Therapeutic Plasma Exchange Rapidly Restores Red Blood Cell Deformability in Waldenström Macroglobulinemia
Naijia Liu 1,2 Shanye Yin 2,3,* Wenjun Deng 4,*
Received: 03 February 2026 Revised: 10 March 2026 Accepted: 20 March 2026 Published: 07 April 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Background
Waldenström macroglobulinemia (WM) is a rare B-cell lymphoproliferative disorder characterized by the overproduction of monoclonal immunoglobulin M (IgM), leading to increased plasma viscosity [1]. The common clinical manifestations of WM, including vision changes, neurological deficits, and mucosal bleeding, are primarily attributed to hyperviscosity syndrome, which impairs blood flow and tissue oxygenation [2]. However, microvascular complications also occur in patients without overt hyperviscosity, suggesting the presence of additional determinants of microcirculation dysfunction beyond plasma viscosity alone [3].
Red blood cells (RBCs) are the primary oxygen carriers in the circulation. RBC deformability, the ability to constantly change shape and recover, is essential for them to traverse tiny capillaries (2–5 μm) smaller than their resting diameter (7–8 μm), thereby ensuring efficient oxygen delivery in microcirculation. Impaired RBC deformability has been implicated in vascular complications across a range of hematologic and inflammatory conditions [4,5]. Prior WM studies reported altered RBC rheology using filtrometer-based approaches; however, these analyses were limited in scope and did not fully assess deformability under physiologically relevant shear stress and osmotic conditions [6,7]. It remained unclear whether extreme IgM elevation directly impairs intrinsic RBC mechanical properties and whether such changes are reversible.
2. Case Report
A 69-year-old man with a history of CD5-negative, CD10-negative low-grade B-cell lymphoma had been receiving zanubrutinib, which was held due to a traumatic left-sided subdural hematoma (SDH) two months prior to the current presentation. The hematoma was managed conservatively. At the current evaluation, interval neuroimaging revealed unexpected expansion of the prior SDH, a new contralateral subdural collection in the absence of additional trauma, and evidence of white matter microvascular ischemic changes.
On admission, the patient was found to have a markedly elevated serum IgM of 7467 mg/dL, consistent with WM. Clinically, he did not exhibit classic features of hyperviscosity syndrome such as mucosal bleeding or focal neurologic deficits, and reported only intermittent headache and increasing fatigue. Complete blood count revealed a white blood cell count of 6.4 × 109/L and platelet count of 178 × 109/L, both within normal limits. However, RBC indices were substantially reduced, with a hemoglobin level of 7.2 g/dL, hematocrit of 22.9%, RBC count of 3.54 × 109/L, mean corpuscular volume of 64.7 fL, and mean corpuscular hemoglobin of 20.3 pg (Table 1).
Given the extreme IgM elevation and concern for potential subclinical microcirculatory impairment, the patient was urgently admitted for inpatient management. Therapeutic plasma exchange (TPE) was initiated to reduce IgM burden despite the absence of overt hyperviscosity symptoms. The patient underwent two TPE sessions over 48 h, resulting in a reduction of serum IgM to 3643 mg/dL, accompanied by improvement in RBC indices and overall clinical stabilization (Table 1). At the 3-month follow-up, the patient reported a general sense of well-being without new symptoms or significant headaches. Repeated neuroimaging demonstrated relatively stable left SDH and minimal progression of the right-sided SDH. Serum IgM remained at 3452 mg/dL, and RBC indices continued to show sustained improvement.
Table 1. Complete blood count results.
|
Parameter |
Pre-TPE |
Post-TPE 1 |
Post-TPE 2 |
Follow-Up |
Reference Range |
|---|---|---|---|---|---|
|
Hemoglobin (g/dL) |
7.2 |
8.1 |
9.0 |
9.8 |
14.0–18.0 |
|
Hematocrit (%) |
22.9 |
24.8 |
26.9 |
30.2 |
42–52 |
|
RBC count (×1012/L) |
3.54 |
3.86 |
4.07 |
4.43 |
4.5–5.9 |
|
Mean Corpuscular Volume (MCV) (fL) |
64.7 |
64.2 |
66.1 |
68.2 |
80.0–96.0 |
|
Mean Corpuscular Hemoglobin (MCH) (pg) |
20.3 |
21.0 |
22.1 |
22.1 |
27.0–33.0 |
|
Mean Corpuscular Hemoglobin Concentration (MCHC) (g/dL) |
31.4 |
32.7 |
33.5 |
32.5 |
33.0–36.0 |
|
Red Blood Cell Distribution Width (RDW) (%) |
16.3 |
15.9 |
15.7 |
17.8 |
12.2–16.4 |
|
WBC count (×109/L) |
6.4 |
9.4 |
7.7 |
6.4 |
4.5–11.0 |
|
Neutrophil (%) |
60.0 |
30.0 |
39.0 |
76.0 |
40.0–70.0 |
|
Lymphocyte (%) |
26.0 |
50.0 |
36.0 |
20.0 |
22.0–44.0 |
|
Monocyte (%) |
7.0 |
11.0 |
11.0 |
2.0 |
3.0–9.0 |
|
Eosinophil (%) |
5.0 |
8.0 |
13.0 |
1.0 |
0.0–5.0 |
|
Basophil (%) |
1.0 |
1.0 |
1.0 |
1.0 |
0.0–3.0 |
|
Platelet count (×109/L) |
178 |
167 |
125 |
194 |
150–400 |
|
Serum IgM (mg/dL) |
7467 |
4508 |
3643 |
3452 |
<40 |
To investigate the effects of extreme IgM on RBCs, we measured RBC mechanical features using LORRCA MaxSis laser diffraction ektacytometry (RR Mechatronics, Hoorn, The Netherlands). RBC deformability was evaluated by measuring the elongation index (EI), an indicator of the ability of RBCs to elongate, under a range of shear stresses (0.3–30 Pa) to generate a deformability curve (Figure 1A), and across an osmotic gradient at a fixed shear stress of 30 Pa to generate an osmoscan profile (Figure 1B). EI was calculated as (A − B)/(A + B), where A and B represent the length and width of the RBC diffraction pattern. Key parameters included maximum EI (EI max), representing maximal deformability; osmolality at minimum deformability (O min), reflecting surface-to-volume ratio and osmotic fragility; and osmolality at half of EI max in the hypertonic region, indicating cellular hydration status. All measurements were performed on the fresh blood obtained before treatment, after each TPE session, at 3-month follow-up, and from a healthy control.
Before treatment, the patient demonstrated markedly reduced RBC deformability, with lower EI values across a wide range of physiologically relevant shear stresses compared to a healthy control (Figure 1A). Osmoscan analysis further revealed a reduced EI max, confirming diminished cellular flexibility (Figure 1B). In addition, the osmoscan curve in the WM patient displayed a leftward shift toward a hypotonic condition, suggesting increased surface-to-volume ratio and relative cellular dehydration (Figure 1B). Following TPE, RBC deformability improved rapidly, with increases in EI values observed in both the deformability curve and osmoscan profile (Figure 1A–C). The beneficial effects were sustained at the 3-month follow-up, with continued restoration in cellular hydration status (Figure 1A–C). These improvements closely paralleled reductions in serum IgM levels (Table 1).
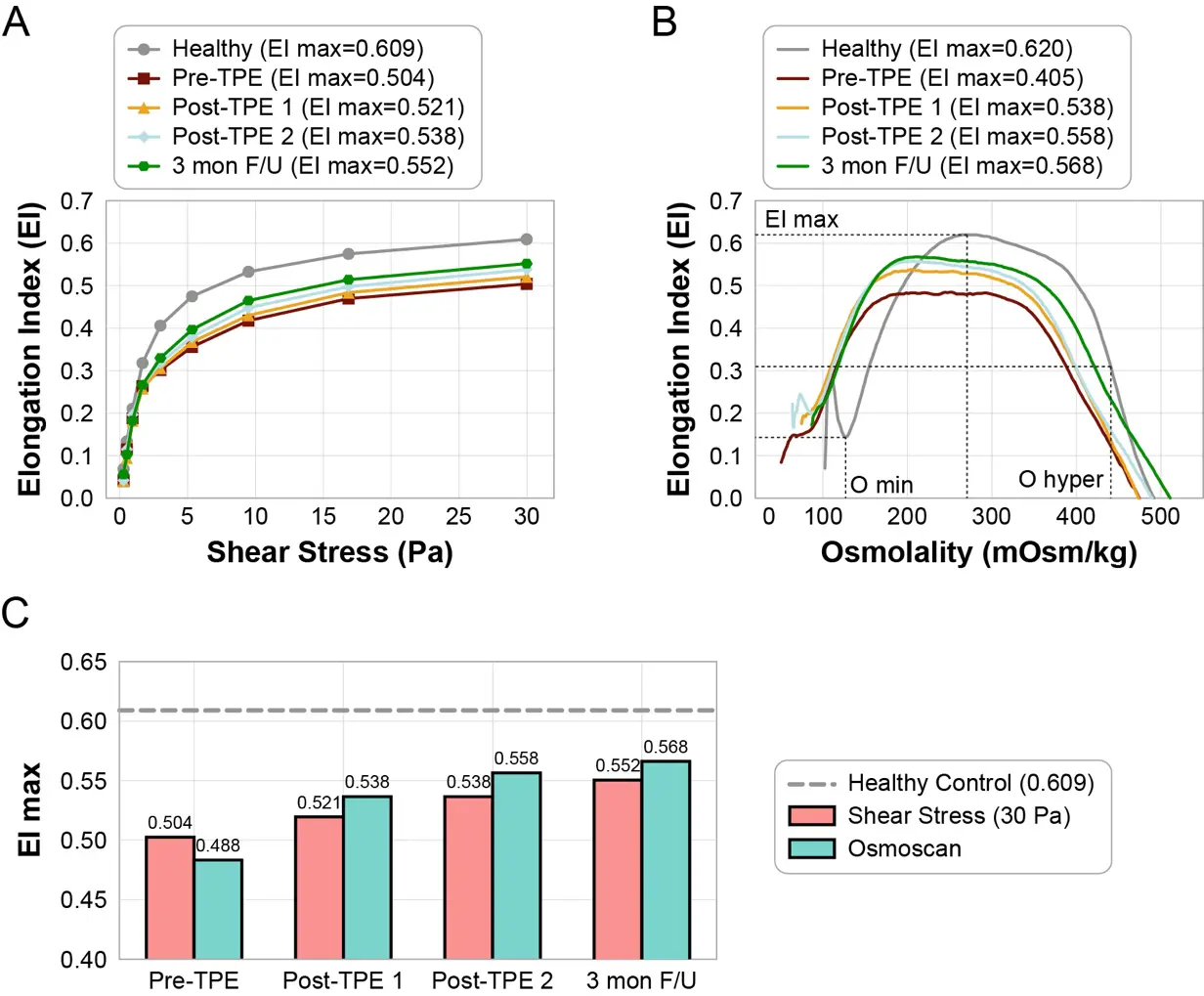
Figure 1. Red blood cell (RBC) mechanical properties in a patient with Waldenström macroglobulinemia (WM). (A) RBC deformability curves in a healthy control and in the WM patient before and after two sessions of therapeutic plasma exchange (TPE) treatment. (B) Osmoscan profiles of RBCs from the healthy control and the WM patients before and after two TPE sessions. (C) Maximal elongation index (EI max) from deformability and osmoscan measurements.
3. Discussion
WM is characterized by the excessive production of monoclonal IgM, which can lead to hyperviscosity syndrome and a spectrum of microcirculatory complications. In this report, we demonstrate that extreme IgM elevation in WM is associated with substantial impairment of RBC deformability. Importantly, these abnormalities were rapidly and partially corrected following IgM reduction with TPE, supporting a reversible IgM-mediated alteration of RBC mechanical properties, rather than irreversible cellular injury.
RBC deformability is critical for maintaining effective oxygen delivery within the microcirculation, particularly in organs with dense capillary networks, such as the brain [4,5]. In the present case, reduced RBC deformability in the setting of elevated IgM may increase microvascular flow resistance and impair tissue perfusion, thereby contributing to microvascular ischemia and injury. Alterations in cellular hydration and surface-to-volume ratio may further compromise RBC mechanical fitness and worsen microcirculatory dysfunction. In line with this, our patient showed imaging evidence of brain microvascular injury and unexpected progression of SDH despite minimal clinical hyperviscosity symptoms, highlighting the potential relevance of subclinical RBC mechanical abnormalities.
The rapid improvement in RBC deformability following TPE closely paralleled the reduction in circulating IgM levels, supporting a direct relationship between IgM burden and RBC mechanical dysfunction. Several mechanisms may underlie this phenomenon. Elevated IgM may directly interact with the RBC membrane, alter membrane fluidity, and disrupt ion and water transport, thereby reducing cellular deformability and increasing resistance to shear stress. In addition, indirect effects, such as changes in plasma osmolality and enhanced RBC aggregation, may further diminish RBC flexibility. However, these mechanisms remain largely unknown and require further systematic investigation.
Our findings suggest that RBC mechanical dysfunction may represent an underrecognized contributor to microcirculatory impairment in WM. While serum IgM levels and plasma viscosity are routinely used to assess hyperviscosity risk and guide clinical management, they do not capture the contribution of RBC rheology to microvascular flow dynamics, especially in patients without overt clinical symptoms but with evidence of end-organ injury. Direct assessment of RBC deformability using ektacytometry may therefore provide additional insight into the pathophysiological consequences of extreme IgM elevation. Compared with earlier filtrometer-based approaches [6,7], ektacytometry offers a comprehensive, quantitative evaluation of RBC mechanical behavior across physiologically relevant conditions, providing a more sensitive measure of RBC functional integrity. Moreover, the rapid improvement in RBC deformability following TPE highlights the reversibility of this process and broadens our understanding of the clinical benefits of IgM reduction. These observations highlight that RBC mechanical features may serve as sensitive markers of subclinical microcirculatory dysfunction, potentially identifying patients who may benefit from early intervention even in the absence of hyperviscosity symptoms.
While the direct functional assessment of RBC mechanics before and after IgM reduction provides compelling evidence linking extreme IgM levels with reversible impairment of RBC deformability, this case has several limitations, including its single-patient design and the absence of longitudinal plasma viscosity measurements. Future studies in an expanded patient cohort, with close assessment of plasma viscosity and RBC deformability, are needed to better define the relationship among IgM, RBC mechanics, and microcirculation. In addition, as current WM treatment often includes chemoimmunotherapy (e.g., bendamustine and rituximab) or targeted agents (e.g., ibrutinib and zanubrutinib) [8,9,10], the potential effects of these therapies on RBC mechanical properties warrant further investigation.
In summary, this case demonstrates that severe IgM elevation in WM can impair RBC deformability and that these abnormalities improve rapidly following TPE. These findings provide mechanistic insight into the microcirculatory complications of WM and suggest that RBC mechanical assessment may represent a useful tool for detecting subclinical hyperviscosity and guiding therapeutic decision-making.
Author Contributions
N.L. collected the clinical data and drafted the manuscript. S.Y. conceived and designed the study. W.D. co-conceived and co-designed the study, performed and analyzed the red blood cell deformability assays. All authors reviewed and approved the final manuscript.
Ethics Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Montefiore Cancer Center Institutional Ethics Committee and Institutional Review Board (Reference committee number: 2014-3056; approval date: 30 September 2015).
Informed Consent Statement
Patient consent was waived due to the retrospective nature of the study.
Data Availability Statement
The raw data supporting the conclusions of this article are not publicly available because of data privacy restrictions but can be accessed upon reasonable request with appropriate institutional approvals.
Funding
This work was supported in part by NIH grants R01NS134819 (W.D.), R21NS140800 (W.D.), R01CA291607 (S.Y.), and R21CA267527 (S.Y.), as well as a grant from the Feldstein Medical Foundation (S.Y.).
Declaration of Competing Interest
W.D. and S.Y. hold equity in Yihui Bio, Inc. All other authors declare no potential conflicts of interest.
References
- Gertz MA. Waldenstrom macroglobulinemia: 2023 update on diagnosis, risk stratification, and management. Am. J. Hematol. 2023, 98, 348–358. DOI:10.1002/ajh.26796 [Google Scholar]
- Treon SP, Sarosiek S, Castillo JJ. Diagnosis and management of Waldenstrom’s macroglobulinemia. Hematol. Oncol. 2025, 43 (Suppl. S2), e70071. DOI:10.1002/hon.70071 [Google Scholar]
- Patel N, Al Hadidi S, Yellapragada S. Pathophysiology and Treatments of Complications of Waldenstrom’s Macroglobulinemia. Clin. Hematol. Int. 2024, 6, 11–18. DOI:10.46989/001c.124268 [Google Scholar]
- Yazdanbakhsh M, Phan C, Patel P, Simpson E, Wingnean K, Acker JP. RBC deformability and clinical relevance in transfusion recipients. Transfus. Apher. Sci. 2025, 64, 104278. DOI:10.1016/j.transci.2025.104278 [Google Scholar]
- Prudinnik DS, Kussanova A, Vorobjev IA, Tikhonov A, Ataullakhanov FI, Barteneva NS. Deformability of Heterogeneous Red Blood Cells in Aging and Related Pathologies. Aging Dis. 2024, 16, 1242–1264. DOI:10.14336/AD.2024.0526 [Google Scholar]
- Ramakrishnan S, Degenhardt R, Vietzke K. Erythrocyte deformability in Waldenström’s macroglobulinemia. Clin. Hemorheol. Microcirc. 2000, 22, 17–20. DOI:10.3233/CHM-2000-212 [Google Scholar]
- Ramakrishnan S, Grebe R, Singh M, Schmid-Schönbein H. Haemorheological behaviour of erythrocytes & risk factor profile in patients suffering from Waldenström’s macroglobulinaemia. Indian J. Med. Res. 1998, 107, 128–133. Available online: https://search.proquest.com/openview/5683395c9e4c3af28f7abd360918ef7c/1?pq-origsite=gscholar&cbl=37533 (accessed on 19 March 2026).
- Gertz MA. Waldenström macroglobulinemia: 2025 update on diagnosis, risk stratification, and management. Am. J. Hematol. 2025, 100, 1061–1073. DOI:10.1002/ajh.27666 [Google Scholar]
- Kapoor P, Rajkumar SV. Current approach to Waldenstrom macroglobulinemia. Blood Rev. 2023, 62, 101129. DOI:10.1016/j.blre.2023.101129 [Google Scholar]
- Zanwar S, Abeykoon JP. Treatment paradigm in Waldenstrom macroglobulinemia: Frontline therapy and beyond. Ther. Adv. Hematol. 2022, 13, 20406207221093962. DOI:10.1177/20406207221093962 [Google Scholar]
