Sustainable Additive Manufacturing of Polymer Composites for Advanced Water Treatment: A Review
Received: 08 January 2026 Revised: 05 February 2026 Accepted: 17 March 2026 Published: 26 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
The global water crisis is intensifying, driven by pervasive pollution from multiple sectors. The World Health Organization stated that approximately 2 billion people still lack access to safe drinking water, which is a dire situation fuelled by widespread contamination. Industrial and municipal discharges are primary contributors; for instance, China’s Henan Province alone releases an estimated 1,445,700 tons of chemical oxygen demand and 46,300 tons of ammonia nitrogen annually [1]. Similar challenges persist in developed nations, including the United States, where up to 28% of the population relies on drinking water systems that violate health standards [2]. The crisis extends beyond freshwater systems. Annually, an estimated 0.5–11 million tons of petroleum enter the oceans, while agricultural runoff threatens nearly 75% of global farmland with pesticide pollution, degrading both surface and groundwater [3,4]. River pollution is equally severe, exemplified by Malaysia, where over 30% of rivers were reported as heavily polluted in 2017, requiring extensive treatment [5]. These threats highlight the urgent need for innovative, efficient, and sustainable water treatment technologies to safeguard human health and restore ecosystems.
Over recent decades, a wide range of water treatment technologies have been developed. Physical methods, such as activated carbon adsorption, are widely used to remove heavy metals, pesticides, volatile organic compounds, and other organic contaminants [6,7]. However, their sustainability is hampered by the energy intensity of regeneration, the risks of handling hazardous spent waste, and competitive interference in complex water matrices [8]. Chemical methods, particularly Advanced Oxidation Processes (AOPs) like ozonation, Fenton oxidation, and photocatalysis, offer a different approach by generating highly reactive species to degrade recalcitrant pollutants. While effective and often synergistic with adsorption, AOPs face practical limitations, including stringent pH requirements, difficulties in catalyst recovery, sludge formation, and high energy consumption [9]. It is precisely these limitations of conventional physical and chemical technologies that have driven intense research toward advanced, customizable materials capable of overcoming such shortcomings.
Among the most prominent emerging water treatment technologies is membrane filtration, particularly reverse osmosis (RO). This pressure-driven process uses a semi-permeable membrane to remove dissolved constituents from feedwater [10]. Since the 1980s, the market has been dominated by polymeric thin-film composites, primarily made from polyamides or cellulose acetate, valued for their high permeability, selectivity, and strong desalination performance [11]. A typical composite structure features a polyester web (120–150 µm) for support, a microporous interlayer (~40 µm), and an ultra-thin polyamide barrier layer (~0.2 µm) with sub-nanometer pores (<0.6 nm), enabling salt rejection rates over 99% [12]. To further enhance these membranes, nanomaterials like graphene oxide, carbon nanotubes, and carbon nanofibers are incorporated into polymer matrices, attributed to their high surface area and mechanical strength [13,14]. Concurrently, natural clays have gained interest as low-cost, effective fillers due to their layered structures and tunable surfaces [15]. For example, a palygorskite/chitosan composite has demonstrated a high Pb(II) ions adsorption capacity of 201.5 mg/g, highlighting the promise of polymer/clay hybrids [16].
The pursuit of sustainable, high-performance materials for water treatment has accelerated the adoption of additive manufacturing (AM), or 3D printing [17]. This technology provides exceptional design freedom, rapid prototyping, and reduced production costs and development time compared to conventional methods such as injection molding [18,19]. Specifically for water treatment, AM enables the precise fabrication of components across several key areas, including polymer membranes, structured catalysts for advanced oxidation processes, custom water treatment modules, biofilm carriers and microfluidic devices. Figure 1 shows the feasible applications of 3D-printed polymers in water treatment.
 |
 |
| (a) | (b) |
Figure 1. Applications of 3D-printed polymers in water treatment. The figure illustrates diverse components fabricated via additive manufacturing: (a) 3D printed polymer membranes for filtration, (b) Assorted 3D printed water treatment components.
Aligning material’s innovation with the 2030 sustainable development goals (SGD) is an urgent priority, AM emerges as a key enabler for Responsible Consumption and Production (SDG 12), primarily through on-demand fabrication and a significant reduction in supply-chain waste [20,21]. The field is actively advancing its own sustainability through both optimization and material innovation. Practical engineering modifications, such as insulating hot ends and heated build chambers, can lower energy use by over 30%, while sealed printer enclosures reduce total energy consumption by approximately 18%. Concurrently, design strategies like lightning infill patterns and the development of advanced foaming polymers can decrease material consumption by up to 51% [22,23,24].
A compelling demonstration of AM-driven innovation in water treatment is the fabrication of functional biofilms via Fused Filament Fabrication (FFF). These structures utilize a sustainable polylactic acid (PLA) matrix, which is surface coated with nanosilver particles (AgNPs). The 3D-printed PLA scaffold provides customizable, biodegradable architecture, while the AgNPs impart potent antimicrobial properties and enhance the degradation of organic dyes like Neutral Red Chloride and Alizarin Yellow R [25,26,27,28,29,30]. This approach exemplifies the powerful synergy achievable by integrating the design freedom of additive manufacturing with advanced multifunctional nanocoatings.
Ultimately, the sustainable development of high-performance polymer composites for water treatment hinges on a fundamental and interdependent relationship: structure–properties–processing–application. This means that achieving environmental sustainability is intrinsically linked to material choices (polymers and fillers), manufacturing methods (e.g., additive manufacturing), and the resulting functional performance. Therefore, this review focuses on the critical role of sustainable polymer and filler selection as the cornerstone of this relationship for advanced water treatment. By focusing the discussion on this materials-centric paradigm, the review provides a targeted roadmap for designing water treatment solutions that are both effective and inherently aligned with the principles of green chemistry and responsible production.
2. Additive Manufacturing for Polymer Composites
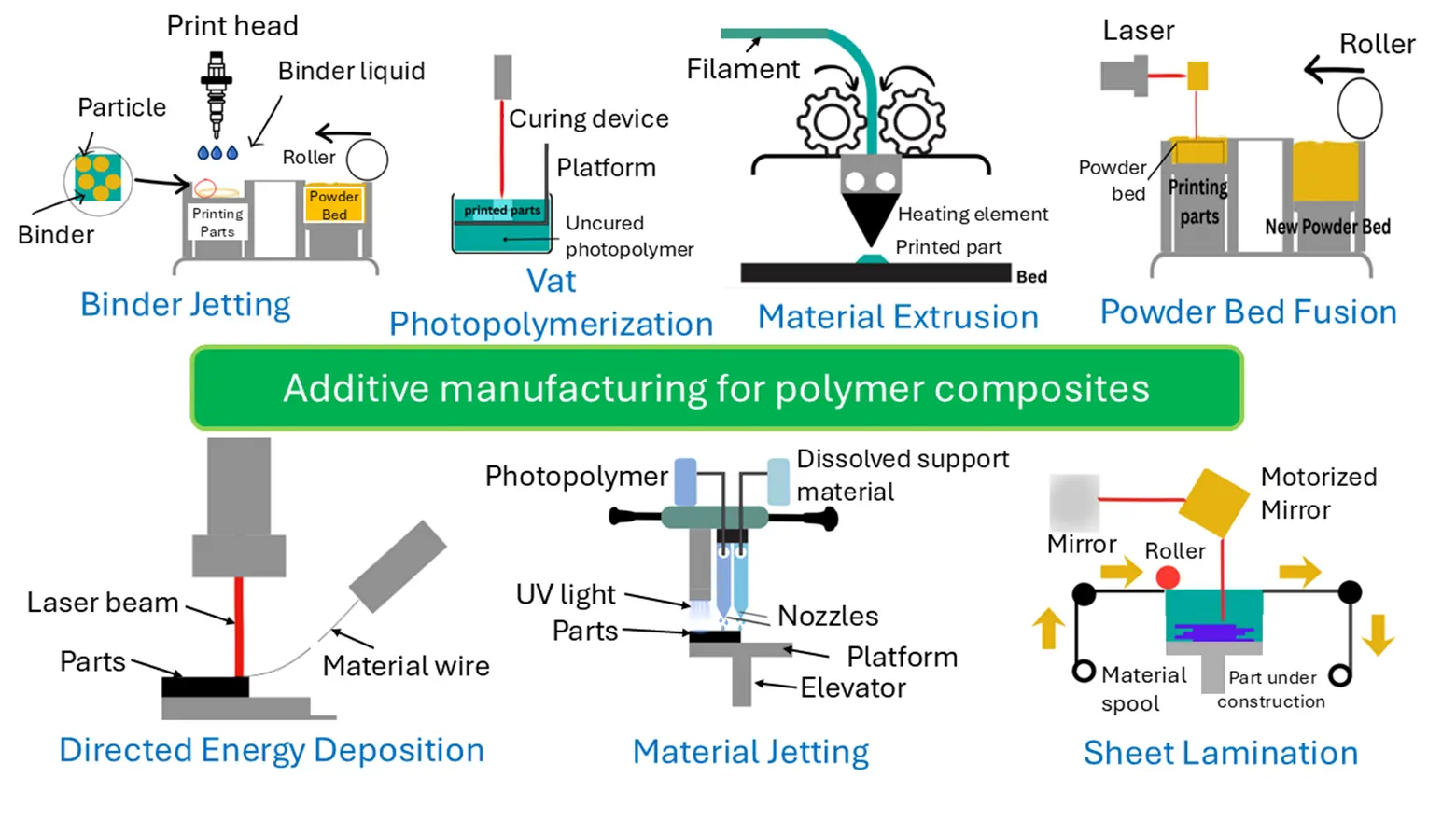
Additive manufacturing (AM) is a process of joining materials to make parts from 3D model data, usually layer upon layer, as opposed to subtractive manufacturing and formative manufacturing methodologies [31]. Compared to conventional manufacturing methods, such as injection molding, additive manufacturing (AM) can offer a cost advantage due to minimal tooling requirements and the elimination of manufacturability constraints imposed by traditional molds or tool paths. This “complexity-for-free” approach enables the integration of topology-optimized features and part consolidation, thereby reducing downstream assembly labor and potentially lowering lifecycle costs through improved use-phase performance [32]. Figure 2 shows several categories of AM, including binder jetting, directed energy deposition, vat photopolymerization, material extrusion, material jetting, powder-bed fusion and sheet lamination.
The selection of an optimal printing technique is fundamentally controlled by the target application and the required performance characteristics of the final part [33]. For water treatment applications, this section critically assesses the feasibility of these AM processes for fabricating advanced polymer composites. The ability to precisely control porosity, including pore size, geometry, and interconnectivity, is a significant advantage of additive manufacturing (AM) in water treatment applications. These structural parameters significantly influence mass transfer behavior, active surface area accessibility, hydraulic performance, and overall treatment efficiency across different mechanisms, including adsorption, filtration, and catalytic degradation. Table 1 shows some studies on AM-fabricated porous materials and the influence of porosity on water treatment. It also demonstrates some correlation between the structural characteristics of AM-fabricated materials (e.g., porosity and pore architecture) and their corresponding treatment performance in selected water remediation applications.
This review covers AM technologies, including binder jetting, material extrusion, vat photopolymerization, etc. While each technique possesses distinct mechanical and material processing principles, the discussion is specifically oriented toward their potential and utility in fabricating advanced polymer composites. The core emphasis is not merely on printing polymers, but on evaluating how these processes enable the integration of functional fillers (e.g., nanosilver, graphene oxide, natural clays) and the creation of complex, tailored architectures that can be useful for water treatment.
Table 1. Some studies on AM-fabricated porous materials and the influence of porosity on water treatment.
|
Materials |
AM Technique |
Porosity (%) |
Pore Size (µm) |
Surface-Area Density |
Adsorption Capacity |
Significant Findings |
Reference |
|---|---|---|---|---|---|---|---|
|
Alginate and cellulose nanocrystal (CNC) |
Micro-extrusion-based 3D coaxial printing |
N.A. |
nm to µm scale (disordered meso- and macropores) |
28.81 m2/g |
~68 mg/g (equilibrium); 97.22 mg/g (maximum at 25 °C) |
The hierarchical grid-like architecture exhibited significantly higher Cu(II) adsorption rate and capacity compared to bulk (solid) hydrogel counterparts. The enhanced performance was primarily attributed to ion exchange, chelation, and electrostatic interactions. |
[34] |
|
Polylactic acid (PLA) filter coated with iron (III) oxide |
Fused Deposition Modeling (FDM) |
N.A. |
800 to 4000 µm (0.8 to 4.0 mm square channels) |
Not explicitly stated (dependent on CAD channel width) |
129.87 mg/g for As (III) (using 0.8 mm channels) |
Narrower channels (0.8 mm) significantly enhanced iron oxide deposition and improved arsenic removal kinetics. The system operates under hydrostatic pressure without the need for electricity, and the filters can be regenerated through acid washing. |
[35] |
|
Acrylate-based UV-curable resin, chitosan |
Stereolithography (SLA) |
Highly porous |
300 µm |
Ranged from 0.183 m2/g (round holes) up to 0.254 m2/g (skewed hexagonal holes) |
3.7 mg/g for Cu(II) |
The material exhibited a rapid initial adsorption rate of 2.2 mg·g−1·min−1. Approximately 92% of the adsorbed metal ions could be desorbed using EDTA, indicating excellent reusability. The optimal operating conditions were 25 °C and pH 5.5. |
[36] |
|
PES/PVP/NMP + PLA spacer |
FDM for molds, followed by NIPS casting |
Flat: 38.4%; D-Patterned: 46.1%; HC-Patterned: 49.4% |
Flat: ~0.015 µm; D-Patterned: ~0.024 µm; HC-Patterned: ~0.023 µm |
+32% (D-Pattern) and +29% (HC-Pattern) larger area vs. flat |
N.A. |
Patterned structures exhibited higher pure water flux, reduced fouling propensity, improved flux recovery, and lower pressure drops compared to flat counterparts incorporating physical spacers. |
[37] |
Remarks: N.A. = Data not available.
2.1. Binder Jetting
Binder Jetting (BJ) is an energy-efficient and versatile additive manufacturing technique, distinguished by its ability to fabricate parts from a wide range of powder materials, including the option to combine multiple powders in a single build [38]. It holds significant promise for the cost-effective production of complex components, largely because it avoids the energy-intensive melting of raw materials [39]. Commercial systems from manufacturers such as ExOne, HP, and Voxeljet support the binder jetting of ceramics, metals, polymers, sand, and biomaterials [40].
Binder jetting offers a unique pathway for creating functional porous structures. For instance, Shen et al. [41] developed a conductive composite by printing graphene ink onto a polyvinyl alcohol (PVOH) powder bed, forming an interconnected porous framework with a density of 0.62 g/cm3. The printed structure exhibited a tensile modulus of 3.59 MPa, demonstrating mechanical properties comparable to silicone rubbers. More directly relevant to filtration, Meenashisundaram et al. [42] produced highly porous (40–60%) 316L stainless steel scaffolds with an interconnected open-cell structure, demonstrating the feasibility of manufacturing binder-jetted porous filters using a polymeric space holder (polymethyl methacrylate, PMMA). The technique’s compatibility with biopolymers like polylactic acid (PLA) and polycaprolactone (PCL), achieved through solvent-based binding, further suggests its potential for fabricating sustainable, custom-shaped biofilm carriers or adsorbent scaffolds [43].
Binder Jetting holds significant potential for fabricating water treatment materials by enabling the creation of complex, highly porous, and multifunctional polymer composites. Its primary advantage is the energy-efficient, multi-material fabrication of intricate monolithic structures, such as customized filter bodies and adsorbent scaffolds, without thermally degrading sensitive functional fillers, such as bio-adsorbents or metal-organic frameworks (MOFs). The technique is uniquely capable of producing parts with designed, interconnected porosity essential for high surface area and flow.
2.2. Directed Energy Deposition
Directed Energy Deposition (DED) is an additive manufacturing process defined by the use of focused thermal energy to fuse materials by melting as they are being deposited [44]. Renowned for its high deposition rates and ability to fabricate large-scale parts (up to ~1 m3), DED is predominantly applied in metalworking for repairing high-value components, constructing large structures (compare with powder bed fusion 3D printing), and depositing specialized coatings [45,46,47].
While DED has been traditionally associated with metals, technological advancements have successfully extended its capabilities to polymer composites [48,49]. Notable demonstrations include the fabrication of polyamide 12 (PA12)/carbon nanotube composites [50], the deposition of polyetheretherketone (PEEK) coatings on metal substrates [51], and the integration of metal–organic frameworks (MOFs) within acrylonitrile-butadiene-styrene (ABS) matrices [52]. These examples highlight DED’s potential for creating high-performance, multifunctional composite structures.
However, the direct application of DED for fabricating polymer composites specifically engineered for water treatment remains a significant and largely unexplored frontier. This gap presents a compelling opportunity to harness DED’s distinctive advantages. The process’s high deposition rates and scalability make it ideal for manufacturing large, durable structural components, such as custom reactor housings. More innovatively, DED’s precision melt-deposition capability enables the single-step fabrication or cladding of surfaces with functional polymer composites. For example, a corrosion-resistant structural substrate could be directly coated with a composite layer embedding photocatalytic nanoparticles (e.g., titanium dioxide, TiO2) for in-situ pollutant degradation, or with a porous polymer matrix containing selective adsorbents, such as metal-organic frameworks (MOFs). This ability to engineer multifunctional, graded structures, where composition, porosity, and functionality are spatially tailored, could pioneer a new class of integrated filtration media that synergistically combine structural integrity, high surface area, and targeted reactivity, thereby overcoming the inherent limitations of conventional homogeneous materials.
2.3. Vat Photopolymerization
Vat photopolymerization (VP) is one of the earliest and most extensively used additive manufacturing technologies, with broad adoption across sectors including electronics, aerospace, automotive, and biomedical applications like tissue engineering and drug delivery [53,54,55]. The process operates by using a light source to selectively cure and solidify photosensitive liquid resins layer by layer. The technology encompasses several categories distinguished by their curing mechanisms and light sources. The primary variants include stereolithography, digital light processing, two-photon polymerization, and volumetric 3D printing [56]. These techniques are particularly well-suited for fabricating polymer composites, typically by incorporating functional nanomaterials, such as nano-metallic particles, into the photocurable resin matrix.
It is worth noting that VP offers significant sustainability advantages in terms of energy efficiency. Among its variants, stereolithography is exceptionally energy-efficient, with a specific energy consumption of approximately 75–148 MJ/kg. This is considerably lower than many other AM processes, including fused deposition modeling (up to 1246 MJ/kg), selective laser melting (83 to 588 MJ/kg), and direct metal laser sintering (241 to 339 MJ/kg) [57]. Despite that, VP also enables precise porosity control in membranes. Hoskins and Zou [58] achieved pores ranging from 0.57–3.73 µm and porosities of 4.5–60% via two-photon polymerization, demonstrating independent tuning of pore size and porosity (e.g., 1.9 µm pores at both 38% and 22% porosity). This capability is essential for optimizing permeability and selectivity independently in water treatment membranes.
Despite the challenge of volatile organic compound (VOC) emissions, the sustainability profile of vat photopolymerization for water treatment composites can be significantly enhanced through material innovation and process control. The environmental impact is primarily governed by resin chemistry. The development and use of bio-based, acrylate-free, or water-developable photopolymer resins directly reduce hazardous emissions at the source. Furthermore, VP’s inherent advantages, i.e., high precision and low energy consumption compared to other AM methods, remain compelling. For water treatment, this precision enables the fabrication of complex, high-resolution structures ideal for advanced applications, such as photocatalyst-loaded monoliths for pollutant degradation, intricately patterned microfluidic devices for contaminant sensing, or membranes with precisely engineered pore architectures. By prioritizing greener resin formulations and tailoring VP’s design capabilities, the technology can produce effective, tailorable polymer composites for purification while aligning with broader environmental goals.
2.4. Material Extrusion
Material extrusion is a dominant, cost-effective additive technique in which material is dispensed through a nozzle and deposited layer-by-layer [59]. It primarily encompasses two methods: Fused Filament Fabrication (FFF)/Filament Deposition Modeling (FDM), which melts solid thermoplastic filaments, and Direct Ink Writing (DIW), which extrudes viscoelastic pastes or inks. The fundamental distinction lies in the feedstock: FFF is constrained to processable filaments, whereas DIW can utilize a broader range of materials provided the ink exhibits suitable rheological properties [60]. A key advantage of DIW is its ability to operate at ambient conditions without heated components, enabling the single-step fabrication of complex, multi-material structures, often with minimal post-processing [61].
From a sustainability perspective, the environmental impact varies considerably between these methods. For FFF, energy use and emissions are directly tied to material and process parameters; for instance, printing with ABS consumes more energy than PLA due to higher temperature requirements [62]. A study on a Stratasys Dimension SST FDM machine reported emissions of approximately 23.7 g CO2 per build [63]. In contrast, DIW generally presents a more favorable profile, as it frequently employs water-based inks and operates at room temperature with gantry-based systems, substantially reducing energy consumption and offering greater flexibility for integrating functional fillers [64].
Both FFF and DIW hold considerable promise for fabricating polymer composites for water treatment. FFF/FDM offers superior process stability, accessibility, and ease of use, making it a robust platform for prototyping and standardized components. DIW emerges as a more sustainable and versatile technology, capable of producing multifunctional composite architectures with lower energy input, though its advancement depends on overcoming challenges in ink development and scalability.
The feasibility of using Fused Filament Fabrication (FFF) and Direct Ink Writing (DIW) to produce sustainable polymer composites for water treatment is strong but distinct. FFF offers practical viability by utilizing commercially available bio-based filaments (e.g., PLA) compounded with sustainable fillers, suitable for fabricating customizable, macro-scale components like filter housings and biofilm carriers with a focus on material circularity. suitable for fabricating customizable, macro-scale components like filter housings and biofilm carriers with controlled macro-porosity and a focus on material circularity. For example, Roy Barman et al. [65] showed that FDM allows direct control of porosity through adjustable infill density, achieving 92% ethylbenzene removal at 50% infill, while PLA-TiO2 filters reached 100% methyl orange degradation at 34–50% infill. DIW provides a transformative, high-potential pathway centred on minimal energy use and advanced functionality. It enables room-temperature printing of aqueous-based inks loaded with sensitive nanomaterials or biological agents, ideal for creating intricate, multi-functional structures such as catalytic monoliths or graded membranes.
2.5. Material Jetting
Material Jetting (MJ), also referred to as PolyJet or Multijet, is widely recognized as one of the most convenient and effective additive manufacturing (AM) methods for producing high-resolution composite materials [66,67,68]. MJ offers excellent scalability, enabling reliable operation from laboratory-scale prototyping to industrial-scale production [69]. Compared with many other AM technologies, MJ provides superior cost-efficiency, printing speed, and material flexibility. For instance, the piezoelectric–pneumatic material jetting (PPMJ) system, an ink-based AM process, can fabricate 3D structures at speeds of up to 500 mm/s and is capable of printing extremely viscous inks (up to 1,000,000 mPa·s), far exceeding the limits of conventional inkjet and aerosol-jet systems [70]. Balavandy et al. [71] reported that the MJ method can fabricate 136 devices in a single 136-min run at an approximate cost of USD 0.60 per device.
In the context of water treatment, MJ has been applied to specialized devices such as microfluidics and biocarriers. Examples include single-material fluidic devices with integrated filters for point-of-collection colorimetric sensing [72] and acrylic-based gyroid biocarriers for biological wastewater treatment [73]. Material Jetting (MJ) has strong potential to produce functional polymer composites for water treatment, provided its materials and processing strategies are carefully optimized. Advancements in resin chemistry, including water-stable, hydrophilic, or hybrid organic–inorganic photopolymers, can significantly increase the chemical durability of MJ-printed components in aqueous environments. Incorporating functional nanomaterials such as TiO2, ZnO, AgNP, graphene oxide, and inorganic fillers (e.g., zeolite, hydroxyapatite) into jettable inks can further impart adsorption, photocatalytic, or antimicrobial properties essential for pollutant removal. Multi-material jetting also enables the fabrication of gradient structures, hybrid filtration layers, and surface-localized catalytic regions, enhancing filtration efficiency and broadening MJ’s applicability beyond prototyping.
Mechanical limitations of MJ can be mitigated through the integration of nanocellulose, short fibers, ceramic microfillers, or polymeric reinforcements, while post-processing treatments, such as UV/thermal curing, plasma activation, dip-coating, or nanocomposite surface films, can improve hydrophilicity, durability, and anti-fouling behavior. By combining these material innovations with MJ’s inherent precision and high-resolution capabilities, the technology can be repositioned as a sustainable platform for fabricating microfluidic devices, catalytic reactors, and modular filtration units. With continued development in resin recovery, reduced support usage, and energy-efficient curing systems, MJ could become a viable and environmentally responsible method for producing polymer composites tailored for water and wastewater treatment.
2.6. Powder Bed Fusion
Among the major additive manufacturing (AM) technologies, Powder Bed Fusion (PBF) stands out as a highly suitable technique for fabricating large, complex components [74]. Similar to other AM methods, PBF enables the creation of intricate geometries with excellent dimensional accuracy and supports a wide variety of materials, including polymers, ceramics, and metals [75]. A major advantage of PBF is its solvent-free, layer-wise processing approach. Additionally, the unsintered powder surrounding the part acts as a natural support, making the process effectively “support-free”. Most importantly, PBF is renowned for producing structures with controlled porosity and highly interconnected pore networks, which are particularly beneficial for filtration, adsorption, and catalytic applications [76].
PBF technologies operate by selectively fusing powder particles layer by layer using a thermal energy source [77]. This general category includes Selective Heat Sintering (SHS), Selective Laser Sintering (SLS), Selective Laser Melting (SLM), Direct Metal Laser Sintering (DMLS), Electron Beam Melting (EBM), and Multi Jet Fusion (MJF). While many of these systems are optimized for metals or ceramics, Selective Laser Sintering (SLS) and Multi Jet Fusion (MJF) are the primary PBF techniques capable of processing polymer composites.
A wide range of polymer composite systems has been produced using SLS and MJF. Notable examples include polypropylene (PP)/Dowex 21K resin composite filters for palladium and platinum adsorption [78], gyroid nanostructured polyamide components, and polyamide-12 (PA12) parts coated with silver-based antibacterial layers [79]. The sustainability of PBF varies considerably with the type of material processed; metal-based PBF typically requires far more energy than polymer-based systems. For instance, machining 1 kg of aluminium alloy, Al7075 uses only 16.3 MJ/kg, whereas laser powder bed fusion (L-PBF) consumes approximately 560 MJ/kg to deposit 1 kg of AlSi10Mg alloy and up to 1678.13 MJ/kg to fabricate a complete part [80]. By comparison, energy consumption for PA12 printing is substantially lower, at approximately 986.48 MJ/kg [81].
Improving the sustainability of Powder Bed Fusion (PBF) requires a combination of optimized processing parameters, efficient material management, and intentional design strategies. From a processing perspective, one of the most effective sustainability improvements is the optimization of energy usage during laser or lamp exposure. This can be achieved by fine-tuning scan strategies, reducing unnecessary overexposure, and employing adaptive power modulation based on part geometry and thermal history. Reusing and rejuvenating unfused powder is another critical element; controlled sieving, blending with virgin powder, and implementing powder-aging monitoring systems help extend powder life while maintaining consistent mechanical performance. Lower-temperature polymer powders, such as thermoplastic polyurethane (TPU), and newly developed bio-based or recyclable powders, represent a promising pathway to reduce energy consumption compared to high-temperature polymer systems.
2.7. Sheet Lamination
Sheet lamination, commonly termed Laminated Object Manufacturing (LOM), is an additive manufacturing process that constructs parts by sequentially bonding and cutting layers of sheet material [82]. This method shares the advantages of other AM techniques, such as producing complex geometries at relatively low cost, while offering specific benefits like nontoxic processing, inexpensive raw materials, and continuous production capability [83].
Laminated Object Manufacturing (LOM) offers a direct pathway to producing sustainable polymer composites for water treatment by exploiting its unique process advantages. Its inherent nontoxic, solvent-free bonding, typically using heat and pressure alone, aligns with green manufacturing principles by avoiding volatile organic compound (VOC) emissions. The process is highly compatible with recycled, bio-based, or natural material sheets, such as those made from PLA, cellulose, or polymer-fiber mats, which can be laminated with functional interlayers. For water treatment, this could enable the fabrication of large-scale, laminated composite panels or structured packings where each layer serves a distinct purpose: a structural support layer from recycled polymer, an adsorbent layer infused with biochar or clay particles, and a functional top layer with antimicrobial properties. This multi-material, waste-minimizing approach, coupled with LOM’s ability to use inexpensive, non-toxic feedstocks, positions it as a potentially sustainable and scalable method for creating customized filtration components. However, realizing this potential requires focused research to develop suitable recycled or bio-based sheet materials and to design effective, layer-specific functionalities for targeted water treatment.
2.8. Summary Remarks on Printability, Design Flexibility, and Sustainability of AM for Polymer Composites
The additive manufacturing (AM) techniques reviewed herein demonstrate distinct capabilities for printing polymer composites. Fused Filament Fabrication (FFF/FDM) and Direct Ink Writing (DIW) offer high accessibility and broad compatibility with diverse polymer matrices and functional fillers, making them well-suited for fabricating composites with customized adsorption or antimicrobial properties. In contrast, Selective Laser Sintering (SLS) and Multi-Jet Fusion (MJF) provide superior geometric precision and mechanical performance, enabling the production of robust, porous, or architectured composites capable of withstanding the operational stresses of water treatment systems. However, methods such as Directed Energy Deposition (DED) and Laminated Object Manufacturing (LOM) currently show limited direct applicability for polymer-based water treatment composites due to prevailing material constraints and process complexity. Nonetheless, their unique inherent strengths point toward distinct, high-potential niches. DED’s capability for large-scale, high-deposition-rate manufacturing and its ability to clad or coat existing components could be exploited for creating durable, multifunctional surfaces on structural water treatment modules. Similarly, LOM’s solvent-free, layer-by-layer assembly is exceptionally well-suited for fabricating sustainable, multi-material laminates from recycled polymer sheets or bio-based composites, offering a promising route for large-area, panel-type filtration media. Thus, while not yet mature for mainstream application, these methods represent compelling avenues for future research to overcome current limitations and unlock novel, scalable, and sustainable composite architectures for water treatment.
A universal advantage across AM is its inherent design flexibility. Unlike conventional manufacturing, AM facilitates the fabrication of intricate internal geometries, controlled porosity, lattice structures, and surface micro-architectures that can dramatically enhance water treatment performance. These capabilities enable improved fluid dynamics, maximized surface area for adsorption, enhanced mass transfer, and the spatial integration of multifunctional zones (e.g., catalytic or antimicrobial regions). This geometric control allows for the optimized design of 3D-printed components, such as adsorbers, photocatalytic reactors, biofilm carriers, and microfluidic devices with higher efficiency and reduced material consumption.
From a sustainability perspective, AM processes vary significantly in energy consumption and environmental impact. Vat Photopolymerization (SLA/DLP) and DIW, which operate at lower temperatures, generally exhibit lower energy demands. Conversely, powder-based systems such as SLS/MJF require substantial thermal input, leading to a higher carbon footprint. The sustainability profile is further influenced by the choice of polymers, fillers, and build strategies. While AM can minimize material waste through on-demand fabrication and design optimization, certain methods (e.g., material jetting, powder bed fusion) still generate notable waste streams or rely on energy-intensive equipment. Therefore, a balanced consideration of printability, design freedom, and environmental impact is critical when selecting an AM process for sustainable water treatment composites. The diverse capabilities of AM establish a robust foundation for developing polymer composites tailored for advanced water purification. The ability to simultaneously fine-tune geometry, material composition, and process sustainability positions AM as a transformative tool for creating high-performance, resource-efficient, and environmentally conscious water treatment solutions.
Table 2 shows the comparison of AM techniques and potential key modification strategies for water treatment. Although the seven additive manufacturing techniques differ significantly in their baseline printability, energy demand, and sustainability performance, all can be engineered into viable platforms for polymer composite-based water treatment systems through targeted processes and material modifications. For example, Vat Photopolymerization and Material Jetting excel in precision and multifunctionality but require improvements in resin sustainability and waste reduction, whereas Material Extrusion and Binder Jetting offer greater compatibility with recycled and bio-based polymers. Powder Bed Fusion provides unmatched control over interconnected porosity, critical for filtration and adsorption, but must address energy intensity through powder reuse and design optimization.
The comparison reveals that functional performance in water treatment is more strongly governed by post-print functionalization, filler distribution, and structural design than by the printing method itself. Strategies such as surface coating, polymer–nanofiller hybridization, lattice-based eco-design, and modular scaffold fabrication enable even traditionally limited techniques (e.g., DED and Sheet Lamination) to contribute meaningfully. Consequently, additive manufacturing for water treatment should be viewed not as a selection of good or bad techniques, but as a spectrum of modifiable manufacturing platforms aligned through sustainable design and functional intent. Through material reformulation, process optimization, and surface functionalization, all seven additive manufacturing techniques can be transformed into sustainable and application-ready routes for polymer composite water treatment technologies.
While all seven additive manufacturing (AM) techniques can be technically adapted for polymer composite-based water treatment, cost, including capital expenditure, material consumption, energy use, and post-processing requirements, ultimately governs technology selection for real-world implementation. Differences in process efficiency, material utilization, and post-processing demands can significantly influence overall expenses, making some AM techniques more suitable than others for large-scale applications. The cost-effectiveness of different AM techniques is compared qualitatively based on their relative economic practicality. This comparison considers major cost-related factors, including capital investment, feedstock cost, energy consumption, post-processing requirements, material utilization efficiency, and scalability for water treatment applications. Among the AM techniques, Material Extrusion demonstrates the highest cost-effectiveness due to its low equipment cost, inexpensive thermoplastic feedstocks, moderate energy consumption, minimal material waste, and strong scalability for porous monolith fabrication. Sheet Lamination and Binder Jetting follow, benefiting from relatively low energy demand and reasonable material efficiency, although binder costs and post-processing slightly reduce the economic advantage of Binder Jetting. Vat Photopolymerization offers high fabrication precision but is limited by expensive resins and mandatory post-curing, resulting in moderate cost-effectiveness. Powder Bed Fusion requires higher capital and energy input, reducing its cost competitiveness. Material Jetting is constrained by high equipment and material costs, as well as support waste, while Directed Energy Deposition shows the lowest cost-effectiveness due to substantial capital expenditure, high energy consumption, and limited relevance to polymer-based adsorption systems. It should be emphasized that this ranking represents a qualitative comparison and does not constitute a comprehensive quantitative cost analysis.
A detailed lifecycle cost analysis could further quantify these differences and provide a data-driven basis for selecting the most economically viable AM approach. Techniques that combine low material cost, moderate energy demand, minimal post-processing, and compatibility with bio-based or recycled feedstocks are most likely to transition from laboratory demonstrations to scalable water treatment solutions. Future research should shift from technique-centric optimization toward cost-normalized performance metrics, such as adsorption capacity or contaminant removal efficiency per unit cost and energy input, to enable meaningful comparison and accelerate real-world adoption.
Table 2. Comparison of AM techniques and potential key modification strategies for water treatment.
|
AM Technique |
Printability |
Cost-Effectiveness |
Design Flexibility |
Energy Efficiency |
Sustainability |
Functionalization Potential |
Key Modification Strategies for Water Treatment |
|---|---|---|---|---|---|---|---|
|
Binder Jetting |
★★★ |
★★★★ |
★★★★ |
★★★★ |
★★★★ |
★★★★ |
Use polymer–ceramic hybrid powders, bio-based or water-soluble binders, post-print infiltration with alginate, chitosan, GO, or TiO2; low-temperature curing enables porous adsorptive filters |
|
Directed Energy Deposition |
★★ |
★ |
★★★ |
★ |
★★ |
★★★ |
Adapted for polymer use via low-energy laser or extrusion-assisted DED; hybrid deposition of polymer scaffolds followed by functional coatings |
|
Vat Photopolymerization |
★★★★★ |
★★★ |
★★★★★ |
★★★ |
★★★ |
★★★★★ |
Develop low-toxicity, bio-based resins; embed nanofillers (GO, TiO2, ZnO, AgNPs); apply post-curing washing and plasma treatment to remove residual monomers and activate surfaces |
|
Material Extrusion |
★★★★ |
★★★★★ |
★★★★ |
★★★★ |
★★★★ |
★★★★ |
Use recycled or bio-based filaments; compound with clays, lignin, HAp, and biochar; design gyroid/lattice porosity for enhanced mass transfer |
|
Material Jetting |
★★★★★ |
★★ |
★★★★★ |
★★ |
★★ |
★★★★★ |
Reduce waste via voxel-level material control; incorporate functional inks (GO, AgNPs); shift toward water-based or biodegradable photopolymers; post-print solvent extraction |
|
Powder Bed Fusion |
★★★★ |
★★ |
★★★★ |
★★ |
★★★ |
★★★★ |
Optimize laser power and scan strategy; reuse powder; design self-supporting porous architectures; apply surface coating (TiO2, Ag, chitosan) instead of bulk loading |
|
Sheet Lamination |
★★★ |
★★★★ |
★★★ |
★★★★★ |
★★★★★ |
★★★ |
Employ biodegradable polymer sheets; integrate adsorptive interlayers (GO, cellulose, clay films); solvent-free thermal bonding for low-energy fabrication |
Notes: Functionalization potential reflects ease of incorporating adsorptive, photocatalytic, or antimicrobial functions either during or after printing. Rating: ★ = very low; ★★= low; ★★★ = moderate; ★★★★ = high; ★★★★★ = very high.
3. Sustainability Aspects and Considerations in 3D Printing Polymer Composites for Water Treatment
The successful integration of additive manufacturing (AM) into water treatment technologies depends not only on functional performance but also on minimizing environmental impact across the material lifecycle, from sourcing and fabrication to end-of-use. While broader sustainability considerations in AM, such as machine energy consumption, circular economy frameworks, and full lifecycle assessments, are undeniably important, this review adopts a materials-centric perspective to ensure a focused and in-depth analysis.
Consequently, this section concentrates on the foundational material choices that govern the sustainability of 3D-printed composites for water treatment. It is structured around two important aspects: (1) the selection of sustainable polymer matrices and (2) the choice of eco-friendly and functional fillers. By examining these core elements, we establish the principles for designing printable polymer composites that are both effective for water purification and aligned with green material science.
3.1. Sustainable Polymer Selection for Water Treatment
Market data reflects a rapidly accelerating demand for sustainable polymer composites, with the global market value reaching USD 73.56 billion in 2020 and projected to grow to USD 102.79 billion by 2028 [84]. Within this context, sustainable polymers are defined as materials sourced from renewable, recycled, or waste-carbon feedstocks that are designed for end-of-life recyclability, biodegradability, or composting, and which demonstrate a reduced environmental footprint across their entire lifecycle [85].
Polymer composites are engineered materials comprising two primary components: a polymer matrix that forms the continuous phase and a reinforcement or filler that enhances mechanical properties and introduces specific functionalities [86]. Advances in green chemistry and nanotechnology have positioned these composites as highly promising for wastewater treatment, owing to their advantageous combination of lightweight design, excellent mechanical properties, thermal stability, ease of functionalization, and selective adsorption capacity [87,88].
A wide variety of polymers serve as matrices in wastewater treatment composites. Common synthetic thermoplastics include polyvinyl alcohol (PVA), polyether sulfone (PES), polyvinylidene fluoride (PVDF), polyvinyl chloride (PVC), polypropylene (PP), polyethylene (PE), and polyacrylonitrile (PAN) [89]. Thermosets such as epoxies and polyurethanes, along with polymers like polyethylene glycol (PEG) and various polyacrylates, are also utilized [90]. Concurrently, biopolymers represent an important and expanding category. This includes natural polymers like chitosan, alginate, casein, cellulose, and chitin [89,90,91], as well as biodegradable polyesters such as polycaprolactone (PCL), polylactic acid (PLA), polyhydroxybutyrate (PHB), and polyhydroxybutyrate-co-valerate (PHBV) [92,93,94].
The final performance of these composites depends on synergistic interactions between the matrix and functional reinforcements. To enhance adsorption capacity, catalytic activity, selectivity, and reusability, contemporary composites frequently incorporate nanofillers or nanocoatings [95,96,97].
In application, polymer composites for water treatment are most commonly engineered into two functional forms: hydrogels and membranes. Polymeric membranes remain highly attractive due to their low cost, high contaminant rejection, low energy consumption, small carbon footprint, and material availability [89]. Hydrogels represent another emerging class with significant advantages, including fast-swelling kinetics, tunable surface charge, tailored pore structure, catalytic functionality, and broad chemical compatibility, making them effective for adsorbing diverse pollutants [98,99,100]. Based on this overview, the following subsections examine high-potential polymer matrices for additive manufacturing in wastewater treatment.
3.1.1. Poly(lactic acid) (PLA)
Poly(lactic acid) (PLA) is currently one of the most versatile and widely adopted thermoplastics for polymer additive manufacturing [101]. Its broad usage stems from several sustainability and performance advantages, including its inherent biodegradability, its environmental friendliness characterized by lower emissions of volatile organic compounds and nanoparticles compared to materials like acrylonitrile-butadiene-styrene (ABS), and its excellent biocompatibility [102,103,104]. In wastewater treatment, PLA-based composites have emerged as promising materials due to their ability to serve as effective carriers or matrices for active components used in heavy metal removal, dye adsorption, photocatalysis, and antimicrobial treatment. This potential is attributed to the uniqueness of PLA. First, PLA exhibits excellent printability across various AM techniques, enabling the precise fabrication of complex, architectured geometries such as lattices, spirals, gyroids, and structures with gradient porosity. These designed surfaces enhance mass transfer, increase the available active surface area, and improve interactions with contaminants. Second, certain PLA composites facilitate energy-efficient treatment; for example, 3D-printed PLA–hydroxyapatite filters have demonstrated high water flux driven solely by gravity or capillary action, eliminating the need for external pressure or electrical input [105]. Third, PLA possesses favorable surface chemistry, allowing straightforward functionalization via methods such as plasma treatment, photografting, alkaline hydrolysis, and atom transfer radical polymerization. These surface modifications promote strong bonding of active materials to the polymer matrix, which is essential for maximizing performance and minimizing the leaching of functional agents during water treatment operations [106].
3.1.2. Polyhydroxyalkanoates (PHA)
Polyhydroxyalkanoates (PHAs) are a versatile family of biodegradable and biocompatible polyesters synthesized by various microorganisms as intracellular carbon and energy storage compounds [107,108,109]. Despite their excellent inherent biodegradability and biocompatibility, commercial variants like PHB and PHBV are hindered by low impact strength, high brittleness, and limited flexibility, reducing their suitability for many industrial applications when compared to conventional or other biodegradable polymers [110]. Furthermore, high manufacturing costs, driven partly by substrate expenses accounting for approximately 40% of total production, continue to constrain large-scale adoption [111]. Nevertheless, PHAs possess several intrinsic advantages that support their use as sustainable alternatives. Their biodegradation profile is particularly noteworthy, as reported by Koller et al. [112], showing that PHAs meet all biodegradability standards specified in EN 13432 across diverse environments, including soil, freshwater, seawater, and composting systems, and crucially degrade without leaving behind persistent microplastic residues.
Recent work has explored the performance of PHA-based composites in diverse water treatment processes, demonstrating their functional versatility. Notable examples include heavy metal removal, where a PLA/PHBV/graphene oxide composite produced via melt compounding and compression molding achieved 98% Pb2+ removal [113]. For nickel adsorption, a PHB/cellulose acetate film fabricated by solvent casting demonstrated 86.44% Ni2+ removal with an adsorption capacity of 5.042 mg/g [114]. In dye and oil remediation, a cost-efficient PHBV/cellulose membrane produced by solution casting removed up to 95.15% of vegetable oil and 83.79% of Congo red dye, while retaining more than 40% of its adsorption capacity after three reuse cycles [115]. Beyond these specific applications, PHA-based systems have also shown promise for bacterial filtration, nanoparticle rejection, landfill leachate treatment, desalination, and solvent dehydration, demonstrating their broad potential as functional and sustainable water treatment materials [116].
For successful 3D printing, PHA often requires formulation into a composite feedstock to overcome its inherent brittleness and improve thermal processability. This can involve blending with other biopolymers, such as PLA, melt-compounding with plasticizers, or incorporating functional fillers, such as biochar, clay, or graphene oxide, directly into the filament or resin. These modifications simultaneously enhance printability and introduce adsorption or catalytic sites for water purification. The design freedom of additive manufacturing can then be exploited to fabricate geometrically optimized structures, such as high-surface-area scaffolds and/or porous monoliths from these PHA composites, maximizing contact with contaminated water and creating efficient, biodegradable filtration media.
3.1.3. Cellulose-Based Polymers
Cellulose is a naturally occurring polysaccharide and the most abundant biodegradable biopolymer [117]. It is primarily derived from plant cell walls, but is also produced by algae, fungi, bacteria, and marine organisms such as tunicates [118]. The numerous hydroxyl groups along its molecular chains enable strong hydrogen bonding with water, causing cellulose to swell or disperse easily in aqueous environments, a characteristic widely exploited in processing steps such as dissolution, regeneration, and chemical modification [119].
In recent years, nanocellulose-based composites have emerged as one of the most promising classes of materials for water purification, exhibiting high performance in dye removal, heavy metal adsorption, general water purification, and oil absorption [120,121,122,123]. Nanocellulose exists in three principal forms, each with distinct structural and functional properties. Cellulose nanocrystals are rigid, rod-like nanocrystals less than 100 nanometers in thickness, known for a high surface-to-volume ratio and exceptional mechanical properties, including tensile strength of approximately 10 GPa, which make them excellent for adsorption, catalysis, and composite reinforcement [124]. Cellulose nanofibers consist of long, flexible fibers typically 5–60 nanometers in diameter and 0.1–2 micrometers in length, produced via mechanical fibrillation often preceded by chemical or enzymatic pretreatment; their lower crystallinity (50–80%) and high aspect ratio contribute to a strong network-forming ability, making them well-suited for applications in membrane filtration and flocculation. Bacterial cellulose is biosynthesized by microorganisms such as Gluconacetobacter hansenii and is characterized by ultra-high purity and crystallinity (84–89%), an exceptional water-holding capacity (up to 99%), and outstanding mechanical strength, biocompatibility, and biodegradability, rendering it an ideal material for advanced filtration and high-value water treatment applications. Together, these nanocellulose variants offer extensive tunability and functionality for fabricating sustainable, high-performance water treatment composites, a potential that is significantly amplified when integrated with the design freedom of additive manufacturing [125].
Cellulose, particularly in its nanofibrillated or nanocrystalline forms, can be incorporated into 3D printing for water treatment by formulating it into functional bio-inks for Direct Ink Writing (DIW) or as a reinforcement in thermoplastic filaments for Fused Filament Fabrication (FFF). As a highly hydrophilic and modifiable biopolymer, cellulose provides an excellent, sustainable matrix for loading with active fillers such as metal-organic frameworks (MOFs) or graphene oxide. When 3D printed, cellulose-based composites can be shaped into high-surface-area architectures, such as porous lattices, monoliths, or structured membranes, that maximize contact with contaminated water for efficient adsorption of dyes, heavy metals, and oils, exploiting both the material’s distinctive affinity for pollutants and the geometric advantages of additive manufacturing.
3.1.4. Recycled and Upcycled Polymers
The global transition toward a circular economy, a regenerative model designed to eliminate waste and keep materials in continuous use, has intensified interest in recycling and upcycling polymers [126]. Within this paradigm, AM has emerged as a promising platform for valorizing post-consumer plastic waste by converting it into functional feedstock for 3D printing. A wide variety of recycled plastics have been successfully processed via AM, including recycled polyethylene terephthalate (rPET), polypropylene (rPP), high-density polyethylene (rHDPE), polylactic acid (rPLA), acrylonitrile butadiene styrene (rABS), high-impact polystyrene (rHIPS), and polycarbonate (rPC) [127].
To restore or enhance the properties of these recycled materials, a common strategy is to blend them with virgin polymers. This approach improves printability and mechanical strength while reducing the demand for new petroleum-based plastics, thereby conserving resources and extending material lifecycles [128]. From an environmental perspective, recycling plastics for AM offers substantial sustainability benefits by diverting waste from landfills and incineration, thereby mitigating associated CO2 emissions and toxic releases [129]. This integration supports sustainable manufacturing goals by reducing raw material extraction, minimizing waste, and lowering the carbon footprint of production. Recycled and upcycled polymers represent a vital and growing material class for sustainable 3D printing. They offer a compelling combination of environmental, economic, and functional advantages, positioning them as foundational components in the development of eco-friendly polymer composites for advanced water treatment systems.
The application of recycled polymers in 3D printing for water treatment offers a direct pathway to fabricating sustainable, customized components such as filter housings, structural packings, biofilm carriers, and modular reactor elements. By processing materials like rPET or rPLA into functional filaments or inks, AM can transform post-consumer waste into geometrically complex parts designed to optimize hydraulic flow, maximize surface area, and integrate functional zones for adsorption or catalysis. For instance, a recycled polymer matrix can serve as a durable scaffold, into which sustainable fillers (e.g., biochar or clay minerals) are compounded, creating a composite that exploits both the circularity of the matrix and the remediation capability of the filler. This approach can divert plastic from the waste stream as well as enables the on-demand, localized production of treatment components, reducing supply chain emissions and material surplus.
However, several technical and quality concerns must be addressed to ensure reliability and performance. The primary challenge is the inconsistent properties of recycled feedstocks, which often exhibit batch-to-batch variations in molecular weight, contamination levels, and thermal stability, leading to unpredictable printability and mechanical performance. There is also the risk of leaching of residual additives or adsorbed contaminants from the recycled plastic into the treated water, necessitating rigorous cleaning protocols and material certification for safe use. Overcoming these barriers requires advanced sorting and purification technologies, and the development of standardized recycling and printing guidelines specifically tailored for water treatment applications.
3.2. Sustainable Fillers and Functional Additives
The selection of fillers and functional additives profoundly influences the performance and sustainability of 3D-printed polymer composites for water treatment. While the polymer matrix provides the structural framework and printability, the incorporated filler phase imparts critical functionalities, such as adsorption, catalysis, antimicrobial activity, and mechanical reinforcement, tailoring the composite for specific purification tasks. This section examines the critical role of sustainable fillers, often energy-intensive, which focus on alternatives that align with green chemistry principles and circular economy goals. These additives can enhance the efficacy of water treatment composites as well as determine their overall environmental footprint, lifecycle, and potential for safe end-of-use scenarios.
Fillers for sustainable composites can be broadly categorized into several main classes based on their origin and function: bio-based fillers, inorganic fillers, and functionalized fillers. Bio-based fillers, derived from renewable biological resources, offer low environmental impact and inherent biocompatibility. Bio-fillers represent a diverse class of sustainable additives derived from renewable biological sources, generally categorized into several groups: cellulose-based, starch-based, protein-based, mineral-based fillers, and emerging materials derived from sources such as microalgae and paper sludge residues [130]. Their adoption in polymer composites is propelled by compelling advantages, including cost-effectiveness, reduced environmental impact, renewability, and their capacity to enhance specific material properties. The inherently low density of many bio-fillers also contributes to the development of lightweight composite structures. As a result, bio-reinforced polymer composites have garnered significant industrial interest and are increasingly applied in sectors ranging from biomedicine and construction to electronics and automotive manufacturing [131]. For water treatment applications, bio-fillers are particularly valuable as they can introduce or enhance functionalities such as contaminant adsorption, biodegradability, and biocompatibility, aligning material performance with the principles of a circular economy. Inorganic fillers, including various mineral and engineered particles, provide robust mechanical and thermal properties, often with high natural abundance. Functionalized fillers represent an advanced category in which any base material, whether bio-based or inorganic, is chemically or physically modified to introduce targeted, high-performance properties for contaminant removal.
The integration of these fillers into polymer matrices for additive manufacturing presents unique opportunities and challenges. The chosen filler must be compatible with the printing process, whether dispersed in a filament for Fused Filament Fabrication (FFF) or suspended in a resin for Vat Photopolymerization, without compromising rheology or curing kinetics. Furthermore, the filler-matrix interface must be engineered to ensure strong adhesion, load transfer, and long-term stability in aqueous environments.
While biopolymers such as cellulose, chitosan, and lignin can serve as the primary matrix in 3D-printed composites for water treatment, they are also highly effective as functional fillers or minor components within a synthetic or other biopolymeric matrix. Their suitability for this secondary role stems from their distinct physics and properties. When processed into specific forms, such as nanocellulose fibrils, chitosan nanoparticles, or micronized lignin powder, these materials provide high surface area, abundant functional groups (e.g., hydroxyl, amine, phenolic), and intrinsic adsorption capacity. Incorporating them as fillers, even at low loadings, can significantly enhance the composite’s performance by introducing active sites for pollutant binding, improving mechanical reinforcement, or imparting antimicrobial properties. This approach utilizes the unique functionality of biopolymers while mitigating potential limitations in their printability or mechanical strength when used alone as the matrix, thereby enabling the design of hybrid composites that optimize both processability and water treatment efficacy. The following subsections will detail each potential filler, evaluating their performance, characteristic properties, mechanisms of action in water treatment, and their specific suitability for use in sustainable, 3D-printed composite systems.
3.2.1. Lignin (as Filler)
Lignin is the most abundant renewable source of aromatic biopolymers, constituting approximately 15–40% of the dry weight of most plants [132]. Structurally, it is a highly complex, amorphous heteropolymer composed of three major phenylpropanoid monomers: p-coumaryl alcohol, guaiacyl alcohol, and syringyl alcohol. Its molecular architecture varies with biomass source; for example, softwood lignin is rich in guaiacyl (G) units, forming a densely cross-linked network, while hardwood lignin contains a mixture of guaiacyl (G) and syringyl (S) units, resulting in a more linear and less condensed structure [133].
This chemical versatility, characterized by abundant hydroxyl, methoxyl, and other functional groups, enables strong interactions with various polymer matrices. Consequently, lignin is widely employed as a filler, stabilizer, compatibilizer, and reinforcing agent in polymer composites [134]. In recent years, these functional characteristics have also positioned lignin as a promising, low-cost adsorbent for wastewater treatment. However, its inherent structural heterogeneity and low porosity often result in limited adsorption efficiency in its native form [135]. To overcome this limitation, various modifications, including sulfonation, oxidation, graft polymerization, and carbonization, have been employed to enhance its adsorption capacity and selectivity. These tailored treatments have successfully enabled lignin-based materials to remove heavy metals, dyes, pharmaceuticals, and other organic pollutants effectively. To realize the full commercial potential of lignin-based materials, several defined challenges must be addressed. These include standardizing feedstock variability, refining extraction processes to improve cost-effectiveness and product consistency, and fostering stronger collaboration across the value chain, from research institutions to industrial producers and end-users. Successfully overcoming these hurdles presents a significant opportunity to unlock lignin’s promise as a scalable, high-performance, and sustainable material for advanced applications [136].
Lignin’s complex aromatic structure, rich in phenolic and methoxy groups, provides a distinct mechanism for water treatment through π-π interactions, hydrophobic attraction, and redox activity, making it particularly effective for adsorbing and potentially degrading persistent organic pollutants like pharmaceuticals, endocrine disruptors, and synthetic dyes. In 3D printing, lignin’s dual role is to act as a sustainable reinforcement, improving the thermal stability and UV resistance of biopolymer matrices, while simultaneously introducing active sites for contaminant capture. When formulated into composite filaments or resins, lignin enables the additive manufacturing of robust, multifunctional structures, such as photocatalytic supports or filters for non-polar contaminant removal, demonstrating its natural affinity for aromatic and hydrophobic compounds and offering a valorization pathway for a major biomass byproduct into tailored water purification media.
3.2.2. Chitosan (as Filler)
Chitosan is the second most abundant natural polymer after cellulose, derived from the deacetylation of chitin (a biopolymer) widely sourced from the exoskeletons of crustaceans, insects, and mollusks [137,138]. As a biocompatible, biodegradable, and non-toxic biomolecule, chitosan possesses a flexible polysaccharide backbone rich in reactive amine (–NH2) and hydroxyl (–OH) groups [139]. These functional groups are central to its interactions with contaminants, establishing chitosan as one of the most versatile and effective bio-fillers for water treatment applications. The amine and hydroxyl groups enable chitosan to form stable coordination complexes with heavy metal ions, functioning as a highly efficient bio-adsorbent. Under acidic conditions, the polymer becomes protonated and polycationic, facilitating strong electrostatic attraction to negatively charged pollutants such as anionic dyes, phosphates, and organic acids. Its ability to form hydrogen bonds further broadens its affinity for a wide range of inorganic and organic contaminants [140].
Owing to its chemical versatility, chitosan has been extensively studied as a sustainable filler for the development of advanced polymer composites, membranes, hydrogels, and adsorbent beads, with strong potential for wastewater remediation. Chitosan’s unique polycationic nature and abundant functional groups make it particularly valuable for creating 3D-printed components designed for selective contaminant removal and antimicrobial action. In water treatment, these properties are exploited by formulating chitosan into composite inks for Direct Ink Writing (DIW) or by incorporating it into thermoplastic filaments for Fused Filament Fabrication (FFF). Chitosan can actively participate in the purification process, in which its protonated amine groups in acidic or neutral water provide strong electrostatic binding sites for anionic pollutants like phosphates, nitrates, and certain dyes, while also exhibiting intrinsic antibacterial activity that inhibits biofilm formation on the printed structures.
The integration of chitosan into additive manufacturing enables the fabrication of complex, high-surface-area architectures, such as porous scaffolds, helical mixers, or gradient-density filters that are optimized for specific water treatment mechanisms. For instance, a 3D-printed chitosan-based hydrogel monolith can be designed to swell in water, exposing a vast internal network of active sites for rapid contaminant adsorption. Moreover, chitosan’s biocompatibility allows it to be combined with other bioactive agents or enzymes within a printable matrix, paving the way for advanced smart filters capable of degrading specific organic pollutants. This synergy between chitosan’s inherent chemical functionality and the geometric design freedom of 3D printing facilitates the development of highly efficient, customizable, and sustainable treatment modules that target complex water matrices beyond the capability of traditional adsorbents.
3.2.3. Cellulose (as Filler)
Cellulose exhibits excellent properties for water treatment, most notably as an effective adsorbent for removing metal ions, dyes, and various organic and inorganic pollutants. Beyond its adsorption capabilities, cellulose and its derivatives also serve as highly effective reinforcement materials, owing to their high Young’s modulus and intrinsic mechanical robustness [141]. Nanocellulose, in particular, demonstrates exceptional potential as a composite filler due to its nanoscale dimensions, high crystallinity, and exceptionally large specific surface area. Its incorporation into polymer matrices significantly enhances dry and wet mechanical strength, improves interfacial bonding, and promotes filler flocculation, leading to more uniform dispersion and composite integrity [142]. These attributes improve the material’s overall stability and durability in aqueous environments.
This reinforcement capability is clearly demonstrated by Kelly et al. [143], where the addition of just 0.5 wt% nanocellulose nearly doubled the tensile strength of a polymer composite to 17.20 MPa and substantially increased flexural strength from 19.31 MPa to 24.51 MPa. Furthermore, nanocellulose reduced voids and interlayer porosity in the 3D-printed structure, thereby improving interlayer adhesion and yielding a denser, more continuous final part. Comparable enhancements were reported by Müller et al. [144], who evaluated various cellulose derivatives, including cotton flakes, miscanthus fibers, and walnut particles, within a stereolithography (SLA)-printed resin. These reinforcements significantly increased tensile strength while improving elongation at break by 63% to 323%, depending on the cellulose type, demonstrating nanocellulose’s unique ability to simultaneously enhance stiffness, strength, and ductility in polymer composites. To fully harness cellulose’s reinforcing and functional potential, chemical modifications, such as grafting, esterification, etherification, and acylation, are often employed. These treatments improve compatibility with hydrophobic polymer matrices, introduce targeted functional groups, and enhance stability and selectivity for water treatment applications [145].
In water treatment, cellulose is primarily valued for its high surface area and abundance of hydroxyl groups, which provide active sites for the adsorption of heavy metals, dyes, and oils. When used in additive manyfacturing, cellulose is incorporated into polymer composite filaments or resins to create structured adsorbents and filters with precisely engineered geometries. 3D-printed structures from cellulose composites maximize hydraulic contact and internal surface area, significantly enhancing pollutant capture efficiency compared to conventional forms. Furthermore, cellulose can be chemically modified or combined with other functional agents within the printable feedstock to introduce additional capabilities, such as photocatalytic degradation or selective ion exchange, thereby creating multifunctional, sustainable purification devices through additive manufacturing.
3.2.4. Biochar
Biochar is a carbon-rich solid material produced from organic biomass through thermochemical decomposition under oxygen-limited conditions [145]. Its feedstock sources are highly diverse, encompassing agricultural residues, forestry by-products, wood chips, algae, sewage sludge, animal manure, and various types of organic municipal waste [146]. Production methods primarily include pyrolysis, as well as hydrothermal carbonization, gasification, and torrefaction. In wastewater treatment, biochar has emerged as an attractive adsorbent for removing heavy metals, nutrients, and organic contaminants, owing to its advantageous physiochemical properties [147]. These include a large specific surface area, well-developed micro- and meso-porosity, high carbon content, abundant surface functional groups, and alkaline surface chemistry, which contribute to a strong affinity for pollutants. Numerous studies have demonstrated the high efficacy of modified biochars in adsorbing heavy metals such as Pb2+ and Cd2+ [148,149,150]. Furthermore, modified biochars often exhibit excellent selectivity toward specific metal ions and maintain strong performance over multiple regeneration cycles.
Beyond heavy metal adsorption, biochar is increasingly recognized for its ability to remove excess nutrients, particularly nitrogen (N) and phosphorus (P), which are key contributors to eutrophication [151]. Treatment strategies such as electro-oxidation, microalgae-based membrane bioreactors, and adsorption have been employed to enhance nutrient removal, with metal-modified biochars showing promise by substantially reducing nutrient leaching. For example, eggshell-modified corn-stalk-derived biochar exhibited a remarkably high phosphorus uptake capacity of 557 mg/g [152]. Biochar also demonstrates significant potential in absorbing diverse organic contaminants, including dyes, phenols, pesticides, and antibiotics, thereby broadening its applicability in comprehensive water purification [153].
In additive manufacturing, biochar is typically incorporated as a functional filler within printable matrices such as hydrogels or polymer-based inks. Nevertheless, maintaining printability requires careful control of filler loading and particle size. Current studies suggest that relatively low biochar contents (<5–10 wt.%) and small particle sizes are necessary to preserve rheological stability and extrusion fidelity [154]. For example, incorporating 10 wt.% biochar increased the ink viscosity, thereby enhancing printing resolution. The fabricated hydrogel demonstrated a threefold increase in pore volume, accompanied by a significant improvement in contaminant adsorption capacity [155].
3.2.5. Zeolite
Zeolites are inorganic polymeric materials characterized by a three-dimensional tetrahedral framework, where each tetrahedron (denoted as TO4) contains a central trivalent, tetravalent, or pentavalent element, such as aluminum, silicon, or phosphorus. These tetrahedra connect through shared oxygen atoms, forming highly ordered networks of [SiO4]4− and [AlO4]5− units [99]. This unique crystalline structure imparts the defining properties of zeolites, e.g., high porosity, ion-exchange capacity, and molecular sieving behavior, making them cost-effective and environmentally compatible materials for separation processes [156].
Zeolites are fundamentally divided into natural and synthetic classes [157]. Natural zeolites, such as clinoptilolite, typically exhibit irregular, plate-like morphologies with rough, porous surfaces, resulting in high specific surface areas [158]. Owing to their intrinsic porosity and cation-exchange capacity, natural zeolites have been widely applied in water filtration, heavy-metal and dye adsorption, oil removal, and antimicrobial treatment [159,160,161]. In contrast, synthetic zeolites are engineered via hydrothermal synthesis to achieve highly controlled pore structures, uniform crystal size, and tailored purity, making them ideal for precise catalytic and separation applications [162,163,164]. The superior performance of synthetic zeolites comes with the challenge of higher resource intensity, highlighting a critical research imperative to develop more sustainable synthesis pathways that balance exceptional functionality with improved economic and environmental metrics [165].
Polymer/zeolite composites have gained significant attention by merging the high adsorption capacity of zeolites with the mechanical durability and processability of polymer matrices. These composites are commonly fabricated through coating or extrusion molding techniques [166]. Their integration offers key operational advantages, including reduced pressure drop at high fluid velocities, enhanced mass transfer from increased external surface area, improved product selectivity, and longer operational lifetimes due to reduced fouling [167].
Recent studies have reported the effectiveness of polymer/zeolite composites for water treatment. For instance, electrospun polylactic acid (PLA) fibers containing 4 wt% clinoptilolite (a type of zeolite) have demonstrated multi-functional filtration, achieving complete microplastic rejection, over 90% removal of methylene blue dye, and significant antimicrobial activity [168]. According to Kennedy et al. [169], melt-extruded PLA/zeolite composites have shown 40–60% ammonia removal within 48 hours. For heavy metal remediation, polyvinyl alcohol (PVA)/zeolite composites exhibit strong affinity and selectivity, typically following the adsorption order: Pb > Cr > Cu > Zn > Cd > Ni [170,171]. Furthermore, polysulfone blended with zeolite has achieved 96.8% copper ion removal via crossflow microfiltration under low pressure [172]. These studies highlight zeolite filled polymer composites as a promising and scalable platform for advanced water purification, combining high contaminant selectivity with enhanced mechanical performance and processability.
3.2.6. Clay
Clays and their associated oxides (e.g., Al, Fe, Si) have gained prominence in contaminant remediation due to their natural abundance, low cost, and eco-friendly profile. Their effectiveness stems from key physicochemical properties, notably high surface area and strong ion-exchange capacity, which make them potent adsorbents for diverse pollutants [173]. Structurally, clays belong to the phyllosilicate family, characterized by layered architectures of alternating tetrahedral silica and octahedral alumina sheets [174].
Among these, montmorillonite (MMT) is one of the most extensively studied clays for water treatment. MMT is a 2:1 phyllosilicate, consisting of an octahedral aluminum/magnesium oxide sheet sandwiched between two tetrahedral silica layers. Isomorphic substitution within its structure generates a permanent negative surface charge, making it particularly effective for binding cationic heavy metals [175,176]. Beyond its charge characteristics, MMT exhibits notable anisotropy, excellent barrier properties, high thermal stability, and can be exfoliated into nanosheets with extremely high aspect ratios [177,178]. These properties enable diverse applications; for example, polyacrylamide/MMT composites have achieved removal efficiencies of 98.7% for Co2+ and 99.3% for Ni2+ [179], while 3D-printed gelatin/alginate hydrogels containing MMT demonstrated a Pb2+ adsorption capacity of 134 mg/g [180]. MMT is also effective in dye removal and enhances membrane performance, in which, polysulfone/MMT (loading = 2 wt%) membrane achieved 79% diclofenac removal with strong antifouling properties [181]. In additive manufacturing, MMT has been integrated into polyethylene terephthalate glycol (PETG)-based composites, though surface modification remains essential to preserve mechanical performance in the final printed structures [182].
Kaolinite is another prominent clay mineral, valued as a cost-effective alternative to MMT [183]. It is a 1:1 dioctahedral phyllosilicate, typically composed of approximately 46% SiO2 and 38% Al2O3, and belongs to the aluminum silicate group, which is obtained by processing kaolin [184,185]. Unlike the swelling structure of MMT, kaolinite adopts a rigid, non-swelling arrangement stabilized by strong interlayer hydrogen bonds, resulting in lower cation-exchange capacity and more uniform particle dimensions [186]. When exfoliated, its plate-like structure provides a large surface area that enhances gas-barrier properties and mechanical reinforcement in polymers. Its surface aluminol and siloxane groups can also be chemically modified to improve interfacial bonding with organic matrices [187].
In water treatment, polymer/kaolinite composites demonstrate strong performance, particularly in dye adsorption. A composite assembled from natural chitosan and exfoliated kaolinite nanosheets achieved a maximum adsorption capacity of 261.69 mg/g for Cu2+ ions, demonstrating superior selectivity for copper in mixed metal systems (Cu2+ > Ni2+ > Co2+ >Zn2+ > Mn2+) driven by a triple synergistic mechanism of coordination bonding, ion exchange, and electrostatic attraction. The incorporation of kaolinite nanosheets served as physical cross-linkers, significantly enhancing thermal stability and mechanical flexibility (tensile strain >300%), while the material retained over 85% removal efficiency after five adsorption-desorption cycles [188]. In another research, a poly(aniline-co-o-phenylenediamine)/kaolinite microcomposite synthesized via in-situ chemical oxidative copolymerization exhibited enhanced thermal stability compared to the pure copolymer, while achieving significant removal efficiencies for heavy metals from lake water with a selectivity order of Pb2+ > Cu2+ > Cd2+ (for 10% clay loading), driven by the chelating properties of amino and imino groups in the polymer chain [189]. Beyond environmental applications, kaolinite also enhances additive manufacturing processes by acting as a nucleating agent, improving crystallization, increasing the melt flow index, and reducing warping, thereby enhancing the dimensional stability of 3D-printed parts [190,191].
3.2.7. Hydroxyapatite
Hydroxyapatite (HAp, Ca10(PO4)6(OH)2) is the predominant calcium phosphate ceramic and closely mimics the mineral composition of natural bone [192]. HAp has emerged as a potential adsorbent for diverse water pollutants attributed to its high biocompatibility, bioactivity, large specific surface area (~100 m2·g−1), and intrinsic porosity [193]. It exhibits a strong affinity for organic contaminants such as dyes and antibiotics [194] and functions as an effective “green” photocatalyst in wastewater treatment [195].
HAp demonstrates notable efficacy in heavy metal remediation, effectively removing ions like Cd2+ (99%) and Pb2+ (97%) through surface complexation, ion exchange, and dissolution–precipitation mechanisms [196]. However, its wide band gap (~5.5 eV) limits its inherent photocatalytic activity, resulting in minimal degradation of certain antibiotics under photolysis [197,198]. To address these constraints, HAp is frequently engineered into nanocomposites with enhanced optical and surface properties, achieving significantly improved adsorption capacities, for example, up to 769 mg/g for Congo red [199].
In additive manufacturing, HAp is a well-established filler in biomedical applications, particularly for fabricating bone scaffolds, where it enhances antibacterial activity, thermal stability, and mechanical properties such as flexural modulus [200,201]. Based on this foundation, recent research has adapted HAp-based composites for water remediation. Fijoł et al. [105] used a direct compounding approach via thermally induced phase separation to embed HAp into PLA filaments, producing monolithic filters with exceptional selectivity for Pb2+ and Cd2+ and adsorption capacities reaching 360.5 mg/g through a dissolution–precipitation mechanism. Wang et al. [202] employed a scaffold-filling technique, injecting a chitosan/HAp mixture into a 3D-printed PLA frame; the composite achieved a Cu2+ uptake of 119 mg/g through synergistic interactions with chitosan’s functional groups.
3.2.8. Silver Nanoparticles (AgNPs)
Silver nanoparticles (AgNPs) are metallic nanostructures typically ranging in size from 1 to 100 nm [203]. Their exceptionally high surface-to-volume ratio and large proportion of surface atoms impart unique optical, electrical, and functional properties [204]. AgNPs exhibit broad-spectrum antimicrobial activity, demonstrating potent inhibitory and bactericidal effects against a wide range of pathogens, including Gram-positive and Gram-negative bacteria, viruses, and fungi [205]. The high biocidal performance has established AgNPs as a leading nanomaterial for disinfection and microbial control.
In environmental and water treatment applications, AgNPs are valued for their strong affinity for halogens and their inherent antimicrobial efficacy. They are utilized for iodide removal in drinking water production and for capturing radioactive iodine in nuclear wastewater streams [206,207]. An approach developed by Polo et al. [208] employed a magnetically assisted oxidation–precipitation process using Ag-nanoparticle-functionalized magnetic microparticles. In their research, ~20 nm AgNPs anchored on polymer-coated Fe3O4 cores are activated by hydrogen peroxide to oxidize Ag to Ag+, which subsequently precipitates halides. The method achieved removal efficiencies of 67.01% for bromide and 56.92% for chloride at neutral pH, though performance was notably influenced by pH and the presence of organic scavengers.
Owing to their ease of synthesis, stability, and proven antimicrobial action, AgNPs are increasingly integrated into additive manufacturing processes [209]. Their incorporation into 3D-printed polymer composites significantly enhances the disinfection capability of fabricated structures, extending their utility to advanced water purification, filtration, and surface-contact pathogen control. However, the efficacy of these structures is often limited by silver ion leaching, which occurs via surface oxidation followed by dissolution into the water stream. This leaching process is primarily influenced by water chemistry factors, such as low pH, which promotes Ag+ dissolution, and by hydrodynamic conditions, including flow rate and residence time, which affect release kinetics [210]. To ensure safety and environmental sustainability, these systems must be optimized so that silver concentrations do not exceed the WHO and US-EPA regulatory limit of 180 µg/L [211].
3.2.9. Titanium Dioxide (TiO2)
Titanium dioxide nanoparticles (TiO2) are among the most extensively studied photocatalysts for water treatment [212]. TiO2 can be synthesized through various routes, including sol–gel, hydrothermal, and green chemistry methods [213]. TiO2 generally exhibits low toxicity, making them a safer and more environmentally compatible option for remediation [214]. In alignment with this safety profile, the U.S. Food and Drug Administration (FDA) classifies TiO2 as “Generally Recognized as Safe” (GRAS). It is approved for use as a color additive in food products, provided its concentration does not exceed 1% of the food’s weight, as specified in FDA regulations (21 CFR 73.575) [215]. The photocatalytic performance of TiO2 is profoundly influenced by its crystalline phase. TiO2 exists in three primary polymorphs: the tetragonal Anatase and Rutile phases, and the orthorhombic Brookite [216]. Among these, Anatase is typically preferred for organic pollutant degradation due to its high surface reactivity and favorable charge carrier dynamics. Rutile, the most thermodynamically stable phase, possesses a high refractive index that enhances light scattering and harvesting efficiency. Brookite, a metastable phase with a high absorption coefficient, is less common and can transform into Rutile under prolonged hydrothermal conditions [217,218,219,220].
The photocatalytic efficacy of TiO2 polymorphs in aqueous systems is well-documented. For instance, Anatase slurries have been reported to remove ~98% of cationic methylene blue within 150 min [221], while Anatase-based nanocomposites can degrade 99.7% of anionic methyl orange in 30 min [222]. Mixed-phase systems often exhibit synergistic effects; the commercial P25 formulation (contains 80.3% anatase and 19.7% rutile) achieves over 95% methylene blue degradation under optimized conditions [223].
Recent research has explored the integration of TiO2 into additive manufacturing via the polymer/nanoparticle composites technique. However, achieving a balance between photocatalytic efficiency and printability remains a key challenge, as performance is highly dependent on filler loading. For example, Bansiddhi et al. [224] incorporated 5 wt% mixed-phase TiO2 into a photocurable resin, achieving significant dye removal (81.9% methylene blue, 60% Rhodamine B), though the dominant mechanism was adsorption rather than photocatalysis. Sangiorgi et al. [225] observed that high loadings (30 wt%) of surface-treated TiO2 in a PLA matrix led to printing defects, such as pore collapse, yet the composite still achieved complete methyl orange removal after 24 hours due to increased surface roughness. Han et al. [226] demonstrated that the TiO2-coated 3D-printed Acrylonitrile Butadiene Styrene (ABS) scaffold achieved >99% oil rejection in oil–water separation systems and exhibited excellent regenerability.
3.2.10. Zinc Oxide (ZnO)
Zinc oxide (ZnO) nanoparticles can crystallize in three polymorphs, i.e., hexagonal wurtzite, cubic zincblende, and cubic rocksalt, which are widely recognized for their advantageous physicochemical properties, including low cost, environmental compatibility, and biocompatibility [227]. A defining feature is their wide bandgap, which confers strong UV absorption and scattering capabilities, making ZnO an effective ultraviolet radiation blocker [228]. In addition to its functional efficacy, ZnO is classified by the U.S. Food and Drug Administration (FDA) as Generally Recognized as Safe (GRAS) when used as a nutrient or dietary supplement (21 CFR 182.8991) [229]. For environmental safety and water quality, the World Health Organization (WHO) and the U.S. Environmental Protection Agency (EPA) have established a secondary maximum contaminant level for zinc at 5.0 mg/L (5 ppm) [230]. This threshold is primarily based on organoleptic properties, such as taste and water turbidity, as zinc is an essential element and generally exhibits low toxicity to humans at these concentrations.
ZnO NPs also exhibit broad-spectrum antimicrobial activity, demonstrating high efficacy against pathogens such as Escherichia coli, Staphylococcus aureus, and bacteriophages [231]. This biocidal performance is strongly size-dependent, as smaller nanoparticles provide a larger reactive surface area for generating reactive oxygen species (ROS), such as hydrogen peroxide. These ROS penetrate microbial cell walls, inducing oxidative damage and cell death [232]. Beyond antimicrobial applications, ZnO NPs serve as multifunctional additives in polymer composites, enhancing toughness, mechanical integrity, and anti-aging properties in polymer matrices [233]. Furthermore, their photocatalytic efficiency can be significantly improved through doping with metals (e.g., Ag, Cu) or non-metals (e.g., N, C), which enhance charge separation and electron mobility [234].
In additive manufacturing, ZnO has attracted significant interest in importing advanced functional properties. Kumar et al. [235] demonstrated that PLA filaments containing 2 wt% ZnO, produced via melt extrusion, achieved a shape recovery rate of 99.64% at 40 °C, increased crystallinity, and produced smoother surfaces suitable for biosensing. Additionally, certain ZnO formulations exhibit room-temperature ferromagnetism, enabling magnetic-assisted wastewater treatment; magnetic ZnO composites have achieved approximately 70% methylene blue removal with good reusability over multiple cycles [236].
The integration of ZnO into polymer matrices for additive manufacturing presents a defined opportunity to enhance functional performance, which can be fully realized by addressing interfacial compatibility [237]. By advancing these material engineering approaches, the inherent functional benefits of ZnO, such as antimicrobial activity, UV responsiveness, and photocatalytic capability, can be successfully integrated into 3D-printed components without compromising their structural integrity.
3.2.11. Graphene-Based Filler
Graphene-based nanomaterials have gained significant attention in membrane engineering due to their two-dimensional morphology, lightweight structure, and characteristic honeycomb lattice [238]. This family of materials includes graphene oxide (GO), reduced graphene oxide (rGO), graphene nanosheets, and multilayer graphene, each offering distinct properties based on chemical composition, layer count, and defect density. Among these, graphene oxide (GO) is particularly significant for water treatment applications due to its abundance of oxygen-containing functional groups, large specific surface area, and excellent colloidal stability. These characteristics enable GO to disperse effectively in polymer matrices and function as a highly versatile nanofiller [239].
GO enhances membrane performance through multiple mechanisms. Its hydrophilic surface chemistry improves water transport, while its functional groups enhance interfacial compatibility with polymers and increase pollutant adsorption capacity [240]. These combined effects lead to superior separation efficiency, as demonstrated by the incorporation of up to 3 wt.% GO into polyethersulfone (PES) membranes, which significantly improved heavy-metal removal to 85–90% for Zn, 70–75% for Cu, and 65–70% for Cd [241]. Saeedi-Jurkuyeh et al. [242] fabricated a thin-film nanocomposite forward osmosis membrane by embedding GO nanosheets within a polyamide active layer, which increased water flux to 34.3 L·m−2·h−1 and achieved exceptional heavy-metal rejection (Pb: 99.9%, Cd: 99.7%, Cr: 98.3%). GO also exhibits strong potential for dye adsorption via π–π interactions, electrostatic forces, and binding to surface functional groups, effectively removing a broad spectrum of anionic and cationic dyes depending on its oxidation state and surface chemistry [243,244].
Recent advances in additive manufacturing have further expanded GO’s role as a multifunctional filler. For example, Mohd Nizam et al [245] incorporated 10–25 wt.% GO into a polyurethane acrylate (PUA) matrix using digital light processing, significantly enhancing hydrophilicity and mechanical strength while imparting antimicrobial activity and high adsorption capacity for heavy metals. In another approach, Feng et al. [246] integrated 0.7 wt.% nano-GO into a methacrylated chitosan/gelatin matrix via UV-assisted direct ink writing, which increased the Young’s modulus nearly tenfold and produced a membrane with superhydrophilicity and underwater superoleophobicity, enabling high permeation flux (>39,500 L·m⁻2·h−1) and >99.5% oil–water separation efficiency.
These studies demonstrate that graphene-based materials, particularly GO, are among the most versatile and high-performance nanofillers for water purification. They enable the development of advanced membranes and 3D-printed structures that simultaneously enhance mechanical performance, hydrophilicity, antifouling resistance, and multi-contaminant removal efficiency.
3.2.12. Regulatory Considerations for Filler Materials in Water Treatment
Table 3 presents an assessment of several filler materials commonly employed for water treatment applications. We can evaluate each filler according to its application roles, leaching or migration behavior, and the water chemistry parameters that influence its stability and performance. A critical observation is the diversity of leaching mechanisms across materials, ranging from the oxidative dissolution of silver ions (Ag⁺) to the release of dissolved organic carbon (DOC) from biochar and the protonation-induced dissolution of chitosan under acidic conditions. This variability highlights the importance of matching filler selection not only to treatment goals but also to the specific chemical characteristics of the target water matrix to minimize unintended migration of filler components into treated water.
While certain fillers demonstrate promising functional properties for water treatment, such as high adsorption capacity or filtration efficiency, their practical feasibility is contingent upon compliance with stringent regulatory standards for water quality and material safety. A significant challenge arises from the heterogeneity of regulatory frameworks across material classes, which complicates direct comparisons among fillers. For instance, some materials are governed by health-based guidelines (e.g., WHO provisional values for silver), while others are subject to operational use limits (e.g., NSF/ANSI 60 standards) or indirect regulation through finished water quality parameters (e.g., aluminum and turbidity limits for clay and zeolite). Despite these inconsistencies, Table 3 provides valuable insights into the regulatory landscape governing filler materials, offering researchers a foundational understanding of safety considerations. This information can help guide future research directions in polymer-filler composites for water treatment, emphasizing the need to design materials that not only enhance performance but also demonstrate compatibility with existing regulatory frameworks and prioritize minimal leaching or migration of filler constituents into treated water.
Table 3. Some assessment of filler materials for water treatment applications, including leaching behavior, water chemistry influences, regulatory status, and risk mitigation strategies.
|
Filler |
Application Roles |
Leaching/Migration Behaviour |
Water Chemistry Influence |
Regulatory Status |
Risk Level (Qualitative) |
Mitigation Strategies |
|---|---|---|---|---|---|---|
|
Ag (Silver NPs) |
Strong antimicrobial agent |
Ag+ ion release (oxidative dissolution) |
Oxidation of silver ion; Aggregation |
WHO guideline ≈0.1 mg/L Not recommended for disinfection of drinking-water |
Moderate to High |
Encapsulation; Immobilization; Alternative disinfection methods |
|
Biochar |
Heavy metal adsorption; Organic removal |
DOC leaching; Fine particle migration |
Dissolved Organic Content (DOC): High DOC levels can detrimentally affect the removal of E. coli. Redox Activity: Biochar’s conductivity and surface functional groups (quinones/phenolics) mediate microbial electron transfer. |
Follow EBC (European Biochar Foundation) and IBI (International Biochar Initiative) standard guidelines for safe use in water treatment. |
Low to Moderate |
Controlled pyrolysis; Pre-ozonation; Washing prior to use |
|
Cellulose |
Structural reinforcement; Hydrophilicity |
Volumetric swelling; Fiber release |
pH; Ionic strength |
FDA/Material Safety Standard Generally Recognized as Safe (GRAS) |
Low |
Surface modification (TEMPO-oxidation); Crosslinking |
|
Chitosan |
Adsorption of metals; Coagulation |
Protonation and dissolution in acidic media |
pH < 6.3: Dissolution; pH > 7.0: Stability increases; High salinity affects swelling |
Malaysia (SPAN/MOH) pH 6.5–8.5; Turbidity < 5 NTU; Standard A BOD < 20 mg/L. |
Low |
Covalent crosslinking; Polymer composites formation |
|
Clay (bentonite, kaolinite) |
Barrier properties; Adsorption; Turbidity removal |
Aluminum leaching; Particle release |
Ionic strength affects dispersion; pH extremes increase aluminum leaching |
Aluminum: Secondary MCL 0.05–0.2 mg/L (EPA). EU standard: 0.2 mg/L. Turbidity: Must achieve <1 NTU before system integration. |
Low |
Organic modification (organoclays); Strong matrix bonding |
|
GO (Graphene Oxide) |
Adsorption enhancement |
Sheet detachment under poor crosslinking |
Ionic strength and pH affect stability |
Explicitly not approved for public water supply use in UK. “There are no approvals nor applications under consideration for the use of graphene oxide products” under Regulation 31 |
Moderate |
Covalent crosslinking; Reduction to rGO; Layer confinement |
|
Hydroxyapatite |
Phosphate removal; Metal sequestration |
Minimal Ca2⁺/PO43⁻ release |
Dissolution increases under acidic conditions |
NSF/ANSI 60 maximum use level: 1207 mg/L (operational limit for treatment chemicals).
|
Low |
Sintering at high temperatures; Stable matrix incorporation |
|
TiO2 |
Photocatalysis; Contaminant degradation |
Very low ionic dissolution; Particulate migration |
Mechanical shear; UV exposure; pH |
No WHO or national drinking water standards for ingestion. Inhalation toxicity is primary concern. NSF/ANSI 60 specifies max use level of 90 mg/L as total Ti (operational limit, not health-based). |
Low–Moderate |
Matrix bonding; Sintering; Immobilization on supports |
|
Zeolite |
Ion exchange; Adsorption; Filtration media |
Low; Potential Aluminium leaching at pH extremes |
pH-dependent cation exchange capacity; Ionic strength |
Max Use Level: Up to 66 g/L (NSF/ANSI 60). Aluminium leaching: EPA SMCL of 0.05–0.2 mg/L; WHO/MOH limit of 0.2 mg/L. |
Low |
Acid-washing prior to use; Particle size control; pH monitoring |
3.3. Selection of AM-Fabricated Polymer-Filler Composites in Water Treatment
Figure 3 presents the key criteria guiding the selection of polymer matrices and functional fillers for the fabrication of 3D-printed sustainable polymer composites for water treatment applications. From the polymer perspective, emphasis is placed on 3D printability and process compatibility to ensure reliable fabrication using AM techniques. In addition, the water stability (e.g., water resistance) of the 3D printed polymer composite’s structure should be considered as an essential factor in order to maintain their performance during prolonged exposure to an aqueous environment. Sustainability-related considerations, including the use of bio-based materials, low carbon footprint, and compatibility with circular economy principles, are highlighted as increasingly important factors in the design of water treatment materials.
The filler selection is primarily governed by functional performance and physicochemical properties that directly influence contaminant removal efficiency and overall treatment effectiveness. Functional fillers such as adsorbents, photocatalysts, or antimicrobial agents are selected to impart targeted capabilities, including adsorption, separation, or microbial inhibition, while maintaining compatibility with the polymer matrix and the printing process. Sustainability considerations, including filler abundance, environmental safety, and the potential for reuse or regeneration, further ensure that the resulting 3D-printed composites function as effective and environmentally responsible materials for water treatment.
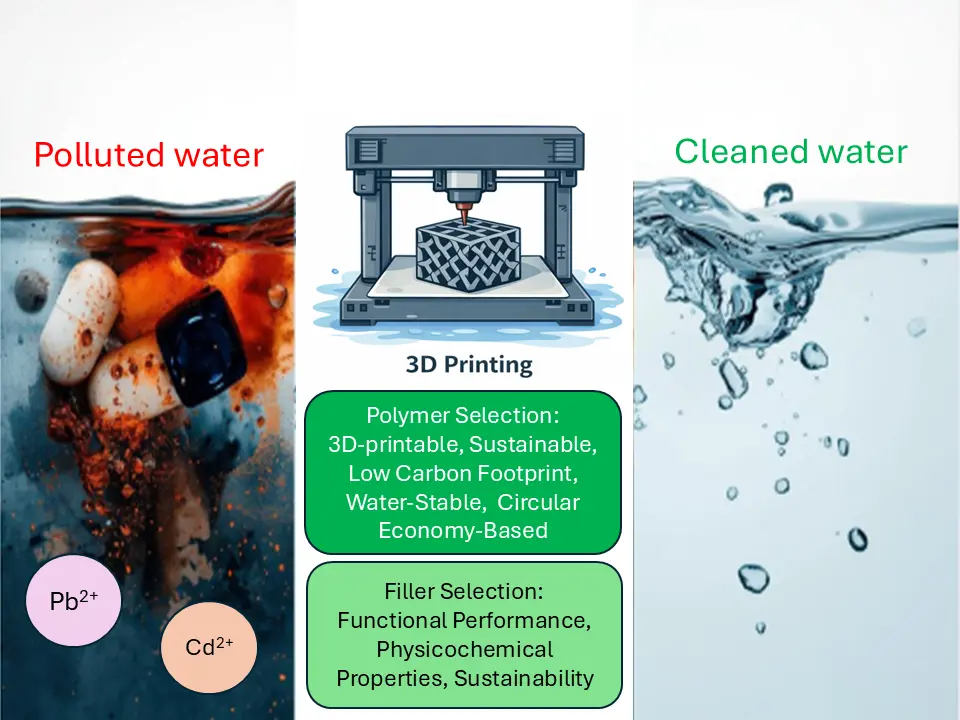
Figure 3. Key criteria for polymer and filler selection in 3D-printed sustainable polymer composites for water treatment applications.
Table 4 summarizes some examples of 3D-printed polymer composites that demonstrate strong potential for water treatment applications. Although some of the reported studies were originally designed for general or multifunctional purposes, their findings provide valuable insights that can be translated into water purification and environmental remediation systems. The table highlights how different additive manufacturing techniques, including DIW, DLP, FDM, FFF, and SLA, have been applied to prepare polymer composites with tailored functionalities and targeted applications.
Table 4. Additive manufacturing of polymer composites with potential applications in water treatment.
|
Polymer Matrix |
Fillers/Loading |
Printing Technique |
Significant Finding |
Reference |
|---|---|---|---|---|
|
PLA |
Zeolite (~16 wt%) |
FFF |
40–65% ammonia removal in 48 h |
[169] |
|
PLA |
Laccase immobilized enzyme |
FFF |
Removed ~40% of estrogens from wastewater. Retained 80% activity after 20 days. |
[247] |
|
PLA |
Hydroxyapatite (15wt%) |
FFF |
High adsorption capacity for cadmium (360.5 mg/g) and lead (112.6 mg/g). Achieved high water flux without external pressure. |
[105] |
|
PLA |
Cu-doped TiO2/rGO (applied as a coating using 4 mg/mL suspension) |
FDM |
Achieved >90% removal of methylene blue (30 cycles) and high stability (200% reduction in leaching via PDA bioadhesive) |
[248] |
|
PLA |
Silver nanoparticles (0.01–5 wt%) |
FDM |
Achieved >99% reduction of S. aureus, E. coli, and P. aeruginosa |
[249] |
|
PLA |
SiO2 nanoparticles + PDMS |
FFF |
Achieved super hydrophobicity (WCA 158.2°) and 88.6% continuous oil–water separation efficiency. |
[250] |
|
PLA |
Silver nitrate (0.5, 1.0, 2.0, 2.5 wt%) |
DLP |
Showed inhibition zones of S. aureus and E. coli |
[251] |
|
PVA/sodium alginate |
Graphene oxide (~33 wt%) |
DIW |
Achieved a high Cu2⁺ adsorption capacity of 208 mg/g. |
[252] |
|
Acrylate base photocurable resin |
TiO2 (5 wt%) + SiO2 (5 wt%) |
SLA |
Removal of methylene blue (81.9%) and Rhodamine B (60%). |
[224] |
|
Chitosan |
Zeolitic imidazolate framework-67 (~20 wt%) |
SLA |
Effective removal in ternary systems (Pb, Cd, Cu) |
[253] |
|
Dimethylacetamide modified Chitosan |
AgNPs (0.3–1.2 mL) with GO (0.3–1.2 mL) |
UV Inkjet 3D Printing |
Inhibited >95% of Coliforms and 99.9% of E. coli |
[254] |
|
ABS |
AgNPs |
FDM |
Eliminated all tested bacteria within 4h and fungi within 19 h. Maintained efficacy for up to 8 usage cycles. |
[255] |
|
Polycaprolactone |
Sodium alginate (5–30 wt%) |
FFF |
Achieved copper adsorption capacity (93.3 mg/g) |
[256] |
Remarks: DIW = Direct Ink Writing; DLP = Digital Light Processing; FDM = Fused Deposition Modeling; FFF = Fused Filament Fabrication; SLA = Stereolithography.
A variety of polymer matrices, such as PLA, PVA, chitosan, ABS, and PCL, have been explored, in combination with functional fillers including zeolites, hydroxyapatite, TiO2, silver nanoparticles, graphene oxide, and other inorganic or carbon-based additives at different loading levels. These material combinations enable the targeting of diverse water contaminants, including ammonia, heavy metal ions, dyes, pharmaceuticals, oils, and pathogenic microorganisms.
There is a growing interest in sustainable, biodegradable, and low-carbon materials for environmental applications. Meanwhile, hydrophilic polymers such as PVA and chitosan are particularly attractive for adsorption- and bio-interaction-driven processes, while photocatalytic or antimicrobial fillers (e.g., TiO2, silver nanoparticles, graphene-based materials) impart additional functionality beyond simple physical filtration. The incorporation of functional fillers demonstrates that additive manufacturing can serve as a versatile platform for tuning surface chemistry, adsorption capacity, antimicrobial activity, and separation performance without compromising geometric complexity or design freedom.
3.4. Practical Implications
The transition of AM-fabricated water treatment modules from laboratory-scale prototypes to industrial systems requires a careful evaluation of operational performance, structural reliability, and environmental impact. Additive manufacturing enables the creation of diverse functional modules, including adsorbents, membranes, porous filters, photocatalytic reactors, ion-exchange units, and bioreactors, each of which benefits from tunable porosity, hierarchical architectures, and tailored material properties. While laboratory studies highlight AM’s capacity for precise control over geometry and functionality, industrial deployment demands attention to continuous flow operation, mechanical stability, reproducibility, and process integration. Material selection, energy consumption, post-processing, and recyclability collectively influence sustainability and cost-effectiveness. Pilot-scale studies incorporating AM modules can help optimize flow dynamics, regeneration cycles, fouling mitigation, and overall process efficiency, providing actionable guidance for practical implementation.
3.4.1. Hydrodynamics
Hydrodynamic performance is critical for all AM-fabricated water treatment systems. Whether for porous adsorbents, flow-through membranes, catalytic reactors, or bioreactors, structures must satisfy constraints related to pressure drop, flow distribution, mass-transfer efficiency, and fouling resistance. AM facilitates geometric innovations that improve fluid dynamics, such as Triply Periodic Minimal Surfaces (TPMS), lattice networks, or hexagonal/honeycomb channels, which are difficult or impossible to produce via conventional fabrication [257,258,259]. Optimized flow enhances mixing, reduces dead zones, and improves contact between water and functional surfaces, benefiting diverse applications from filtration to catalytic degradation.
3.4.2. Fouling and Material Functionality
Fouling, caused by the accumulation of organic, inorganic, or biological matter, can degrade the performance of membranes, filters, and reactor surfaces [260,261,262]. AM allows functionalization of module surfaces, embedding antimicrobial agents, designing superhydrophobic or Slippery Liquid-Infused Porous Surfaces (SLIPS), or creating shear-optimized geometries to mitigate fouling while maintaining high throughput [263,264,265]. These strategies apply broadly across water treatment modules, enhancing performance in microfiltration, ultrafiltration, bioreactors, and other flow-through systems.
3.4.3. Scaling Up
Scaling AM from lab prototypes to industrial modules requires consideration of structural integrity, reproducibility, and integration into process units. Challenges include printer design, material rheology, interlayer bonding, and geometric fidelity [266]. Successful examples, such as the replacement of traditional filter components with 3D-printed modules, illustrate cost and lead-time reductions while demonstrating the importance of performance-driven scale-up. Geometries effective at small scales may require optimization to maintain efficiency, minimize hydraulic resistance, and resist fouling in larger-scale modules [267]. Standardized metrics for volumetric productivity, regeneration robustness, and mechanical stability are essential to bridge the gap from lab-scale demonstrations to full-scale operation.
3.4.4. Environmental and Economic Considerations
AM offers potential environmental advantages through waste reduction, localized manufacturing, and design optimization [268,269,270]. However, life-cycle analyses indicate that benefits depend strongly on production volume, material choice, and design optimization [271,272,273]. For example, AM can outperform conventional manufacturing for small batch production or for modules specifically designed to control AM’s unique capabilities, achieving better “performance-per-environmental-cost” outcomes. Evaluating sustainability and cost-effectiveness must therefore account for the functional performance of the water treatment module, not just production metrics.
3.5. Deployment-Oriented Screening Matrix for AM Techniques in Water Treatment Applications
Recall that Table 1 suggests the potential feasibility of various AM methods for fabricating polymer-filler composites; nevertheless, translating laboratory-scale success to practical water treatment applications requires a deployment-oriented screening framework that considers scalability, material compatibility, and process constraints. Table 5 addresses this need by presenting a decision matrix that evaluates several AM techniques against criteria critical for real-world deployment, namely scalability for high-volume production, suitability for polymer-filler composite formulations, primary operational constraints, and post-processing requirements.
The comparative analysis reveals Material Extrusion as the most deployment-ready technique, offering high scalability and excellent compatibility with polymer-filler systems. Vat Photopolymerization and Binder Jetting follow as moderate-to-high suitability options, though each presents distinct challenges, resin toxicity and filler sedimentation for the former, and the mandatory post-infiltration step for the latter. At the lower end of the suitability spectrum, Directed Energy Deposition and Powder Bed Fusion face significant thermal degradation risks that limit their applicability for polymer-based composites, while Material Jetting remains constrained by high costs and nozzle reliability issues at scale. This screening framework provides researchers with a structured approach for method selection aligned with their target application scale and material system, ultimately supporting more efficient translation from laboratory innovation to deployed water treatment solutions.
Table 5. Deployment-oriented screening matrix for AM techniques in water treatment applications.
|
Deployment Suitability |
AM Techniques |
Scalability |
Suitability for Polymer-Filler Composites |
Primary Constraint |
Post-Processing Requirement |
Best Suited For |
|---|---|---|---|---|---|---|
|
High |
Material Extrusion |
★★★★★ |
★★★★★ |
Nozzle clogging with excessive filler loading; filament consistency |
Minimal—support removal only |
Pilot-to-industrial scale; high filler loadings; complex geometries |
|
Moderate-High |
Vat Photopolymerization |
★★★ |
★★★★★ |
Resin toxicity; filler sedimentation in vat |
High—washing + post-curing required |
High-resolution components; moderate production volumes |
|
Binder Jetting |
★★★★ |
★★★★ |
Limited polymer options; post-infiltration needed |
Moderate—depowdering + infiltration + curing |
||
|
Moderate |
Material Jetting |
★★ |
★★★★★ |
High material cost; nozzle reliability with fillers |
Moderate—support removal + solvent extraction |
Specialized applications; prototype development; surface-functionalized devices |
|
Powder Bed Fusion |
★★★ |
★★★ |
High thermal degradation risk for polymers; material waste |
Moderate—depowdering + surface coating |
||
|
Sheet Lamination |
★★★★ |
★★★ |
Limited geometric complexity; layer delamination risk |
Minimal—trimming only |
||
|
Low |
Directed Energy Deposition |
★ |
★★ |
Polymer incompatibility; thermal degradation |
High—machining often required |
Research only; metal-polymer hybrids; coating applications |
Rating: ★ = very low; ★★= low; ★★★ = moderate; ★★★★ = high; ★★★★★ = very high.
4. Conclusions and Future Perspectives
This review has highlighted the strong potential of sustainable polymer composites fabricated via additive manufacturing (AM) for advanced water treatment applications. By integrating environmentally friendly polymer matrices, bio-based fillers, and/or functional nanomaterials, AM enables the fabrication of customized, high-performance structures with controlled porosity, high surface area, and multifunctionality. Compared with conventional manufacturing routes, AM offers clear advantages in material efficiency, design flexibility, and the ability to tailor structures for specific water treatment functions such as adsorption, catalysis, filtration, and antimicrobial action. However, significant technological gaps remain, particularly concerning the trade-off between printing resolution and material durability. Current additive manufacturing techniques face challenges in achieving the sub-micron precision required for advanced membranes, such as reverse osmosis (RO) and nanofiltration (NF), while using materials that can withstand the harsh chemical and thermal environments typical of industrial water treatment [274]. These processes are prone to structural defects, including void formation and “stair-stepping” [275], nanoparticle agglomeration that creates stress concentrations [276], and thermal instabilities, such as warping and poor molecular interfusion, during large-scale production [277]. Addressing these interrelated challenges is therefore essential to improving the reliability and performance of additively manufactured polymer structures for practical water purification applications.
The widespread adoption of these promising technologies offers exciting opportunities for targeted research and development. Key areas for advancement include expanding the library of durable, water-stable sustainable feedstocks, optimizing composite formulations to balance functionality with printability and strength, and enhancing the energy efficiency of AM processes [278]. Addressing performance consistency, developing strategies for the circularity of multi-material composites, and establishing standardized evaluation frameworks will be crucial to accelerate industrial translation. Furthermore, conducting comprehensive life cycle assessments, particularly for novel hybrid and multi-material systems, will provide the essential data to validate and guide their sustainable development for water treatment applications.
Future research should focus on developing low-energy AM processes, recyclable or reprocessable composite systems, and bio-derived (or waste-based fillers) with minimal environmental impact. Advances in digital design, such as topology optimization, lattice architectures, and gradient porosity, will play an important role in improving treatment efficiency while reducing material usage. For real-world implementation, scaling up requires transitioning from laboratory-scale prototypes to modular, repeatable, and easily maintainable systems compatible with existing treatment infrastructure. By addressing these challenges through interdisciplinary collaboration, additive-manufactured sustainable polymer composites can evolve from promising laboratory concepts into practical, scalable solutions for global water treatment, supporting circular economic principles and long-term environmental sustainability.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this manuscript, the authors used Google Gemini and ChatGPT to improve language clarity and correct grammatical errors. In addition, Google Gemini and Canva were partially used to generate the graphical abstract, Figure 1, Figure 2, Figure 3, and Table 5. Following the use of these tools, the authors reviewed all content and took full responsibility for the accuracy, originality, and integrity of the published work.
Acknowledgement
The authors would like to acknowledge the financial support provided by Universiti Sains Malaysia through the Academic Research Grant (Grant Account: R502-KR-ARU005-0000000598-K134).
Author Contributions
Y.S.O.: Writing original draft and manuscript preparation; W.S.C.: Conceptualization, review and editing.
Ethics Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data sharing is not applicable to this article.
Funding
This work was funded by Universiti Sains Malaysia Academic Research Grant (Geran Penyelidikan Akademik; Grant Number: R502-KR-ARU005-0000000598-K134).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Wei H, Qiu H, Liu J, Li W, Zhao C, Xu H. Evaluation and source identification of water pollution. Ecotoxicol. Environ. Saf. 2025, 289, 117499. DOI:10.1016/j.ecoenv.2024.117499 [Google Scholar]
- Keiser DA, Shapiro JS. US water pollution regulation over the past half century: Burning waters to crystal springs? J. Econ. Perspect. 2019, 33, 51–75. DOI:10.1257/jep.33.4.51 [Google Scholar]
- Tang FHM, Lenzen M, McBratney A, Maggi F. Risk of pesticide pollution at the global scale. Nat. Geosci. 2021, 14, 206–210. DOI:10.1038/s41561-021-00712-5 [Google Scholar]
- Majeed LR, Sharma D, Rautela KS, Kumar M. Sustainable agriculture, aquaculture and phytoremediation through freshwater macrophytes: A comprehensive review of mineral uptake, soil health, and water quality dynamics. Discover Water 2025, 5, 1. DOI:10.1007/s43832-024-00188-5 [Google Scholar]
- Anual ZF. Drinking water quality in Malaysia: A review on its current status. Int. J. Environ. Sci. Nat. Res. 2020, 24. DOI:10.19080/IJESNR.2020.24.556132 [Google Scholar]
- Demirbas A. Agricultural based activated carbons for the removal of dyes from aqueous solutions: A review. J. Hazard. Mater. 2009, 167, 1–9. DOI:10.1016/j.jhazmat.2008.12.114 [Google Scholar]
- Shah HH, Amin M, Pepe F, Mancusi E, Fareed AG. Overview of environmental and economic viability of activated carbons derived from waste biomass for adsorptive water treatment applications. Environ. Sci. Poll. Res. 2023, 32, 19084–19109. DOI:10.1007/s11356-023-30540-6 [Google Scholar]
- Jevremović A, Ranković M, Ležajić AJ, Uskoković-Marković S, Vasiljević BN, Gavrilov N, et al. Regeneration or repurposing of spent pollutant adsorbents in energy-related applications: A sustainable choice? Sustain. Chem. 2025, 6, 28. DOI:10.3390/suschem6030028 [Google Scholar]
- Satyam S, Patra S. The evolving landscape of advanced oxidation processes in wastewater treatment: Challenges and recent innovations. Processes 2025, 13, 987. DOI:10.3390/pr13040987 [Google Scholar]
- Malaeb L, Ayoub GM. Reverse osmosis technology for water treatment: State of the art review. Desalination 2011, 267, 1–8. DOI:10.1016/j.desal.2010.09.001 [Google Scholar]
- Yang Z, Zhou Y, Feng Z, Rui X, Zhang T, Zhang Z. A review on reverse osmosis and nanofiltration membranes for water purification. Polymers 2019, 11, 1252. DOI:10.3390/polym11081252 [Google Scholar]
- Lee KP, Arnot TC, Mattia D. A review of reverse osmosis membrane materials for desalination: Development to date and future potential. J. Memb. Sci. 2011, 370, 1–22. DOI:10.1016/j.memsci.2010.12.036 [Google Scholar]
- Younas F, Mustafa A, Farooqi ZUR, Wang X, Younas S, Mohy-Ud-din W, et al. Current and emerging adsorbent technologies for wastewater treatment: Trends, limitations, and environmental implications. Water 2021, 13, 215. DOI:10.3390/w13020215 [Google Scholar]
- Jatoi AS, Ahmed J, Bhutto AA, Jeyapaul AS. Recent advances and future perspectives of carbon-based nanomaterials for environmental remediation. Braz. J. Chem. Eng. 2024, 42, 151–169. DOI:10.1007/s43153-024-00439-x [Google Scholar]
- Berber MR. Current advances of polymer composites for water treatment and desalination. J. Chem. 2020, 2020, 7608423. DOI:10.1155/2020/7608423 [Google Scholar]
- Amari A, Mohammed Alzahrani F, Mohammedsaleh Katubi K, Salem Alsaiari N, Tahoon MA, Ben Rebah F. Clay-polymer nanocomposites: Preparations and utilization for pollutants removal. Materials 2021, 14, 1365. DOI:10.3390/ma14061365 [Google Scholar]
- Subramani R, Mustafa MA, Ghadir GK, Al-Tmimi HM, Alani ZK, Haridas D, et al. Advancements in 3D printing materials: A comparative analysis of performance and applications. Appl. Chem. Eng. 2024, 7, 3867. DOI:10.59429/ace.v7i2.3867 [Google Scholar]
- Anbarasu K, Devarajan Y. Nanomaterials-based wastewater treatment: Addressing challenges and advancing sustainable solutions. BioNanoScience 2025, 15, 149. DOI:10.1007/s12668-024-01780-8 [Google Scholar]
- Wang D, Ling D, Lin W, Wang K, Feng Y, Liu L, et al. Potential and challenges of 3D printing technology in membrane-based water treatment research. ACS ES&T Water 2024, 4, 3619–3622. DOI:10.1021/acsestwater.4c00616 [Google Scholar]
- Cheng HN, Gross RA. Sustainability and green polymer chemistry-an overview. In ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2020; pp. 1–11. DOI:10.1021/bk-2020-1372.ch001 [Google Scholar]
- Tarazona NA, Machatschek R, Balcucho J, Castro-Mayorga JL, Saldarriaga JF, Lendlein A. Opportunities and challenges for integrating the development of sustainable polymer materials within an international circular (bio)economy concept. MRS Energy Sustain. 2022, 9, 28–34. DOI:10.1557/s43581-021-00015-7 [Google Scholar]
- Nguyen ND, Ashraf I, Kim W. Compact model for 3d printer energy estimation and practical energy-saving strategy. Electronics 2021, 10, 483. DOI:10.3390/electronics10040483 [Google Scholar]
- Harding OJ, Griffiths CA, Rees A, Pletsas D. Methods to reduce energy and polymer consumption for fused filament fabrication 3D printing. Polymers 2023, 15, 1874. DOI:10.3390/polym15081874 [Google Scholar]
- Safeer S, Pandey RP, Rehman B, Safdar T, Ahmad I, Hasan SW, et al. A review of artificial intelligence in water purification and wastewater treatment: Recent advancements. J. Water Process Eng. 2022, 49, 102974. DOI:10.1016/j.jwpe.2022.102974 [Google Scholar]
- Fijoł N, Aguilar-Sánchez A, Ruiz-Caldas MX, Redlinger-Pohn J, Mautner A, Mathew AP. 3D printed polylactic acid (PLA) filters reinforced with polysaccharide nanofibers for metal ions capture and microplastics separation from water. Chem. Eng. J. 2023, 457, 141153. DOI:10.1016/j.cej.2022.141153 [Google Scholar]
- Park SS, Lee YS, Lee SW, Repo E, Kim TH, Park Y, et al. Facile surface treatment of 3D-printed PLA filter for enhanced graphene oxide doping and effective removal of cationic dyes. Polymers 2023, 15, 269. DOI:10.3390/polym15020269 [Google Scholar]
- Khaldoun K, Khizar S, Saidi-Besbes S, Zine N, Errachid A, Elaissari A. Synthesis of silver nanoparticles as an antimicrobial mediator. J. Umm Al-Qura Univ. Appl. Sci. 2025, 11, 274–293. DOI:10.1007/s43994-024-00159-5 [Google Scholar]
- Arenas García MA, Hidouri S, Hao X, de Medeiros Dantas JM, Dorval Courchesne NM. Biological synthesis of reusable silver nanoparticle-protein antimicrobial films. Adv. Mater. Interfaces 2025, 12, 2400649. DOI:10.1002/admi.202400649 [Google Scholar]
- Nayak MR, Kamble RR, Nadoni VB, Shettar AK, Hoskeri JH, Keri RS. Adroit green synthesis of silver nanoparticles: Multivariate photocatalytic degradation of Alizarin Yellow R dye and biomedical applications. Appl. Organomet. Chem. 2025, 39, e70397. DOI:10.1002/aoc.70397 [Google Scholar]
- Hafizza Mohd Amran N, Khairul Amri Kamarudin M, Abd Wahab N, Hafiz Md Saad M, Hafiz Rosli M, Armi Abu Samah M, et al. Environmental management on natural river based on water quality deterioration in Terengganu River Basin, Terengganu, Malaysia. Int. J. Eng. Technol. 2018, 7, 132–136. DOI:10.14419/ijet.v7i4.34.23844 [Google Scholar]
- Zhou L, Miller J, Vezza J, Mayster M, Raffay M, Justice Q, et al. Additive manufacturing: A comprehensive review. Sensors 2024, 24, 2668. DOI:10.3390/s24092668 [Google Scholar]
- Jung S, Kara LB, Nie Z, Simpson TW, Whitefoot KS. Is additive manufacturing an environmentally and economically preferred alternative for mass production? Environ. Sci. Technol. 2023, 57, 6373–6386. DOI:10.1021/acs.est.2c04927 [Google Scholar]
- Ma T, Zhang Y, Ruan K, Guo H, He M, Shi X, et al. Advances in 3D printing for polymer composites: A review. InfoMat 2024, 6, e12568. DOI:10.1002/inf2.12568 [Google Scholar]
- Wu Y, Tang C, Si P, Zhang D. 3D coaxial printing of porous construct stacked with hollow filaments for heavy metal removal. Sci. Total Environ. 2023, 887, 164120. DOI:10.1016/j.scitotenv.2023.164120 [Google Scholar]
- Kim K, Ratri MC, Choe G, Nam M, Cho D, Shin K. Three-dimensional, printed water-filtration system for economical, on-site arsenic removal. PLoS ONE 2020, 15, e0231475. DOI:10.1371/journal.pone.0231475 [Google Scholar]
- Ibrahim Y, Hilal N. Enhancing ultrafiltration membrane permeability and antifouling performance through surface patterning with features resembling feed spacers. Clean Water 2023, 6, 60. DOI:10.1038/s41545-023-00277-3 [Google Scholar]
- Zhang D, Xiao J, Guo Q, Yang J. 3D-printed highly porous and reusable chitosan monoliths for Cu(II) removal. J. Mater. Sci. 2019, 54, 6728–6741. DOI:10.1007/s10853-019-03332-y [Google Scholar]
- Yuan S, Li S, Zhu J, Tang Y. Additive manufacturing of polymeric composites from material processing to structural design. Compos. Part B Eng. 2021, 219, 108903. DOI:10.1016/j.compositesb.2021.108903 [Google Scholar]
- Li E, Yan W. Binder-powder interactions in binder jetting: Binder drying, layer shifting, and inter-layer binding. Addit. Manuf. 2025, 110, 104951. DOI:10.1016/j.addma.2025.104951 [Google Scholar]
- Garzón EO, Alves JL, Neto RJL. Post-process influence of infiltration on binder jetting technology. In Materials Design and Applications; Silva LFM, Ed.; Springer: Cham, Switzerland, 2017; pp. 233–255. DOI:10.1007/978-3-319-50784-2_19 [Google Scholar]
- Shen X, Chu M, Hariri F, Vedula G, Naguib HE. Binder jetting fabrication of highly flexible and electrically conductive graphene/PVOH composites. Addit. Manuf. 2020, 36, 101565. DOI:10.1016/j.addma.2020.101565 [Google Scholar]
- Meenashisundaram GK, Xu Z, Nai MLS, Lu S, Ten JS, Wei J. Binder jetting additive manufacturing of high porosity 316L stainless steel metal foams. Materials 2020, 13, 3744. DOI:10.3390/ma13173744 [Google Scholar]
- Wu BM, Borland SW, Giordano RA, Cima LG, Sachs EM, Cima MJ. Solid free-form fabrication of drug delivery devices. J. Control. Release 1996, 40, 77–87. DOI:10.1016/0168-3659(95)00173-5 [Google Scholar]
- Ahn DG. Directed energy deposition (DED) process: State of the art. Int. J. Precis. Eng. Manuf.-Green Tech. 2021, 8, 703–742. DOI:10.1007/s40684-020-00302-7 [Google Scholar]
- Svetlizky D, Das M, Zheng B, Vyatskikh AL, Bose S, Bandyopadhyay A, et al. Directed energy deposition (DED) additive manufacturing: Physical characteristics, defects, challenges and applications. Mater. Today 2021, 49, 271–295. DOI:10.1016/j.mattod.2021.03.020 [Google Scholar]
- Zhang N, Wang Z, Zhao Z, Zhang D, Feng J, Yu L, et al. 3D printing of micro-nano devices and their applications. Microsyst. Nanoeng. 2025, 11, 35. DOI:10.1038/s41378-024-00812-3 [Google Scholar]
- Zhao K, Ye Z, Su Z, Cao W, Shi D, Hao X, et al. A diffusion-controlled kinetic model for binder burnout in a green part fabricated by binder jetting based on the thermal decomposition kinetics of TEG-DMA. Addit. Manuf. 2025, 105, 104793. DOI:10.1016/j.addma.2025.104793 [Google Scholar]
- Ligon SC, Liska R, Stampfl J, Gurr M, Mülhaupt R. Polymers for 3D printing and customized additive manufacturing. Chem. Rev. 2017, 117, 10212–10290. DOI:10.1021/acs.chemrev.7b00074 [Google Scholar]
- Hüner B, Kıstı M, Uysal S, Uzgören İN, Özdoğan E, Süzen YO, et al. An overview of various additive manufacturing technologies and materials for electrochemical energy conversion applications. ACS Omega 2022, 7, 40638–40658. DOI:10.1021/acsomega.2c05096 [Google Scholar]
- Kutlu Y, Wencke YL, Luinstra GA, Esen C, Ostendorf A. Directed energy deposition of PA12 carbon nanotube composite powder using a fiber laser. Procedia CIRP 2020, 94, 128–133. DOI:10.1016/j.procir.2020.09.025 [Google Scholar]
- Wittmann A, Chamandani SA, Hentschel O, Sommereyns A, Kopp SP, Schmidt M. Functional polyetheretherketone coatings fabricated by laser-based directed energy deposition. Surf. Coat. Technol. 2025, 496, 131629. DOI:10.1016/j.surfcoat.2024.131629 [Google Scholar]
- Chadha U, Selvaraj SK, Lamsal AS, Maddini Y, Ravinuthala AK, Choudhary B, et al. Directed energy deposition via artificial intelligence-enabled approaches. Complexity 2022, 2022. DOI:10.1155/2022/2767371 [Google Scholar]
- Xiao J, Zhang D, Ma Z, Chen L, Sun Y, Jia P, et al. Vat photopolymerization 3D printing application in electronics. In Vat Photopolymerization Additive Manufacturing: 3D Printing Processes, Materials, and Applications; Wang X, Ed.; Elsevier: Amsterdam, The Netherlands, 2024; pp. 365–405. DOI:10.1016/B978-0-443-15487-4.00012-1 [Google Scholar]
- Chartrain NA, Williams CB, Whittington AR. A review on fabricating tissue scaffolds using vat photopolymerization. Acta Biomater. 2018, 74, 90–111. DOI:10.1016/j.actbio.2018.05.010 [Google Scholar]
- Al Rashid A, Ahmed W, Khalid MY, Koç M. Vat photopolymerization of polymers and polymer composites: Processes and applications. Addit. Manuf. 2021, 47, 102279. DOI:10.1016/j.addma.2021.102279 [Google Scholar]
- Pagac M, Hajnys J, Ma QP, Jancar L, Jansa J, Stefek P, et al. A review of vat photopolymerization technology: Materials, applications, challenges, and future trends of 3D printing. Polymers 2021, 13, 598. DOI:10.3390/polym13040598 [Google Scholar]
- Khalid M, Peng Q. Sustainability and environmental impact of additive manufacturing: A literature review. Comput. Aided Des. Appl. 2021, 18, 1210–1232. DOI:10.14733/cadaps.2021.1210-1232 [Google Scholar]
- Hoskins JK, Zou M. 3D Printing of High-Porosity Membranes with Submicron Pores for Microfluidics. Nanomanufacturing 2024, 4, 120–137. DOI:10.3390/nanomanufacturing4030009 [Google Scholar]
- Abdalla H, Fattah KP, Abdallah M, Tamimi AK. Environmental footprint and economics of a full-scale 3D-printed house. Sustainability 2021, 13, 11978. DOI:10.3390/su132111978 [Google Scholar]
- Saadi MASR, Maguire A, Pottackal NT, Thakur MSH, Ikram MM, Hart AJ, et al. Direct ink writing: A 3D printing technology for diverse mMaterials. Adv. Mater. 2022, 34, 2108855. DOI:10.1002/adma.202108855 [Google Scholar]
- Neumann TV, Dickey MD. Liquid metal direct write and 3D printing: A review. Adv. Mater. Technol. 2020, 5. DOI:10.1002/admt.202000070 [Google Scholar]
- Warke S, Puranik VS. Comparison of energy consumption of ABS and PLA while 3 D printing with fused deposition modeling process. Mater. Today Proc. 2022, 66, 2098–2103. DOI:10.1016/j.matpr.2022.05.509 [Google Scholar]
- Balogun VA, Kirkwood N, Mativenga PT. Energy consumption and carbon footprint analysis of fused deposition modelling: A case study of RP Stratasys Dimension SST FDM. Int. J. Sci. Eng. Res. 2015, 6, 442–447. [Google Scholar]
- Rocha VG, Saiz E, Tirichenko IS, García-Tuñón E. Direct ink writing advances in multi-material structures for a sustainable future. J. Mater. Chem. A 2020, 8, 15646–15657. DOI:10.1039/D0TA04181E [Google Scholar]
- Roy Barman S, Gavit P, Chowdhury S, Chatterjee K, Nain A. 3D-Printed Materials for Wastewater Treatment. JACS Au 2023, 3, 2930–2947. DOI:10.1021/jacsau.3c00409 [Google Scholar]
- Udroiu R, Braga IC. Polyjet technology applications for rapid tooling. In Proceedings of the 21st Innovative Manufacturing Engineering & Energy International Conference (IManE&E 2017), Iaşi, Romania, 24–27 May 2017. DOI:10.1051/matecconf/201711203011 [Google Scholar]
- Walczak R, Adamski K, Lizanets D. Inkjet 3D printed check microvalve. J. Micromech. Microeng. 2017, 27, 47002. DOI:10.1088/1361-6439/aa6152 [Google Scholar]
- Gülcan O, Günaydın K, Tamer A. The state of the art of material jetting—A critical review. Polymers 2021, 13, 2829. DOI:10.3390/polym13162829 [Google Scholar]
- Elkaseer A, Chen KJ, Janhsen JC, Refle O, Hagenmeyer V, Scholz SG. Material jetting for advanced applications: A state-of-the-art review, gaps and future directions. Addit. Manuf. 2022, 60, 103270. DOI:10.1016/j.addma.2022.103270 [Google Scholar]
- Jabari E, Liravi F, Davoodi E, Lin L, Toyserkani E. High speed 3D material-jetting additive manufacturing of viscous graphene-based ink with high electrical conductivity. Addit. Manuf. 2020, 35, 101330. DOI:10.1016/j.addma.2020.101330 [Google Scholar]
- Balavandy SK, Li F, Maya F, Breadmore MC. 3D printed monolithic microfluidic device with an integrated porous filter: Application to pH measurement of water. Talanta Open 2024, 9, 100280. DOI:10.1016/j.talo.2023.100280 [Google Scholar]
- Keshan Balavandy S, Li F, Macdonald NP, Maya F, Townsend AT, Frederick K, et al. Scalable 3D printing method for the manufacture of single-material fluidic devices with integrated filter for point of collection colourimetric analysis. Anal. Chim. Acta 2021, 1151, 238101. DOI:10.1016/j.aca.2020.11.033 [Google Scholar]
- Proano-Pena G, Carrano AL, Blersch DM. Analysis of very-high surface area 3D-printed media in a moving bed biofilm reactor for wastewater treatment. PLoS ONE 2020, 15, e0238386. DOI:10.1371/journal.pone.0238386 [Google Scholar]
- Joshua RJN, Raj SA, Hameed Sultan MT, Łukaszewicz A, Józwik J, Oksiuta Z, et al. Powder bed fusion 3D printing in precision manufacturing for biomedical aApplications: A comprehensive review. Materials 2024, 17, 769. DOI:10.3390/ma17030769 [Google Scholar]
- Dzogbewu TC, de Beer D. Powder bed fusion of multimaterials. J. Manuf. Mater. Process. 2023, 7, 15. DOI:10.3390/jmmp7010015 [Google Scholar]
- Park S, Deng K, Fu KK. Additive manufacturing including laser-based manufacturing. In Sustainable Manufacturing Processes; Narayanan RG, Gunasekera JS, Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 285–311. DOI:10.1016/B978-0-323-99990-8.00010-2 [Google Scholar]
- Adamov I, Stanojević G, Pavlović SM, Medarević D, Ivković B, Kočović D, et al. Powder bed fusion–laser beam (PBF-LB) three-dimensional (3D) printing: Influence of laser hatching distance on the properties of zolpidem tartrate tablets. Int J. Pharm. 2024, 657, 124161. DOI:10.1016/j.ijpharm.2024.124161 [Google Scholar]
- Lahtinen E. Chemically Functional 3D Printing: Selective Laser Sintering of Customizable Metal Scavengers. Doctoral Dissertation, University of Jyväskylä, Jyväskylä, Finland, 20 December 2019. [Google Scholar]
- Bazan A, Turek P, Zakręcki A. Influence of antibacterial coating and mechanical and chemical treatment on the surface properties of PA12 parts manufactured with SLS and MJF techniques in the context of medical applications. Materials 2023, 16, 2405. DOI:10.3390/ma16062405 [Google Scholar]
- Priarone PC, Lunetto V, Atzeni E, Salmi A. Laser powder bed fusion (L-PBF) additive manufacturing: On the correlation between design choices and process sustainability. Procedia CIRP 2018, 67, 85–90. DOI:10.1016/j.procir.2018.09.058 [Google Scholar]
- Anwar T, Lopes AC, Silva EC, Mould ST, Sampaio AM, Pontes AJ. Cradle-to-gate life cycle assessment: A comparison of polymer and metal-based powder bed fusion for the production of a robot end-effector with internal conformal channels. Prog. Addit. Manuf. 2025, 10, 561–579. DOI:10.1007/s40964-024-00640-x [Google Scholar]
- Tan LJ, Zhu W, Zhou K. Recent progress on polymer materials for additive manufacturing. Adv. Funct. Mater. 2020, 30, 2003062. DOI:10.1002/adfm.202003062 [Google Scholar]
- Dermeik B, Travitzky N. Laminated object manufacturing of ceramic-based materials. Adv. Eng. Mater. 2020, 22, 2000256. DOI:10.1002/adem.202000256 [Google Scholar]
- Sahayaraj AF, Dhamotharan S, Sandeep D, Ramachandran P, Jenish I, Divakaran D et al. Sustainable smart polymer composite materials: A comprehensive review. In Proceedings of the 2023 Research, Invention, and Innovation Congress (RI2C 2023), Bangkok (Online), Thailand, 24–25 August 2023. DOI:10.1051/e3sconf/202342802014 [Google Scholar]
- Mohanty AK, Wu F, Mincheva R, Hakkarainen M, Raquez JM, Mielewski DF, et al. 3 Sustainable polymers. Nat. Rev. Methods Primers 2022, 2, 46. DOI:10.1038/s43586-022-00124-8 [Google Scholar]
- Bukvić M, Milojević S, Gajević S, Đorđević M, Stojanović B. Production technologies and application of polymer composites in engineering: A review. Polymers 2025, 17, 2187. DOI:10.3390/polym17162187 [Google Scholar]
- Mora P, Rimdusit S, Jubsilp C. Development, characteristics, and potential applications of green composites from bio-derived polybenzoxazine and glass fiber. Polym. Compos. 2025, 46, 9319–9333. DOI:10.1002/pc.29558 [Google Scholar]
- Jaspal D, Malviya A. Composites for wastewater purification: A review. Chemosphere 2020, 246, 125788. DOI:10.1016/j.chemosphere.2019.125788 [Google Scholar]
- Divya S, Oh TH. Polymer nanocomposite membrane for wastewater treatment: A critical review. Polymers 2022, 14, 1732. DOI:10.3390/polym14091732 [Google Scholar]
- Nguyen-Tri P, Nguyen TA, Carriere P, Ngo Xuan C. Nanocomposite coatings: Preparation, characterization, properties, and applications. Int. J. Corros. 2018, 2018, 4749501. DOI:10.1155/2018/4749501 [Google Scholar]
- Dongre RS, Sadasivuni KK, Deshmukh K, Mehta A, Basu S, Meshram JS, et al. Natural polymer based composite membranes for water purification: A review. Polym.-Plast. Technol. Mater. 2019, 58, 1295–1310. DOI:10.1080/25740881.2018.1563116 [Google Scholar]
- Chu L, Wang J. Denitrification of groundwater using PHBV blends in packed bed reactors and the microbial diversity. Chemosphere 2016, 155, 463–470. DOI:10.1016/j.chemosphere.2016.04.090 [Google Scholar]
- Luo G, Hou Z, Tian L, Tan H. Comparison of nitrate-removal efficiency and bacterial properties using PCL and PHBV polymers as a carbon source to treat aquaculture water. Aquac. Fish. 2020, 5, 92–98. https://doi.org/10.1016/j.aaf.2019.04.002 [Google Scholar]
- Rossi MM, Alfano S, Amanat N, Andreini F, Lorini L, Martinelli A, et al. A Polyhydroxybutyrate (PHB)-biochar reactor for the adsorption and biodegradation of trichloroethylene: Design and startup phase. Bioengineering 2022, 9, 192. DOI:10.3390/bioengineering9050192 [Google Scholar]
- Saleem H, Zaidi SJ. Developments in the application of nanomaterials for water treatment and their impact on the environment. Nanomaterials 2020, 10, 1764. DOI:10.3390/nano10091764 [Google Scholar]
- Jani M, Arcos-Pareja JA, Ni M. Engineered zero-dimensional fullerene/carbon dots-polymer based nanocomposite membranes for wastewater treatment. Molecules 2020, 25, 4934. DOI:10.3390/molecules25214934 [Google Scholar]
- Areche FO, Mamani CMC, Cárdenas JAL, Sumarriva-Bustinza LA, Pastrana PAP, Porras-Roque MS, et al. A comprehensive review on monitoring and purification of water through tunable 2D nanomaterials. Braz. J. Biol. 2023, 83, e273843. DOI:10.1590/1519-6984.273843 [Google Scholar]
- Sinha V, Chakma S. Advances in the preparation of hydrogel for wastewater treatment: A concise review. J. Environ. Chem. Eng. 2019, 7, 103295. DOI:10.1016/j.jece.2019.103295 [Google Scholar]
- Silva EC, Soares VR, Nörnberg AB, Fajardo AR. Recyclable 3D-printed composite hydrogel containing rice husk biochar for organic contaminants adsorption in tap water. ACS Appl. Polym. Mater. 2023, 5, 8415–8429. DOI:10.1021/acsapm.3c01534 [Google Scholar]
- Miller DJ, Dreyer DR, Bielawski CW, Paul DR, Freeman BD. Surface modification of water purification membranes. Angew. Chem. Int. Ed. 2016, 56, 4662–4711. DOI:10.1002/anie.201601509 [Google Scholar]
- Bergaliyeva S, Sales DL, Delgado FJ, Bolegenova S, Molina SI. Manufacture and characterization of polylactic acid filaments recycled from real waste for 3D printing. Polymers 2023, 15, 2165. DOI:10.3390/polym15092165 [Google Scholar]
- da Silva D, Kaduri M, Poley M, Adir O, Krinsky N, Shainsky-Roitman J, et al. Biocompatibility, biodegradation and excretion of polylactic acid (PLA) in medical implants and theranostic systems. Chem. Eng. J. 2018, 340, 9–14. DOI:10.1016/j.cej.2018.01.010 [Google Scholar]
- García H, López Pola T. Health and safety in 3D printing. Int. J. Occup. Environ. Saf. 2022, 6, 14–25. DOI:10.24840/2184-0954_006.001_0003 [Google Scholar]
- Joseph TM, Kallingal A, Suresh AM, Mahapatra DK, Hasanin MS, Haponiuk J, et al. 3D printing of polylactic acid: Recent advances and opportunities. Int. J. Adv. Manuf. Technol. 2023, 125, 1015–1035. DOI:10.1007/s00170-022-10795-y [Google Scholar]
- Fijoł N, Abdelhamid HN, Pillai B, Hall SA, Thomas N, Mathew AP. 3D-printed monolithic biofilters based on a polylactic acid (PLA)-hydroxyapatite (HAp) composite for heavy metal removal from an aqueous medium. RSC Adv. 2021, 11, 32408–32418. DOI:10.1039/D1RA05202K [Google Scholar]
- Baran EH, Erbil HY. Surface modification of 3D printed PLA objects by fused deposition modeling: A review. Colloids Interfaces 2019, 3, 43. DOI:10.3390/colloids3020043 [Google Scholar]
- Panaksri A, Tanadchangsaeng N. Polyhydroxyalkanoate biopolyesters as extracellular matrix scaffolds by 3D printing technology. Polym. Int. 2025, 74, 874–887. DOI:10.1002/pi.6740 [Google Scholar]
- Kovalcik A. Recent Advances in 3D printing of polyhydroxyalkanoates: A review. Eurobiotech J. 2021, 5, 48–55. DOI:10.2478/ebtj-2021-0008 [Google Scholar]
- Jayalath SU, de Alwis AP. PHA, the greenest plastic so far: Advancing microbial synthesis, recovery, and sustainable applications for circularity. ACS Omega 2025, 10, 32564–32586. DOI:10.1021/acsomega.5c00684 [Google Scholar]
- Gautam S, Gautam A, Pawaday J, Kanzariya RK, Yao Z. Current status and challenges in the commercial production of polyhydroxyalkanoate-based bioplastic: A review. Processes 2024, 12, 1720. DOI:10.3390/pr12081720 [Google Scholar]
- Meereboer KW, Misra M, Mohanty AK. Review of recent advances in the biodegradability of polyhydroxyalkanoate (PHA) bioplastics and their composites. Green Chem. 2020, 22, 5519–5558. DOI:10.1039/D0GC01647K [Google Scholar]
- Koller M, Heeney D, Mukherjee A. Biodegradability of polyhydroxyalkanoate (PHA) biopolyesters in nature: A review. Biodegradation 2025, 36, 76. DOI:10.1007/s10532-025-10164-y [Google Scholar]
- Bankole MT, Abdulkareem AS, Mohammed IA, Ochigbo SS, Tijani JO, Abubakre OK, et al. Selected heavy metals removal from electroplating wastewater by purified and polyhydroxylbutyrate functionalized carbon nanotubes adsorbents. Sci. Rep. 2019, 9, 4475. . DOI:10.1038/s41598-018-37899-4 [Google Scholar]
- Tejada-Tovar C, Villabona-Ortíz Á, Toro-Madrid O, Ortega-Toro R, Bonilla Mancilla H. Cellulose acetate–PHB biocomposite from Saccharum Officinarum for Ni (II) Adsorption: Equilibrium and kinetics. J. Compos. Sci. 2025, 9, 376. DOI:10.3390/jcs9070376 [Google Scholar]
- Choonut A, Reungsang A. Poly[hydroxybutyrate-co-hydroxyvalerate] and cellulose biocomposite membrane derived from sugarcane bagasse for Congo red dye and vegetable oil removal in water remediation. PLoS ONE 2025, 20, e0336293. DOI:10.1371/journal.pone.0336293 [Google Scholar]
- Sen Gupta R, Samantaray PK, Bose S. Going beyond cellulose and chitosan: Synthetic biodegradable membranes for drinking water, wastewater, and oil-water remediation. ACS Omega 2023, 8, 24695–24717. DOI:10.1021/acsomega.3c01699 [Google Scholar]
- Manyatshe A, Sibali LL. A review on the recovery of cellulose, lignin, and hemicellulose biopolymers from the same source of lignocellulosic biomass—Methodology, characterization and applications. J. Water Process Eng. 2025, 70, 107037. DOI:10.1016/j.jwpe.2025.107037 [Google Scholar]
- Shaghaleh H, Xu X, Wang S. Current progress in production of biopolymeric materials based on cellulose, cellulose nanofibers, and cellulose derivatives. RSC Adv. 2018, 8, 825–842. DOI:10.1039/C7RA11157F [Google Scholar]
- Etale A, Onyianta AJ, Turner SR, Eichhorn SJ. Cellulose: A review of water interactions, applications in composites, and water treatment. Chem. Rev. 2023, 123, 2016–2048. DOI:10.1021/acs.chemrev.2c00477 [Google Scholar]
- Akhter F, Pinjaro MA, Ahmed J, Ahmed M, Arain HJ, Ahsan MJ, et al. Recent advances and synthesis approaches for enhanced heavy metal adsorption from wastewater by silica-based and nanocellulose-based 3D structured aerogels: A state of the art review with adsorption mechanisms and prospects. Biomass Convers. Biorefin. 2025, 15, 6585–6614. DOI:10.1007/s13399-024-05469-6 [Google Scholar]
- Suleimenova M, Tabynbayeva A, Toshtay K, Tauanov Z. Composite membranes based on MXene and nanocellulose for water purification: Structure, efficiency, and future prospects. Membranes 2025, 15, 293. DOI:10.3390/membranes15100293 [Google Scholar]
- Wei T, Zhu Y, Duan G, Han J, Han X, Zhang C, et al. Recent advances in nanocellulose-derived materials for dyes adsorption: A review. Int. J. Biol. Macromol. 2025, 306, 141770. DOI:10.1016/j.ijbiomac.2025.141770 [Google Scholar]
- Yan C, Luo J, Huang C, Liu L, Sun S, Zhou X. Fabrication of nanocellulose-based high-mechanical and super-hydrophobic xerogels for speedy oil absorbents. Carbohydr. Polym. 2025, 351, 123097. DOI:10.1016/j.carbpol.2024.123097 [Google Scholar]
- Shojaeiarani J, Bajwa DS, Chanda S. Cellulose nanocrystal based composites: A review. Compos. Part C Open Access 2021, 5, 100164. DOI:10.1016/j.jcomc.2021.100164 [Google Scholar]
- Das R, Lindström T, Sharma PR, Chi K, Hsiao BS. Nanocellulose for sustainable water purification. Chem. Rev. 2022, 122, 8936–9031. DOI:10.1021/acs.chemrev.1c00683 [Google Scholar]
- Robaina M, Murillo K, Rocha E, Villar J. Circular economy in plastic waste—Efficiency analysis of European countries. Sci. Total Environ. 2020, 730, 139038. DOI:10.1016/j.scitotenv.2020.139038 [Google Scholar]
- Pongsathit S, Chanthot P, Rwei SP, Pattamaprom C. Upcycling waste polyethylene terephthalate bottles into eco-friendly filaments for material extrusion 3D printing. Eng. Sci. 2025, 35, 1495. DOI:10.30919/es1495 [Google Scholar]
- Hasan MR, Davies IJ, Paramanik A, John M, Biswas WK. Fabrication and characterisation of sustainable 3D-printed parts using post-consumer PLA plastic and virgin PLA blends. Processes 2024, 12, 760. DOI:10.3390/pr12040760 [Google Scholar]
- Ibrahim I, Ashour AG, Zeiada W, Salem N, Abdallah M. A systematic review on the technical performance and sustainability of 3D printing filaments using recycled plastic. Sustainability 2024, 16, 8247. DOI:10.3390/su16188247 [Google Scholar]
- Latos-Brozio M, Rułka K, Masek A. Review of bio-fillers dedicated to polymer compositions. Chem. Biodivers. 2025, 22, e202500406. DOI:10.1002/cbdv.202500406 [Google Scholar]
- Divakaran D, Suyambulingam I, Somasundaram R, Gehitha TR, Rangappa SM, Siengchin S. Futuristic prospects of bio-based fillers for industrial application. In Sustainable Fillers/Plasticizers for Polymer Composites: Promising Resources; Suyambulingam I, Divakaran D, Rangappa SM, Siengchin S, Eds.; Elsevier: Amsterdam, The Netherlands, 2024; pp. 491–514. DOI:10.1016/B978-0-443-15630-4.00020-8 [Google Scholar]
- Cao L, Zhang C, Chen H, Tsang DCW, Luo G, Zhang S, et al. Hydrothermal liquefaction of agricultural and forestry wastes: State-of-the-art review and future prospects. Bioresour. Technol. 2017, 245, 1184–1193.DOI:10.1016/j.biortech.2017.08.196 [Google Scholar]
- Cao Y, Chen SS, Zhang S, Ok YS, Matsagar BM, Wu KCW, et al. Advances in lignin valorization towards bio-based chemicals and fuels: Lignin biorefinery. Bioresour. Technol. 2019, 291, 121878. DOI:10.1016/j.biortech.2019.121878 [Google Scholar]
- Ridho MR, Agustiany EA, Rahmi Dn M, Madyaratri EW, Ghozali M, Restu WK, et al. Lignin as green filler in polymer composites: Development methods, characteristics, and potential applications. Adv. Mater. Sci. Eng. 2022, 2022, 1363481. DOI:10.1155/2022/1363481 [Google Scholar]
- Wang B, Sun D, Yuan TQ, Song G, Sun RC. Recent advances in lignin modification and its application in wastewater treatment. In Lignin Utilization Strategies: From Processing to Applications; Yoo CG, Ragauskas A, Eds.; American Chemical Society: Washington, DC, USA, 2021; Volume 1377, pp. 143–173. DOI:10.1021/bk-2021-1377.ch007 [Google Scholar]
- Wenger J, Haas V, Stern T. Why can we make anything from lignin except money? Towards a broader economic perspective in lignin research. Curr. For. Rep. 2020, 6, 294–308. DOI:10.1007/s40725-020-00126-3 [Google Scholar]
- Keshvardoostchokami M, Majidi M, Zamani A, Liu B. A review on the use of chitosan and chitosan derivatives as the bio-adsorbents for the water treatment: Removal of nitrogen-containing pollutants. Carbohydr. Polym. 2021, 273, 118625. DOI:10.1016/j.carbpol.2021.118625 [Google Scholar]
- Iber BT, Kasan NA, Torsabo D, Omuwa JW. A review of various sources of chitin and chitosan in nature. J. Renew. Mater. 2022, 10, 1097–1123. DOI:10.32604/jrm.2022.018142 [Google Scholar]
- Fila K, Podkościelna B, Szymczyk K. The application of chitosan as an eco-filler of polymeric composites. Adsorption 2024, 30, 157–165. DOI:10.1007/s10450-023-00403-0 [Google Scholar]
- Mohan K, Karthick Rajan D, Rajarajeswaran J, Divya D, Ramu Ganesan A. Recent trends on chitosan based hybrid materials for wastewater treatment: A review. Curr. Opin. Environ. Sci. Health 2023, 33, 100473. DOI:10.1016/j.coesh.2023.100473 [Google Scholar]
- Li VCF, Kuang X, Mulyadi A, Hamel CM, Deng Y, Qi HJ. 3D printed cellulose nanocrystal composites through digital light processing. Cellulose 2019, 26, 3973–3985. DOI:10.1007/s10570-019-02353-9 [Google Scholar]
- Lourenço AF, Gamelas JAF, Sarmento P, Ferreira PJT. Cellulose micro and nanofibrils as coating agent for improved printability in office papers. Cellulose 2020, 27, 6001–6010. DOI:10.1007/s10570-020-03184-9 [Google Scholar]
- Kelly PV, Shams Es-Haghi S, Ahmad AAL, Lamm ME, Copenhaver K, Alyamac-Seydibeyoglu E, et al. High-strength 3D printed poly(lactic acid) composites reinforced by shear-aligned polymer-grafted cellulose nanofibrils. RSC Appl. Polym. 2024, 3, 111–124. DOI:10.1039/D4LP00283K [Google Scholar]
- Müller M, Urban J, Svobodová J, Mishra RK. Modification of the mechanical properties of photosensitive resin by using biobased fillers during stereolithography (SLA) 3D printing. Materials 2025, 18, 2699. DOI:10.3390/ma18122699 [Google Scholar]
- Aziz T, Farid A, Haq F, Kiran M, Ullah A, Zhang K, et al. A review on the modification of cellulose and its applications. Polymers 2022, 14, 3206. DOI:10.3390/polym14153206 [Google Scholar]
- Wang J, Wang S. Preparation, modification and environmental application of biochar: A review. J. Clean. Prod. 2019, 227, 1002–1022. DOI:10.1016/j.jclepro.2019.04.282 [Google Scholar]
- Xiang W, Zhang X, Chen J, Zou W, He F, Hu X, et al. Biochar technology in wastewater treatment: A critical review. Chemosphere 2020, 252, 126539. DOI:10.1016/j.chemosphere.2020.126539 [Google Scholar]
- Xie Y, Wang L, Li H, Westholm LJ, Carvalho L, Thorin E, et al. A critical review on production, modification and utilization of biochar. J. Anal. Appl. Pyrolysis 2022, 161, 105405. DOI:10.1016/j.jaap.2021.105405 [Google Scholar]
- Hao P, Fu H, Ma S, Xue W, Xiong S, Li G, et al. MgO-embedded S-doped porous biochar composites for efficient removal Cd(II) and Pb(II) in water: DFT studies and mechanistic insights. Sep. Purif. Technol. 2025, 363, 132079. DOI:10.1016/j.seppur.2025.132079 [Google Scholar]
- Zhu X, Zhi Y, Zhang Y, Liu T, Ashraf S, Xu D, et al. Activated biochar from co-hydrothermal carbonization of municipal sludge and enteromorpha for high-efficiency Pb(II) removal: Activation optimization and adsorption mechanism. Biomass Bioenergy 2026, 204, 108434. DOI:10.1016/j.biombioe.2025.108434 [Google Scholar]
- Ghaedi S, Rajabi H, Hadi Mosleh M, Spencer BF, Sedighi M. Assessing the efficiency and reusability of zirconium-based MOF-biochar composite for the removal of Pb (II) and Cd (II) in single and multi-ionic systems. J. Environ. Manag. 2025, 380, 125122. DOI:10.1016/j.jenvman.2025.125122 [Google Scholar]
- Liu G, Zhang T. Advances in hydrothermal carbonization for biomass wastewater valorization: Optimizing nitrogen and phosphorus nutrient management to enhance agricultural and ecological outcomes. Water 2025, 17, 800. DOI:10.3390/w17060800 [Google Scholar]
- Wu X, Quan W, Chen Q, Gong W, Wang A. Efficient adsorption of nitrogen and phosphorus in wastewater by biochar. Molecules 2024, 29, 1005. DOI:10.3390/molecules29051005 [Google Scholar]
- Liang L, Xi F, Tan W, Meng X, Hu B, Wang X. Review of organic and inorganic pollutants removal by biochar and biochar-based composites. Biochar 2021, 3, 255–281. DOI:10.1007/s42773-021-00101-6 [Google Scholar]
- Day R, Han N, Adhikari S, Wie JJ, Yoo CG, Zhao X, et al. Biochar–polymer composites for 3D printing: A review. Biochar 2026, 8, 18. DOI:10.1007/s42773-025-00520-9 [Google Scholar]
- Bahmanzadegan F, Ghaemi A. A comprehensive review on novel zeolite-based adsorbents for environmental pollutant. J. Hazard. Mater. Adv. 2025, 17, 100617. DOI:10.1016/j.hazadv.2025.100617 [Google Scholar]
- Du J, Sun J, Zhang D. Preparation of modified zeolite/chitosan composites and study on the adsorption performance of Pb2+. Polym. Eng. Sci. 2024, 64, 196–206. DOI:10.1002/pen.26539 [Google Scholar]
- Yadav VK, Choudhary N, Tirth V, Kalasariya H, Gnanamoorthy G, Algahtani A, et al. A short review on the utilization of incense sticks ash as an emerging and overlooked material for the synthesis of zeolites. Crystals 2021, 11, 1255. DOI:10.3390/cryst11101255 [Google Scholar]
- Salih AM, Abdulrahman RR, Hussein HHS, Aivas SA, Yaqub KQ, Latif AD. Characterization of natural zeolite and evaluation of its adsorption capacity. Br. J. Interdiscip. Res. 2025, 2, 31–47. DOI:10.31039/bjir.v2i4.26 [Google Scholar]
- Kalbuadi DN, Goenadi DH, Santi LP, Nurtjahja LR. The potential use of natural clinoptilolite zeolite for crude oil spill removal from sea water. J. Miner. Mater. Charact. Eng. 2019, 7, 446–453. DOI:10.4236/jmmce.2019.76031 [Google Scholar]
- Gadore V, Mishra SR, Yadav N, Yadav G, Ahmaruzzaman M. Advances in zeolite-based materials for dye removal: Current trends and future prospects. Inorg. Chem. Commun. 2024, 166, 112606. DOI:10.1016/j.inoche.2024.112606 [Google Scholar]
- Hrenović J, Rajić N. Application of modified natural zeolite—clinoptilolite for bacterial control in the environment. Materials 2025, 18, 2411. DOI:10.3390/ma18102411 [Google Scholar]
- Poorkhalil A, Tayefehseyfi E, Farrokhzad H, Mohsenzadeh A. Natural and synthetic zeolites for arsenic removal from water: A comprehensive review of mechanisms, performance, and future perspectives. J. Hazard. Mater. Adv. 2025, 19, 100866. DOI:10.1016/j.hazadv.2025.100866 [Google Scholar]
- Roth WJ, Gil B, Tarach KA, Góra-Marek K. Top-down engineering of zeolite porosity. Chem. Soc. Rev. 2025, 54, 7484–7560. DOI:10.1039/D5CS00319A [Google Scholar]
- Król M. Natural vs. synthetic zeolites. Crystals 2020, 10, 622. DOI:10.3390/cryst10070622 [Google Scholar]
- Rammal J, Daou A, El Badan D, Baki ZA, Darwich S, Rammal W, et al. Comparative analysis of natural and synthetic materials for wastewater treatment: Plant powders, activated carbon, biochar, zeolite, and nanomaterials. J. Chem. Rev. 2025, 7, 566–590. DOI:10.48309/jcr.2025.511327.1421 [Google Scholar]
- Li X, Rezaei F, Rownaghi AA. Methanol-to-olefin conversion on 3D-printed ZSM-5 monolith catalysts: effects of metal doping, mesoporosity and acid strength. Microporous Mesoporous Mater. 2019, 276, 1–12. DOI:10.1016/j.micromeso.2018.09.016 [Google Scholar]
- Kahraman S, Katırcı A, Aytaç A, Veli S, Nigiz FU. Fabrication and characterization electrospun clinoptilolite filled polylactic acid composite membrane: Purification of multiple impurities from water. Appl. Clay Sci. 2025, 268, 107756. DOI:10.1016/j.clay.2025.107756 [Google Scholar]
- Kennedy AJ, Ballentine ML, Das A, Griggs CS, Klaus K, Bortner MJ. Additive manufacturing for contaminants: ammonia removal using 3D printed polymer-zeolite composites. ACS ES&T Water 2021, 1, 621–629. DOI:10.1021/acsestwater.0c00131 [Google Scholar]
- Senila M, Neag E, Cadar O, Kovacs ED, Aschilean I, Kovacs MH. Simultaneous removal of heavy metals (Cu, Cd, Cr, Ni, Zn and Pb) from aqueous solutions using thermally treated Romanian zeolitic volcanic tuff. Molecules 2022, 27, 3938. DOI:10.3390/molecules27123938 [Google Scholar]
- Buzukashvili S, Sommerville R, Hu W, Brooks O, Kökkılıç O, Ouzilleau P, et al. Zeolite synthesis from coal fly ash and its application to heavy metals remediation from water contaminated with Pb, Cu, Zn and Ni ions. Miner. Eng. 2024, 209, 108619. DOI:10.1016/j.mineng.2024.108619 [Google Scholar]
- Yıldız Yiğit M, Baran ES, Moral ÇK. A polymer—zeolite composite for mixed metal removal from aqueous solution. Water Sci. Technol. 2021, 83, 1152–1166. DOI:10.2166/wst.2021.057 [Google Scholar]
- Sudhakar P, Rao R, Dave H, Vasveliya V. Clays and their polymer nanocomposites. In Clay Composites: Environmental Applications; Vithanage M, Lazzara G, Rajapaksha AU, Eds.; Springer: Singapore, 2023; pp. 319–340. DOI:10.1007/978-981-99-2544-5_15 [Google Scholar]
- Ebrahim SA, Hassabo AG, Othman H. Enhancing polymer matrix reinforcements: Exploring the potential of biologically modified clay minerals in dyeing, pigment dyes, and wastewater treatment. J. Text. Color. Polym. Sci. 2025, 22, 389–417. DOI:10.21608/jtcps.2024.288767.1371 [Google Scholar]
- Shi Y, Zhong S, Wang X, Feng C. A review of the removal of heavy metal ions in wastewater by modified montmorillonite. Water Policy 2022, 24, 1590–1609. DOI:10.2166/wp.2022.033 [Google Scholar]
- Heryanto H, Tahir D, Akmal A, Setiawan V. Advancements in clay mineral adsorbents of montmorillonite and kaolinite for effective water contamination remediation. Int. J. Environ. Sci. Technol. 2025, 22, 14693–14718. DOI:10.1007/s13762-025-06630-y [Google Scholar]
- Alias AH, Norizan MN, Sabaruddin FA, Asyraf MRM, Norrrahim MNF, Ilyas AR, et al. Hybridization of MMT/lignocellulosic fiber reinforced polymer nanocomposites for structural applications: a review. Coatings 2021, 11, 1355. DOI:10.3390/coatings11111355 [Google Scholar]
- Gao Y, Wang Y, Chen C, Zhou J, Cheng Y, Shi L. Preparation of montmorillonite nanosheets with a high aspect ratio through heating/rehydrating and gas-pushing exfoliation. Langmuir 2022, 38, 10520–10529.DOI:10.1021/acs.langmuir.2c01320 [Google Scholar]
- Moreno-Sader K, García-Padilla A, Realpe A, Acevedo-Morantes M, Soares JBP. Removal of heavy metal water pollutants (Co2+ and Ni2+) using polyacrylamide/sodium montmorillonite (PAM/Na-MMT) nanocomposites. ACS Omega 2019, 4, 10834–10844. DOI:10.1021/acsomega.9b00981 [Google Scholar]
- Miao Y, Peng W, Wang W, Cao Y, Li H, Chang L, et al. 3D-printed montmorillonite nanosheets based hydrogel with biocompatible polymers as excellent adsorbent for Pb(Ⅱ) removal. Sep. Purif. Technol. 2022, 283, 120176. DOI:10.1016/j.seppur.2021.120176 [Google Scholar]
- Mortezapour F, Shadjou N, Mahmoudian M. Effective removal of anionic dyes from aqueous solution using polyethersulfone based membrane reinforced by montmorillonite. BMC Chem. 2025, 19, 91. DOI:10.1186/s13065-025-01464-8 [Google Scholar]
- Markowski M, Piasecki A, Andrzejewski J. The application of montmorillonite (MMT), halloysite (HNT), and carbon nanotubes (CNT) in toughened polyethylene terephthalate glycol/polycarbonate (PETG/PC) blends: The critical view on the use of nanosized fillers as phase structure modifiers. Polymers 2025, 17, 1463. DOI:10.3390/polym17111463 [Google Scholar]
- Bangar SP, Chaudhary V, Khubber S, Whiteside WS. Kaolinite-based nanocomposites for enhancing starch and other biodegradable polymer applications in food packaging. Int. J. Biol. Macromol. 2025, 320, 145889. DOI:10.1016/j.ijbiomac.2025.145889 [Google Scholar]
- Buyondo KA, Kasedde H, Kirabira JB. A comprehensive review on kaolin as pigment for paint and coating: Recent trends of chemical-based paints, their environmental impacts and regulation. Case Stud. Chem. Environ. Eng. 2022, 6, 100244. DOI:10.1016/j.cscee.2022.100244 [Google Scholar]
- Garcia-Lodeiro I, Palomo A, Fernández-Jiménez A. Crucial insights on the mix design of alkali-activated cement-based binders. In Handbook of Alkali-Activated Cements, Mortars and Concretes, 1st ed.; Pacheco-Torgal F, Labrincha J, Leonelli C, Palomo A, Chindaprasit P, Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 49–73. DOI:10.1533/9781782422884.1.49 [Google Scholar]
- Al-Ameri OB, Alzuhairi M, Bailón-García E, Carrasco-Marín F, Amaro-Gahete J. Transforming petrochemical processes: Cutting-edge advances in kaolin catalyst fabrication. Appl. Sci. 2024, 14, 9080. DOI:10.3390/app14199080 [Google Scholar]
- de Macedo Neto JC, do Nascimento NR, Bello RH, de Verçosa LA, Neto JE, da Costa JCM, et al. Kaolinite review: Intercalation and production of polymer nanocomposites. Eng. Sci. 2022, 17, 28–44. DOI:10.30919/es8d499 [Google Scholar]
- Fan X, Wang Z, Lv Z, Han S, Fan G, Wang W, et al. Chitosan/kaolinite composite hydrogel with high mechanical strength for efficient and selective removal of copper ions: Toward sustainable resource valorization. Carbohydr. Polym. 2025, 366, 123831. DOI:10.1016/j.carbpol.2025.123831 [Google Scholar]
- Al-Hussaini AS, Elias AM, Abd El-Ghaffar MA. New poly(aniline-co-o-phenylenediamine)/kaolinite microcomposites for water decontamination. J. Polym. Environ. 2017, 25, 35–45. DOI:10.1007/s10924-016-0782-7 [Google Scholar]
- Chaka KT, Fambri L, Govindan N. Kaolinite/polypropylene nanocomposites. Part 3: 3D printing. Int. Res. J. Eng. Technol. 2017, 4, 132–147. [Google Scholar]
- Menčík P, Přikryl R, Krobot Š, Melčová V, Kontárová S, Plavec R, et al. Evaluation of the properties of PHB composite filled with kaolin particles for 3D printing applications using the design of experiment. Int. J. Mol. Sci. 2022, 23, 14409. DOI:10.3390/ijms232214409 [Google Scholar]
- Mohd Pu’ad NAS, Abdul Haq RH, Mohd Noh H, Abdullah HZ, Idris MI, Lee TC. Synthesis method of hydroxyapatite: A review. Mater. Today Proc. 2020, 29, 233–239. DOI:10.1016/j.matpr.2020.05.536 [Google Scholar]
- Etinosa PO, Osuchukwu OA, Anisiji EO, Lawal MY, Mohammed SA, Ibitoye OI, et al. In-depth review of synthesis of hydroxyapatite biomaterials from natural resources and chemical regents for biomedical applications. Arab. J. Chem. 2024, 17, 106010. DOI:10.1016/j.arabjc.2024.106010 [Google Scholar]
- Manikandan K, Kumar ND, Velmurugan R, Gokulnath M, Parvathy S, Ayyar M, et al. Enhanced efficiency of zinc oxide hydroxyapatite nanocomposite in photodegradation of methylene blue, ciprofloxacin, and in wastewater treatment. Sci. Rep. 2025, 15, 31819. DOI:10.1038/s41598-025-14310-7 [Google Scholar]
- Chen Y, Zou R, Wu Q. Facile synthesis of hollow ZnO/HAp microsphere photocatalysts for efficient removal of tetracycline under full light irradiation. Inorg. Chem. Commun. 2025, 174, 114084. DOI:10.1016/j.inoche.2025.114084 [Google Scholar]
- Hatim A, Khaddoudi D, Abida F, Ait Ali Ouydir H, Kouzbour S, Agunaou M, et al. Low-cost and sustainable hydroxyapatite synthesis using technical-grade phosphoric acid for environmental applications: Structural and morphological insights. Open Ceram. 2025, 24, 100870. DOI:10.1016/j.oceram.2025.100870 [Google Scholar]
- Yan C, Zeng G, Zhu H, Zhang S, Yang A, Cheng Z, et al. Biogenic Cu-based hydroxyapatite nanocomposites for efficient clean of antibiotics: Performance and mechanism. Sep. Purif. Technol. 2025, 360, 131123. DOI:10.1016/j.seppur.2024.131123 [Google Scholar]
- Labrag J, Abbadi M, Hnini M, Bekkali CE, Bouziani A, Robert D, et al. Antibiotic photocatalysis and antimicrobial activity of low-cost multifunctional Fe3O4@HAp nanocomposites. J. Environ. Health Sci. Eng. 2023, 21, 429–440. DOI:10.1007/s40201-023-00869-8 [Google Scholar]
- Pai S, Kini MS, Selvaraj R. Environmental and energy management: A review on adsorptive removal of dyes from wastewater by hydroxyapatite nanocomposites. Environ. Sci. Pollut. Res. 2021, 28, 11835–11849. DOI:10.1007/s11356-019-07319-9 [Google Scholar]
- Kim BS, Yang SS, Kim CS. Incorporation of BMP-2 nanoparticles on the surface of a 3D-printed hydroxyapatite scaffold using an ε-polycaprolactone polymer emulsion coating method for bone tissue engineering. Colloids Surf. B Biointerfaces 2018, 170, 421–429. DOI:10.1016/j.colsurfb.2018.06.043 [Google Scholar]
- Thirugnanasambandam A, Nallamuthu R, Rangappa SM, Siengchin S, Vijay Kumar V. Material extrusion of the poly(lactic acid)/HAp nanocomposite scaffold for bone tissue applications: A comprehensive investigation. ACS Omega 2025, 10, 44948–44959. DOI:10.1021/acsomega.5c02055 [Google Scholar]
- Wang Y, Wang Y, Qiu S, Wang C, Zhang H, Guo J, et al. 3D-printed filters for efficient heavy metal removal from water using PLA@CS/HAP composites. Polymers 2023, 15, 4144. DOI:10.3390/polym15204144 [Google Scholar]
- Fahmy HM, Mosleh AM, Elghany AA, Shams-Eldin E, Abu Serea ES, Ali SA, et al. Coated silver nanoparticles: Synthesis, cytotoxicity, and optical properties. RSC Adv. 2019, 9, 20118–20136. DOI:10.1039/c9ra02907a [Google Scholar]
- Dasauni K, Divya, Mathpal P, Nailwal TK. Polymeric nanoparticle-based insecticide: A critical review of agriculture production. In Nano-Enabled Agrochemicals in Agriculture, 1st ed.; Ghorbanpour M, Shahid MA, Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 445–466. DOI:10.1016/B978-0-323-91009-5.00015-X [Google Scholar]
- Bahcelioglu E, Unalan HE, Erguder TH. Silver-based nanomaterials: A critical review on factors affecting water disinfection performance and silver release. Crit. Rev. Environ. Sci. Technol. 2021, 51, 2389–2423. DOI:10.1080/10643389.2020.1784666 [Google Scholar]
- Chen X, Wang L, Ding C, Xie H, Zou H, Deng J, et al. Highly efficient removal of radioactive iodine anions by nano silver modified activated carbon fiber. Appl. Surf. Sci. 2024, 643, 158644. DOI:10.1016/j.apsusc.2023.158644 [Google Scholar]
- Li L, Yu SJ, Zheng RG, Li P, Li QC, Liu JF. Removal of iodide anions in water by silver nanoparticles supported on polystyrene anion exchanger. J. Environ. Sci. 2023, 128, 45–54. DOI:10.1016/j.jes.2022.08.012 [Google Scholar]
- Polo AMS, Lopez-Peñalver JJ, Sánchez-Polo M, Rivera-Utrilla J, López-Ramón MV, Rozalén M. Halide removal from water using silver doped magnetic-microparticles. J. Environ. Manag. 2020, 253, 109731. DOI:10.1016/j.jenvman.2019.109731 [Google Scholar]
- N’Gatta KM, Assanvo EF, El Hayek J, Masquelez N, Kamgang Syapnjeu P, Deabate S, et al. 3D-printed polycaprolactone scaffolds reinforced with cellulose nanocrystals and silver nanoparticles for bone tissue engineering. ACS Appl. Mater. Interfaces 2025, 17, 41028–41043. DOI:10.1021/acsami.5c06504 [Google Scholar]
- Zafar MS, Ejaz S, Ahmad F, Ibrahim AH, Al-Rawi SS, Manzoor F, et al. Silver nanoparticles in water disinfection: A comprehensive overview on their mechanisms, benefits, and limitations. Arch. Microbiol. 2026, 208, 166.DOI:10.1007/s00203-026-04719-7 [Google Scholar]
- Sharma R, Kumar A. Human health risk assessment and uncertainty analysis of silver nanoparticles in water. Environ. Sci. Pollut. Res. 2024, 31, 13739–13752. DOI:10.1007/s11356-024-32006-9 [Google Scholar]
- Mahlambi MM, Ngila CJ, Mamba BB. Recent developments in environmental photocatalytic degradation of organic pollutants: The case of titanium dioxide nanoparticles—A review. J. Nanomater. 2015, 2015, 790173. DOI:10.1155/2015/790173 [Google Scholar]
- Blessymol B, Yasotha P, Kalaiselvi V, Gopi S. An antioxidant study of titanium dioxide (TiO2) nanoparticles against mace of nutmeg in Myristica fragrans houtt, rhizomes of Curcuma longa linn and Kaempferia galanga extracts. Results Chem. 2024, 7, 101291. DOI:10.1016/j.rechem.2023.101291 [Google Scholar]
- García A, Delgado L, Torà JA, Casals E, González E, Puntes V, et al. Effect of cerium dioxide, titanium dioxide, silver, and gold nanoparticles on the activity of microbial communities intended in wastewater treatment. J. Hazard. Mater. 2012, 199–200, 64–72. DOI:10.1016/j.jhazmat.2011.10.057 [Google Scholar]
- Manzoor Q, Sajid A, Ali Z, Nazir A, Sajid A, Imtiaz F, et al. Toxicity spectrum and detrimental effects of titanium dioxide nanoparticles as an emerging pollutant: A review. Desalin. Water Treat. 2024, 317, 100025. DOI:10.1016/j.dwt.2024.100025 [Google Scholar]
- Armaković SJ, Savanović MM, Armaković S. Titanium dioxide as the most used photocatalyst for water purification: An overview. Catalysts 2023, 13, 26. DOI:10.3390/catal13010026 [Google Scholar]
- Reyes-Coronado D, Rodríguez-Gattorno G, Espinosa-Pesqueira ME, Cab C, De Coss R, Oskam G. Phase-pure TiO2 nanoparticles: Anatase, brookite and rutile. Nanotechnology 2008, 19, 145605. DOI:10.1088/0957-4484/19/14/145605 [Google Scholar]
- Shanmugam S, Madheswaran B, Soga T. Synthesis of titanium dioxide nanoparticles with desired ratio of anatase and rutile phases and the effect of high temperature annealing. Trans. Mater. Res. Soc. Jpn. 2018, 43, 255–261. DOI:10.14723/tmrsj.43.255 [Google Scholar]
- Mittireddi RT, Dotiyal M, Roy R, Prajapati D, Pandey PK, Mishra A, et al. Highly active brookite titania nanoparticles for degradation of toxic organic pollutants. Powder Technol. 2025, 466, 121373. DOI:10.1016/j.powtec.2025.121373 [Google Scholar]
- Eddy DR, Permana MD, Sakti LK, Sheha GAN, Solihudin, Hidayat S, et al. Heterophase polymorph of TiO2 (anatase, rutile, brookite, TiO2 (B)) for efficient photocatalyst: Fabrication and activity. Nanomaterials 2023, 13, 704. DOI:10.3390/nano13040704 [Google Scholar]
- Fouad SM, El-Shazly YMS, Alyoubi MA, Nosier SA, Abdel-Aziz MH. Enhanced photocatalytic degradation of cationic dyes using slurry of anatase titania in a falling film reactor. Case Stud. Chem. Environ. Eng. 2023, 8, 100518. DOI:10.1016/j.cscee.2023.100518 [Google Scholar]
- Liu G, Pan X, Li J, Li C, Ji C. Facile preparation and photocatalytic performance of anatase TiO2/nanocellulose composite. Cellulose 2021, 28, 57. DOI:10.3762/bxiv.2021.57.v1 [Google Scholar]
- Tichapondwa S, Newman J, Kubheka O. Effect of TiO2 phase on the photocatalytic degradation of methylene blue dye. Phys. Chem. Earth Parts A/B/C 2020, 118–119, 102900. DOI:10.1016/j.pce.2020.102900 [Google Scholar]
- Bansiddhi A, Panomsuwan G, Hussakan C, Htet TL, Kandasamy B, Janbooranapinij K, et al. Ecofriendly 3D printed TiO2/SiO2/polymer scaffolds for dye removal. Top. Catal. 2023, 66, 1662–1673. DOI:10.1007/s11244-023-01864-x [Google Scholar]
- Sangiorgi A, Gonzalez Z, Ferrandez-Montero A, Yus J, Sanchez-Herencia AJ, Galassi C, et al. 3D printing of photocatalytic filters using a biopolymer to immobilize TiO2 nanoparticles. J. Electrochem. Soc. 2019, 166, H3239–H3248. DOI:10.1149/2.0341905jes [Google Scholar]
- Han L, Shen L, Lin H, Huang Z, Xu Y, Li R, et al. 3D printing titanium dioxide-acrylonitrile-butadiene-styrene (TiO2-ABS) composite membrane for efficient oil/water separation. Chemosphere 2023, 315, 137791. DOI:10.1016/j.chemosphere.2023.137791 [Google Scholar]
- Mousa MA, Khairy M. Synthesis of nano-zinc oxide with different morphologies and its application on fabrics for UV protection and microbe-resistant defense clothing. Text. Res. J. 2020, 90, 2492–2503. DOI:10.1177/0040517520920952 [Google Scholar]
- Dai K, Cao S, Yuan J, Wang Z, Li H, Yuan C, et al. Recent advances of sustainable UV shielding materials: Mechanisms and applications. ACS Appl. Mater. Interfaces 2025, 17, 30402–30422. DOI:10.1021/acsami.5c04539 [Google Scholar]
- Araki SM, Baby AR. New perspectives on titanium dioxide and zinc oxide as inorganic UV filters: Advances, safety, challenges, and environmental considerations. Cosmetics 2025, 12, 77. DOI:10.3390/cosmetics12020077 [Google Scholar]
- Samad A, Din MI, Ahmed M. Synthesis of zinc oxide nanoparticles impregnated clay tablets and their potential applications as point of use water purification intervention. Desalin. Water Treat. 2020, 181, 251–257. DOI:10.5004/dwt.2020.25123 [Google Scholar]
- Jin SE, Jin HE. Antimicrobial activity of zinc oxide nano/microparticles and their combinations against pathogenic microorganisms for biomedical applications: From physicochemical characteristics to pharmacological aspects. Nanomaterials 2021, 11, 263. DOI:10.3390/nano11020263 [Google Scholar]
- Mirhosseini F, Amiri M, Daneshkazemi A, Zandi H, Javadi ZS. Antimicrobial effect of different sizes of nano zinc oxide on oral microorganisms. Front. Dent. 2019, 16, 105–112. DOI:10.18502/fid.v16i2.1361 [Google Scholar]
- Jiang J, Pi J, Cai J. The advancing of zinc oxide nanoparticles for biomedical applications. Bioinorg. Chem. Appl. 2018, 2018, 1062562. DOI:10.1155/2018/1062562 [Google Scholar]
- Aina ARN, Patel H, Aich S, Roy B, Samanta NS, Pal B. Recent advances in ZnO based photocatalysts for industrial dye degradation. Discover Appl. Sci. 2025, 7, 977. DOI:10.1007/s42452-025-07530-z [Google Scholar]
- Kumar R, Singh R, Singh M, Kumar P. On ZnO nano particle reinforced PVDF composite materials for 3D printing of biomedical sensors. J. Manuf. Process. 2020, 60, 268–282. DOI:10.1016/j.jmapro.2020.10.027 [Google Scholar]
- Kumar R, Singh R, Singh M, Kumar P. ZnO nanoparticle-grafted PLA thermoplastic composites for 3D printing applications: Tuning of thermal, mechanical, morphological and shape memory effect. J. Thermoplast. Compos. Mater. 2022, 35, 799–825. DOI:10.1177/0892705720925119 [Google Scholar]
- Billings C, Cai C, Liu Y. Utilization of antibacterial nanoparticles in photocurable additive manufacturing of advanced composites for improved public health. Polymers 2021, 13, 2616. DOI:10.3390/polym13162616 [Google Scholar]
- Sharma N, Dev Gupta R, Chandmal Sharma R, Dayal S, Singh Yadav A. Graphene: An overview of its characteristics and applications. Mater. Today Proc. 2021, 47, 2752–2755. DOI:10.1016/j.matpr.2021.03.086 [Google Scholar]
- Obayomi KS, Lau SY, Danquah M, Chiong T, Takeo M. Advances in graphene oxide based nanobiocatalytic technology for wastewater treatment. Environ. Nanotechnol. Monit. Manag. 2022, 17, 100647. DOI:10.1016/j.enmm.2022.100647 [Google Scholar]
- Ntone EPN, Rahman SA, Samah RA, Fauzi MA, Ngouangna EN, Hasbullah H, et al. Advancing wastewater treatment: A review on the cutting-edge graphene oxide-enhanced polymeric membranes. Rev. Chem. Eng. 2026, 42, 1–29. DOI:10.1515/revce-2025-0016 [Google Scholar]
- Marjani A, Nakhjiri AT, Adimi M, Jirandehi HF, Shirazian S. Effect of graphene oxide on modifying polyethersulfone membrane performance and its application in wastewater treatment. Sci. Rep. 2020, 10, 2049. DOI:10.1038/s41598-020-58472-y [Google Scholar]
- Saeedi-Jurkuyeh A, Jafari AJ, Kalantary RR, Esrafili A. A novel synthetic thin-film nanocomposite forward osmosis membrane modified by graphene oxide and polyethylene glycol for heavy metals removal from aqueous solutions. React. Funct. Polym. 2020, 146, 104397. DOI:10.1016/j.reactfunctpolym.2019.104397 [Google Scholar]
- Das L, Das P, Bhowal A, Bhattachariee C. Synthesis of hybrid hydrogel nano-polymer composite using graphene oxide, chitosan and PVA and its application in waste water treatment. Environ. Technol. Innov. 2020, 18, 100664. DOI:10.1016/j.eti.2020.100664 [Google Scholar]
- Velusamy S, Roy A, Sundaram S, Kumar Mallick T. A review on heavy metal ions and containing dyes removal through graphene oxide-based adsorption strategies for textile wastewater treatment. Chem. Rec. 2021, 21, 1570–1610. DOI:10.1002/tcr.202000153 [Google Scholar]
- Mohd Nizam NH, Badrol Hisam ZA, Zazani AH. Innovation of 3D-printed waste-derived graphene oxide for water treatment via digital light processing. In Proceedings of the International Undergraduates Get Together 2024 (IUGeT 2024) Undergraduates’ Digital Engagement Towards Global Ingenuity 2nd Edition 2024, pp. 196-205. Available online: https://ir.uitm.edu.my/id/eprint/118743 (accessed on 5 January 2026).
- Feng Z, Xu Y, Ding W, Li Q, Zhao X, Wei X, et al. Nano graphene oxide creates a fully biobased 3D-printed membrane with high-flux and anti-fouling oil/water separation performance. Chem. Eng. J. 2024, 485, 149603. DOI:10.1016/j.cej.2024.149603 [Google Scholar]
- Rybarczyk A, Smułek W, Grzywaczyk A, Kaczorek E, Jesionowski T, Nghiem LD, et al. 3D printed polylactide scaffolding for laccase immobilization to improve enzyme stability and estrogen removal from wastewater. Bioresour. Technol. 2023, 381, 129144. DOI:10.1016/j.biortech.2023.129144 [Google Scholar]
- Majooni Y, Sava SP, Fayazbakhsh K, Yousefi N. Sustainable 3D-printed platforms with durable photocatalytic coatings for efficient water treatment. Adv. Sustain. Syst. 2025, 9, e00135. DOI:10.1002/adsu.202500135 [Google Scholar]
- Podstawczyk D, Skrzypczak D, Połomska X, Stargała A, Witek-Krowiak A, Guiseppi-Elie A, et al. Preparation of antimicrobial 3D printing filament: In situ thermal formation of silver nanoparticles during the material extrusion. Polym. Compos. 2020, 41, 4692–4705. DOI:10.1002/pc.25743 [Google Scholar]
- Wang X, An J, Hassan A, Gao Q, Liu X, Boudaoud H. Simplified 3D printing method for fabrication of superhydrophobic oil–water separation membrane with controllable pore size. J. Appl. Polym. Sci. 2025, 142, e57217. DOI:10.1002/app.57217 [Google Scholar]
- Laksemana IKA, Adi RK, Handayani M, Rahmayanti YD, Anggoro MA, Muflikhun MA, et al. Antibacterial enhancement and mechanical characterization of PLA resin through the addition of AgNO3 for DLP 3D printing. Mater. Lett. 2025, 399, 139105. DOI:10.1016/j.matlet.2025.139105 [Google Scholar]
- Zhang H, Shi Z, Liu X, Wang B, Niu W, Cai W, et al. Insights into graphene oxide double-network macro-monothlic adsorbent through 3D printing: Mechanical properties and Cu2+ adsorption mechanism. Sep. Purif. Technol. 2025, 363, 132140. DOI:10.1016/j.seppur.2025.132140 [Google Scholar]
- Wu T, Karimi-Maleh H, Li Y, Zhang D, Zhang Z, Zhong N, et al. 3D printed porous chitosan/metal–organic framework composites as effective adsorbents to remove heavy metals from wastewater. Chem. Eng. J. 2024, 493, 152780. DOI:10.1016/j.cej.2024.152780 [Google Scholar]
- Ogazi AC, Osifo PO. Inhibition of Coliforms and Escherichia coli bacterial strains in water by 3D printed CS/GO/AgNP filtration membranes. J. Polym. Environ. 2023, 31, 4448–4467. DOI:10.1007/s10924-023-02889-9 [Google Scholar]
- Tse I, Jay A, Na I, Murphy S, Niño-Martínez N, Martínez-Castañon GA, et al. Antimicrobial activity of 3D-printed acrylonitrile butadiene styrene (ABS) polymer-coated with silver nanoparticles. Materials 2021, 14, 7681. DOI:10.3390/ma14247681 [Google Scholar]
- Liakos IL, Mondini A, Del Dottore E, Filippeschi C, Pignatelli F, Mazzolai B. 3D printed composites from heat extruded polycaprolactone/sodium alginate filaments and their heavy metal adsorption properties. Mater. Chem. Front. 2020, 4, 2472–2483. DOI:10.1039/D0QM00159G [Google Scholar]
- Boo C, Elimelech M, Hong S. Fouling control in a forward osmosis process integrating seawater desalination and wastewater reclamation. J. Membr. Sci. 2013, 444, 148–156. DOI:10.1016/j.memsci.2013.05.004 [Google Scholar]
- Singh RK, Fu Y, Zeng C, Nguyen DT, Roy P, Bao J, et al. Hydrodynamics of countercurrent flow in an additive-manufactured column with triply periodic minimal surfaces for carbon dioxide capture. Chem. Eng. J. 2022, 450, 138124. DOI:10.1016/j.cej.2022.138124 [Google Scholar]
- Park S, Jeong YD, Lee JH, Kim J, Jeong K, Cho KH. 3D printed honeycomb-shaped feed channel spacer for membrane fouling mitigation in nanofiltration. J. Membr. Sci. 2020, 620, 118665. DOI:10.1016/j.memsci.2020.118665 [Google Scholar]
- Nguyen T, Roddick F, Fan L. Biofouling of water treatment membranes: A review of the underlying causes, monitoring techniques and control measures. Membr. 2012, 2, 804–840. DOI:10.3390/membranes2040804 [Google Scholar]
- Navarro-Tovar R, Zoumpouli GA, Gorgojo P, Martin P, Chew YJ, Mattia D, et al. Mitigation of organic fouling in membrane distillation via 3D printed wavy composite membranes. Desalination.2025, 614, 119201. DOI:10.1016/j.desal.2025.119201 [Google Scholar]
- Lepowsky E, Tasoglu S. Emerging Anti-Fouling Methods: Towards reusability of 3D-printed devices for biomedical applications. Micromachines 2018, 9, 196. DOI:10.3390/mi9040196 [Google Scholar]
- Bushnaq H, Balakrishnan HK, Burton T, Montoya JC, Rodriguez-Andres J, Mackenzie J, et al. 3D printed photo-sensitized microfiltration membranes for simultaneous water filtration and pathogen management. Mater. Hori. 2026. DOI:10.1039/D5MH01431J [Google Scholar]
- Chen T, Xu P, Qiu M, Chen X, Zhong Z, Fan Y. Construction of anti-fouling ceramic tubular membranes with corrugated inner surfaces using DLP 3D printing. J. Membr. Sci. 2024, 706, 122941. DOI:10.1016/j.memsci.2024.122941 [Google Scholar]
- Castillo EHC, Thomas N, Al-Ketan O, Rowshan R, Al-Rub RKA, Nghiem LD, et al. 3D printed spacers for organic fouling mitigation in membrane distillation. J. Membr. Sci. 2019, 581, 331–343. DOI:10.1016/j.memsci.2019.03.040 [Google Scholar]
- Al Jassmi H, Al Najjar F, Mourad AHI. Large-scale 3D printing: The way forward. IOP Conf. Ser. Mater. Sci. Eng. 2018, 324, 012088. DOI:10.1088/1757-899X/324/1/012088 [Google Scholar]
- Koo JW, Ho JS, An J, Zhang Y, Chua CK, Chong TH. A review on spacers and membranes: conventional or hybrid additive manufacturing? Water Res. 2021;188, p.116497. DOI:10.1016/j.watres.2020.116497 [Google Scholar]
- Jayawardane H, Davies IJ, Gamage JR, John M, Biswas WK. Investigating the ‘techno-eco-efficiency’ performance of pump impellers: Metal 3D printing vs. CNC machining. Int. J. Adv. Manuf. Technol. 2022, 121, 6811–6836. DOI:10.1007/s00170-022-09748-2 [Google Scholar]
- Ulkir O. Energy-consumption-based life cycle assessment of additive-manufactured product with different types of materials. Polymers 2023, 15, 1466. DOI:10.3390/polym15061466 [Google Scholar]
- Alex Y, Divakaran NC, Pattanayak I, Lakshyajit B, Ajay PV, Mohanty S. Comprehensive study of PLA material extrusion 3D printing optimization and its comparison with PLA injection molding through life cycle assessment. Sustain. Mater. Technol. 2025, 43, e01222. DOI:10.1016/j.susmat.2024.e01222 [Google Scholar]
- Bezzina CM, Refalo P. Fused Filament Fabrication and Injection Moulding of Plastic Packaging: An Environmental and Financial Comparative Assessment. Machines 2023, 11(6), p.634. DOI:10.3390/machines11060634 [Google Scholar]
- Wan Harun WS, Ahmad F, Mohd Foudzi F, Tsumori F, Thirugnanasambandam A, Saber M. Sustainable additive manufacturing of polymer composites: A review. Nexus Sustain. Energy Technol. J. 2025, 1, 75–92. DOI:10.63100/3080-1915.1006 [Google Scholar]
- Garcia FL, Nunes AO, Martins MG, Belli MC, Saavedra YM, Silva DA, et al. Comparative LCA of conventional manufacturing vs. additive manufacturing: the case of injection moulding for recycled polymers. Int. J. Sustain. Eng. 2021, 14(6), pp.1604-1622. DOI:10.1080/19397038.2021.1990435 [Google Scholar]
- Zhang Y, Li J, Zou Y, Liu Y, Li L, Xu J, et al. 3D printing technologies in water treatment: Applications, challenges, and emerging trends. Chem. Eng. J. 2024, 500, 157184. DOI:10.1016/j.cej.2024.157184 [Google Scholar]
- Saroia J, Wang Y, Wei Q, Lei M, Li X, Guo Y, et al. A review on 3D printed matrix polymer composites: Its potential and future challenges. Int. J. Adv. Manuf. Technol. 2020, 106, 1695–1721. DOI:10.1007/s00170-019-04534-z [Google Scholar]
- Zeinedini A, Shokrieh MM. Agglomeration phenomenon in graphene/polymer nanocomposites: Reasons, roles, and remedies. Appl. Phys. Rev. 2024, 11, 041301. DOI:10.1063/5.0223785 [Google Scholar]
- Goh GD, Wong KK, Tan N, Seet HL, Nai ML. Large-format additive manufacturing of polymers: A review of fabrication processes, materials, and design. Virtual Phys. Prototyp. 2024, 19, e2336160. DOI:10.1080/17452759.2024.2336160 [Google Scholar]
- Chow WS. Polymer engineering at the nexus of additive manufacturing, artificial intelligence, and responsible innovation. Express Polym. Lett. 2026, 20, 324–325. DOI:10.3144/expresspolymlett.2026.25 [Google Scholar]