Eco-Friendly Synthesis of Spherical Lanthanum-Doped Hydroxyapatite from Phosphogypsum Waste: A High-Efficiency Strategy for Fluoride Removal in Aqueous Solutions
Shanzhu Jiang
1,2,†
Zhaobo Wang
2,*,†
 Yangjie Qin
2
Bensanglang Cao
2
Ruan Chi
1,2
Yangjie Qin
2
Bensanglang Cao
2
Ruan Chi
1,2
Received: 12 January 2026 Revised: 27 January 2026 Accepted: 23 March 2026 Published: 03 April 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Phosphogypsum (PG), a byproduct of phosphoric acid production via the wet process, contains calcium sulfate dihydrate (CaSO4·2H2O) along with impurities such as residual phosphate rock (3–8% P2O5), fluoride (1.2–4.5%), heavy metals (e.g., Cd, As), and trace radionuclides [1]. China, the world’s largest PG producer, generates over 80 million tons annually, with cumulative stockpiles exceeding 800 million tons as of 2022. As China’s leading phosphate chemical hub, Hubei holds the nation’s largest phosphate rock reserves and fertilizer output. It generates nearly 30 million tons of phosphogypsum each year, making it the country’s leading source of accumulated waste. The primary factors restricting the comprehensive utilization of phosphogypsum are its high impurity content and the low economic competitiveness of its related products. Only 50% of the PG is currently utilized, primarily in low-value applications like cement retardants (33%) and soil amendments (2%), while the rest is landfilled, posing severe ecological risks through acidic leachate (pH 1.5–4.5) and fluoride migration into groundwater [2,3]. Fluoride contamination from PG stockpiles and natural sources affects 37 million people in China, particularly in Shaanxi and Xinjiang, where groundwater F− concentrations reach 6–30 mg/L [4]. Chronic exposure to 3–6 mg/L F− causes dental fluorosis (mottled enamel) and skeletal fluorosis (bone deformation), as confirmed by epidemiological studies showing 42% prevalence in endemic areas [5].
Conventional PG recycling faces three key bottlenecks: (1) The application of building materials (such as gypsum board) is facing market resistance due to user acceptance issues, approximately 3000 tons of phosphogypsum are consumed annually through this pathway [6,7]; (2) Soil amendment requires costly transportation and shows inconsistent crop yield improvement (vs. commercial fertilizers), around 600 tons of phosphogypsum are consumed annually through this pathway [8,9]; (3) Chemical conversion to high-value products (e.g., (NH4)2SO4) demands energy-intensive fluoride removal (>90% purity required), approximately 400 tons of phosphogypsum are consumed annually through this pathway [10]. Despite the above approaches, the full utilization of phosphogypsum remains challenging given its annual production volume. In order to achieve “waste for waste” comprehensive treatment model, HAP synthesized by chemical conversion of PG (PGHAP) to adsorb and remove F− in water is a novel resourceful utilization. HAP is a calcium phosphate biomaterial with the chemical formula Ca10(PO4)6(OH)2. There are two different binding sites C and P on the surface, the C site composed of Ca2+ is positively charged, and the P site composed of PO43− group is negatively charged [11]. The adsorption of F− by HAP is mainly based on its unique physicochemical properties and structural characteristics, and it has become a research hotspot in recent years as a new type of environmentally functional material. Specifically, it can be summarized as Ca2+ in HAP reacting with F− in solution to produce CaF2 precipitation, or F− replacing some of the hydroxyl group (-OH) in HAP to form fluorapatite (Ca5(PO4)3F), or F− adsorbing on the surface of HAP by electrostatic and van der Waals forces. However, pure HAP exhibits limited adsorption capacity and selectivity for fluoride under complex aqueous conditions, necessitating structural modifications to enhance its performance. Experimental studies reveal that unmodified HAP achieves an adsorption capacity of only 6.88–10.9 mg/g [12,13]. This constraint arises from the inherent structural limitations of pure HAP, including its low specific surface area, limited active sites for F− binding, and susceptibility to surface charge reversal in acidic or alkaline media [14].
To address these challenges, structural modifications of HAP have emerged as a critical strategy to enhance both adsorption capacity and selectivity. Spherical HAP achieved a synergistic improvement in specific surface area, diffusion kinetics, and active site density through geometric morphology optimization, and its adsorption capacity and rate were significantly better than non spherical structures [15]. In addition, doping with multivalent cations (e.g., La3+, Zn2+, Mg2+) has been shown to optimize the crystallinity and surface chemistry of HAP. For example, La3+-doped HAP introduces strong Lewis acid-base interactions between La3+ and F−, increasing adsorption capacity while maintaining stability across a pH range of 4–10 [16]. Based on this, this work pioneers the integration of industrial solid waste recycling with advanced adsorbent design, offering a dual solution to fluoride pollution and phosphogypsum accumulation. Advanced characterization techniques (XRD, XPS, and BET) are employed to elucidate the structural evolution and fluoride binding mechanisms. The La-PGHAP composite material is expected to exhibit excellent adsorption capacity compared to conventional HAP, while maintaining stability across a wide pH range. The findings are expected to advance the development of eco-friendly water treatment materials and provide actionable insights for scaling up PG-based adsorbents in industrial applications.
2. Materials and Methods
2.1. Materials and Reagents
A chemical enterprise in Hubei Province, China supplied PG. Potassium phosphate monobasic, hydrochloric acid, sodium hydroxide, and sodium fluoride were purchased from China National Medicines Group Chemical Reagents Co., Ltd. (Beijing, China).
2.2. Synthesis Process of Adsorbent Materials
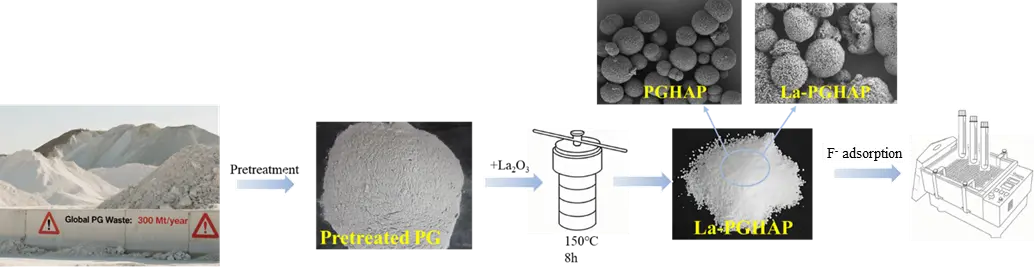
Raw phosphogypsum (50 g) was subjected to acid leaching by stirring with 250 mL of 2 mol/L hydrochloric acid at 80 °C for 2 h, followed by vacuum filtration through a 0.45 μm polyethersulfone membrane to remove insoluble impurities (e.g., silica and residual phosphate rock). The filtrate was rapidly cooled to 25 °C in an ice-water bath to precipitate calcium-rich solids, which were collected via centrifugation (6000 rpm, 10 min) and redispersed in deionized water to form a homogeneous Ca2+ suspension. This calcium source was combined with a 0.5 mol/L potassium dihydrogen phosphate (KH2PO4) solution at a stoichiometric Ca:P molar ratio of 1.67, followed by sequential addition of urea (urea:Ca = 10:1 mol/mol) and EDTA (0.03 mol/L final concentration) to regulate crystal growth kinetics. The mixture pH was adjusted to 3.0 ± 0.1 using diluted HNO3 to prevent premature hydroxyapatite nucleation [17]. For La-doped samples, predetermined quantities of La2O3 (corresponding to La:Ca molar ratios of 1%, 3%, and 5%) were dissolved in 5 mL concentrated HNO3 before incorporation into the reaction system. The precursor solution was transferred into a 100 mL Teflon-lined stainless steel autoclave and hydrothermally treated at 150 °C for 8 h under autogenous pressure (0.85 bar), during which urea decomposition (NH2CONH2 → 2NH3 + CO2) gradually elevated the pH to ~9.5, enabling controlled crystallization. Post-synthesis, the products were vacuum-filtered, washed alternately with deionized water and ethanol to eliminate ionic residues and organic byproducts, and dried overnight at 60 °C in a vacuum oven to obtain PGHAP and La-PGHAP powders (see Figure 1).
2.3. Static Batch Adsorption Test
In the static batch adsorption test, the control variable method was used to control only variables, including adsorption time, F− initial concentration, temperature, pH, and adsorbent dosing. Specifically, a certain mass of adsorbent (10–50 mg) was added to a 50 mL conical flask, and then 20 mL of F− solutions (10, 20, 30, 40, and 50 mg/L) with different concentrations having different pH (2, 4, 6, 8, and 10) were added. The static adsorption test was carried out in a thermostatic water bath shaker set at 200 rpm, and the thermostatic oscillator was set at different temperatures (25, 30, 35, 40, and 45 °C), and the concentration of F− in the supernatant was determined by ion chromatography. The above experiments were repeated three times to take the average value.
2.4. Calculation and Analytical Methods
The amount of F− adsorbed by the adsorbent at time t (qt, mg/g) and the amount adsorbed at the equilibrium moment (qe, mg/g) were calculated by Equation (1) and Equation (2), respectively.
where c0 is the initial F− concentration, mg/L; ct is the concentration of F at time t, mg/L; ce is the F− concentration at the equilibrium moment, mg/L; M is the adsorbent dosage, g; V is the volume of F-solution, L.
The dynamic curves were fitted by pseudo-first-order and pseudo-second-order kinetic models, and the kinetic parameters, such as pseudo-first-order rate constant (k1, h−1), pseudo-second-order rate constant (g/mg·h), the theoretical adsorption capacity (qe0, mg/g), and the number of data points (n) were calculated. The isotherm curves were fitted by the Freundlich and Langmuir model, and the thermodynamic parameters, such as Gibbs energy (ΔG, kJ·mol−1), enthalpy (ΔH, kJ·mol−1) and entropy [ΔS, J·(mol·K)−1] were calculated according to Equation (3), Equation (4) and Equation (5). R is the ideal-gas constant, 8.314 J·(mol·K)−1.
3. Results and Discussion
3.1. Morphological and Compositional Analysis of La-PGHAP
The SEM images in Figure 2 vividly illustrate the series of dynamic morphological changes induced by the gradual increase of La doping concentration. The undoped PGHAP (Figure 2a) serves as a reference, exhibiting well-defined, smooth spherical particles approximately 2–3 μm in diameter. Upon introducing a low La doping level (1%, Figure 2b), the first significant morphological transition occurs. The originally smooth surfaces of the microspheres are uniformly decorated with sub-100 nm protrusions. This phenomenon is attributed to La3+-mediated heterogeneous nucleation, marking the initial stage where La incorporation begins to alter the crystal growth habit. As the doping concentration is raised to 3% (Figure 2c), a more profound transformation takes place. The primary particles lose their individual spherical identity and aggregate into larger clusters ranging from 5 to 7 μm. This aggregation is primarily driven by the charge imbalance created by the substitution of Ca2+ with La3+ (as evidenced by the decrease in Ca content from 11.61 at.% to 8.09 at.% in Table 1), which is consistent with the charge compensation mechanism (3Ca2+ → 2La3+ + vacancy). Notably, at the highest doping level of 5% (Figure 2d), the morphology undergoes a final, distinct evolution. The structure develops pronounced porosity with pore sizes around 200 nm, and the overall particle size is reduced to 1–5 μm. This unique architecture is likely the result of synergistic effects involving surface precipitation of La(OH)3 and significant lattice strain due to the ionic radius mismatch between Ca2+ and La3+. Importantly, the highly porous and rugged morphology observed at 3% and 5% La doping is anticipated to provide a larger specific surface area and more active sites, which is highly favorable for the subsequent fluoride adsorption process discussed in the following sections [17,18].
EDS data (Table 1) quantify doping inefficiency: La atomic percentages (0.16–0.24 at.%) remain far below theoretical values (1–5%), indicating limited lattice incorporation (<25% doping efficiency) and preferential La3+ surface enrichment. Concurrently, phosphorus depletion at 5% La (2.97 at.% vs. 6.70 at.% in 1% La-PGHAP) suggests LaPO4 phase formation, while oxygen content stability (26.01–26.98 at.%) confirms maintained apatitic structure. Carbon contamination (47.81–65.06 at.%) likely originates from urea decomposition residues. These findings collectively demonstrate that La doping modifies both surface chemistry (La–O–F coordination sites) and textural properties (mesoporosity), synergistically enhancing fluoride adsorption potential despite suboptimal dopant integration [19,20].

Figure 2. SEM images of PGHAP and La-PGHAP with different La contents: (a) 0%; (b) 1%; (c) 3%; (d) 5%.
Table 1. Average atom percentage of cations of samples by EDS.
|
Element Content (at.%) |
|||||
|---|---|---|---|---|---|
|
Doping Amount |
C |
O |
P |
Ca |
La |
|
PGHAP |
60.25 |
21.28 |
7.05 |
11.42 |
0 |
|
1% La-PGHAP |
55.61 |
26.01 |
6.70 |
11.61 |
0.16 |
|
3% La-PGHAP |
47.81 |
38.67 |
5.20 |
8.09 |
0.23 |
|
5% La-PGHAP |
65.06 |
26.98 |
2.97 |
4.75 |
0.24 |
3.2. Structural and Textural Characterization of PGHAP and La-PGHAP
XRD analysis (Figure 3a) confirms the dual-phase composition of pristine PGHAP, exhibiting characteristic diffraction peaks of both CaSO4·2H2O (2θ = 11.6°, 20.7°, 23.4°) and hydroxyapatite (HAP, JCPDS 09-0432, 2θ = 25.9°, 31.8°, 49.5°), demonstrating incomplete conversion of phosphogypsum during synthesis. The coexistence of these phases suggests residual sulfate groups from the PG precursor, consistent with its industrial origin. Upon La doping (Figure 3b), the HAP phase remains dominant across all La-PGHAP samples (1–5% La), indicating successful La3+ incorporation into the HAP lattice. Notably, the (002) peak at 25.9° progressively broadens (FWHM increases from 0.38° to 0.61°) and shifts to lower angles (25.9° → 25.6°) with higher La loading, confirming La3+ substitution at Ca2+ sites (ionic radius mismatch) and resultant lattice expansion along the c-axis.
N2 adsorption-desorption isotherms (Figure 3c) reveal a systematic enhancement in textural properties with La doping. All samples display Type IV isotherms with H3 hysteresis loops (P/P0 = 0.45–0.95), indicative of mesoporous structures formed by nanoparticle aggregation. The BET surface area (Table 2) increases dramatically from 24.55 m2/g (PGHAP) to 53.11 m2/g (5% La-PGHAP), while total pore volume rises from 0.1412 cm3/g to 0.2491 cm3/g, attributable to La3+-induced inhibition of HAP crystallite growth and formation of interparticle voids. Corresponding pore size distributions (Figure 3d) demonstrate a progressive shift from broad macroporous dominance (PGHAP: the average pore size of the mesopores is 36.59 nm) to narrower mesoporous regimes (5% La-PGHAP: the average pore size of the mesopores is 18.97 nm), corroborated by decreasing average mesopore size (22.99 → 18.76 nm) and micropore volume reduction (0.00562 → 0.00197 cm3/g). This textural evolution (enhanced mesoporosity coupled with reduced microporosity) optimizes mass transfer kinetics for adsorbate diffusion while maximizing accessible active sites.
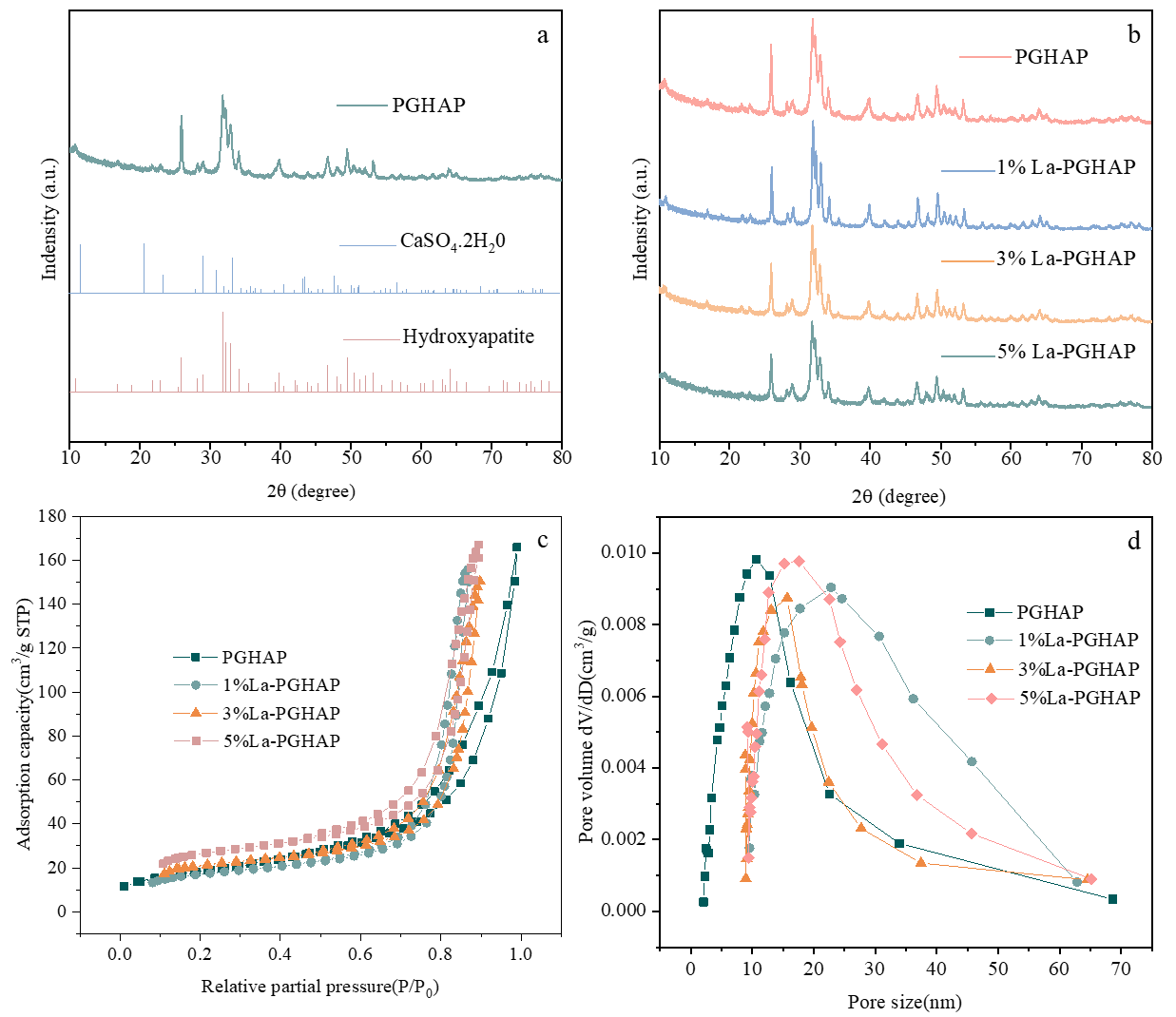
Figure 3. (a) XRD characterization of PGHAP, CaSO4·2H2O, hydroxyapatite; (b) XRD characterization of PGHAP and 1–5% La-PGHAP; (c) N2 adsorption-desorption diagram of PGHAP and 1–5% La-PGHAP; (d) pore size distribution of PGHAP and 1–5% La-PGHAP.
Table 2. The specific surface area and pore structure table before and after material synthesis.
|
Samples |
BET Specific Surface Area |
Specific Surface Area in Micropores |
Extraporeal Specific Surface Area |
Total Pore Volume |
Micropore Volume |
Average Pore Size |
Average Pore Size of Mesopores |
|---|---|---|---|---|---|---|---|
|
m2/g |
m2/g |
m2/g |
cm3/g |
cm3/g |
nm |
nm |
|
|
PGHAP |
24.5501 |
13.4632 |
11.0869 |
0.1412 |
0.00562 |
22.9979 |
36.5950 |
|
1% La-PGHAP |
38.6148 |
3.8452 |
34.7696 |
0.2412 |
0.00115 |
24.9903 |
26.4742 |
|
3% La-PGHAP |
46.8563 |
4.9055 |
41.9507 |
0.2549 |
0.00140 |
21.7560 |
23.4647 |
|
5% La-PGHAP |
53.1108 |
5.9231 |
47.1877 |
0.2491 |
0.00197 |
18.7590 |
18.9746 |
3.3. Multivariate Adsorption Performance of PGHAP and La-PGHAP
As evidenced by the adsorption results (Figure 4), La doping significantly enhances the fluoride (F−) uptake capacity of phosphogypsum-derived hydroxyapatite (PGHAP) in a dose-dependent but non-linear manner. At 0% La doping (pristine PGHAP), the adsorption capacity reaches 2.10 mg/g (under non optimal adsorption conditions), attributed to conventional mechanisms including ion exchange (OH− → F−) and electrostatic attraction at Ca2+ sites. Upon introducing 1% La, the capacity surges to 2.55 mg/g (under the same adsorption conditions), demonstrating that La3+ doping creates additional Lewis acid-base interaction sites—La3+ coordinates strongly with F− through inner-sphere complexation. This aligns with studies showing La–O–F bond formation enhances chemisorption efficiency. The incremental gain diminishes at 3% La doping (3.1 mg/g, +23.5% vs. 1% La-PGHAP), and the adsorption capacity for fluoride ions reaches its maximum. Notably, the plateau at 5% La (2.9 mg/g) indicates two competing effects: (1) Excessive La loading (>3%) may induce particle agglomeration, reducing effective surface area; (2) Formation of La(OH)3 surface precipitates at high doping levels could block mesopores, as evidenced by the unchanged capacity despite increased La content. The observed performance hierarchy (5% ≈ 3% > 1% > 0% La) under fixed experimental conditions (10 mg/L F−, 25 °C, 50 mg adsorbent) highlights optimal doping at 3% La. This balance maximizes La3+ active site density while maintaining structural integrity. The pH-dependent zeta potential would strengthen electrostatic F− attraction, though competitive anion effects (e.g., Cl−) warrant further investigation. Therefore, 3% lanthanum doping was set as the optimum condition (all subsequent references to La-PGHAP are considered to be 3% lanthanum doping).
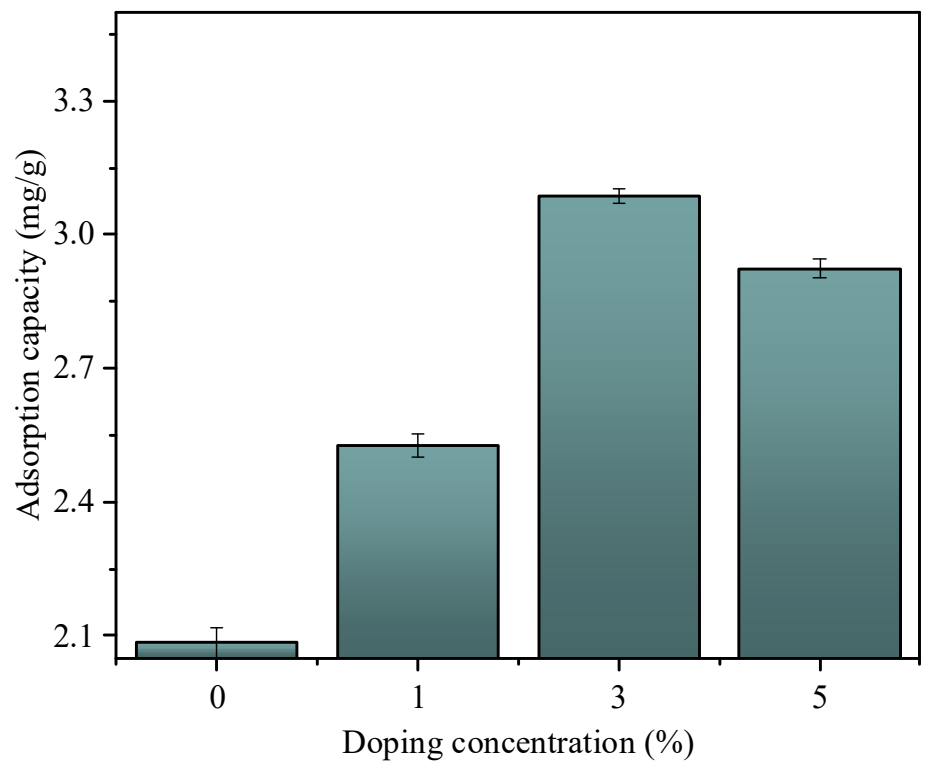
Figure 4. Adsorption capacity of adsorbent materials with different La doping levels for fluoride ions under the same conditions (pH 7.0, 50 mL F− solution at 10 mg/L, 50 mg adsorbent, 25 °C).
In order to investigate the influence of different factors on adsorption properties, the multivariate adsorption behavior of PGHAP and La-PGHAP toward fluoride ions (F−) was systematically investigated under six operational parameters (Figure 5), revealing fundamental divergences in adsorption mechanisms mediated by La3+ doping. La-PGHAP exhibits significantly enhanced fluoride adsorption performance compared to pristine PGHAP across all tested conditions, with mechanistic divergences rooted in La3+ doping effects. Adsorption kinetics (Figure 5a) show that both materials exhibit rapid initial adsorption (0–50 min), La-PGHAP achieves a higher saturation capacity, attributed to La3+-mediated chemisorption, reflecting optimized pore accessibility in La-doped mesostructures [21]. Adsorption capacity inversely correlates with dosage due to particle aggregation effects. Dosage dependence (Figure 5b) demonstrates La-PGHAP’s superior site accessibility, La-PGHAP achieves 8.10 mg/g, 3.3× higher than PGHAP’s 2.44 mg/g, demonstrating superior site availability, while its mesoporous structure mitigates aggregation-induced capacity loss slope.
pH sensitivity (Figure 5c) was studied in the range of 2–10, the adsorption of F− by PGHAP and La-PGHAP (collectively referred to here as HAP) reached a maximum value at pH 4. Thereafter, the adsorption of F− by HAP decreased as the pH continued to increase, which may be attributed to the fact that the hydroxyl groups on the HAP surface can release hydroxide ions in acidic solution, thereby making the surface positively charged. At this time, HAP and negatively charged F− ions were attracted to each other by electrostatic attraction, leading to an increase in adsorption in the solution. However, in neutral or alkaline environments, the charge on the surface of HAP may gradually become neutral or negative, thus reducing the adsorption of F− due to the presence of electrostatic repulsion. Meanwhile, at low pH, F− in solution existed mainly as free ions, which was due to the high concentration of H+ produced by the ionization of water under acidic conditions, making F− less likely to form complexes with other ions. At high pH, F− may form HF2− with OH− in the water, this may also contribute to the decrease in adsorption [22,23].
F− concentration response (Figure 5d) shows La-PGHAP’s linear capacity scaling to its maximum at 30 mg/L, outperforming PGHAP’s plateau at this point due to La3+’s multidentate binding capability [24]. Temperature activation (Figure 5e) confirms the endothermic nature of both materials. Adsorption increases endothermically for both materials. At 40 °C, La-PGHAP achieves 29.35 mg/g (3.7× PGHAP’s 7.90 mg/g), as thermal energy facilitates La–F inner-sphere complexation and pore expansion [25]. Ionic strength effects (Figure 5f) reveal La-PGHAP’s exceptional NaCl tolerance, leveraging charge screening and high-selectivity ligand exchange, whereas PGHAP plateaus at 0.15 mg/L NaCl [26]. These results collectively demonstrate that the optimum adsorption conditions of F− on La-PGHAP were 30 mg/L F−, 0.2 g/L HAP dosage, 0.2 mg/L NaCl, pH 4, 40 °C, 150 min, and the maximum adsorption capacity could reach up to 31.98 mg/g, which was better than the results in other reports [23,27]. La doping transforms PGHAP from a diffusion-limited, pH-sensitive adsorbent into a robust multifunctional material with rapid kinetics, high capacity, and exceptional environmental resilience, positioning it as a next-generation solution for industrial fluoride remediation.
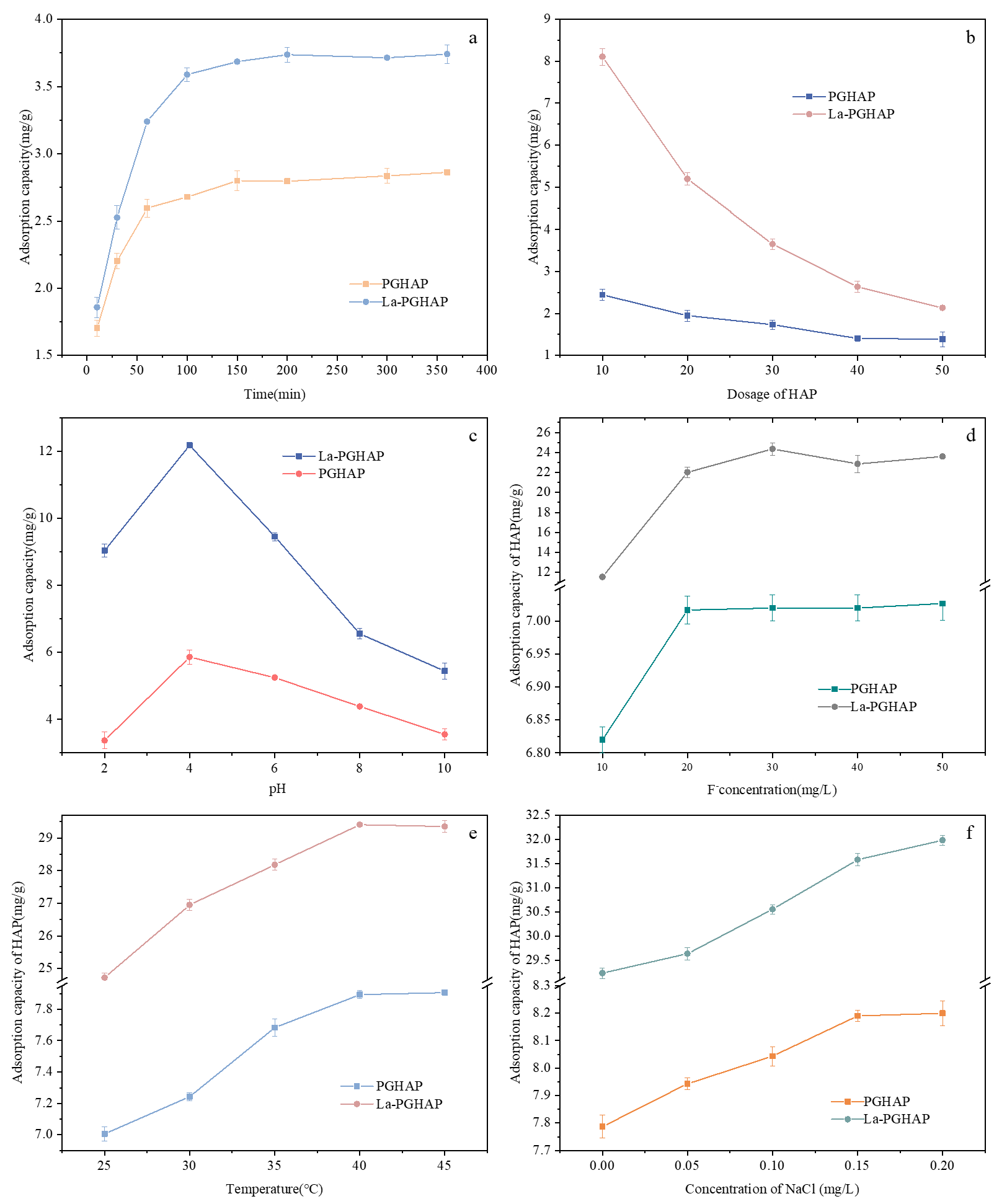
Figure 5. Effects of different factors on the F− adsorption: (a) adsorption time; (b) dosage of adsorbent; (c) pH; (d) concentration of F−; (e) temperature; (f) concentration of NaCl.
3.4. Adsorption Kinetics Analysis of PGHAP and La-PGHAP
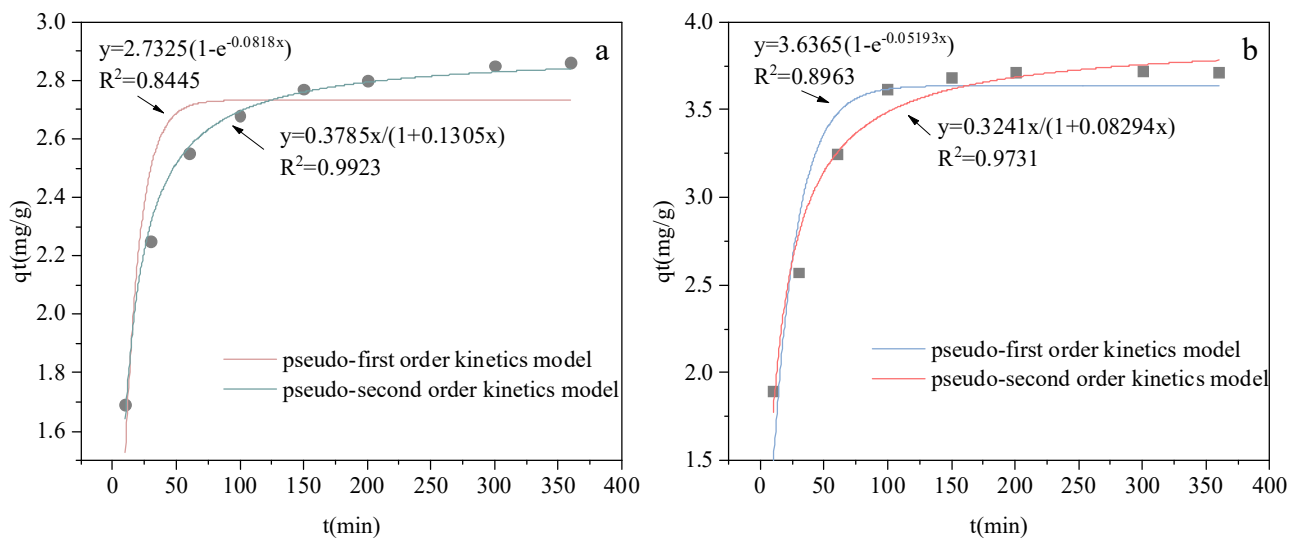
In order to explore the adsorption mechanism of F− on PGHAP and La-PGHAP, the adsorption kinetics of PGHAP and La-doped PGHAP were systematically investigated using pseudo-first-order (PFO) and pseudo-second-order (PSO) models (non-optimal conditions), as demonstrated in Figure 6 and Table 3. For pristine PGHAP, the experimental equilibrium adsorption capacity (qe,exp) reached 2.86 mg/g, while La-PGHAP exhibited significantly enhanced performance (qe,exp = 3.71 mg/g), confirming the critical role of La3+ doping in optimizing active sites for fluoride capture [23,28].
The PSO model demonstrated superior fitting for both adsorbents, as evidenced by higher determination coefficients (PGHAP: R2 = 0.9923 for PSO vs. R2 = 0.8445 for PFO; La-HAP: R2 = 0.9731 for PSO vs. R2 = 0.8963 for PFO). This dominance of PSO kinetics implies chemisorption-driven mechanisms, likely involving strong La3+-F− coordination bonds and ion exchange at Ca2+ vacancies generated during La3+ substitution. The PSO rate constant (K2) decreased from 0.04498 g/(mg·min) for PGHAP to 0.02123 g/(mg·min) for La-PGHAP, suggesting La doping slightly slowed the initial adsorption rate due to increased surface complexity and potential pore-blocking effects. However, the 30% enhancement in qe for La-PGHAP compensates for this kinetic trade-off, aligning with its higher density of high-affinity La–O–F binding sites [23,28,29].
Table 3. Adsorption kinetic parameters of PGHAP and La-PGHAP.
|
Model |
Actual Adsorption Capacity |
Pseudo-First-Order Kinetic |
Pseudo-Second Order Kinetic |
||||
|---|---|---|---|---|---|---|---|
|
Samples |
qe (mg/g) |
K1 |
R2 |
S |
K2 |
R2 |
S |
|
PGHAP |
2.86 |
0.0819 |
0.8445 |
0.1491 |
0.04498 |
0.9923 |
0.5249 |
|
La-PGHAP |
3.71 |
0.05193 |
0.8963 |
0.2001 |
0.02123 |
0.9731 |
0.5958 |
3.5. Adsorption Isotherm Analysis of PGHAP and La-PGHAP
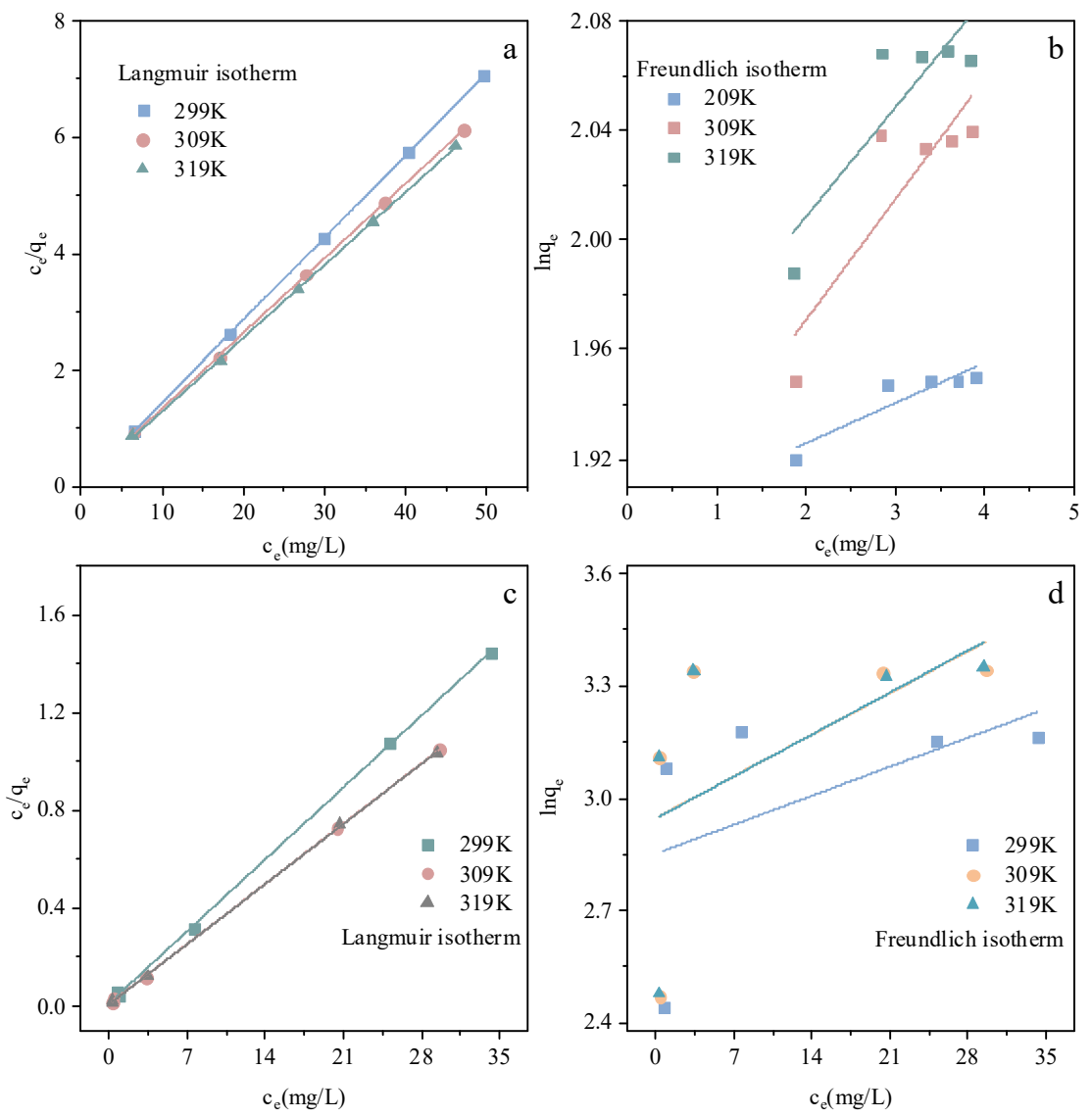
The adsorption equilibrium behavior of PGHAP and La-PGHAP was rigorously evaluated using Langmuir and the Freundlich isotherm models, as depicted in Figure 7 and Table 4. The Langmuir model exhibited exceptional fitting accuracy for both adsorbents across all temperatures (R2 > 0.9995), suggesting monolayer adsorption dominated by homogeneous binding sites. In contrast, the Freundlich model showed significantly lower correlation coefficients (R2 = 0.28–0.33 for La-PGHAP; R2 = 0.69–0.88 for PGHAP), indicating limited applicability for describing multilayer or heterogeneous adsorption processes. The F− on La-PGHAP adsorption process was more consistent with the assumption of monolayer adsorption. The Langmuir model assumed that the adsorbent surface was homogeneous, and that only one adsorbent molecule can be adsorbed at each adsorption site, and that cascading adsorption does not occur after saturation. The high degree of fit suggested that the adsorption sites on the adsorbent surface were independent and unresponsive to each other.
Remarkable qmax enhancement of La-PGHAP was observed (23.81–28.49 mg/g), with KL increasing from 3.36 (298 K) to 4.62 (318 K). The positive temperature dependence of KL aligns with chemisorption mechanisms, where La3+–F− coordination bonds strengthen through endothermic activation. In addition, it also implied that the adsorption energy was homogeneous rather than energetically diverse as described by the Freundlich model. This may be related to the crystal structure and surface properties of La-PGHAP, allowing F− to form a uniform and energetically homogeneous monolayer adsorption on the surface [30,31,32].
Table 4. Adsorption isotherm model parameters of PGHAP and La-PGHAP.
|
Sample |
Model |
Langmuir Isotherm Model |
Freundlich Isotherm Model |
||||
|---|---|---|---|---|---|---|---|
|
Temperature (K) |
qmax (mg/g) |
KL (L/mg) |
R2 |
KF (mg/g(L/mg)1/n) |
n |
R2 |
|
|
PGHAP |
298 |
19.22 |
5.56 |
0.9999 |
6.6619 |
67.80 |
0.8775 |
|
308 |
15.55 |
1.99 |
0.9998 |
6.5693 |
22.65 |
0.7699 |
|
|
318 |
21.53 |
2.69 |
0.9998 |
6.8806 |
25.04 |
0.6933 |
|
|
La-PGHAP |
298 |
23.8095 |
3.36 |
0.9995 |
16.5196 |
8.3612 |
0.2822 |
|
308 |
28.4900 |
4.2289 |
0.9997 |
19.7379 |
8.0386 |
0.3206 |
|
|
318 |
28.4900 |
4.6184 |
0.9996 |
19.8518 |
7.9936 |
0.3308 |
|
3.6. Adsorption Thermodynamics Analysis of PGHAP and La-PGHAP
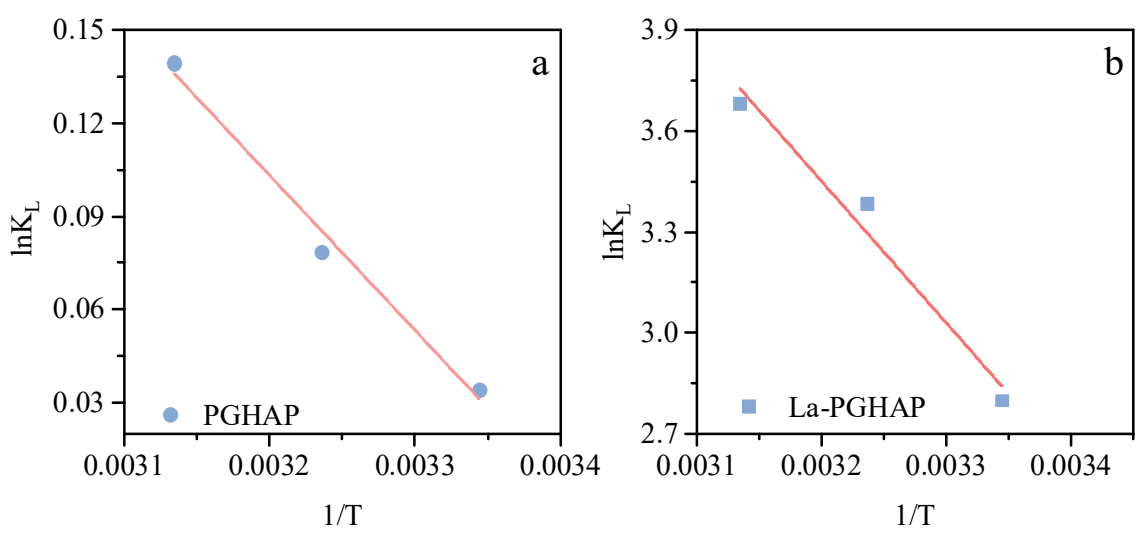
The thermodynamic parameters derived from Van’t Hoff plots (Figure 8a,b) and Table 5 reveal distinct adsorption mechanisms for PGHAP and La-PGHAP. For pristine PGHAP, the positive enthalpy change (ΔH = +4.16 kJ/mol) and modest entropy gain (ΔS = +14.17 J/(mol·K)) indicate a weakly endothermic process dominated by physical interactions (e.g., electrostatic attraction or ion exchange), where the slight spontaneity (ΔG = −0.085 to −0.370 kJ/mol) and low equilibrium constants (lnKL = 0.034–0.139) reflect limited F− binding affinity. In stark contrast, La-PGHAP exhibits dramatically enhanced thermodynamic driving forces, with ΔH surging to +35.06 kJ/mol and ΔS escalating to +140.89 J/(mol·K), signifying a chemisorption-dominated mechanism involving high-energy La3+–F− coordination bonds. The strongly negative ΔG values (−6.96 to −9.76 kJ/mol) and the exponential lnKL dependence on temperature (2.80–3.68) confirm that La doping transforms adsorption into a thermally activated process, in which elevated temperatures (299–319 K) facilitate La3+ site activation and F− desolvation. The 8.4-fold ΔH amplification and 9.9-fold ΔS enhancement upon La incorporation quantitatively validate synergistic effects of chemical bond formation (La–F) and interfacial restructuring, establishing La-PGHAP as a temperature-responsive adsorbent optimized for high-temperature fluoride remediation [33,34,35,36,37,38].
Table 5. Thermodynamic parameters of adsorption onto PGHAP and La-PGHAP at different temperatures.
|
Sample |
T/K |
lnKL |
R2 |
ΔG/kJ·mol−1 |
ΔS/J·(mol·K)−1 |
ΔH/kJ·mol−1 |
|---|---|---|---|---|---|---|
|
PGHAP |
299 |
0.0343 |
0.9879 |
−0.0852 |
14.1687 |
4.1592 |
|
309 |
0.0785 |
−0.2016 |
||||
|
319 |
0.1394 |
−0.3697 |
||||
|
La-PGHAP |
299 |
2.799891 |
0.9719 |
−6.9602 |
140.889 |
35.0610 |
|
309 |
3.385237 |
−8.6967 |
||||
|
319 |
3.681351 |
−9.7635 |
3.7. Reusability Analysis of PGHAP and La-PGHAP
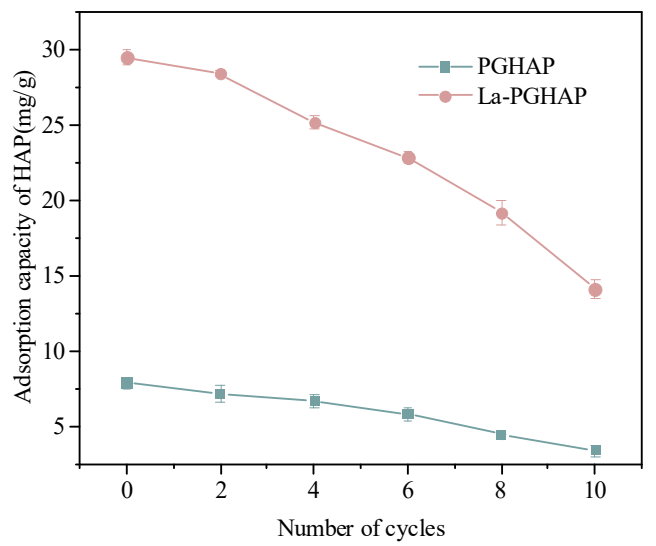
Reusability was an important performance parameter of adsorbents, and multiple reuse can reduce the cost of industrial applications [39]. The reusability evaluation over 10 adsorption-desorption cycles was shown in Figure 9. For pristine PGHAP, the adsorption capacity exhibited a continuous decline from 7.89 mg/g (Cycle 1) to 6.68 (Cycle 4) and 3.41 mg/g (Cycle 10), retaining 84.6% and 43.2% of its initial capacity. La-PGHAP demonstrated markedly enhanced cyclic stability, with capacity decreasing from 29.48 mg/g to 25.17 (Cycle 4) and 14.15 mg/g (Cycle 10), retaining 85.4% and 48.0% of its initial capacity, despite its higher initial adsorption performance [40,41]. The divergence in decay patterns (PGHAP’s rapid linear decline versus La-PGHAP’s biphasic decay) reveals La3+’s dual role in enhancing durability: (1) La–O bonds resist acid corrosion more effectively than Ca–O bonds, reducing active site loss; (2) La3+ doping introduces lattice distortions that inhibit hydroxyapatite recrystallization during regeneration, preserving mesoporous structures. While La-PGHAP’s absolute capacity loss exceeds PGHAP’s, its residual capacity remains higher than PGHAP’s, demonstrating the critical trade-off between initial performance and long-term usability [42,43].
4. Conclusions
This study successfully synthesized lanthanum-doped hydroxyapatite (La-PGHAP) from phosphogypsum (PG) waste, demonstrating its efficacy as a high-performance adsorbent for fluoride removal. Key findings include the acid precipitation-hydrothermal method enabled the conversion of PG into La-PGHAP with a well-defined spherical structure, high crystallinity, and a mesoporous architecture. La3+ doping enhanced surface reactivity and introduced additional adsorption sites via strong La3+–F− interactions. Maximum F− adsorption capacity reached 31.98 mg/g under optimal conditions (pH 4, 40 °C), outperforming conventional hydroxyapatite. Adsorption mechanisms were dominated by chemisorption, as evidenced by the fits to pseudo-second-order kinetics and the Langmuir isotherm model. In addition, La-PGHAP exhibited robust performance over a broad pH range (2–10) and maintained 85.4% of its adsorption capacity after 4 regeneration cycles, highlighting its stability and reusability. The “waste-to-waste” strategy not only addresses PG disposal challenges but also provides a cost-effective solution for remediation of fluoride-contaminated water. The integration of industrial waste recycling with advanced adsorbent design positions La-PGHAP as a promising candidate for large-scale environmental applications, aligning with the Sustainable Development Goals.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this manuscript, the author(s) used Deepseek in order to optimize the logical flow of the manuscript structure, refine English expression accuracy, and check for grammatical and typographical errors. After using these tools, the author(s) thoroughly reviewed and edited the content, verified all scientific data, experimental details, and conclusion validity, and take(s) full responsibility for the content of the published article.
Acknowledgements
The authors gratefully acknowledge financial support from the Natural Science Foundation of Hubei Province (Grant No. 2025AFD245), and the Natural Science Foundation of Yichang Municipality (Grant No. A24-3-043). The authors extend their gratitude to Scientific Compass (www.shiyanjia.com, accessed on 8 July 2025) for providing invaluable assistance with characterization analysis.
Author Contributions
Conceptualization, Z.W.; Methodology, S.J.; Investigation, S.J. and Y.Q.; Resources, Z.W., B.C. and R.C.; Data Curation, S.J. and Y.Q.; Writing—Original Draft Preparation, S.J.; Writing—Review & Editing, Z.W.; Visualization, B.C.; Supervision, Z.W. and R.C.; Project Administration, Z.W.; Funding Acquisition, Z.W.
Ethics Statement
“Not applicable” for studies not involving humans or animals.
Informed Consent Statement
“Not applicable” for studies not involving humans.
Data Availability Statement
The data used in this study are not publicly accessible and cannot be made available due to internal research policies.
Funding
This research was funded by the Natural Science Foundation of Hubei Province grant number 2025AFD245 and the Natural Science Foundation of Yichang Municipality grant number A24-3-043.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
-
Akfas F, Elghali A, Aboulaich A, Munoz M, Benzaazoua M, Bodinier J-L. Exploring the potential reuse of phosphogypsum: A waste or a resource? Sci. Total Environ. 2024, 908, 168196. DOI:10.1016/j.scitotenv.2023.168196 [Google Scholar]
-
Fang K, Xu L, Yang M, Chen Q. One-step wet-process phosphoric acid by-product CaSO4 and its purification. Sep. Purif. Technol. 2023, 309, 123048. DOI:10.1016/j.seppur.2022.123048 [Google Scholar]
-
Wang M, Guo Z, Du J, Lu H, Liu L, Wang T, et al. Assessing the hepatotoxicity of phosphogypsum leachate in zebrafish (Danio rerio). Sci. Total Environ. 2024, 926, 172018. DOI:10.1016/j.scitotenv.2024.172018 [Google Scholar]
-
Li B, Zhang L, Cheng M, Chen L, Fang W, Liu S, et al. Evaluation of fluoride emissions and pollution from an electrolytic aluminum plant located in Yunnan province. J. Hazard. Mater. 2024, 478, 135500. DOI:10.1016/j.jhazmat.2024.135500 [Google Scholar]
-
Johnston NR, Strobel SA. Principles of fluoride toxicity and the cellular response: A review. Arch. Toxicol. 2020, 94, 1051–1069. DOI:10.1007/s00204-020-02687-5 [Google Scholar]
-
Zeng L, Bian X, Weng J, Zhang T. Wetting-drying effect on the strength and microstructure of cement-phosphogypsum stabilized soils. J. Rock Mech. Geotech. Eng. 2024, 16, 1049–1058. DOI:10.1016/j.jrmge.2023.06.022 [Google Scholar]
-
Ou L, Li R, Zhu H, Zhao H, Chen R. Upcycling waste phosphogypsum as an alternative filler for asphalt pavement. J. Clean. Prod. 2023, 420, 138332. DOI:10.1016/j.jclepro.2023.138332 [Google Scholar]
-
Zhang J, Yu K, Yu M, Dong X, Tariq Sarwar M, Yang H. Facet-engineering strategy of phosphogypsum for production of mineral slow-release fertilizers with efficient nutrient fixation and delivery. Waste Manag. 2024, 182, 259–270. DOI:10.1016/j.wasman.2024.04.041 [Google Scholar]
-
Liu X, Wu C, Lv W, Dong E, Fu S, Zhang L, et al. Regulating sulfur migration and transformation in low water-binder ratio cementitious system incorporating phosphogypsum aggregate: Environmentally friendly clean materials. J. Build. Eng. 2024, 91, 109586. DOI:10.1016/j.jobe.2024.109586 [Google Scholar]
-
Sazali NN, Mohamed MA, Mohd Yusoff SF, Hasnan NSN, Nordin NA, Anuar NA, et al. Conversion of waste phosphogypsum into value-added 1D/2D homojunction hydroxyapatite with enhanced structural, morphology, and photoelectrochemical performance. J. Environ. Chem. Eng. 2024, 12, 112784. DOI:10.1016/j.jece.2024.112784 [Google Scholar]
-
Adhikara AG, Maharani AP, Puspitasari A, Nuswantoro NF, Juliadmi D, Maras MAJ, et al. Bovine hydroxyapatite for bone tissue engineering: Preparation, characterization, challenges, and future perspectives. Eur. Polym. J. 2024, 214, 113171. DOI:10.1016/j.eurpolymj.2024.113171 [Google Scholar]
-
Balasooriya IL, Chen J, Korale Gedara SM, Han Y, Wickramaratne MN. Applications of Nano Hydroxyapatite as Adsorbents: A Review. Nanomaterials 2022, 12, 2324. DOI:10.3390/nano12142324 [Google Scholar]
-
Rathnayake A, Hettithanthri O, Sandanayake S, Mahatantila K, Rajapaksha AU, Vithanage M. Essence of hydroxyapatite in defluoridation of drinking water: A review. Environ. Pollut. 2022, 311, 119882. DOI:10.1016/j.envpol.2022.119882 [Google Scholar]
-
Russo V, D’Angelo A, Salvi C, Paparo R, Fortunato ME, Cepollaro EM, et al. Fluoride adsorption on hydroxyapatite: From batch to continuous operation. J. Environ. Chem. Eng. 2024, 12, 111973. DOI:10.1016/j.jece.2024.111973 [Google Scholar]
-
Qin Y, Wang Z, Jiang S, Chi R, Huang S, Ma H, et al. Synthesis of solid micro-spherical hydroxyapatite by hard-template method: Optimization and characterization. J. Porous Mater. 2025, 32, 741–751. DOI:10.1007/s10934-024-01731-4 [Google Scholar]
-
Wimalasiri AKDVK, Fernando MS, Dziemidowicz K, Williams GR, Koswattage KR, Dissanayake DP, et al. Structure–Activity Relationship of Lanthanide-Incorporated Nano-Hydroxyapatite for the Adsorption of Fluoride and Lead. ACS Omega 2021, 6, 13527–13543. DOI:10.1021/acsomega.0c05935 [Google Scholar]
-
Jiang S, Wang Z, Qin Y, Chi R, Huang S, Ma H, et al. Acid precipitation-hydrothermal synthesis of needle-like hydroxyapatite for protein adsorption from waste phosphogypsum. Environ. Technol. 2025, 46, 1718–1729. DOI:10.1080/09593330.2024.2402099 [Google Scholar]
-
Wang C, Yuan X, Kang H, Yin R, Cai A, Yue Z, et al. Structural characterization, photothermal effect and biological properties of La/Ga ion co-doped hydroxyapatite coated with polydopamine. J. Ind. Eng. Chem. 2024, 139, 573–586. DOI:10.1016/j.jiec.2024.05.033 [Google Scholar]
-
Gao L, Li Y, Li G, Huo J, Jia L. Interface charge induced self-assembled (Co(OH)2)4@La(OH)3 heterojunction derived from Co4-MOF@La(HCO2)3 to boost oxygen evolution reaction. Chem. Eng. J. 2023, 451, 138743. DOI:10.1016/j.cej.2022.138743 [Google Scholar]
-
Zhao G, Li M, Li H, Ping Z, Wang P, Wu Y, et al. La-doped micro-angular cube ZnSnO3 with nano-La2O3 decoration for enhanced ethylene glycol sensing performance at low temperature. Sens. Actuators A Phys. 2023, 362, 114649. DOI:10.1016/j.sna.2023.114649 [Google Scholar]
-
Ganta DD, Hirpaye BY, Raghavanpillai SK, Menber SY, Hahn JR. Green Synthesis of Hydroxyapatite Nanoparticles Using Monoon longifolium Leaf Extract for Removal of Fluoride from Aqueous Solution. J. Chem. 2022, 2022, 4917604. DOI:10.1155/2022/4917604 [Google Scholar]
-
Scheverin VN, Horst MF, Lassalle VL. Novel hydroxyapatite-biomass nanocomposites for fluoride adsorption. Results Eng. 2022, 16, 100648. DOI:10.1016/j.rineng.2022.100648 [Google Scholar]
-
Sekar S, Panchu SE, Kolanthai E, Rajaram V, Subbaraya NK. Enhanced stability of hydroxyapatite/sodium alginate nanocomposite for effective fluoride adsorption. Mater. Today Proc. 2022, 58, 909–917. DOI:10.1016/j.matpr.2021.12.064 [Google Scholar]
-
Jeyaseelan A, Viswanathan NJI, Research EC. Investigation of Hydroxyapatite-Entrenched Cerium Organic Frameworks Incorporating Biopolymeric Beads for Efficient Fluoride Removal. Ind. Eng. Chem. Res. 2022, 61, 7911–7925. DOI:10.1021/acs.iecr.2c00487 [Google Scholar]
-
Huang S, Hu M, Li D, Wang L, Zhang C, Li K, et al. Fluoride sorption from aqueous solution using Al(OH)3-modified hydroxyapatite nanosheet. Fuel 2020, 279, 118486. DOI:10.1016/j.fuel.2020.118486 [Google Scholar]
-
Laonapakul T, Suthi T, Otsuka Y, Mutoh Y, Chaikool P, Chindaprasirt P. Fluoride Adsorption Enhancement of Calcined-Kaolin/Hydroxyapatite composite. Arab. J. Chem. 2022, 15, 104220. DOI:10.1016/j.arabjc.2022.104220 [Google Scholar]
-
Yapo NZS, Aw S, Briton BGH, Drogui P, Yao KB, Adouby K. Removal of fluoride in groundwater by adsorption using hydroxyapatite modified Corbula trigona shell powder. Chem. Eng. J. Adv. 2022, 12, 100386. DOI:10.1016/j.ceja.2022.100386 [Google Scholar]
-
Huang J, Liu T, Zhang Y, Hu P. Reinforced adsorption mechanism of fluorine ions by calcium-depleted hydroxyapatite and application in the raffinate from the vanadium industry. Chem. Eng. J. 2023, 452, 139379. DOI:10.1016/j.cej.2022.139379 [Google Scholar]
-
Nijhawan A, Butler EC, Sabatini DA. Fluoride Adsorption on Porous Hydroxyapatite Ceramic Filters: A Study of Kinetics. Environ. Eng. Sci. 2020, 37, 409–416. DOI:10.1089/ees.2019.0392 [Google Scholar]
-
Mchich Z, Aziz K, Kjidaa B, Saffaj N, Saffaj T, Mamouni R. Eco-friendly engineering of micro composite-based hydroxyapatite bio crystal and polyaniline for high removal of OG dye from wastewater: Adsorption mechanism and RSM@BBD optimization. Environ. Res. 2024, 257, 119289. DOI:10.1016/j.envres.2024.119289 [Google Scholar]
-
Cheikh S, Imessaoudene A, Bollinger JC, Manseri A, Bouzaza A, Hadadi A, et al. Adsorption behavior and mechanisms of the emerging antibiotic pollutant norfloxacin on eco-friendly and low-cost hydroxyapatite: Integrated experimental and response surface methodology optimized adsorption process. J. Mol. Liq. 2023, 392, 123424. DOI:10.1016/j.molliq.2023.123424 [Google Scholar]
-
Li S, Du K. Self-sacrificing template synthesis of ultralight porous hydroxyapatite microtube-based ceramics for hemoglobin adsorption. Chem. Eng. J. 2024, 492, 152404. DOI:10.1016/j.cej.2024.152404 [Google Scholar]
-
Wang Y, Wang C, Huang X, Zhang Q, Wang T, Guo X. Guideline for modeling solid-liquid adsorption: Kinetics, isotherm, fixed bed, and thermodynamics. Chemosphere 2024, 349, 140736. DOI:10.1016/j.chemosphere.2023.140736 [Google Scholar]
-
Mokhati A, Kecira Z, Benturki O, Bernardo M, Sellaoui L, Mechi N, et al. Insights into nitrobenzene adsorption mechanism on apricot stone activated carbon: A study via statistical physics models and thermodynamic analysis. Colloids Surf. A Physicochem. Eng. Asp. 2024, 691, 133864. DOI:10.1016/j.colsurfa.2024.133864 [Google Scholar]
-
Wu G, Zhao R, Li X. Response to “Comment on Magnetic tubular nickel@silica-graphene nanocomposites with high preconcentration capacity for organothiophosphate pesticide removal in environmental water: Fabrication, magnetic solid-phase extraction, and trace detection” [J. Hazard. Mater. 457 (2023) 131788]. J. Hazard. Mater. 2024, 464, 132926. DOI:10.1016/j.jhazmat.2023.132926 [Google Scholar]
-
Tran HN, Lima EC, Juang R-S, Bollinger J-C, Chao H-P. Thermodynamic parameters of liquid–phase adsorption process calculated from different equilibrium constants related to adsorption isotherms: A comparison study. J. Environ. Chem. Eng. 2021, 9, 106674. DOI:10.1016/j.jece.2021.106674 [Google Scholar]
-
Hamri N, Imessaoudene A, Bouchelkia N, Boudraa R, Cheikh S, Bollinger JC, et al. Innovative surface modification of Kaolinite by direct grafting of an anionic surfactant to improve pollutant adsorption and effluent decontamination. J. Dispers. Sci. Technol. 2024, 1–20. DOI:10.1080/01932691.2024.2448761 [Google Scholar]
-
Imessaoudene A, Cheikh S, Bollinger JC, Belkhiri L, Tiri A, Bouzaza A, et al. Zeolite Waste Characterization and Use as Low-Cost, Ecofriendly, and Sustainable Material for Malachite Green and Methylene Blue Dyes Removal: Box–Behnken Design, Kinetics, and Thermodynamics. Appl. Sci. 2022, 12, 7587. DOI:10.3390/app12157587 [Google Scholar]
-
Adamu DB, Tufa LT, Lee J, Zereffa E, Segne TA, Razali MH. Facile synthesis of bismuth and iron co-doped hydroxyapatite nanomaterials for high-performance fluoride ions adsorption. J. Environ. Chem. Eng. 2023, 11, 111196. DOI:10.1016/j.jece.2023.111196 [Google Scholar]
-
Mahmood BK, Kareem RO, Bulut N, Ates T, Keser S, Kaygili O, et al. Structural, magnetic, thermal, biocompatibility, and electronic properties of lanthanum doped-magnesium hydroxyapatite. Ceram. Int. 2025, 51, 18607–18616. DOI:10.1016/j.ceramint.2025.02.041 [Google Scholar]
-
Li B, Chen H, Yuan X. Influence of different La2O3 loading on hydroxyapatite supported nickel catalysts in the dry reforming of methane. Fuel 2024, 369, 131687. DOI:10.1016/j.fuel.2024.131687 [Google Scholar]
-
Sun X, Ke C, Lu Q, Wang Q, Zhou S, Liang L, et al. Recent Advances in Adsorption Techniques for the Removal of Fluoride Ions from Wastewater: A Review. Ind. Eng. Chem. Res. 2024, 63, 19936–19954. DOI:10.1021/acs.iecr.4c02734 [Google Scholar]
-
Sinharoy A, Chung CM. Fluoride removal from wastewater and potential for resource recovery: Comparative studies between different treatment technologies. Environ. Eng. Res. 2024, 29, 240179-0. DOI:10.4491/eer.2024.179 [Google Scholar]