MITRA-FR (304) |
TEER (MitraClip) + GDMT |
GDMT alone |
Composite of all-cause death or unplanned HF hospitalization at 12 months |
No significant difference (54.6% vs. 51.3%, HR 1.16, 95% CI 0.73–1.84) |
Broad MR inclusion, larger LV volumes, less severe MR, and less intensive GDMT |
|
2018 |
COAPT (614) |
TEER (MitraClip) + GDMT |
GDMT alone |
HF hospitalization 24 months |
Significant reduction (35.8% vs. 67.9%, HR 0.53, 95% CI 0.40–0.70); all-cause mortality also reduced (29.1% vs. 46.1%, HR 0.62, 95% CI 0.46–0.82) |
Rigorous GDMT, more severe MR, smaller LV, strict selection, high procedural success |
|
2024 |
RESHAPE-HF2 (505) |
TEER (MitraClip) + GDMT |
GDMT alone |
Composite of first/recurrent HF hospitalization or CV death at 24 months |
Significant reduction (rate ratio 0.64, 95% CI 0.48–0.85, p = 0.002); improved KCCQ-OS, lower recurrent HF hospitalizations |
Broader MR severity, international sites, robust endpoints, and low device-related adverse events |
Abbreviations: TEER = Transcatheter edge-to-edge repair; MR = Mitral regurgitation; GDMT = Guideline-directed medical therapy; HF = Heart failure; LV = Left ventricle/ventricular; CV = Cardiovascular; HR = Hazard ratio; CI = Confidence interval; KCCQ-OS = Kansas City Cardiomyopathy Questionnaire–Overall Summary score.
The MITRA-FR trial randomized 304 patients with severe FMR and heart failure to MitraClip plus medical therapy versus medical therapy alone [16]. The trial found no significant difference in the composite endpoint of all-cause death or unplanned heart failure hospitalization at 1 or 2 years [16]. A critical analysis of the cohort reveals that MITRA-FR patients had larger LV volumes but less severe MR (mean EROA 0.31 cm2) relative to that ventricular size, a phenotype often described as “proportionate” MR [44,45,149]. Furthermore, the optimization of GDMT prior to enrollment was less stringent than in subsequent trials [45,150]. These factors suggest that in patients where MR is merely a biomarker of advanced ventricular disease rather than a driver, valve intervention may be futile [10,16].
In contrast, the COAPT trial enrolled 614 patients with symptomatic heart failure and moderate-to-severe or severe FMR who remained symptomatic despite a rigorous “run-in” period of maximally tolerated GDMT [48,151]. In this cohort, TEER with MitraClip led to significant reductions in heart failure hospitalizations (HR 0.53) and all-cause mortality (HR 0.62) at 24 months compared to GDMT alone, with durable benefits observed up to 36 months, along with improved quality of life [48,151]. The COAPT population differed fundamentally from MITRA-FR: patients had more severe MR (mean EROA 0.41 cm2) but less severe LV dilation [44]. This “disproportionate” MR phenotype implies that the valvular lesion contributed excessively to the pathophysiology, making it a prime target for repair [44].
Most recently, the RESHAPE-HF2 trial has expanded the evidence base. It included 505 patients with moderate-to-severe FMR (mean EROA 0.25 cm2) and symptomatic heart failure [13]. TEER significantly reduced first and recurrent heart failure hospitalizations and cardiovascular death at 24 months, with consistent benefits across subgroups [13,148,152]. Notably, the benefit was pronounced in patients with recent heart failure hospitalizations [148,152]. The results of RESHAPE-HF2 support the broader application of TEER, suggesting that clinical benefit extends to patients with MR severity that may be lower than the strict COAPT thresholds, provided they remain symptomatic despite medical therapy [13,152].
Real-world data, specifically regarding AFMR, also demonstrate encouraging outcomes. Registry data indicate procedural success rates ranging from 87% to 94%, with durable symptomatic improvement. In the Spanish Mitraclip series, 94% of AFMR patients had MR reduced to grade ≤2+, and 80% improved to a NYHA functional class I–II at 12 months [153,154]. The MITRA-TUNE registry reported mid-term MR recurrence in only 11% [155,156,157]. However, anatomical phenotype matters: patients with “hamstrung” or atriogenic tethering, characterized by massive atrial enlargement causing marked posterior leaflet angulation, often experience higher residual MR and worse outcomes, highlighting the need for careful anatomical screening [157,158].
Technological evolution continues to refine these outcomes. Beyond the MitraClip, the Edwards PASCAL system offers novel design features, including independent leaflet capture and a central spacer, which enable enhanced adaptation to complex anatomy [15,159]. In the CLASP study, PASCAL achieved MR grade ≤2+ in 96% of cases with a 92% one-year survival rate [160]. A head-to-head analysis from the CLASP IID randomized trial demonstrated non-inferiority of PASCAL compared to MitraClip in both safety (3.4% vs. 4.8% major events) and efficacy, with numerically higher rates of trace or no MR at six months (87% vs. 84%) [161,162,163].
Successful procedural planning relies heavily on 3D TEE and CT to assess jet location, leaflet length, and annular dimensions [164,165]. In VFMR, broad central jets may necessitate multiple clips, whereas AFMR often requires centrally placed devices, unless atriogenic tethering is present [164]. Residual trans-mitral gradients > 5 mmHg, severe annular calcification, and flail gaps > 10 mm remain relative contraindications [37,166]. Following TEER, particularly in AFMR, aggressive rhythm control may offer additional remodeling benefits; however, this effect diminishes in the setting of extensive atrial fibrosis [19,166]. Overall, TEER offers higher procedural success and lower morbidity than surgery, positioning it as the preferred intervention for appropriate candidates [12,15,152].
6.3.2. Indirect Coronary-Sinus Annuloplasty
Indirect coronary sinus annuloplasty represents a minimally invasive, anatomically guided strategy for treating FMR, exploiting the proximity of the coronary sinus (CS) and the great cardiac vein to the posterior mitral annulus [167,168]. Among available technologies, the Carillon Mitral Contour System is the most extensively studied device [13,52,167,169,170,171,172]. It is deployed percutaneously via the right internal jugular or femoral vein and implanted within the coronary sinus, where it applies circumferential tension to reduce the septal-lateral annular dimension, thereby enhancing leaflet coaptation and mitigating mitral regurgitation [167,169,170].
The physiological rationale for this technique lies in the unique anatomical relationship between the coronary sinus and the adjacent mitral annulus [173,174,175]. By anchoring and “cinching” the coronary sinus, the posterior annulus can be effectively constricted without entering the left atrium or ventricle [174]. However, this anatomical relationship varies among patients, and pre-procedural imaging plays a crucial role in determining procedural feasibility [176]. Optimal outcomes are associated with a coronary sinus-to-annulus distance of less than 7.8 mm and an angle of less than 14.2°, as determined by pre-procedural cardiac CT angiography [177]. In addition, the proximity of the coronary sinus to the left circumflex artery raises the risk of coronary compression, making detailed anatomic evaluation and intraprocedural monitoring essential to avoid vascular complications [173,174,175].
Clinical trials and pooled analyses have consistently shown that the Carillon device reduces MR severity, facilitates left ventricular reverse remodeling, and improves functional capacity, particularly in patients with proportionate FMR, in which annular dilation is the dominant pathophysiologic mechanism (Table 6) [13,167,170,171,172,178]. In this subgroup, indirect annuloplasty reduces left ventricular volumes and enhances forward stroke volume, providing symptomatic relief without the need for left heart access [52]. Among patients with AFMR, the technique has also proven safe and effective, demonstrating reductions in left atrial volume and improvements in NYHA functional class; however, the degree of MR reduction may be less dramatic than with TEER [142,144,176]. Importantly, the procedural success rate is high, and major adverse events are relatively infrequent if patients are appropriately selected [169,176,179].
Table 6. Clinical Studies of Transcatheter Direct Annuloplasty Devices for Functional Mitral Regurgitation.
|
Annuloplasty Type |
Year |
Trial (N) |
Intervention |
Comparator |
Primary Outcome |
Result Summary |
Quality Considerations |
|---|---|---|---|---|---|---|---|
|
Indirect |
2009 |
AMADEUS (48) |
Carillon device |
None (single arm) |
MR reduction (echo), 6MWD, QoL |
Significant reduction in MR (22–32% by echo), improved 6MWD and QoL at 6 months, 13% 30-day major adverse event rate |
Early feasibility, small n, anatomical exclusion, procedural learning curve |
|
Indirect |
2012 |
TITAN (53) |
Carillon device |
Device recapture (non-implant) |
Change in regurgitant volume, LV volumes, 6MWD |
Significant reduction in MR and LV volumes, improved 6MWD in implant group vs. recapture; 1.9% 30-day major adverse event |
Non-randomized, device recapture as comparator, moderate sample size |
|
Indirect |
2022 |
Rottländer et al. (41) |
Carillon device |
MitraClip |
MR reduction, LA volume, NYHA class |
Both safe/feasible; MitraClip greater MR reduction, Carillon greater LA volume reduction at 12 months |
Retrospective, single-center, small n, AFMR focus |
|
Direct |
2016 |
Maisano et al. (31) |
Cardioband |
None (single arm) |
Technical success, annular reduction, MR grade at 30 days |
100% device delivery; significant annular reduction; MR ≤ 2+ in 88% at 30 days; no device/procedure-related deaths |
Early feasibility, high-risk, short follow-up |
|
Direct |
2016 |
Nickenig et al. (31) |
Cardioband |
None (single arm) |
Annular reduction, MR grade, NYHA, 6MWD at 6–7 months |
Annular septolateral dimension ↓ > 30%; MR ≥ 3+ from 77% to 13.6%; NYHA III/IV from 95.5% to 18.2%; 6MWD ↑; 9.7% mort. at 7 months |
Feasibility, moderate sample, no control, short follow-up |
|
Direct |
2016 |
Nickenig et al. (71) |
Direct annuloplasty system |
None (single arm) |
Device success, MR reduction, LV remodeling at 6 months |
Device success 70%; MR grade ↓ by mean 1.3; annular and LV dimensions ↓; 6MWD and NYHA improved; 12.2% mortality at 6 month |
Feasibility, moderate sample, no control, short follow-up |
|
Direct |
2019 |
Messika-Zeitoun et al. (60) |
Cardioband |
None (multicenter) |
Technical/device/procedural success, MR, NYHA, QoL, 6MWD at 1 year |
Technical success 97%; device/procedural 72/68%; MR ≤ 2+ in 61% (95% of echo-followed); NYHA I/II 79%; QoL, 6MWD improved; 87% survival at 1 year |
Multicenter, moderate n, no control, device modification mid-study |
Abbreviations: MR = Mitral regurgitation; NYHA = New York Heart Association (functional class); 6MWD = 6-min walk distance; QoL = Quality of life; LV = Left ventricle/ventricular; ↓ = Decrease; ↑ = Increase.
However, the anatomical suitability of the coronary sinus remains a major determinant of procedural efficacy [168,173,175,176]. Not all patients have favorable CS anatomy, and suboptimal alignment or excessive CS annulus separation can limit the capacity for effective annular remodeling [173,175,176,180]. As such, comprehensive multimodality imaging, including echocardiography, cardiac CT, and, where necessary, coronary angiography, is indispensable for case planning [38,83,181]. In recognition of this, both the American Society of Echocardiography and the European Association of Echocardiography recommend detailed pre-procedural evaluation to assess MR severity, annular geometry, and the spatial relationship of the coronary sinus to both the mitral annulus and coronary arteries [38,83,181].
In summary, indirect coronary sinus annuloplasty is a promising, catheter-based intervention for patients with FMR, particularly those with annular-dominant pathology, including both proportionate VFMR and atrial FMR. It offers a safe and less invasive alternative to direct mitral interventions, especially in patients with contraindications to TEER or surgery. While early results are encouraging, ongoing clinical trials and longer-term follow-up are necessary to fully define its place in the therapeutic algorithm, particularly in relation to emerging transcatheter and surgical techniques.
6.3.3. Direct Annuloplasty (Cardioband, Millipede)
Direct annuloplasty techniques aim to replicate the surgical principle of annular reduction through a fully transcatheter approach [182]. This strategy directly targets annular dilatation, a central pathophysiologic driver in both atrial and select forms of ventricular FMR [182]. Among the devices currently under clinical evaluation, Cardioband (Edwards Lifesciences) and Millipede represent the most advanced platforms [182,183].
The Cardioband system is an adjustable, transcatheter direct annuloplasty device designed to mimic the effect of surgical ring annuloplasty [182,183]. Under combined fluoroscopic TEE guidance, the device is delivered transvenously and sequentially anchored along the posterior mitral annulus, from the lateral to the medial fibrous trigone [147,184,185]. Once in place, the band is cinched in the beating heart, dynamically reducing the septolateral annular dimension until optimal regurgitation reduction is achieved, while carefully monitoring trans-mitral gradient to avoid iatrogenic stenosis [184]. This real-time adjustability allows for individualized optimization of leaflet coaptation and hemodynamics [147,184,186]. Clinical studies have demonstrated that Cardioband implantation is both feasible and safe in high-risk patients, resulting in significant reductions in annular area, mitral regurgitation grade, and left ventricular volume, as well as improvements in NYHA class, exercise capacity, and quality of life at 6 to 12 months (Table 5) [184,185,187]. Device success rates across studies range from 70% to 97%, with most patients achieving moderate or less moderate MR at 1 year [185,187]. In a multicenter feasibility study involving 60 high-surgical-risk patients with moderate-to-severe secondary FMR, Cardioband achieved 87% one-year survival, 66% freedom from heart failure rehospitalization, and 78% freedom from mitral reintervention, with sustained MR reduction and symptomatic improvement [187]. Follow-up registries have confirmed durable MR ≤ 2+ at 12–24 months, with the degree of annular shrinkage correlating with the clinical response [185,187]. Ideal candidates for Cardioband therapy exhibit predominantly annular dilatation, preserved leaflet mobility, and an effective mitral valve area greater than 4.0 cm2 [182,183]. Relative contraindications include heavy posterior annular calcification, inter-commissural gaps > 1 cm, and basal ventricular aneurysms, which may impair anchor seating or limit efficacy [182,183].
The Millipede annuloplasty system employs a modular approach, providing a semi-rigid, complete Nitinol ring with eight adjustable anchors, each of which can be repositioned to achieve symmetrical annular capture and reshaping [188]. The system incorporates intracardiac echo guidance, offering precise visualization during deployment [188]. In the first-in-human feasibility study involving seven patients, Millipede achieved a 31% reduction in septolateral annular diameter, with baseline MR grades of 3–4+ reduced to ≤1+ at both discharge and 30 days post-procedure, without any major periprocedural complications [188]. While large-scale data is still pending, the design allows for resheathing and resizing, offering potential advantages in anatomically complex or heterogeneous annular geometries [188]. The system is also compatible with future TMVR, positioning it as a versatile platform for staged or combined interventions [188].
As with all transcatheter mitral therapies, comprehensive pre-procedural imaging is essential for patient selection and procedural planning [37,189]. Both the American Society of Echocardiography and the European Association of Cardiovascular Imaging emphasize detailed evaluation of annular geometry, leaflet motion, and MR severity using echocardiography and cardiac CT [37,189]. Direct annuloplasty is generally most effective in patients with AFMR or milder forms of VFMR, where leaflet tethering is limited [144,166,190]. In contrast, for patients with advanced ventricular remodeling and a tenting height greater than 10 mm, edge-to-edge repair or combined strategies may be more suitable [29,37]. The hemodynamic goals of direct annuloplasty include achieving a residual trans-mitral gradient <5 mmHg and limiting residual MR to no more than mild [191]. In AFMR, where annular dilation is often atrial in origin and may be reversible, post-procedural rhythm control and hemodynamic unloading are critical adjuncts to promote sustained reverse remodeling and functional improvement [166].
In summary, direct annuloplasty with devices such as Cardioband and Millipede offers an anatomically precise, minimally invasive therapy for selected patients with FMR, particularly those with annular-dominant pathology and preserved leaflet motion. While current data support safety and early efficacy, randomized trials remain ongoing. IDE studies comparing Cardioband and Millipede with medical therapy, edge-to-edge repair, or combination strategies will be essential in defining their long-term durability, impact on remodeling, and appropriate place within the evolving treatment algorithm for FMR. As device design, imaging integration, and operator experience continue to advance, direct annuloplasty is poised to become a key tool in personalized, mechanism-driven mitral valve therapy.
6.3.4. TMVR
TMVR is an emerging therapeutic option for patients with severe FMR who are at high or prohibitive surgical risk and are either unsuitable for transcatheter repair or have failed prior repair attempts [192,193,194]. TMVR offers a mechanistically definitive solution, replacing the native mitral valve with a bioprosthetic implant that eliminates regurgitation regardless of underlying annular or subvalvular distortion, an important advantage over repair-based strategies, whose success may be limited by complex or severely remodeled anatomy [195,196,197]. Currently available TMVR platforms include transapically delivered devices, such as the Tendyne valve (Abbott) and the Intrepid valve (Medtronic), as well as fully transseptal systems, like the Sapien M3 and Evoque valves, which are under investigation in pivotal trials [162,198,199]. These systems are designed to anchor securely within the mitral annulus and accommodate the dynamic, elliptical, and noncalcified anatomy characteristic of FMR [196].
Clinical studies of TMVR have demonstrated high procedural success rates, reaching up to 97%, with near-complete elimination of mitral regurgitation in most patients (Table 7) [191,192,194,200]. Improvements in symptoms, exercise capacity, and quality of life are typically sustained through 1 to 2 years of follow-up [191,192,194,200]. In the CTSN trial, moderate-to-severe MR recurred in 32.6% of patients who underwent surgical repair, compared to only 2.3% after valve replacement, despite similar survival rates and comparable degrees of left ventricular reverse remodeling [139,193]. These findings underscore the durability advantage of replacement, particularly in anatomies prone to recurrent tethering or annular dilation. In one prospective study of high-risk patients, TMVR reduced heart failure hospitalizations from 1.3 to 0.5 events per year, with 93% of survivors exhibiting no residual MR at two years [139,193]. However, despite these promising outcomes, early mortality remains substantial, particularly within the first 90 days, reflecting the advanced clinical status of patients typically selected for this therapy [193,194,200].
TMVR is not without challenges. Potential complications include obstruction, prosthetic thrombosis, paravalvular leak, and difficulty anchoring the device in the setting of heavy mitral annular calcification [201,202,203]. Pre-procedural planning is critical, with multimodality imaging playing a central role [204,205]. Cardiac computed tomography (CT) is used to evaluate annular dimensions, simulate device deployment, and, crucially, estimate the neo–left ventricular outflow tract (neo-LVOT) area [114,116,117,206]. A neo-LVOT <2.0 cm2 is strongly associated with increased risk of LVOT obstruction [206,207]. As a result, ongoing randomized trials, most notably SUMMIT and APOLLO, are evaluating TMVR in comparison with transcatheter edge-to-edge repair to determine its relative safety, efficacy, and appropriate clinical niche [195,208].
In summary, TMVR currently remains reserved for patients with severe symptomatic FMR who are not candidates for surgery or edge-to-edge repair, and its use is predicated on a multidisciplinary heart team assessment and advanced imaging-based procedural planning. As device technology matures, imaging integration improves, and operator experience expands, the role of TMVR is expected to broaden, potentially offering a durable, anatomically agnostic treatment option for an increasing number of patients with FMR.
To assist clinicians in navigating these interventional strategies, Table 8 provides a clinical guide for selecting the most appropriate therapeutic approach based on the dominant imaging phenotype and the underlying pathophysiology of FMR.
Table 7. Clinical Studies of Transcatheter Mitral Valve Replacement for Functional Mitral Regurgitation.
|
Year |
Trial (N) |
Intervention |
Comparator |
Primary Outcome |
Result Summary |
Quality Considerations |
|---|---|---|---|---|---|---|
|
2015 |
Abdul-Jawad Altisent et al. (3) |
Fortis TMVR (transapical) |
None (case series) |
Procedural success, MR reduction, 6-mo outcomes |
100% device success, no major complications, all alive at 6 month, NYHA ≤ II, no HF readmission |
Compassionate use, very high surgical risk, small n, short follow-up |
|
2017 |
Muller et al. (30) |
Tendyne TMVR (transapical) |
None (feasibility) |
Device success, MR reduction, 30-day safety |
93% device success, 0 acute deaths/strokes, 1 death at 13d, 75% NYHA I/II at 30d, 1+ MR in 1 pt |
Early feasibility, high surgical risk, short follow-up |
|
2018 |
Bapat et al. (50) |
Intrepid TMVR (transapical) |
None (pilot) |
Device success, MR reduction, 30-day safety |
96% device success, 14% 30-day mortality, 79% NYHA I/II at follow-up, all mild/no MR |
High/extreme risk, short follow-up, pilot study |
|
2019 |
Sorajja et al. (100) |
Tendyne TMVR (transapical) |
None (feasibility) |
Technical success, 1-yr survival, MR reduction |
96% technical success, 6% 30d mortality, 72% 1-yr survival, 88.5% NYHA I/II, 73% KCCQ ↑ ≥ 10 pts |
High/prohibitive risk, nonrandomized, 1-yr follow-up |
|
2021 |
Muller et al. (100) |
Tendyne TMVR (transapical) |
None (prospective) |
2-yr all-cause mortality, HF hospitalization, MR reduction |
97% device success, 39% 2-yr mortality (44% in first 90d), HFH 0.51/yr vs. 1.3/yr pre-TMVR, 93% no MR at 2 yr |
High surgical risk, nonrandomized, 2-yr follow-up |
|
2023 |
Ludwig et al. (97 TMVR, 97 GDMT) |
TMVR (CHOICE-MI registry, mostly transapical) |
GDMT (COAPT control) |
2-yr HF hospitalization, mortality, MR reduction |
2-yr HFH 32.8% TMVR vs. 54.4% GDMT (HR 0.59); 2-yr mortality similar (36.8% vs. 40.8%); all TMVR ≤ 1+ MR |
Propensity-matched, registry vs RCT control, device heterogeneity, high risk |
Abbreviations: TMVR = Transcatheter mitral valve replacement; MR = Mitral regurgitation; NYHA = New York Heart Association (functional class); HF = Heart failure; HFH = Heart failure hospitalization; KCCQ = Kansas City Cardiomyopathy Questionnaire; GDMT = Guideline-directed medical therapy; RCT = Randomized controlled trial; ↑ = Increase.
Table 8. Imaging-Guided Interventional Decision-Making in Functional Mitral Regurgitation.
|
Dominant Imaging Phenotype |
Key Imaging Findings |
Pathophysiologic Interpretation |
Preferred Therapeutic Strategy |
Rationale |
|---|---|---|---|---|
|
Advanced VFMR with severe tethering and myocardial disease |
|
Ventricular-dominant disease with irreversible remodeling and reduced closing forces |
Surgery (chordal-sparing replacement) or TMVR in high-risk surgical candidates |
TEER durability is limited when tethering and scar burden are extensive; valve replacement offers more complete and durable MR elimination |
|
Disproportionate VFMR with preserved myocardial viability |
|
Valvular lesion is a major driver of symptoms and outcomes |
TEER (MitraClip or PASCAL) after optimized GDMT ± CRT |
MR reduction improves outcomes when myocardium remains responsive and remodeling is potentially reversible |
|
VFMR with electrical dyssynchrony |
|
Functional MR amplified by electromechanical delay |
CRT first, reassess MR; TEER if MR persists |
CRT can restore synchrony, reduce tethering, and obviate valve intervention in responders |
|
Ischemic VFMR with revascularizable myocardium |
|
MR partially driven by ischemia and regional remodeling |
CABG ± mitral intervention (repair or replacement depending on tethering severity) |
Revascularization may reduce MR; severe tethering favors replacement over repair |
|
Classic AFMR with annular-dominant disease |
|
Annular-driven MR with preserved ventricular mechanics |
Annuloplasty (surgical or transcatheter) ± TEER; rhythm control |
Annular reduction restores coaptation; TEER adjunctive if residual MR persists |
|
AFMR with atriogenic tethering (“hamstrung” posterior leaflet) |
|
Combined annular dilation and atrial-induced leaflet mal-coaptation |
Annuloplasty-first strategy, cautious use of TEER |
Isolated TEER may leave residual MR due to persistent annular pathology |
|
Mixed/Dual FMR |
|
Combined atrial and ventricular mechanisms |
Individualized Heart Team approach: TEER vs surgery vs TMVR |
Single-modality therapy is often insufficient; imaging defines dominant driver |
|
End-stage remodeling with prohibitive anatomy for repair |
|
Unsuitable for repair strategies |
TMVR (selected cases) or palliative GDMT |
CT-guided planning essential to avoid LVOT obstruction |
Abbreviations: VFMR = ventricular functional mitral regurgitation; AFMR = atrial functional mitral regurgitation; MR = mitral regurgitation; LV = left ventricle; LA = left atrium; EROA = effective regurgitant orifice area; LGE = late gadolinium enhancement; ECV = extracellular volume; CRT = cardiac resynchronization therapy; TEER = transcatheter edge-to-edge repair; TMVR = transcatheter mitral valve replacement; CABG = coronary artery bypass grafting; CAD = coronary artery disease; ↑ = increase.
7. Future Directions
The management of FMR has reached a critical juncture. While the success of TEER in highly selected patients has definitively demonstrated the prognostic importance of reducing MR, the conflicting results of landmark trials such as COAPT and MITRA-FR underscore a fundamental challenge: the lack of a universally applicable patient selection tool. Consequently, the future of FMR care is rapidly evolving, with a focus on enhanced, personalized risk stratification, the adoption of innovative technologies, and a deeper understanding of disease heterogeneity.
A primary research objective is to refine the timing and criteria for intervention. Recent studies, including RESHAPE-HF2 and MATTERHORN, suggest a paradigm shift toward earlier use of TEER in well-selected, symptomatic patients, moving beyond the current approach of only intervening after GDMT has failed. This emphasis on earlier, individualized intervention makes it critical to clarify whether FMR is primarily a driver or merely a marker of heart failure progression, as this distinction dictates which patients will derive the greatest benefit from valve intervention versus intensified medical therapy alone. To achieve this precision, future algorithms must move beyond traditional parameters to incorporate dynamic assessment from stress echocardiography and detailed myocardial tissue characterization from CMR, which enables the identification of viable versus scarred myocardium.
Artificial intelligence (AI) is emerging as a critical enabling technology in this effort [209,210]. AI-based tools are already being applied to automate chamber segmentation, annular geometry assessment, and volumetric analysis across echocardiography, CMR, and CT, reducing inter-operator variability and improving reproducibility of MR severity grading [211]. In echocardiography, deep learning algorithms can automatically quantify ventricular and atrial volumes, strain, and regurgitant jet parameters [212], while AI-assisted CT and CMR workflows facilitate annular sizing, neo-LVOT prediction, and myocardial scar quantification for procedural planning [213]. By harmonizing quantitative imaging across modalities, AI has the potential to address longstanding limitations in FMR phenotyping and trial generalizability.
Ultimately, future research must extend beyond procedural success to evaluate the durability of reverse remodeling, patient-centered outcomes, and cost-effectiveness. As diagnostic complexity and therapeutic options continue to expand, integrating advanced imaging, AI-assisted analytics, and clinical expertise within a multidisciplinary Heart Team framework will be essential to delivering individualized, evidence-based care for patients with functional mitral regurgitation.
8. Conclusions
FMR remains a highly prevalent and morbid condition, inextricably linked to adverse outcomes in patients with both HFrEF and preserved (HFpEF) ejection fraction. As detailed throughout this review, FMR is not a singular entity, but a spectrum defined by two mechanistically distinct subtypes: VFMR, driven by LV remodeling and leaflet tethering, and AFMR, caused by left atrial LA and annular dilation. This heterogeneity mandates a mechanism-specific approach to evaluation and management as detailed in Figure 2 (Management process of FMR).
The central clinical dilemma, determining which patients with severe secondary MR benefit from valve intervention, has been partially resolved by the landmark COAPT trial, which demonstrated that in highly selected patients, aggressive MR reduction with TEER improves prognosis. However, the discordant results with MITRA-FR underscore that the field has moved beyond simply treating all severe MR to identifying the specific mechanical and myocardial environment in which intervention is beneficial. Patient selection is paramount.
Multi-modality imaging is therefore indispensable for precision medicine in FMR. Echocardiography serves as the diagnostic cornerstone, grading severity and identifying the primary mechanism, while CMR provides the gold standard for volumetric quantification and, critically, myocardial tissue characterization via late gadolinium enhancement and T1 mapping. Furthermore, CT offers unparalleled anatomical detail, making it essential for pre-procedural planning, particularly for TMVR. These imaging data sets enable the application of advanced risk stratification tools, such as differentiating FMR phenotypes to the proportionate/disproportionate VFMR framework, which helps rationalize the divergent results observed in therapeutic trials. Effective management, as summarized in our comparative Table 1 (Mechanistic Characteristics) and Table 2 (Diagnostic Modalities), defined a carefully integrated approach.
Ultimately, FMR care is rapidly transitioning from a one-size-fits-all model to an individualized model. First-line therapy remains robust, which includes GDMT, followed by the judicious consideration of transcatheter or surgical therapies for persistent, symptomatic MR. Future efforts, guided by the multidisciplinary Heart Team, will focus on integrating artificial intelligence, next-generation devices, and refined risk stratification to target earlier, more individualized interventions that maximize myocardial recovery and substantially improve the long-term outcomes for this challenging patient population.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
The authors used Google Gemini and ChatGPT 5.2 to generate ideas for visualizing and presenting the illustrations described in the paper. Grammarly was used to improve grammar and edit paragraphs in the manuscript. After using these tools, the authors reviewed and made necessary edits to the content, taking full responsibility for the final article published.
Author Contributions
Conceptualization, T.K.M.W. and S.-W.L.; Methodology (literature search strategy), S.-W.L.; Validation (critical review of arguments), J.E.R., Y.-F.T., C.T. and T.K.M.W.; Investigation (literature collection), S.-W.L. and J.E.R.; Resources, T.K.M.W.; Writing—Original Draft Preparation, S.-W.L.; Writing—Review & Editing, S.-W.L., J.E.R., Y.-F.T., C.T. and T.K.M.W.; Visualization, S.-W.L.; Supervision, T.K.M.W.; Project Administration, T.K.M.W.; Funding Acquisition, T.K.M.W.
Ethics Statement
Not applicable. This is a review article that synthesizes and summarizes previously published literature and does not involve any new studies with human participants or animals performed by any of the authors.
Informed Consent Statement
Not applicable. This review article does not contain any studies with human participants performed by any of the authors.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article. All information discussed in this review is based on previously published studies, which are cited in the reference list.
Funding
This research received no external funding.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
-
Watanabe N, Maltais S, Nishino S, O’Donoghue TA, Hung J. Functional Mitral Regurgitation: Imaging Insights, Clinical Outcomes and Surgical Principles. Prog. Cardiovasc. Dis. 2017, 60, 351–360. DOI:10.1016/j.pcad.2017.11.006 [Google Scholar]
-
O’Gara PT, Mack MJ. Secondary Mitral Regurgitation. N. Engl. J. Med. 2020, 383, 1458–1467. DOI:10.1056/NEJMcp1903331 [Google Scholar]
-
Bax JJ, Di Carli M, Narula J, Delgado V. Multimodality imaging in ischaemic heart failure. Lancet 2019, 393, 1056–1070. DOI:10.1016/S0140-6736(18)33207-0 [Google Scholar]
-
Zoghbi WA, Levine RA, Flachskampf F, Grayburn P, Gillam L, Leipsic J, et al. Atrial Functional Mitral Regurgitation: A JACC: Cardiovascular Imaging Expert Panel Viewpoint. JACC Cardiovasc. Imaging 2022, 15, 1870–1882. DOI:10.1016/j.jcmg.2022.08.016 [Google Scholar]
-
Nishimura RA, Vahanian A, Eleid MF, Mack MJ. Mitral valve disease—Current management and future challenges. Lancet 2016, 387, 1324–1334. DOI:10.1016/S0140-6736(16)00558-4 [Google Scholar]
-
Debonnaire P, Al Amri I, Leong DP, Joyce E, Katsanos S, Kamperidis V, et al. Leaflet remodelling in functional mitral valve regurgitation: Characteristics, determinants, and relation to regurgitation severity. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 290–299. DOI:10.1093/ehjci/jeu216 [Google Scholar]
-
Farhan S, Silbiger JJ, Halperin JL, Zhang L, Dukkipati SR, Vogel B, et al. Pathophysiology, Echocardiographic Diagnosis, and Treatment of Atrial Functional Mitral Regurgitation: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2022, 80, 2314–2330. DOI:10.1016/j.jacc.2022.09.046 [Google Scholar]
-
Reid A, Ben Zekry S, Naoum C, Takagi H, Thompson C, Godoy M, et al. Geometric differences of the mitral valve apparatus in atrial and ventricular functional mitral regurgitation. J. Cardiovasc. Comput. Tomogr. 2022, 16, 431–441. DOI:10.1016/j.jcct.2022.02.008 [Google Scholar]
-
Kumar M, Thompson PD, Chen K. New Perspective on Pathophysiology and Management of Functional Mitral Regurgitation. Trends Cardiovasc. Med. 2023, 33, 386–392. DOI:10.1016/j.tcm.2022.03.001 [Google Scholar]
-
Senni M, Adamo M, Metra M, Alfieri O, Vahanian A. Treatment of functional mitral regurgitation in chronic heart failure: Can we get a ‘proof of concept’ from the MITRA-FR and COAPT trials? Eur. J. Heart Fail. 2019, 21, 852–861. DOI:10.1002/ejhf.1491 [Google Scholar]
-
Asgar AW, Mack MJ, Stone GW. Secondary mitral regurgitation in heart failure: Pathophysiology, prognosis, and therapeutic considerations. J. Am. Coll. Cardiol. 2015, 65, 1231–1248. DOI:10.1016/j.jacc.2015.02.009 [Google Scholar]
-
Maddox TM, Januzzi JL, Allen LA, Breathett K, Brouse S, Butler J, et al. 2024 ACC Expert Consensus Decision Pathway for Treatment of Heart Failure With Reduced Ejection Fraction: A Report of the American College of Cardiology Solution Set Oversight Committee. J. Am. Coll. Cardiol. 2024, 83, 1444–1488. DOI:10.1016/j.jacc.2023.12.024 [Google Scholar]
-
Anker SD, Friede T, von Bardeleben RS, Butler J, Khan MS, Diek M, et al. Transcatheter Valve Repair in Heart Failure with Moderate to Severe Mitral Regurgitation. N. Engl. J. Med. 2024, 391, 1799–1809. DOI:10.1056/NEJMoa2314328 [Google Scholar]
-
Asch FM, Medvedofsky D. Functional mitral regurgitation. Curr. Opin. Cardiol. 2020, 35, 464–473. DOI:10.1097/HCO.0000000000000770 [Google Scholar]
-
Davidson LJ, Davidson CJ. Transcatheter Treatment of Valvular Heart Disease: A Review. JAMA 2021, 325, 2480–2494. DOI:10.1001/jama.2021.2133 [Google Scholar]
-
Obadia JF, Messika-Zeitoun D, Leurent G, Iung B, Bonnet G, Piriou N, et al. Percutaneous Repair or Medical Treatment for Secondary Mitral Regurgitation. N. Engl. J. Med. 2018, 379, 2297–2306. DOI:10.1056/NEJMoa1805374 [Google Scholar]
-
Dziadzko V, Dziadzko M, Medina-Inojosa JR, Benfari G, Michelena HI, Crestanello JA, et al. Causes and mechanisms of isolated mitral regurgitation in the community: Clinical context and outcome. Eur. Heart J. 2019, 40, 2194–2202. DOI:10.1093/eurheartj/ehz314 [Google Scholar]
-
Figlioli G, Sticchi A, Christodoulou MN, Hadjidemetriou A, Amorim Moreira Alves G, De Carlo M, et al. Global Prevalence of Mitral Regurgitation: A Systematic Review and Meta-Analysis of Population-Based Studies. J. Clin. Med. 2025, 14, 2749. DOI:10.3390/jcm14082749 [Google Scholar]
-
Naser JA, Michelena HI, Lin G, Scott CG, Lee E, Kennedy AM, et al. Incidence, risk factors, and outcomes of atrial functional mitral regurgitation in patients with atrial fibrillation or sinus rhythm. Eur. Heart J. Cardiovasc. Imaging 2023, 24, 1450–1457. DOI:10.1093/ehjci/jead199 [Google Scholar]
-
Tamargo M, Obokata M, Reddy YNV, Pislaru SV, Lin G, Egbe AC, et al. Functional mitral regurgitation and left atrial myopathy in heart failure with preserved ejection fraction. Eur. J. Heart Fail. 2020, 22, 489–498. DOI:10.1002/ejhf.1699 [Google Scholar]
-
Milwidsky A, Mathai SV, Topilsky Y, Jorde UP. Medical Therapy for Functional Mitral Regurgitation. Circ. Heart Fail. 2022, 15, e009689. DOI:10.1161/CIRCHEARTFAILURE.122.009689 [Google Scholar]
-
Chen QF, Zhou X, Katsouras CS, Ni C, Zhu H, Liu C, et al. Atrial and ventricular functional mitral regurgitation: Prevalence, characteristics, outcomes, and disease progression. Eur. Heart J. Cardiovasc. Imaging 2025, 26, 545–556. DOI:10.1093/ehjci/jeae309 [Google Scholar]
-
Dziadzko V, Clavel MA, Dziadzko M, Medina-Inojosa JR, Michelena H, Maalouf J, et al. Outcome and undertreatment of mitral regurgitation: A community cohort study. Lancet 2018, 391, 960–969. DOI:10.1016/S0140-6736(18)30473-2 [Google Scholar]
-
Benfari G, Antoine C, Essayagh B, Batista R, Maalouf J, Rossi A, et al. Functional Mitral Regurgitation Outcome and Grading in Heart Failure With Reduced Ejection Fraction. JACC Cardiovasc. Imaging 2021, 14, 2303–2315. DOI:10.1016/j.jcmg.2021.05.017 [Google Scholar]
-
Bartko PE, Heitzinger G, Pavo N, Heitzinger M, Spinka G, Prausmüller S, et al. Burden, treatment use, and outcome of secondary mitral regurgitation across the spectrum of heart failure: Observational cohort study. BMJ 2021, 373, n1421. DOI:10.1136/bmj.n1421 [Google Scholar]
-
Malagoli A, Rossi L, Zanni A, Sticozzi C, Piepoli MF, Benfari G. Quantified mitral regurgitation and left atrial function in heart failure with reduced ejection fraction: Interplay and outcome implications. Eur. J. Heart Fail. 2022, 24, 694–702. DOI:10.1002/ejhf.2429 [Google Scholar]
-
Nappi F. Assessing emerging causes of mitral regurgitation: Atrial functional mitral regurgitation. J. Int. Med. Res. 2024, 52, 03000605241240583. DOI:10.1177/03000605241240583 [Google Scholar]
-
Nappi F. Atrial functional mitral regurgitation in cardiology and cardiac surgery. J. Thorac. Dis. 2024, 16, 5435–5456. DOI:10.21037/jtd-24-189 [Google Scholar]
-
Praz F, Beyersdorf F, Haugaa K, Prendergast B. Valvular heart disease: From mechanisms to management. Lancet 2024, 403, 1576–1589. DOI:10.1016/S0140-6736(23)02755-1 [Google Scholar]
-
Kochav JD, Kim J, Judd R, Tak KA, Janjua E, Maciejewski AJ, et al. Myocardial Contractile Mechanics in Ischemic Mitral Regurgitation: Multicenter Data Using Stress Perfusion Cardiovascular Magnetic Resonance. JACC Cardiovasc. Imaging 2022, 15, 1212–1226. DOI:10.1016/j.jcmg.2022.03.014 [Google Scholar]
-
Kochav JD, Kim J, Judd R, Kim HW, Klem I, Heitner J, et al. Ischemia-Mediated Dysfunction in Subpapillary Myocardium as a Marker of Functional Mitral Regurgitation. JACC Cardiovasc. Imaging 2021, 14, 826–839. DOI:10.1016/j.jcmg.2021.01.007 [Google Scholar]
-
Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP, III, Gentile F, et al. 2020 ACC/AHA Guideline for the Management of Patients with Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2021, 77, e25–e197. DOI:10.1016/j.jacc.2020.11.018 [Google Scholar]
-
Silbiger JJ. Mechanistic insights into atrial functional mitral regurgitation: Far more complicated than just left atrial remodeling. Echocardiography 2019, 36, 164–169. DOI:10.1111/echo.14249 [Google Scholar]
-
Akamatsu K, Abe Y, Matsumura Y, Shimeno K, Naruko T, Takahashi Y, et al. Etiology of Atrial Functional Mitral Regurgitation: Insights from Transthoracic Echocardiography in 159 Consecutive Patients with Atrial Fibrillation and Preserved Left Ventricular Ejection Fraction. Cardiology 2020, 145, 511–521. DOI:10.1159/000508279 [Google Scholar]
-
Hoit BD. Atrial functional mitral regurgitation. Curr. Opin. Cardiol. 2020, 35, 474–481. DOI:10.1097/HCO.0000000000000761 [Google Scholar]
-
Kagiyama N, Mondillo S, Yoshida K, Mandoli GE, Cameli M. Subtypes of Atrial Functional Mitral Regurgitation: Imaging Insights into Their Mechanisms and Therapeutic Implications. JACC Cardiovasc. Imaging 2020, 13, 820–835. DOI:10.1016/j.jcmg.2019.01.040 [Google Scholar]
-
Bonow RO, O’Gara PT, Adams DH, Badhwar V, Bavaria JE, Elmariah S, et al. 2020 Focused Update of the 2017 ACC Expert Consensus Decision Pathway on the Management of Mitral Regurgitation: A Report of the American College of Cardiology Solution Set Oversight Committee. J. Am. Coll. Cardiol. 2020, 75, 2236–2270. DOI:10.1016/j.jacc.2020.02.005 [Google Scholar]
-
Zoghbi WA, Adams D, Bonow RO, Enriquez-Sarano M, Foster E, Grayburn PA, et al. Recommendations for Noninvasive Evaluation of Native Valvular Regurgitation: A Report from the American Society of Echocardiography Developed in Collaboration with the Society for Cardiovascular Magnetic Resonance. J. Am. Soc. Echocardiogr. 2017, 30, 303–371. DOI:10.1016/j.echo.2017.01.007 [Google Scholar]
-
Bertrand PB, Schwammenthal E, Levine RA, Vandervoort PM. Exercise Dynamics in Secondary Mitral Regurgitation: Pathophysiology and Therapeutic Implications. Circulation 2017, 135, 297–314. DOI:10.1161/CIRCULATIONAHA.116.025260 [Google Scholar]
-
Lancellotti P, Pellikka PA, Budts W, Chaudhry FA, Donal E, Dulgheru R, et al. The Clinical Use of Stress Echocardiography in Non-Ischaemic Heart Disease: Recommendations from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2017, 30, 101–138. DOI:10.1016/j.echo.2016.10.016 [Google Scholar]
-
Faletra FF, La Franca E, Mulè M, Carvelli A, Parisi F, Di Stefano G, et al. Functional Mitral Valve Regurgitation, Pathophysiology, Leaflet ReModeling, and the Role of Imaging. Echocardiography 2025, 42, e70101. DOI:10.1111/echo.70101 [Google Scholar]
-
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP, Guyton RA, et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2014, 63, 2438–2488. DOI:10.1016/j.jacc.2014.02.537 [Google Scholar]
-
Pierard LA, Carabello BA. Ischaemic mitral regurgitation: Pathophysiology, outcomes and the conundrum of treatment. Eur. Heart J. 2010, 31, 2996–3005. DOI:10.1093/eurheartj/ehq411 [Google Scholar]
-
Grayburn PA, Sannino A, Packer M. Proportionate and Disproportionate Functional Mitral Regurgitation: A New Conceptual Framework That Reconciles the Results of the MITRA-FR and COAPT Trials. JACC Cardiovasc. Imaging 2019, 12, 353–362. DOI:10.1016/j.jcmg.2018.11.006 [Google Scholar]
-
Packer M, Grayburn PA. New Evidence Supporting a Novel Conceptual Framework for Distinguishing Proportionate and Disproportionate Functional Mitral Regurgitation. JAMA Cardiol. 2020, 5, 469–475. DOI:10.1001/jamacardio.2019.5971 [Google Scholar]
-
Adamo M, Pagnesi M, Ajmone Marsan N, Bauersachs J, Hausleiter J, Zieroth S, et al. Heart failure with reduced ejection fraction and ventricular secondary mitral regurgitation: A holistic approach. Eur. Heart J. 2026, 47(11), 1294–1303. DOI:10.1093/eurheartj/ehaf480 [Google Scholar]
-
Coats AJS, Anker SD, Baumbach A, Alfieri O, von Bardeleben RS, Bauersachs J, et al. The management of secondary mitral regurgitation in patients with heart failure: A joint position statement from the Heart Failure Association (HFA), European Association of Cardiovascular Imaging (EACVI), European Heart Rhythm Association (EHRA), and European Association of Percutaneous Cardiovascular Interventions (EAPCI) of the ESC. Eur. Heart J. 2021, 42, 1254–1269. DOI:10.1093/eurheartj/ehab086 [Google Scholar]
-
Stone GW, Abraham WT, Lindenfeld J, Kar S, Grayburn PA, Lim DS, et al. Five-Year Follow-up after Transcatheter Repair of Secondary Mitral Regurgitation. N. Engl. J. Med. 2023, 388, 2037–2048. DOI:10.1056/NEJMoa2300213 [Google Scholar]
-
Stone GW, Lindenfeld J, Abraham WT, Kar S, Lim DS, Mishell JM, et al. Transcatheter Mitral-Valve Repair in Patients with Heart Failure. N. Engl. J. Med. 2018, 379, 2307–2318. DOI:10.1056/NEJMoa1806640 [Google Scholar]
-
Hagendorff A, Knebel F, Helfen A, Stobe S, Doenst T, Falk V. Disproportionate mitral regurgitation: Another myth? A critical appraisal of echocardiographic assessment of functional mitral regurgitation. Int. J. Cardiovasc. Imaging 2021, 37, 183–196. DOI:10.1007/s10554-020-01975-6 [Google Scholar]
-
Ooms JF, Bouwmeester S, Debonnaire P, Nasser R, Voigt JU, Schotborgh MA, et al. Transcatheter Edge-to-Edge Repair in Proportionate Versus Disproportionate Functional Mitral Regurgitation. J. Am. Soc. Echocardiogr. 2022, 35, 105–115.e8. DOI:10.1016/j.echo.2021.08.002 [Google Scholar]
-
Kalmucki P, Lipiecki J, Witte KK, Goldberg SL, Baszko A, Siminiak T. Percutaneous mitral annuloplasty with the Carillon device: Outcomes in proportionate and disproportionate functional mitral regurgitation. Am. Heart J. 2023, 265, 137–142. DOI:10.1016/j.ahj.2023.07.010 [Google Scholar]
-
Kałmucki P, Lipiecki J, Dankowski R, Witte KK, Baszko A, Starling RC, et al. Improvement in echocardiographic mitral regurgitation parameters correlates with left ventricular reverse remodeling after percutaneous mitral annuloplasty. Kardiol. Pol. 2025, 83, 725–733. DOI:10.33963/v.phj.105726 [Google Scholar]
-
Bai W, Chen Y, Zhong Y, Deng L, Li D, Zhu W, et al. Assessment of mitral valve geometry in nonvalvular atrial fibrillation patients with or without ventricular dysfunction: Insights from high volume rate three-dimensional transesophageal echocardiography. Int. J. Cardiovasc. Imaging 2023, 39, 2427–2436. DOI:10.1007/s10554-023-02940-9 [Google Scholar]
-
Chang S, Zeng D, Zhang X, Huang L, Cai Y, Huang T, et al. Impact of Non-Valvular Atrial Fibrillation on Mitral Valve Anatomic Features: A Study of Three-Dimensional Mitral Valve by Transesophageal Echocardiography and Automatic Analysis Software. Echocardiography 2024, 41, e15943. DOI:10.1111/echo.15943 [Google Scholar]
-
Machino-Ohtsuka T, Seo Y, Ishizu T, Sato K, Sugano A, Yamamoto M, et al. Novel Mechanistic Insights into Atrial Functional Mitral Regurgitation—3-Dimensional Echocardiographic Study. Circ. J. 2016, 80, 2240–2248. DOI:10.1253/circj.CJ-16-0435 [Google Scholar]
-
Kim DH, Heo R, Handschumacher MD, Lee S, Choi YS, Kim KR, et al. Mitral Valve Adaptation to Isolated Annular Dilation: Insights Into the Mechanism of Atrial Functional Mitral Regurgitation. JACC Cardiovasc. Imaging 2019, 12, 665–677. DOI:10.1016/j.jcmg.2017.09.013 [Google Scholar]
-
Deferm S, Bertrand PB, Verbrugge FH, Verhaert D, Rega F, Thomas JD, et al. Atrial Functional Mitral Regurgitation: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2019, 73, 2465–2476. DOI:10.1016/j.jacc.2019.02.061 [Google Scholar]
-
Tang Z, Fan YT, Wang Y, Jin CN, Kwok KW, Lee AP. Mitral Annular and Left Ventricular Dynamics in Atrial Functional Mitral Regurgitation: A Three-Dimensional and Speckle-Tracking Echocardiographic Study. J. Am. Soc. Echocardiogr. 2019, 32, 503–513. DOI:10.1016/j.echo.2018.11.009 [Google Scholar]
-
Deferm S, Bertrand PB, Verhaert D, Verbrugge FH, Dauw J, Thoelen K, et al. Mitral Annular Dynamics in AF Versus Sinus Rhythm: Novel Insights into the Mechanism of AFMR. JACC Cardiovasc. Imaging 2022, 15, 1–13. DOI:10.1016/j.jcmg.2021.05.019 [Google Scholar]
-
Ye Q, Zhao Y, Bai C, Liu K, Zhao C, Liu Y, et al. Outcome of mitral repair combined with Cox-maze procedure for atrial functional mitral regurgitation with heart failure with recovered ejection fraction. Eur. J. Cardiothorac. Surg. 2023, 64, ezad273. DOI:10.1093/ejcts/ezad273 [Google Scholar]
-
Riccardi M, Cikes M, Adamo M, Pagnesi M, Lombardi CM, Solomon SD, et al. Functional Mitral Regurgitation and Heart Failure With Preserved Ejection Fraction: Clinical Implications and Management. J. Card. Fail. 2024, 30, 929–939. DOI:10.1016/j.cardfail.2024.02.024 [Google Scholar]
-
Otto CM. Clinical practice. Evaluation and management of chronic mitral regurgitation. N. Engl. J. Med. 2001, 345, 740–746. DOI:10.1056/NEJMcp003331 [Google Scholar]
-
Hirji SA, Cote CL, Javadikasgari H, Malarczyk A, McGurk S, Kaneko T. Atrial functional versus ventricular functional mitral regurgitation: Prognostic implications. J. Thorac. Cardiovasc. Surg. 2022, 164, 1808–1815.e4. DOI:10.1016/j.jtcvs.2020.12.098 [Google Scholar]
-
Konstantinou DM, Papadopoulou K, Giannakoulas G, Kamperidis V, Dalamanga EG, Damvopoulou E, et al. Determinants of functional mitral regurgitation severity in patients with ischemic cardiomyopathy versus nonischemic dilated cardiomyopathy. Echocardiography 2014, 31, 21–28. DOI:10.1111/echo.12304 [Google Scholar]
-
Recke S, Marienhagen J, Feistel H, Rein J, von der Emde J. Role of the electrocardiogram in assessing irreversibly impaired left ventricular systolic function in chronic mitral regurgitation. Eur. Heart J. 1991, 12, 770–776. DOI:10.1093/eurheartj/12.7.770 [Google Scholar]
-
Chow SL, Maisel AS, Anand I, Bozkurt B, De Boer RA, Felker GM, et al. Role of Biomarkers for the Prevention, Assessment, and Management of Heart Failure: A Scientific Statement From the American Heart Association. Circulation 2017, 135, e1054–e1091. DOI:10.1161/CIR.0000000000000490 [Google Scholar]
-
Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2022, 79, e263–e421. DOI:10.1016/j.jacc.2021.12.012 [Google Scholar]
-
Seghatol FF, Martin KD, Haj-Asaad A, Xie M, Prabhu SD. Relation of Cardiorenal Syndrome to Mitral and Tricuspid Regurgitation in Acute Decompensated Heart Failure. Am. J. Cardiol. 2022, 168, 99–104. DOI:10.1016/j.amjcard.2021.12.024 [Google Scholar]
-
Cammalleri V, Antonelli G, De Luca VM, Carpenito M, Nusca A, Bono MC, et al. Functional Mitral and Tricuspid Regurgitation across the Whole Spectrum of Left Ventricular Ejection Fraction: Recognizing the Elephant in the Room of Heart Failure. J. Clin. Med. 2023, 12, 3316. DOI:10.3390/jcm12093316 [Google Scholar]
-
Lee J, Mitter SS, Van Assche L, Huh H, Wagner GJ, Wu E, et al. Impact of assuming a circular orifice on flow error through elliptical regurgitant orifices: Computational fluid dynamics and in vitro analysis of proximal flow convergence. Int. J. Cardiovasc. Imaging 2023, 39, 307–318. DOI:10.1007/s10554-022-02729-2 [Google Scholar]
-
Bartko PE, Arfsten H, Heitzinger G, Pavo N, Toma A, Strunk G, et al. A Unifying Concept for the Quantitative Assessment of Secondary Mitral Regurgitation. J. Am. Coll. Cardiol. 2019, 73, 2506–2517. DOI:10.1016/j.jacc.2019.02.075 [Google Scholar]
-
Karam N, Orban M, Kalbacher D, Butter C, Praz F, Lubos E, et al. Impact of effective regurgitant orifice area on outcome of secondary mitral regurgitation transcatheter repair. Clin. Res. Cardiol. 2021, 110, 732–739. DOI:10.1007/s00392-021-01807-0 [Google Scholar]
-
Kron IL, LaPar DJ, Acker MA, Adams DH, Ailawadi G, Bolling SF, et al. 2016 update to The American Association for Thoracic Surgery (AATS) consensus guidelines: Ischemic mitral valve regurgitation. J. Thorac. Cardiovasc. Surg. 2017, 153, e97–e114. DOI:10.1016/j.jtcvs.2017.01.031; Erratum in J. Thorac. Cardiovasc. Surg. 2017, 153, 1076–1079. DOI:10.1016/j.jtcvs.2016.11.068 [Google Scholar]
-
Lee APW, Hsiung MC, Salgo IS, Fang F, Xie JM, Zhang YC, et al. Quantitative analysis of mitral valve morphology in mitral valve prolapse with real-time 3-dimensional echocardiography: Importance of annular saddle shape in the pathogenesis of mitral regurgitation. Circulation 2013, 127, 832–841. DOI:10.1161/CIRCULATIONAHA.112.118083 [Google Scholar]
-
Zilberszac R, Gleiss A, Massetti M, Wisser W, Binder T, Gabriel H, et al. Left atrial size predicts outcome in severe but asymptomatic mitral regurgitation. Sci. Rep. 2023, 13, 3892. DOI:10.1038/s41598-023-31163-0 [Google Scholar]
-
Mihos CG, Liu JE, Anderson KM, Pernetz MA, O’Driscoll JM, Aurigemma GP, et al. Speckle-Tracking Strain Echocardiography for the Assessment of Left Ventricular Structure and Function: A Scientific Statement From the American Heart Association. Circulation 2025, 152, e96–e109. DOI:10.1161/CIR.0000000000001354 [Google Scholar]
-
Flint N, Raschpichler M, Rader F, Shmueli H, Siegel RJ. Asymptomatic Degenerative Mitral Regurgitation: A Review. JAMA Cardiol. 2020, 5, 346–355. DOI:10.1001/jamacardio.2019.5466 [Google Scholar]
-
Ueyama H, Kuno T, Takagi H, Krishnamoorthy P, Prandi FR, Palazzuoli A, et al. Prognostic value of left ventricular global longitudinal strain in mitral regurgitation: A systematic review. Heart Fail. Rev. 2023, 28, 465–483. DOI:10.1007/s10741-022-10265-3 [Google Scholar]
-
Marwick TH, Shah SJ, Thomas JD. Myocardial Strain in the Assessment of Patients with Heart Failure: A Review. JAMA Cardiol. 2019, 4, 287–294. DOI:10.1001/jamacardio.2019.0052 [Google Scholar]
-
Nagueh SF, Sanborn DY, Oh JK, Anderson B, Billick K, Derumeaux G, et al. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography and for Heart Failure with Preserved Ejection Fraction Diagnosis: An Update From the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2025, 38, 537–569. DOI:10.1016/j.echo.2025.03.011 [Google Scholar]
-
Seo J, Yu HT, Kim IS, Kim TH, Joung B, Pak HN, et al. Novel algorithm for non-invasive estimation of left atrial pressure in patients with atrial fibrillation. Eur. Heart J. Cardiovasc. Imaging 2025, 26, 414–421. DOI:10.1093/ehjci/jeae311 [Google Scholar]
-
Holte E, Podlesnikar T, Carvalho F, Demirkiran A, Manka R, Martínez GG, et al. European Association of Cardiovascular Imaging survey on the evaluation of mitral regurgitation. Eur. Heart J. Cardiovasc. Imaging 2024, 25, 573–578. DOI:10.1093/ehjci/jeae053 [Google Scholar]
-
El-Tallawi KC, Messika-Zeitoun D, Zoghbi WA. Assessment of the severity of native mitral valve regurgitation. Prog. Cardiovasc. Dis. 2017, 60, 322–333. DOI:10.1016/j.pcad.2017.11.005 [Google Scholar]
-
Taub CC, Stainback RF, Abraham T, Forsha D, Garcia-Sayan E, Hill JC, et al. Guidelines for the Standardization of Adult Echocardiography Reporting: Recommendations From the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2025, 38, 735–774. DOI:10.1016/j.echo.2025.06.001 [Google Scholar]
-
Cimino S, Guarracino F, Valenti V, Frati G, Sciarretta S, Miraldi F, et al. Echocardiography and Correction of Mitral Regurgitation: An Unbreakable Link. Cardiology 2020, 145, 110–120. DOI:10.1159/000504248 [Google Scholar]
-
Grayburn PA, Thomas JD. Basic Principles of the Echocardiographic Evaluation of Mitral Regurgitation. JACC Cardiovasc. Imaging 2021, 14, 843–853. DOI:10.1016/j.jcmg.2020.06.049 [Google Scholar]
-
Uretsky S, Argulian E, Narula J, Wolff SD. Use of Cardiac Magnetic Resonance Imaging in Assessing Mitral Regurgitation: Current Evidence. J. Am. Coll. Cardiol. 2018, 71, 547–563. DOI:10.1016/j.jacc.2017.12.009 [Google Scholar]
-
Myerson SG. CMR in Evaluating Valvular Heart Disease: Diagnosis, Severity, and Outcomes. JACC Cardiovasc. Imaging 2021, 14, 2020–2032. DOI:10.1016/j.jcmg.2020.09.029 [Google Scholar]
-
Garg P, Swift AJ, Zhong L, Carlhäll CJ, Ebbers T, Westenberg J, et al. Assessment of mitral valve regurgitation by cardiovascular magnetic resonance imaging. Nat. Rev. Cardiol. 2020, 17, 298–312. DOI:10.1038/s41569-019-0305-z [Google Scholar]
-
Gajjar K, Kashyap K, Badlani J, Williams RB, Biederman RWW. A review of the pivotal role of cardiac MRI in mitral valve regurgitation. Echocardiography 2021, 38, 128–141. DOI:10.1111/echo.14941 [Google Scholar]
-
Rajiah PS, Francois CJ, Leiner T. Cardiac MRI: State of the Art. Radiology 2023, 307, e223008. DOI:10.1148/radiol.223008 [Google Scholar]
-
Mehta NK, Kim J, Siden JY, Rodriguez-Diego S, Alakbarli J, Di Franco A, et al. Utility of cardiac magnetic resonance for evaluation of mitral regurgitation prior to mitral valve surgery. J. Thorac. Dis. 2017, 9, S246–S256. DOI:10.21037/jtd.2017.03.54 [Google Scholar]
-
Fidock B, Archer G, Barker N, Elhawaz A, Al-Mohammad A, Rothman A, et al. Standard and emerging CMR methods for mitral regurgitation quantification. Int. J. Cardiol. 2021, 331, 316–321. DOI:10.1016/j.ijcard.2021.01.066 [Google Scholar]
-
Le Goffic C, Toledano M, Ennezat PV, Binda C, Castel AL, Delelis F, et al. Quantitative Evaluation of Mitral Regurgitation Secondary to Mitral Valve Prolapse by Magnetic Resonance Imaging and Echocardiography. Am. J. Cardiol. 2015, 116, 1405–1410. DOI:10.1016/j.amjcard.2015.07.064 [Google Scholar]
-
Myerson SG, d’Arcy J, Christiansen JP, Dobson LE, Mohiaddin R, Francis JM, et al. Determination of Clinical Outcome in Mitral Regurgitation With Cardiovascular Magnetic Resonance Quantification. Circulation 2016, 133, 2287–2296. DOI:10.1161/CIRCULATIONAHA.115.017888 [Google Scholar]
-
Gupta AN, Avery R, Soulat G, Allen BD, Collins JD, Choudhury L, et al. Direct mitral regurgitation quantification in hypertrophic cardiomyopathy using 4D flow CMR jet tracking: Evaluation in comparison to conventional CMR. J. Cardiovasc. Magn. Reson. 2021, 23, 138. DOI:10.1186/s12968-021-00828-y [Google Scholar]
-
Gorecka M, Bissell MM, Higgins DM, Garg P, Plein S, Greenwood JP. Rationale and clinical applications of 4D flow cardiovascular magnetic resonance in assessment of valvular heart disease: A comprehensive review. J. Cardiovasc. Magn. Reson. 2022, 24, 49. DOI:10.1186/s12968-022-00882-0 [Google Scholar]
-
Fernández-Golfín C, De Agustin A, Manzano MC, Bustos A, Sánchez T, Pérez de Isla L, et al. Cardiac magnetic resonance determinants of functional mitral regurgitation in ischemic and non ischemic left ventricular dysfunction. Int. J. Cardiovasc. Imaging 2011, 27, 539–546. DOI:10.1007/s10554-010-9696-y [Google Scholar]
-
Lenon M, Arias-Cartin R, Barras F. The Fe–S proteome of Escherichia coli: Prediction, function, and fate. Metallomics 2022, 14, mfac022. DOI:10.1093/mtomcs/mfac022 [Google Scholar]
-
Helali J, Ramesh K, Brown J, Preciado-Ruiz C, Nguyen T, Silva LT, et al. Late gadolinium enhancement on cardiac MRI: A systematic review and meta-analysis of prognosis across cardiomyopathies. Int. J. Cardiol. 2025, 419, 132711. DOI:10.1016/j.ijcard.2024.132711 [Google Scholar]
-
Wang TKM, Kocyigit D, Choi H, Anthony CM, Chan N, Bullen J, et al. Prognostic Power of Quantitative Assessment of Functional Mitral Regurgitation and Myocardial Scar Quantification by Cardiac Magnetic Resonance. Circ. Cardiovasc. Imaging 2023, 16, e015134. DOI:10.1161/CIRCIMAGING.122.015134 [Google Scholar]
-
Badau Riebel CI, Agoston-Coldea L. Left Ventricular Fibrosis by Cardiac Magnetic Resonance Tissue Characterization in Chronic Mitral Regurgitation Patients. J. Clin. Med. 2024, 13, 3877. DOI:10.3390/jcm13133877 [Google Scholar]
-
Puntmann VO, Peker E, Chandrashekhar Y, Nagel E. T1 Mapping in Characterizing Myocardial Disease: A Comprehensive Review. Circ. Res. 2016, 119, 277–299. DOI:10.1161/CIRCRESAHA.116.307974 [Google Scholar]
-
Zhang H, Yang W, Zhou D, Wang Y, Zhu L, Jiang M, et al. Prognostic Value of Myocardial T1 Mapping and Extracellular Volume Fraction in Heart Failure: A Meta-Analysis. JACC Cardiovasc. Imaging 2025, 18, 1203–1217. DOI:10.1016/j.jcmg.2025.06.017 [Google Scholar]
-
Kitkungvan D, Yang EY, El Tallawi KC, Nagueh SF, Nabi F, Khan MA, et al. Extracellular Volume in Primary Mitral Regurgitation. JACC Cardiovasc. Imaging 2021, 14, 1146–1160. DOI:10.1016/j.jcmg.2020.10.010 [Google Scholar]
-
Markousis-Mavrogenis G, Bacopoulou F, Vlachakis D, Mavrogeni S. Tissue Characterization in Cardiology: Moving Beyond Function. Adv. Exp. Med. Biol. 2021, 1337, 89–97. DOI:10.1007/978-3-030-78771-4_11 [Google Scholar]
-
Weir-McCall JR, Blanke P, Naoum C, Delgado V, Bax JJ, Leipsic J. Mitral Valve Imaging with CT: Relationship with Transcatheter Mitral Valve Interventions. Radiology 2018, 288, 638–655. DOI:10.1148/radiol.2018172758 [Google Scholar]
-
Hirasawa K, Vo NM, Gegenava T, Pio SM, van Wijngaarden SE, Ajmone Marsan N, et al. Mitral Valve Annulus Dimensions Assessment with Three-Dimensional Echocardiography Versus Computed Tomography: Implications for Transcatheter Interventions. J. Clin. Med. 2021, 10, 649. DOI:10.3390/jcm10040649 [Google Scholar]
-
Thériault-Lauzier P, Dorfmeister M, Mylotte D, Spaziano M, Blanke P, Martucci G, et al. Quantitative multi-slice computed tomography assessment of the mitral valvular complex for transcatheter mitral valve interventions part 2: Geometrical measurements in patients with functional mitral regurgitation. EuroIntervention 2016, 12, e1021–e1030. DOI:10.4244/EIJY15M11_10 [Google Scholar]
-
van Rosendael PJ, van Wijngaarden SE, Kamperidis V, Kong WKF, Leung M, Ajmone Marsan N, et al. Integrated imaging of echocardiography and computed tomography to grade mitral regurgitation severity in patients undergoing transcatheter aortic valve implantation. Eur. Heart J. 2017, 38, 2221–2226. DOI:10.1093/eurheartj/ehw612 [Google Scholar]
-
den Harder AM, de Heer LM, Meijer RCA, Das M, Krestin GP, Maessen JG, et al. Effect of computed tomography before cardiac surgery on surgical strategy, mortality and stroke. Eur. J. Radiol. 2016, 85, 744–750. DOI:10.1016/j.ejrad.2016.01.003 [Google Scholar]
-
Valente T, Bocchini G, Rossi G, Sica G, Davison H, Scaglione M. MDCT prior to median re-sternotomy in cardiovascular surgery: Our experiences, infrequent findings and the crucial role of radiological report. Br. J. Radiol. 2019, 92, 20170980. DOI:10.1259/bjr.20170980 [Google Scholar]
-
Yoon SH, Bleiziffer S, Latib A, Eschenbach L, Ancona M, Vincent F, et al. Predictors of Left Ventricular Outflow Tract Obstruction After Transcatheter Mitral Valve Replacement. JACC Cardiovasc. Interv. 2019, 12, 182–193. DOI:10.1016/j.jcin.2018.12.001 [Google Scholar]
-
Kohli K, Wei ZA, Sadri V, Khan JM, Lisko JC, Netto T, et al. Dynamic nature of the LVOT following transcatheter mitral valve replacement with LAMPOON: New insights from post-procedure imaging. Eur. Heart J. Cardiovasc. Imaging 2022, 23, 650–662. DOI:10.1093/ehjci/jeab074 [Google Scholar]
-
Ranganath P, Moore A, Guerrero M, Collins J, Foley T, Williamson E, et al. CT for Pre- and Postprocedural Evaluation of Transcatheter Mitral Valve Replacement. Radiographics 2020, 40, 1528–1553. DOI:10.1148/rg.2020200027 [Google Scholar]
-
Ge Y, Gupta S, Fentanes E, Aghayev A, Steigner M, Sobieszczyk P, et al. Role of Cardiac CT in Pre-Procedure Planning for Transcatheter Mitral Valve Replacement. JACC Cardiovasc. Imaging 2021, 14, 1571–1580. DOI:10.1016/j.jcmg.2020.12.018 [Google Scholar]
-
Caldonazo T, Kirov H, Dochev I, Fischer J, Runkel A, Dewey M, et al. Invasive Coronary Angiography Versus Noninvasive Computed Tomography Coronary Angiography as Preoperative Coronary Imaging for Valve Surgery. Am. J. Cardiol. 2025, 237, 1–5. DOI:10.1016/j.amjcard.2024.11.015 [Google Scholar]
-
Opolski MP, Staruch AD, Jakubczyk M, Min JK, Gransar H, Staruch M, et al. CT Angiography for the Detection of Coronary Artery Stenoses in Patients Referred for Cardiac Valve Surgery: Systematic Review and Meta-Analysis. JACC Cardiovasc. Imaging 2016, 9, 1059–1070. DOI:10.1016/j.jcmg.2015.09.028 [Google Scholar]
-
Barnes C, Sharma H, Gamble J, Dawkins S. Management of secondary mitral regurgitation: From drugs to devices. Heart 2024, 110, 1099–1106. DOI:10.1136/heartjnl-2022-322001 [Google Scholar]
-
Kron IL, Hung J, Overbey JR, Bouchard D, Gelijns AC, Moskowitz AJ, et al. Predicting recurrent mitral regurgitation after mitral valve repair for severe ischemic mitral regurgitation. J. Thorac. Cardiovasc. Surg. 2015, 149, 752–761.e1. DOI:10.1016/j.jtcvs.2014.10.120 [Google Scholar]
-
Russo V. Extended Cardiac Rhythm Monitoring Detects More Arrhythmias in Myotonic Dystrophy Patients. JACC Clin. Electrophysiol. 2025, 11, 1347. DOI:10.1016/j.jacep.2025.03.041 [Google Scholar]
-
Murphy SP, Ibrahim NE, Januzzi JL, Jr. Heart Failure With Reduced Ejection Fraction: A Review. JAMA 2020, 324, 488–504. DOI:10.1001/jama.2020.10262 [Google Scholar]
-
van Bommel RJ, Marsan NA, Delgado V, Borleffs CJW, van Rijnsoever EPM, Schalij MJ, et al. Cardiac resynchronization therapy as a therapeutic option in patients with moderate-severe functional mitral regurgitation and high operative risk. Circulation 2011, 124, 912–919. DOI:10.1161/CIRCULATIONAHA.110.009803 [Google Scholar]
-
Michalski B, Stankovic I, Pagourelias E, Ciarka A, Aarones M, Winter S, et al. Relationship of Mechanical Dyssynchrony and LV Remodeling With Improvement of Mitral Regurgitation After CRT. JACC Cardiovasc. Imaging 2022, 15, 212–220. DOI:10.1016/j.jcmg.2021.08.010 [Google Scholar]
-
Mihos CG, Yucel E, Upadhyay GA, Orencole MP, Singh JP, Picard MH. Left ventricle and mitral valve reverse remodeling in response to cardiac resynchronization therapy in nonischemic cardiomyopathy. Echocardiography 2020, 37, 1557–1565. DOI:10.1111/echo.14844 [Google Scholar]
-
Al-Hashedi EM, Abdu FA, Ahmad A, Yu Y, Li R, Mohammed AA, et al. Impact of Atrial Fibrillation Catheter Ablation on Functional Mitral Regurgitation. Am. J. Cardiol. 2025, 254, 127–138. DOI:10.1016/j.amjcard.2025.07.034 [Google Scholar]
-
Yamauchi R, Morishima I, Okumura K, Kanzaki Y, Morita Y, Watanabe N, et al. Association Between Catheter Ablation for Nonparoxysmal Atrial Fibrillation and Functional Mitral Regurgitation in Patients With Heart Failure with Preserved Ejection Fraction. Am. J. Cardiol. 2023, 207, 192–201. DOI:10.1016/j.amjcard.2023.08.154 [Google Scholar]
-
Castleberry AW, Williams JB, Daneshmand MA, Honeycutt E, Shaw LK, Samad Z, et al. Surgical revascularization is associated with maximal survival in patients with ischemic mitral regurgitation: A 20-year experience. Circulation 2014, 129, 2547–2556. DOI:10.1161/CIRCULATIONAHA.113.005223 [Google Scholar]
-
Kang DH, Sun BJ, Kim DH, Yun SC, Song JM, Choo SJ, et al. Percutaneous versus surgical revascularization in patients with ischemic mitral regurgitation. Circulation 2011, 124, S156–S162. DOI:10.1161/CIRCULATIONAHA.110.011254 [Google Scholar]
-
Fattouch K, Dioguardi P, Guccione F, Nogara A, Salardino M, Sampognaro R, et al. 10-Year Results of Mitral Repair and Coronary Bypass for Ischemic Regurgitation: A Randomized Trial. Ann. Thorac. Surg. 2022, 113, 816–822. DOI:10.1016/j.athoracsur.2021.03.106 [Google Scholar]
-
Sandoval Y, Sorajja P, Harris KM. Contemporary Management of Ischemic Mitral Regurgitation: A Review. Am. J. Med. 2018, 131, 887–895. DOI:10.1016/j.amjmed.2018.01.048 [Google Scholar]
-
Black L, Li K, Shendell DG. Expanding awareness of climate change, sustainability, and environmental health through an introductory short online course for high school students. Explore 2022, 18, 381–383. DOI:10.1016/j.explore.2022.03.006 [Google Scholar]
-
Birtcher KK, Allen LA, Anderson JL, Bonaca MP, Gluckman TJ, Hussain A, et al. 2022 ACC Expert Consensus Decision Pathway for Integrating Atherosclerotic Cardiovascular Disease and Multimorbidity Treatment: A Framework for Pragmatic, Patient-Centered Care: A Report of the American College of Cardiology Solution Set Oversight Committee. J. Am. Coll. Cardiol. 2023, 81, 292–317. DOI:10.1016/j.jacc.2022.08.754 [Google Scholar]
-
Smith PK, Puskas JD, Ascheim DD, Voisine P, Gelijns AC, Moskowitz AJ, et al. Surgical treatment of moderate ischemic mitral regurgitation. N. Engl. J. Med. 2014, 371, 2178–2188. DOI:10.1056/NEJMoa1410490 [Google Scholar]
-
Michler RE, Smith PK, Parides MK, Ailawadi G, Thourani V, Moskowitz AJ, et al. Two-Year Outcomes of Surgical Treatment of Moderate Ischemic Mitral Regurgitation. N. Engl. J. Med. 2016, 374, 1932–1941. DOI:10.1056/NEJMoa1602003 [Google Scholar]
-
Bakaeen FG, Gaudino M, Whitman G, Doenst T, Ruel M, Taggart DP, et al. 2021: The American Association for Thoracic Surgery Expert Consensus Document: Coronary artery bypass grafting in patients with ischemic cardiomyopathy and heart failure. J. Thorac. Cardiovasc. Surg. 2021, 162, 829–850.e1. DOI:10.1016/j.jtcvs.2021.04.052 [Google Scholar]
-
Acker MA, Parides MK, Perrault LP, Moskowitz AJ, Gelijns AC, Voisine P, et al. Mitral-valve repair versus replacement for severe ischemic mitral regurgitation. N. Engl. J. Med. 2014, 370, 23–32. DOI:10.1056/NEJMoa1312808 [Google Scholar]
-
Goldstein D, Moskowitz AJ, Gelijns AC, Ailawadi G, Parides MK, Perrault LP, et al. Two-Year Outcomes of Surgical Treatment of Severe Ischemic Mitral Regurgitation. N. Engl. J. Med. 2016, 374, 344–353. DOI:10.1056/NEJMoa1512913 [Google Scholar]
-
Sideris K, Burri M, Mayr A, Voss S, Vitanova K, Prinzing A, et al. Functional Mitral Valve Regurgitation: Mitral Valve Repair or Replacement? Our “Road Map” for the Appropriate Strategy. J. Clin. Med. 2024, 13, 3264. DOI:10.3390/jcm13113264 [Google Scholar]
-
Wagner CM, Brescia AA, Watt TMF, Bergquist C, Rosenbloom LM, Ceniza NN, et al. Surgical strategy and outcomes for atrial functional mitral regurgitation: All functional mitral regurgitation is not the same! J. Thorac. Cardiovasc. Surg. 2024, 167, 647–655. DOI:10.1016/j.jtcvs.2022.02.056 [Google Scholar]
-
Bakir NH, Bernabei A, Burns DJP, Blackstone EH, Houghtaling PL, DiPaola LM, et al. Durability of annuloplasty in patients with atrial functional mitral regurgitation associated with atrial fibrillation. J. Thorac. Cardiovasc. Surg. 2025, 170, 177–186.e3. DOI:10.1016/j.jtcvs.2024.08.029 [Google Scholar]
-
Amabile A, Fereydooni S, Mori M, Hameed I, Jung J, Krane M, et al. Variable definitions and treatment approaches for atrial functional mitral regurgitation: A scoping review of the literature. J. Card. Surg. 2022, 37, 1182–1191. DOI:10.1111/jocs.16312 [Google Scholar]
-
Shin JH, Lee SH, Joo HC, Youn YN, Kim JH, Lee S. Long-term Outcomes of Mitral Valve Repair for Atrial Functional Mitral Regurgitation. Ann. Thorac. Surg. 2025, 120, 71–78. DOI:10.1016/j.athoracsur.2024.09.001 [Google Scholar]
-
Deferm S, Bertrand PB, Verhaert D, Dauw J, Van Keer JM, Van De Bruaene A, et al. Outcome and durability of mitral valve annuloplasty in atrial secondary mitral regurgitation. Heart 2021, 107, 1503–1509. DOI:10.1136/heartjnl-2021-319045 [Google Scholar]
-
Bao C, Wei K, Zhao D, Zhang J, Mei J, Ma N. Surgical Strategy for Atrial Functional Mitral Regurgitation With Atrial Fibrillation. J. Am. Heart Assoc. 2025, 14, e039019. DOI:10.1161/JAHA.124.039019 [Google Scholar]
-
Zoghbi WA, Asch FM, Bruce C, Gillam LD, Grayburn PA, Hahn RT, et al. Guidelines for the Evaluation of Valvular Regurgitation After Percutaneous Valve Repair or Replacement: A Report from the American Society of Echocardiography Developed in Collaboration with the Society for Cardiovascular Angiography and Interventions, Japanese Society of Echocardiography, and Society for Cardiovascular Magnetic Resonance. J. Am. Soc. Echocardiogr. 2019, 32, 431–475. DOI:10.1016/j.echo.2019.01.003 [Google Scholar]
-
Ponikowski P, Friede T, von Bardeleben RS, Butler J, Shahzeb Khan M, Diek M, et al. Hospitalization of Symptomatic Patients with Heart Failure and Moderate to Severe Functional Mitral Regurgitation Treated with MitraClip: Insights from RESHAPE-HF2. J. Am. Coll. Cardiol. 2024, 84, 2347–2363. DOI:10.1016/j.jacc.2024.08.027 [Google Scholar]
-
Messika-Zeitoun D, Iung B, Armoiry X, Trochu JN, Donal E, Habib G, et al. Impact of Mitral Regurgitation Severity and Left Ventricular Remodeling on Outcome After MitraClip Implantation: Results from the Mitra-FR Trial. JACC Cardiovasc. Imaging 2021, 14, 742–752. DOI:10.1016/j.jcmg.2020.07.021 [Google Scholar]
-
Mauricio R, Kumbhani DJ. MitraClip: How Do We Reconcile the Inconsistent Findings of MITRA-FR and COAPT? Curr. Cardiol. Rep. 2019, 21, 150. DOI:10.1007/s11886-019-1239-0 [Google Scholar]
-
Mack MJ, Lindenfeld J, Abraham WT, Kar S, Lim DS, Mishell JM, et al. 3-Year Outcomes of Transcatheter Mitral Valve Repair in Patients with Heart Failure. J. Am. Coll. Cardiol. 2021, 77, 1029–1040. DOI:10.1016/j.jacc.2020.12.047 [Google Scholar]
-
Dragona VM, Sideris K, Liori S. The role of transcatheter edge-to-edge repair in functional mitral regurgitation: Key takeaways from the MATTERHORN and RESHAPE-HF2 trials. Heart Fail. Rev. 2025, 30, 1133–1138. DOI:10.1007/s10741-025-10533-y [Google Scholar]
-
Benito-González T, Carrasco-Chinchilla F, Estévez-Loureiro R, Pascual I, Arzamendi D, Garrote-Coloma C, et al. Clinical and echocardiographic outcomes of transcatheter mitral valve repair in atrial functional mitral regurgitation. Int. J. Cardiol. 2021, 345, 29–35. DOI:10.1016/j.ijcard.2021.09.056 [Google Scholar]
-
Doldi P, Stolz L, Orban M, Karam N, Praz F, Kalbacher D, et al. Transcatheter Mitral Valve Repair in Patients With Atrial Functional Mitral Regurgitation. JACC Cardiovasc. Imaging 2022, 15, 1843–1851. DOI:10.1016/j.jcmg.2022.05.009 [Google Scholar]
-
Popolo Rubbio A, Testa L, Grasso C, Sisinni A, Tusa M, Agricola E, et al. Transcatheter edge-to-edge mitral valve repair in atrial functional mitral regurgitation: Insights from the multi-center MITRA-TUNE registry. Int. J. Cardiol. 2022, 349, 39–45. DOI:10.1016/j.ijcard.2021.11.027 [Google Scholar]
-
Masiero G, Montonati C, Rubbio AP, Adamo M, Grasso C, Denti P, et al. Impact of Transcatheter Edge-to-Edge Mitral Valve Repair on Atrial Functional Mitral Regurgitation from the GIOTTO Registry. Am. J. Cardiol. 2024, 211, 219–227. DOI:10.1016/j.amjcard.2023.11.007 [Google Scholar]
-
von Stein P, von Stein J, Hohmann C, Wienemann H, Guthoff H, Körber MI, et al. Atrial Functional Mitral Regurgitation Subtypes Undergoing Transcatheter Edge-to-Edge Repair: Suboptimal Outcomes in Atriogenic Hamstringing. JACC Cardiovasc. Imaging 2025, 18, 16–29. DOI:10.1016/j.jcmg.2024.06.019 [Google Scholar]
-
Omori N, Amano M, Kaneko T, Sato Y, Ohno Y, Obokata M, et al. Factors Associated with and Prognosis of Atriogenic Tethering in Patients with Atrial Functional Mitral Regurgitation. Circ. J. 2025, pp CJ-25. DOI:10.1253/circj.CJ-25-0465 [Google Scholar]
-
Srinivasan A, Brown J, Ahmed H, Daniel M. PASCAL repair system for patients with mitral regurgitation: A systematic review. Int. J. Cardiol. 2023, 376, 108–114. DOI:10.1016/j.ijcard.2023.01.026 [Google Scholar]
-
Webb JG, Hensey M, Szerlip M, Schäfer U, Cohen GN, Kar S, et al. 1-Year Outcomes for Transcatheter Repair in Patients with Mitral Regurgitation From the CLASP Study. JACC Cardiovasc. Interv. 2020, 13, 2344–2357. DOI:10.1016/j.jcin.2020.06.019 [Google Scholar]
-
Lim DS, Smith RL, Gillam LD, Zahr F, Chadderdon S, Makkar R, et al. Randomized Comparison of Transcatheter Edge-to-Edge Repair for Degenerative Mitral Regurgitation in Prohibitive Surgical Risk Patients. JACC Cardiovasc. Interv. 2022, 15, 2523–2536. DOI:10.1016/j.jcin.2022.09.005 [Google Scholar]
-
Zahr F, Smith RL, Gillam LD, Chadderdon S, Makkar R, von Bardeleben RS, et al. One-Year Outcomes From the CLASP IID Randomized Trial for Degenerative Mitral Regurgitation. JACC Cardiovasc. Interv. 2023, 16, 2803–2816. DOI:10.1016/j.jcin.2023.10.002 [Google Scholar]
-
Marcoff L, Koulogiannis K, Aldaia L, Mediratta A, Chadderdon SM, Makar MM, et al. Echocardiographic Outcomes with Transcatheter Edge-to-Edge Repair for Degenerative Mitral Regurgitation in Prohibitive Surgical Risk Patients. JACC Cardiovasc. Imaging 2024, 17, 471–485. DOI:10.1016/j.jcmg.2023.09.015 [Google Scholar]
-
Zygouri A, Rasmeehirun P, L’Official G, Papadopoulos K, Ikonomidis I, Donal E. Screening and Procedural Guidance for Mitral Transcatheter Edge-to-Edge Repair (M-TEER). J. Clin. Med. 2025, 14, 4902. DOI:10.3390/jcm14144902 [Google Scholar]
-
Kaewkes D, Kagawa S, Patel V, Omori T, Koseki K, Koren O, et al. Multidetector Computed Tomography in Patients Who Underwent Transcatheter Mitral Edge-to-Edge Repair. Am. J. Cardiol. 2023, 208, 101–110. DOI:10.1016/j.amjcard.2023.09.027 [Google Scholar]
-
Preda A, Coppi F, Melillo F, Leo G, Margonato D, Sgura FA, et al. Transcatheter Approaches to Atrial Functional Mitral Regurgitation: How Far Have We Come? Catheter. Cardiovasc. Interv. 2025, 106, 28–40. DOI:10.1002/ccd.31368 [Google Scholar]
-
Siminiak T, Hoppe UC, Schofer J, Haude M, Herrman JP, Vainer J, et al. Effectiveness and safety of percutaneous coronary sinus-based mitral valve repair in patients with dilated cardiomyopathy (from the AMADEUS trial). Am. J. Cardiol. 2009, 104, 565–570. DOI:10.1016/j.amjcard.2009.04.021 [Google Scholar]
-
Zamorano JL, Badano LP, Bruce C, Chan KL, Gonçalves A, Hahn RT, et al. EAE/ASE recommendations for the use of echocardiography in new transcatheter interventions for valvular heart disease. J. Am. Soc. Echocardiogr. 2011, 24, 937–965. DOI:10.1016/j.echo.2011.07.003 [Google Scholar]
-
Schofer J, Siminiak T, Haude M, Herrman JP, Vainer J, Wu JC, et al. Percutaneous mitral annuloplasty for functional mitral regurgitation: Results of the CARILLON Mitral Annuloplasty Device European Union Study. Circulation 2009, 120, 326–333. DOI:10.1161/CIRCULATIONAHA.109.849885 [Google Scholar]
-
Siminiak T, Wu JC, Haude M, Hoppe UC, Sadowski J, Lipiecki J, et al. Treatment of functional mitral regurgitation by percutaneous annuloplasty: Results of the TITAN Trial. Eur. J. Heart Fail. 2012, 14, 931–938. DOI:10.1093/eurjhf/hfs076 [Google Scholar]
-
Witte KK, Lipiecki J, Siminiak T, Meredith IT, Malkin CJ, Goldberg SL, et al. The REDUCE FMR Trial: A Randomized Sham-Controlled Study of Percutaneous Mitral Annuloplasty in Functional Mitral Regurgitation. JACC Heart Fail. 2019, 7, 945–955. DOI:10.1016/j.jchf.2019.06.011 [Google Scholar]
-
Goldberg SL, Meredith I, Marwick T, Haluska BA, Lipiecki J, Siminiak T, et al. A randomized double-blind trial of an interventional device treatment of functional mitral regurgitation in patients with symptomatic congestive heart failure-Trial design of the REDUCE FMR study. Am. Heart J. 2017, 188, 167–174. DOI:10.1016/j.ahj.2017.02.032 [Google Scholar]
-
Choure AJ, Garcia MJ, Hesse B, Sevensma M, Maly G, Greenberg NL, et al. In vivo analysis of the anatomical relationship of coronary sinus to mitral annulus and left circumflex coronary artery using cardiac multidetector computed tomography: Implications for percutaneous coronary sinus mitral annuloplasty. J. Am. Coll. Cardiol. 2006, 48, 1938–1945. DOI:10.1016/j.jacc.2006.07.043 [Google Scholar]
-
Maselli D, Guarracino F, Chiaramonti F, Mangia F, Borelli G, Minzioni G. Percutaneous mitral annuloplasty: An anatomic study of human coronary sinus and its relation with mitral valve annulus and coronary arteries. Circulation 2006, 114, 377–380. DOI:10.1161/CIRCULATIONAHA.105.609883 [Google Scholar]
-
Tops LF, Van de Veire NR, Schuijf JD, de Roos A, van der Wall EE, Schalij MJ, et al. Noninvasive evaluation of coronary sinus anatomy and its relation to the mitral valve annulus: Implications for percutaneous mitral annuloplasty. Circulation 2007, 115, 1426–1432. DOI:10.1161/CIRCULATIONAHA.106.677880 [Google Scholar]
-
Rottlander D, Godde M, Degen H, Ogutcu A, Saal M, Haude M. Procedural planning of CS-based indirect mitral annuloplasty using CT-angiography. Catheter. Cardiovasc. Interv. 2021, 98, 1393–1401. DOI:10.1002/ccd.29824 [Google Scholar]
-
Rottländer D, Ballof J, Gödde M, Degen H, Ögütcü A, Alektorov K, et al. CT-Angiography to predict outcome after indirect mitral annuloplasty in patients with functional mitral regurgitation. Catheter. Cardiovasc. Interv. 2021, 97, 495–502. DOI:10.1002/ccd.29107 [Google Scholar]
-
Khan MS, Siddiqi TJ, Butler J, Friede T, Levy WC, Witte KK, et al. Functional outcomes with Carillon device over 1 year in patients with functional mitral regurgitation of Grades 2+ to 4+: Results from the REDUCE-FMR trial. ESC Heart Fail. 2021, 8, 872–878. DOI:10.1002/ehf2.13273 [Google Scholar]
-
Krishnaswamy A, Kapadia SR. Indirect Mitral Annuloplasty Using the Carillon Device. Front. Cardiovasc. Med. 2020, 7, 576058. DOI:10.3389/fcvm.2020.576058 [Google Scholar]
-
Sorgente A, Truong QA, Conca C, Singh JP, Hoffmann U, Faletra FF, et al. Influence of left atrial and ventricular volumes on the relation between mitral valve annulus and coronary sinus. Am. J. Cardiol. 2008, 102, 890–896. DOI:10.1016/j.amjcard.2008.05.044 [Google Scholar]
-
Doherty JU, Kort S, Mehran R, Schoenhagen P, Soman P, Dehmer GJ, et al. ACC/AATS/AHA/ASE/ASNC/HRS/SCAI/SCCT/SCMR/STS 2019 Appropriate Use Criteria for Multimodality Imaging in the Assessment of Cardiac Structure and Function in Nonvalvular Heart Disease: A Report of the American College of Cardiology Appropriate Use Criteria Task Force, American Association for Thoracic Surgery, American Heart Association, American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Computed Tomography, Society for Cardiovascular Magnetic Resonance, and the Society of Thoracic Surgeons. J. Am. Coll. Cardiol. 2019, 73, 488–516. DOI:10.1016/j.jacc.2018.10.038 [Google Scholar]
-
Miller M, Thourani VH, Whisenant B. The Cardioband transcatheter annular reduction system. Ann. Cardiothorac. Surg. 2018, 7, 741–747. DOI:10.21037/acs.2018.10.10 [Google Scholar]
-
Ferrero Guadagnoli A, De Carlo C, Maisano F, Ho E, Saccocci M, Cuevas O, et al. Cardioband system as a treatment for functional mitral regurgitation. Expert. Rev. Med. Devices 2018, 15, 415–421. DOI:10.1080/17434440.2018.1485487 [Google Scholar]
-
Maisano F, Taramasso M, Nickenig G, Hammerstingl C, Vahanian A, Messika-Zeitoun D, et al. Cardioband, a transcatheter surgical-like direct mitral valve annuloplasty system: Early results of the feasibility trial. Eur. Heart J. 2016, 37, 817–825. DOI:10.1093/eurheartj/ehv603 [Google Scholar]
-
Nickenig G, Hammerstingl C, Schueler R, Topilsky Y, Grayburn PA, Vahanian A, et al. Transcatheter Mitral Annuloplasty in Chronic Functional Mitral Regurgitation: 6-Month Results with the Cardioband Percutaneous Mitral Repair System. JACC Cardiovasc. Interv. 2016, 9, 2039–2047. DOI:10.1016/j.jcin.2016.07.005 [Google Scholar]
-
Kuwata S, Taramasso M, Guidotti A, Nietlispach F, Maisano F. Evaluation of Valtech’s transcatheter mitral valve repair device. Expert. Rev. Med. Devices 2017, 14, 189–195. DOI:10.1080/17434440.2017.1292122 [Google Scholar]
-
Messika-Zeitoun D, Nickenig G, Latib A, Kuck KH, Baldus S, Schueler R, et al. Transcatheter mitral valve repair for functional mitral regurgitation using the Cardioband system: 1 year outcomes. Eur. Heart J. 2019, 40, 466–472. DOI:10.1093/eurheartj/ehy424 [Google Scholar]
-
Rogers JH, Boyd WD, Smith TW, Bolling SF. Transcatheter Mitral Valve Direct Annuloplasty with the Millipede IRIS Ring. Interv. Cardiol. Clin. 2019, 8, 261–267. DOI:10.1016/j.iccl.2019.02.001 [Google Scholar]
-
Bonow RO, O'Gara PT, Adams DH, Badhwar V, Bavaria JE, Elmariah S, et al. Multisociety expert consensus systems of care document 2019 AATS/ACC/SCAI/STS expert consensus systems of care document: Operator and institutional recommendations and requirements for transcatheter mitral valve intervention: A Joint Report of the American Association for Thoracic Surgery, the American College of Cardiology, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons. Catheter. Cardiovasc. Interv. 2020, 95, 866–884. DOI:10.1002/ccd.28671 [Google Scholar]
-
Iliakis P, Dimitriadis K, Pyrpyris N, Beneki E, Theofilis P, Tsioufis P, et al. Atrial Functional Mitral Regurgitation: From Diagnosis to Current Interventional Therapies. J. Clin. Med. 2024, 13, 5035. DOI:10.3390/jcm13175035 [Google Scholar]
-
Makkar RR, Chikwe J, Chakravarty T, Chen Q, O’Gara PT, Gillinov M, et al. Transcatheter Mitral Valve Repair for Degenerative Mitral Regurgitation. JAMA 2023, 329, 1778–1788. DOI:10.1001/jama.2023.7089 [Google Scholar]
-
Guerrero ME, Daniels DV, Makkar RR, Thourani VH, Asch FM, Pham M, et al. Percutaneous transcatheter valve replacement in individuals with mitral regurgitation unsuitable for surgery or transcatheter edge-to-edge repair: A prospective, multicountry, single-arm trial. Lancet 2025, 406, 2541–2550. DOI:10.1016/S0140-6736(25)02073-2 [Google Scholar]
-
Muller DWM, Sorajja P, Duncan A, Bethea B, Dahle G, Grayburn P, et al. 2-Year Outcomes of Transcatheter Mitral Valve Replacement in Patients with Severe Symptomatic Mitral Regurgitation. J. Am. Coll. Cardiol. 2021, 78, 1847–1859. DOI:10.1016/j.jacc.2021.08.060 [Google Scholar]
-
Conradi L, Ludwig S, Sorajja P, Duncan A, Bethea B, Dahle G, et al. Clinical outcomes and predictors of transapical transcatheter mitral valve replacement: The Tendyne Expanded Clinical Study. EuroIntervention 2024, 20, e887–e897. DOI:10.4244/EIJ-D-23-00904 [Google Scholar]
-
Alperi A, Granada JF, Bernier M, Dagenais F, Rodes-Cabau J. Current Status and Future Prospects of Transcatheter Mitral Valve Replacement: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2021, 77, 3058–3078. DOI:10.1016/j.jacc.2021.04.051 [Google Scholar]
-
Tartaglia F, Antonelli G, Gitto M, Stankowski K, Donia D, Stefanini G, et al. TMVR for the Treatment of Mitral Regurgitation: A State-of-the-Art Review. Circ. Cardiovasc. Interv. 2025, 18, e015298. DOI:10.1161/CIRCINTERVENTIONS.125.015298 [Google Scholar]
-
Fiorilli PN, Herrmann HC. Transcatheter Mitral Valve Replacement: Rationale and Current Status. Annu. Rev. Med. 2020, 71, 249–261. DOI:10.1146/annurev-med-051418-060028 [Google Scholar]
-
Penteris M, Lampropoulos K. The SAPIEN M3 system for transcatheter mitral valve replacement: A new era begins. Cardiovasc. Revasc Med. 2025, 79, 120–125. DOI:10.1016/j.carrev.2025.06.034 [Google Scholar]
-
Penteris M, Lampropoulos K. Device profile of the EVOQUE Eos system for transcatheter mitral valve replacement: A brief narrative review. Expert. Rev. Med. Devices 2025, 22, 1173–1179. DOI:10.1080/17434440.2025.2561242 [Google Scholar]
-
Ludwig S, Perrin N, Coisne A, Ben Ali W, Weimann J, Adam M, et al. Clinical outcomes of transcatheter mitral valve replacement: Two-year results of the CHOICE-MI Registry. EuroIntervention 2023, 19, 512–525. DOI:10.4244/EIJ-D-22-01037 [Google Scholar]
-
Yoon SH, Whisenant BK, Bleiziffer S, Delgado V, Dhoble A, Schofer N, et al. Outcomes of transcatheter mitral valve replacement for degenerated bioprostheses, failed annuloplasty rings, and mitral annular calcification. Eur. Heart J. 2019, 40, 441–451. DOI:10.1093/eurheartj/ehy590 [Google Scholar]
-
Tiwana J, Aldea G, Levin DB, Johnson K, Don CW, Dvir D, et al. Contemporary Transcatheter Mitral Valve Replacement for Mitral Annular Calcification or Ring. JACC Cardiovasc. Interv. 2020, 13, 2388–2398. DOI:10.1016/j.jcin.2020.07.007 [Google Scholar]
-
Fongrat N, Makhijani U, Vajayakumar N, Mangano A, Iantorno M. Advances in Transcatheter Mitral Valve Replacement (TMVR) in Patients with Mitral Annular Calcification: A Case Report of Acute Hemolytic Anemia and Review of Contemporary Approaches. J. Clin. Med. 2025, 14, 4660. DOI:10.3390/jcm14134660 [Google Scholar]
-
Blanke P, Naoum C, Webb J, Dvir D, Hahn RT, Grayburn P, et al. Multimodality Imaging in the Context of Transcatheter Mitral Valve Replacement: Establishing Consensus Among Modalities and Disciplines. JACC Cardiovasc. Imaging 2015, 8, 1191–1208. DOI:10.1016/j.jcmg.2015.08.004 [Google Scholar]
-
Barreiro-Perez M, Caneiro-Queija B, Puga L, Gonzalez-Ferreiro R, Alarcon R, Parada JA, et al. Imaging in Transcatheter Mitral Valve Replacement: State-of-Art Review. J. Clin. Med. 2021, 10, 5973. DOI:10.3390/jcm10245973 [Google Scholar]
-
van den Dorpel MMP, de Assis LU, van Niekerk J, Nuis R, Daemen J, Ren CB, et al. Accuracy of Three-Dimensional Neo Left Ventricular Outflow Tract Simulations with Transcatheter Mitral Valve Replacement in Different Mitral Phenotypes. Catheter. Cardiovasc. Interv. 2025, 105, 249–257. DOI:10.1002/ccd.31287 [Google Scholar]
-
Besola L, Falcetta G, Ceravolo G, Fiocco A, Colli A. New echocardiographic screening tool for left ventricular tract obstruction risk assessment in TMVR. Int. J. Cardiol. 2024, 408, 132112. DOI:10.1016/j.ijcard.2024.132112 [Google Scholar]
-
Fiocco A, Besola L, De Carlo M, Whitlock RP, Colli A. Transcatheter mitral valve repair and replacement. A critical review of current real-world practice. Eur. J. Cardiothorac. Surg. 2025, 67, ezaf373. DOI:10.1093/ejcts/ezaf373 [Google Scholar]
-
Sengupta PP, Kluin J, Lee SP, Oh JK, Smits A. The future of valvular heart disease assessment and therapy. Lancet 2024, 403, 1590–1602. DOI:10.1016/S0140-6736(23)02754-X [Google Scholar]
-
Armoundas AA, Narayan SM, Arnett DK, Spector-Bagdady K, Bennett DA, Celi LA, et al. Use of Artificial Intelligence in Improving Outcomes in Heart Disease: A Scientific Statement from the American Heart Association. Circulation 2024, 149, e1028–e1050. DOI:10.1161/CIR.0000000000001201 [Google Scholar]
-
Sadeghpour A, Jiang Z, Hummel YM, Frost M, Lam CSP, Shah SJ, et al. An Automated Machine Learning-Based Quantitative Multiparametric Approach for Mitral Regurgitation Severity Grading. JACC Cardiovasc. Imaging 2025, 18, 1–12. DOI:10.1016/j.jcmg.2024.06.011 [Google Scholar]
-
Long A, Finer J, Hartman H, Hartzel D, Jing L, Kelsey C, et al. Deep learning for echocardiographic assessment and risk stratification of aortic, mitral, and tricuspid regurgitation: The DELINEATE-regurgitation study. Eur. Heart J. 2025, 46, 2780–2791. DOI:10.1093/eurheartj/ehaf248 [Google Scholar]
-
Hanneman K, Playford D, Dey D, van Assen M, Mastrodicasa D, Cook TS, et al. Value Creation Through Artificial Intelligence and Cardiovascular Imaging: A Scientific Statement From the American Heart Association. Circulation 2024, 149, e296–e311. DOI:10.1161/CIR.0000000000001202 [Google Scholar]