Starch-Based Edible Coatings for Sustainable Food Packaging: A Comprehensive Review
Received: 25 February 2026 Revised: 09 March 2026 Accepted: 16 March 2026 Published: 26 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Sustainable packaging has emerged as a critical priority in recent years due to its environmental, economic, and social implications. Packaging is indispensable in modern life, especially in the food industry, where it ensures product preservation, prevents microbial contamination, and facilitates handling, transportation, and storage. In addition, packaging serves as a medium for communicating information and instructions to consumers. However, conventional packaging materials, particularly plastics, pose significant environmental challenges, including waste accumulation, greenhouse gas emissions, and limited recyclability. With nearly half of global plastic production dedicated to packaging and only a small fraction effectively recycled, the shift toward sustainable alternatives has become an urgent necessity (Figure 1). Sustainability [1] in packaging emphasizes the use of materials and processes that reduce virgin resource consumption, minimize landfill waste, and promote recyclability, reusability, or biodegradability. It is broadly defined across three interconnected dimensions: environmental protection, economic efficiency, and human well-being. Glass, paper, metals, and bio-based materials each offer potential advantages over traditional plastics, though their sustainability depends on factors such as production processes, functionality, and end-of-life management. Recent innovations in biodegradable and bio-based materials, along with improved recycling and reuse strategies, highlight the potential for sustainable packaging to balance the dual objectives of ensuring food safety and reducing environmental burdens [2].
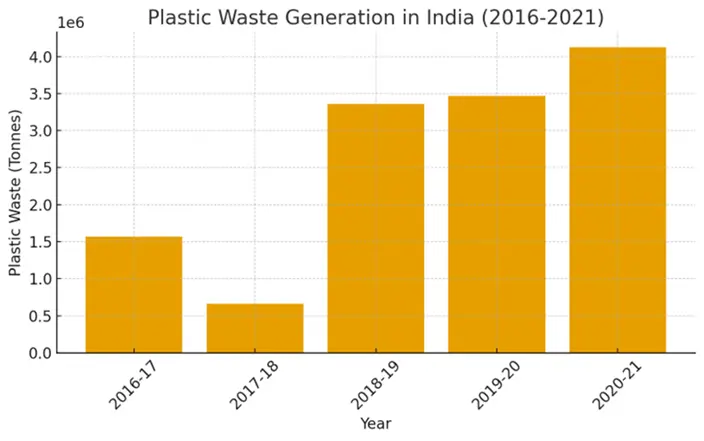
Figure 1. Quantity of plastic waste generated in India (2016–2021), adapted from Central Pollution Control Board (MoEFCC-2022).
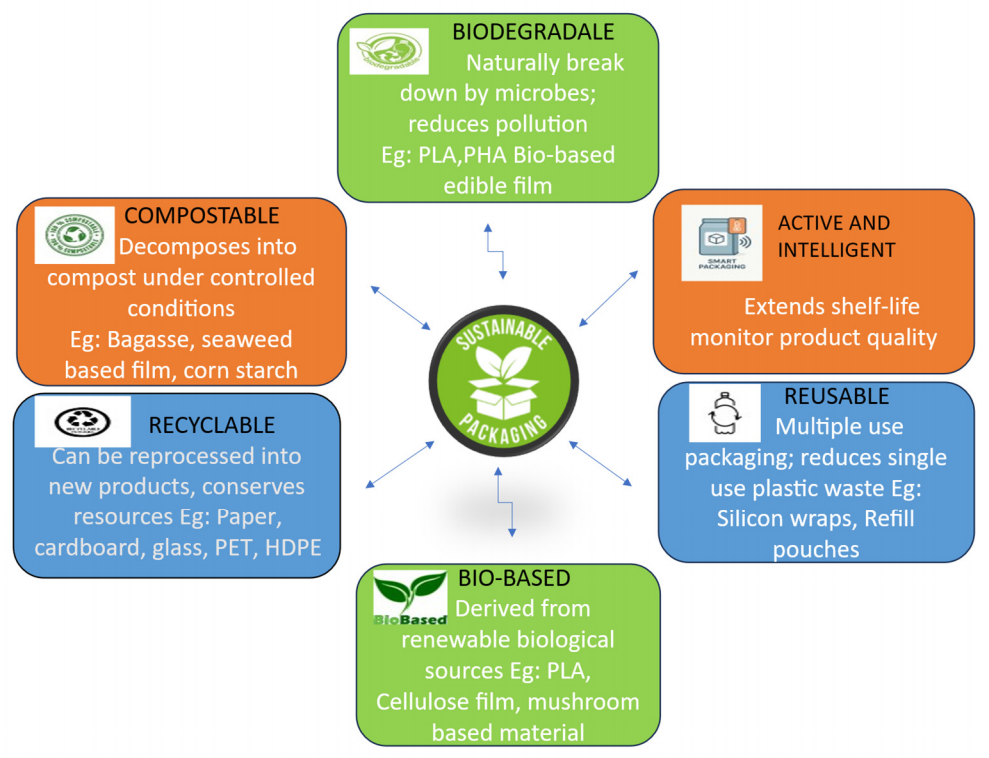
In recent years, rising global awareness of the environmental effects of packaging materials has encouraged the use of food packaging designed to reduce ecological harm, commonly known as sustainable packaging (Figure 2). This approach employs innovative and functional materials that not only promote environmental well-being but also support economic growth [3]. In accordance with the Plastic Waste Management Rules (2022), the Ministry of Environment, Forest, and Climate Change of India formulated guidelines on Extended Producer Responsibility (EPR) for plastic packaging management. The framework outlines measures related to reuse, recycling, the utilization of recycled plastics, and effective end-of-life waste management. It also requires producers, importers, and brand owners to register on a centralized portal and submit an action plan detailing their respective EPR targets. They are further mandated to report annually on the quantities of plastic packaging waste that are collected and managed. In addition, the guidelines advocate for the development of a well-distributed network of collection centers, considering factors such as population density, waste generation patterns, accessibility, and proximity to end users, instead of focusing exclusively on economically profitable areas (MoEFCC, 2022).
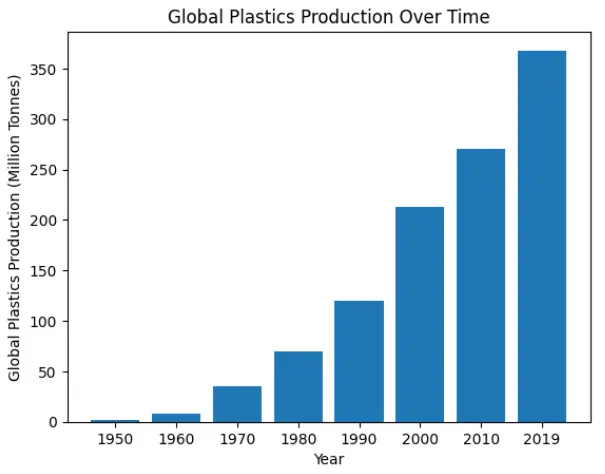
The world witnessed significant changes over the past centuries due to remarkable inventions such as plastics. The fact is undeniable that “plastic is the greatest finding in the millennium”. The International Union of Pure and Applied Chemistry defines plastic as a “polymeric material that may contain other substances to improve performance and/or reduce costs”. Plastic was invented in 1860, and the beginning of the global plastic industry was marked in 1907. However, industrial development happened in the 1920s, and plastic production expanded in the 1940s. About 2 million tons of plastic were produced in 1950, and by 2019, the amount had increased to 368 million tons. Plastic consumption has therefore increased about 180 times from 1950 to 2018 (Figure 2). Thus, an exponential increase in plastic production has been observed [4].
Growing environmental awareness in society has heightened interest in minimizing plastic use and reducing food waste. Within this context, the food industry faces persistent challenges in maintaining both the quality and quantity of products, particularly perishables, throughout the supply chain, from harvest to final consumption. Edible coatings are increasingly recognized as highly effective postharvest measures for preserving the quality of perishable produce, such as fruits and vegetables. These coatings, applied as thin biopolymer films, form a protective barrier that reduces respiration and moisture loss, delays physiological degradation, and suppresses microbial growth (e.g., fungal spoilage, enzymatic browning). Collectively, these effects substantially extend shelf life and better maintain product integrity from harvest to consumption [5]. Edible coatings have emerged as a promising sustainable solution to conventional plastic packaging (Figure 3). Acting as biodegradable protective barriers, they help decrease reliance on synthetic polymers while simultaneously extending the shelf life of food products. Through this dual role, edible coatings play a crucial part in reducing postharvest losses and food waste, aligning with circular economy approaches and advancing environmentally friendly food preservation practices [6]. Edible coatings function as protective barriers controlling moisture, gas exchange, and product–environment interactions, while also enhancing sensory and microbial stability. Their safety, low cost, and feasibility make them a sustainable substitute for plastic packaging [7].
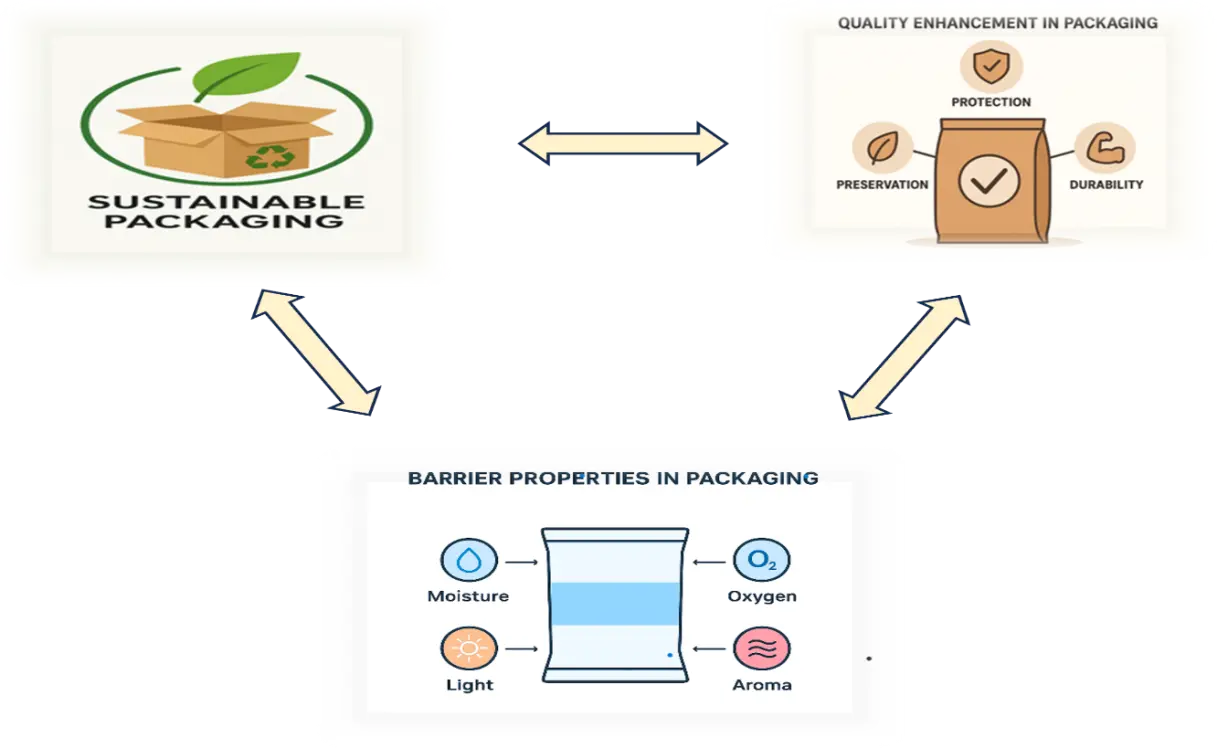
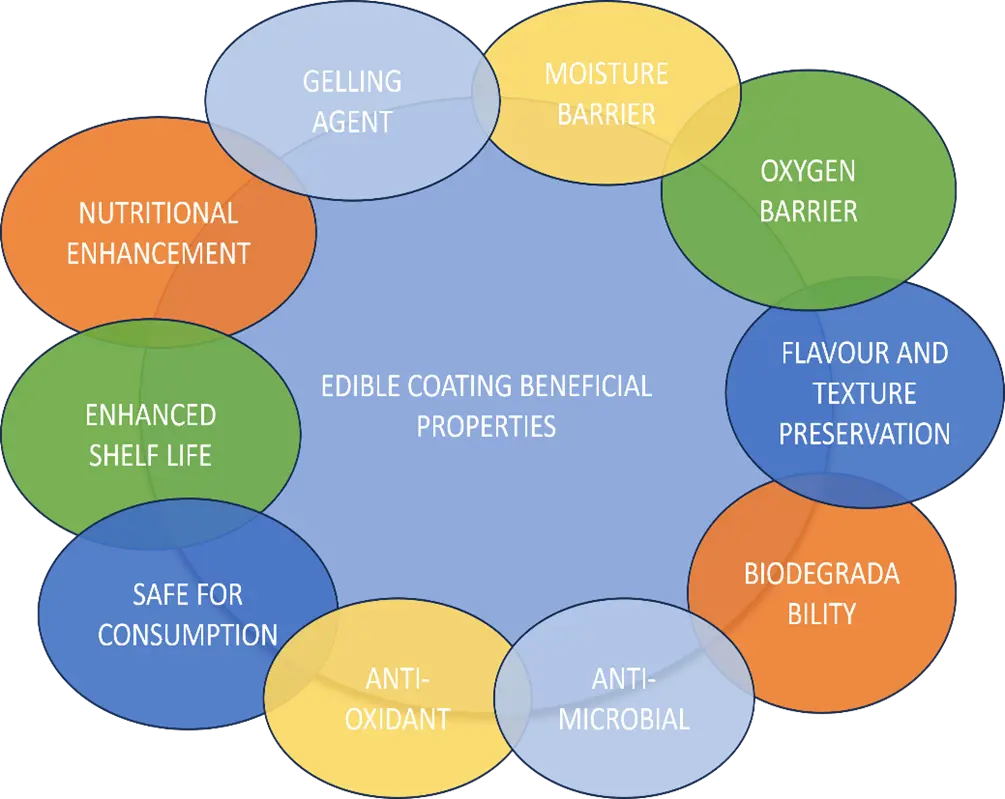
The multifunctional benefits of edible coatings and films can be broadly organized into three categories: barrier characteristics, quality enhancement, and eco-friendly sustainability advantages (Figure 4). In terms of barrier properties, these coatings help control moisture retention, regulate gas exchange, and minimize food–environment interactions, thereby extending shelf life. From a quality enhancement perspective, they improve the appearance of food products, maintain biochemical stability, preserve physicochemical characteristics, and enhance microbial stability. Additionally, the sustainability advantages include their non-toxic and safe nature, low processing cost, and feasibility as an eco-friendly alternative to conventional plastic packaging [8]. Biopolymers act as the principal materials in edible coating formulations and are generally sourced from proteins or polysaccharides. These materials are effective in preventing the transfer of oxygen, lipids, and volatile compounds; however, they generally show inadequate mechanical strength and high permeability to water. Lipid-based components, on the other hand, function as cohesive biomaterials that, upon reaching their transition temperature, provide a glossy appearance and serve as efficient barriers to moisture loss. To enhance the overall physicochemical performance of coatings, composite formulations are often developed. The functional and physicochemical properties of edible coatings can also be improved through the incorporation of active agents such as essential oils, bio-nanocomposites, and inorganic nanoparticles [7]. Starch exhibits a semi-crystalline structure composed of roughly 70% amorphous regions, dominated by water-soluble amylose, and 30% crystalline regions, largely made up of water-insoluble amylopectin. As a polysaccharide, starch is built from glucose monomers arranged in both linear and branched chains. Amylose, accounting for 10–30% of starch, is a straight-chain polymer of more than 100 D-glucose units linked by α-1,4-glycosidic bonds [9]. In contrast, amylopectin constitutes 70–90% of starch and has a branched structure, formed by about 20 glucose units connected through β-1,4-glycosidic bonds [10]. The diverse industrial applications of starch are largely determined by its functional characteristics, particularly gelatinization and rheological properties. In the food sector, it serves as a thickener and binder in products such as puddings, soups, sauces, salad dressings, mayonnaise, and infant formulas. Beyond food, starch finds significant use in pharmaceuticals and cosmetics, where it functions as a binder, disintegrant, diluent, lubricant, and glidant in the production of tablets, capsules, and granules [11].
The global reliance on petroleum-based plastics for food packaging has created a pressing environmental challenge, with waste accumulation, microplastic pollution, and greenhouse gas emissions threatening ecosystems and human health. Although regulatory frameworks such as India’s Plastic Waste Management Rules (2016, 2022) and Extended Producer Responsibility (EPR) guidelines have been introduced to reduce plastic dependency, achieving sustainable packaging solutions remains an unmet challenge. In this context, starch-based edible coatings represent a promising alternative. Derived from renewable, biodegradable, and abundantly available agricultural resources, starch offers a low-cost and eco-friendly substitute for synthetic polymers. Its functional properties such as film-forming ability, transparency, and compatibility with bioactive agents make it highly suitable for extending the shelf life of perishable food products while simultaneously reducing postharvest losses and food waste. Furthermore, starch-based coatings align with circular economy principles by addressing both sustainability goals (biodegradability, reduced plastic use) and food security concerns (enhanced preservation and safety) [12].
A comprehensive review of starch-based edible coatings is therefore essential to consolidate current scientific knowledge, evaluate their physicochemical and functional properties, and examine practical applications in food preservation. Furthermore, addressing challenges associated with large-scale production, such as cost-effectiveness, process standardization, and quality consistency, will provide actionable insights for researchers, industry stakeholders, and policymakers. By bridging scientific advances with practical considerations, this review aims to support the adoption of sustainable, circular-economy-aligned packaging solutions, ultimately reducing plastic dependency, mitigating environmental impacts, and fostering innovation in the food packaging sector.
2. Starch as a Biopolymer for Edible Coatings
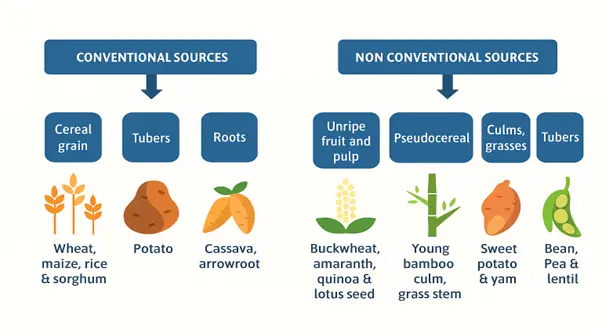
Starch, a widely recognized carbohydrate polymer, is derived from a variety of plant sources that thrive across most temperate regions [13]. These sources can be broadly classified into conventional and non-conventional categories. Conventional sources, which are globally abundant and commercially significant, include cereal grains such as wheat, maize, rice, and sorghum; tuber crops like potato and sweet potato; and root crops such as cassava and arrowroot (Figure 5). Globally, the primary contributors to starch production are corn, wheat, potato, and rice, accounting for approximately 84%, 7%, 4%, and 1% of total starch production, respectively. Besides the major starch-producing crops, numerous alternative plant sources such as tapioca, yam, oat, sweet potato, barley, rye, taro, millet, amaranth, quinoa, legumes (faba beans, lentils, mung beans, peas, and chickpeas), and other plants including palms, chestnuts, acorns, arrowroot, bananas, breadfruit, buckwheat, sorghum, and water chestnuts can also be used for starch extraction [14,15,16].
In recent years, growing attention has been directed toward non-conventional sources of starch to support sustainable production. These include fruits such as peach palm, green banana, and banana kachkal [17,18,19,20,21]; pseudocereals such as buckwheat, amaranth, quinoa, canihua, lotus seed, and faba beans [22,23,24,25]; as well as culms and grasses [26] and plant pulps [20,27]. Starches obtained from these non-conventional sources may exhibit properties that are comparable to or distinct from those of conventional starches, offering opportunities to expand starch applications in food, pharmaceutical, and industrial sectors.
3. Formulation and Processing of Starch-Based Edible Coatings
The effectiveness of edible coatings (EC) in protecting fruits, vegetables, and meat products largely depends on the product’s wettability, which determines the uniformity of the coating layer [28]. Wettability, in turn, is influenced by both the surface characteristics of the food product and the chemical composition and structure of the coating-forming polymers, including the presence of additives such as plasticizers, surfactants, antimicrobials, and antioxidants [29]. Moreover, the effectiveness of applied coating materials is also affected by the application procedure employed, such as dipping, spraying, fluidized-bed, or pan coating, as each method interacts differently with the physical and chemical properties of the food surface. The selection of an appropriate coating technique depends on the nature of the food to be coated, its surface characteristics, and the primary purpose of the coating. Furthermore, the ripening characteristics of food products, including climacteric and non-climacteric behavior, should be taken into account when applying edible coatings [30].
3.1. Formulation of Coating Solution
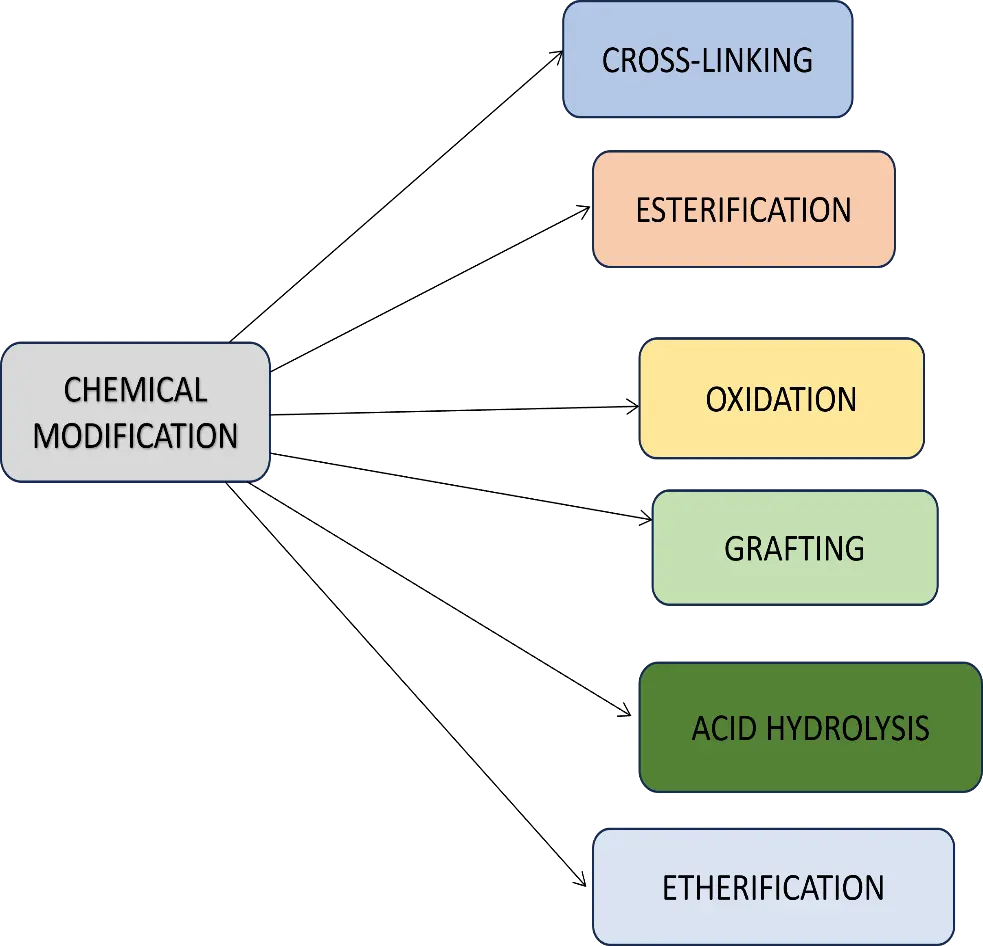
(a) Chemical Modification: The process involves the incorporation of functional groups into starch molecules, resulting in significant alterations in their physicochemical properties, such as pasting characteristics, gelatinization, and retrogradation behaviour (Figure 6). Chemical modification stabilizes the intramolecular and intermolecular bonds of starch granules, changing the physical properties of starch such as retrogradation, salting, and gelatinization. The primary methods for chemical modification of starch include oxidation using various oxidizing agents, etherification using hydroxyethyl, hydroxypropyl, or carboxymethyl to the starch’s hydroxyl groups, esterification using the condensation of certain fatty acids, other carboxylic acids, and phosphates with the active hydroxyl groups of starch, cationization using cationic molecules, cross-linking using different cross-linkers, and graft-polymerizing starch with artificial polymers as mentioned in Table 1. The effectiveness of this modification depends on several factors, including the source of starch and reaction conditions such as concentration, presence of a catalyst, pH, type of substituent, degree of substitution, and the distribution of substituents within the starch structure [31].
Cross-linking: Cross-linking is a widely used technique for modifying native starch in which chemical bonds are created between starch molecules, reinforcing the granule structure and improving hydrogen bonding. The efficiency of this process depends on several factors, including the chemical composition and concentration of the reagent, reaction time, pH, and temperature. Because the degree of cross-linking in food starch is usually very low and difficult to measure chemically, evaluating its physical properties is essential. Cross-linked starch generally exhibits slower retrogradation and higher gelatinization temperatures, as the limited mobility of amorphous chains within the granule restricts their movement [32]. In addition, cross-linking reduces the swelling of starch granules during cooking and may even inhibit gelatinization [33]. The effectiveness of starch cross-linking is also influenced by factors such as the starch source, the type and concentration of the cross-linking reagent, the degree of substitution, temperature, reaction time, and pH [34]. For food-grade starch, the FDA has approved cross-linking agents including adipic acetic mixed anhydride, phosphoryl chloride, sodium trimetaphosphate (STMP), and sodium tripolyphosphate (STPP). Cross-linking is one of the most commonly used techniques for modifying the properties of starch. It is widely used across various fields, including the food and pharmaceutical industries, wastewater treatment, and bioplastic production. Cross-linked starch with sodium trimetaphosphate (STMP) was prepared using the method given by [35] with some modifications.
Esterification: Esterification is an acid-catalyzed substitution reaction in which a nucleophilic acyl compound reacts with a molecule containing an acid anhydride, acid chloride, or carboxylic acid group. This reaction alters the hydroxyl groups in glucose units, thereby decreasing the hydrogen bonding potential of amylose and amylopectin [36]. The rate of esterification is heavily impacted by the structure of the reacting acids, especially their steric, mesomeric, and inductive effects. Additionally, factors such as molar ratio, temperature, and reaction time are crucial in influencing the efficiency of the esterification process [37]. Esterification as a chemical modification also observed improved hydrophobicity, swelling capacity, and lower retrogradation rate. As an example of organic esterification, succinylation increases the thickening power, viscosity, and water retention properties while reducing the gelatinization temperature and retrogradation [38].
Oxidation: Oxidation is a method used to modify starch by converting hydroxyl groups into carbonyl and carboxyl groups, mainly at the C2, C3, and C6 positions of the starch monomer. This process results in the depolymerization of amylose and amylopectin. Oxidative modification increases the hydrophilicity of starch, disrupts its molecular linearity and crystalline structure, and reduces enthalpy and retrogradation due to the formation of carbonyl and carboxyl groups in native starch through the action of oxidizing agents [39]. Different oxidizing agents, such as potassium permanganate, sodium hypochlorite, and hydrogen peroxide, are commonly used for this purpose. The extent and position of oxidation largely depend on factors such as pH, temperature, and the type of reagent used, with reactions mainly occurring at the hydroxyl groups at C2, C3, and C6, producing dialdehyde and dicarboxylic acid derivatives [37]. In recent years, ozone has gained attention as an oxidizing agent because it can react with starch without the need for a catalyst and causes minimal changes to the starch morphology, crystallinity, and internal structure [39].
Grafting: Grafting is a modification technique in which polymer chains (grafting onto) or monomers such as vinyl groups (grafting from) are introduced into starch, leading to the formation of copolymers that combine the properties of both the starch and the added polymer. This copolymerization mainly occurs in the presence of a free radical initiator, typically at the C1–C2 end groups and the C2–C3 positions of the starch molecule [37,40]. Radical initiation can occur through two primary approaches: chemical initiation using agents such as potassium permanganate or ceric ammonium nitrate, and radiation initiation through methods like microwave or UV irradiation. The properties of the resulting grafted starch depend on the type of polymer introduced, but generally include enhanced biodegradability, improved thermal stability, larger hydrodynamic radius, and better flocculation ability [41].
Acid hydrolysis: During acid treatment, hydronium ions attack the oxygen atom in the glycosidic bond, leading to the hydrolysis of that linkage. This modification changes the physicochemical properties of starch without significantly affecting its granule structure, and the resulting characteristics vary depending on the source of the starch [42]. Acid hydrolysis of starch is carried out at temperatures lower than its gelatinization temperature and influences properties such as solubility, pasting viscosity, gelatinization enthalpy, and swelling power. Acid-hydrolyzed starch is widely utilized as a gelling agent in foods such as candies and processed cheese bread. It also functions as a fat replacer in low-fat dairy and confectionery products, helping to reduce syneresis and enhance viscoelastic characteristics [43]. For example, mild hydrolysis does not result in any significant morphological changes, indicating that the starch granules have not undergone boiling [44].
Etherification: Etherification involves the substitution of hydroxyl groups in anhydro-glucose units with functional groups that may carry either positive or negative charges. This process can also lead to the formation of amphoteric and non-ionic starch derivatives. A common example of negatively charged starch is carboxymethyl starch, which is widely used in various industries because of its high hydrophilicity, cold water solubility, and effective flocculation properties, for example, waste water purification as biosorbents to sequestrate heavy metals [37,45].
Table 1. Modification of starch and its applications.
|
Modifications |
Description |
Industrial Sector |
Applications |
References |
|---|---|---|---|---|
|
Cross-linking |
Formation of inter and intra molecular bridges resulting from an interaction between reactive hydroxyl groups in starch and reagents |
Packaging |
Bioplastics |
[46] |
|
Esterification |
Condensation of the carboxylic acids, fatty acids or phosphates with reactive hydroxyl groups of the starch |
Food |
Used as an emulsion stabilizer in refrigerated and frozen food products |
[47] |
|
Oxidation |
It involves the oxidation of primary or secondary hydroxyl groups on the glucose units of starch, forming carbonyl or carboxyl groups with different oxidizing agents |
Food |
Used in batter and bread for coating various foodstuffs |
[48] |
|
Grafting |
Grafting of acrylic monomers with starch via free radicals generated through different free radical initiators |
Bioplastic |
Biodegradable plastics and films |
[41] |
|
Acid hydrolysis |
Hydrolysis of the glycosidic bond occurs due to the attack of hydronium ions on the oxygen atom within the glycosidic linkage |
Food |
Used as gelling agent in the production of gum and processed cheese loaves, fat replacers/fat mimetic |
|
|
Etherification |
Substitution of reactive hydroxyl groups in anhydro-glucose units with functional groups that carry either positive or negative charges |
Wastewater treatment |
Adsorbents of heavy metal ions |
[51] |
(b) Incorporation of Functional Additives: Edible films and coatings often contain additives that contribute desirable characteristics such as antioxidant capacity, antimicrobial activity, and prevention of browning. Additionally, additives are added to improve the rheological qualities, enhance their colour, flavour, and aroma, and to add nutraceuticals [52]. The use of nanoparticles in polymeric-based edible films and coatings can provide unique qualities to packaged products [53].
Antimicrobial agents: According to [54], antimicrobial coatings are composed of antimicrobial compounds that stop germs from developing on a substance’s surface. In order to maintain food safety, the use of coatings in food packaging helps to reduce or inhibit the introduction of pathogenic microbes like viruses and bacteria onto the food’s surface. In order to prevent bacterial and fungal invasion, certain biopolymers like cinnamon oil, Thymus vulgaris essential oil, quercetin starch, and guava leaf extract can be used to develop edible coatings. In traditional approaches, plant-derived extracts and essential oils are used in edible coatings to improve microbial stability by providing antibacterial and antifungal effects in fresh or minimally processed foods. According to a study of [55], black tea extracts with strong antibacterial properties are combined with gelatin-chitosan edible coating to extend the shelf life of minimally treated watermelons. Under refrigerated storage conditions, black tea extract was observed to delay fungal growth by up to 13 days. Physicochemical analysis further demonstrated that this approach effectively reduced weight loss, preserved texture, limited acidity development, and maintained the color and pH of watermelon. Moreover, plant-derived bioactive compounds influenced key properties of the edible coating, including water solubility, tensile strength, elastic modulus, transparency, thickness, and microstructural characteristics.
Antioxidant agents: Natural antioxidants have been utilized to make protective active barriers that can be integrated into food packaging films or applied directly to food items, i.e., edible coatings. It can also be incorporated directly into food. These barriers offer a novel approach to reducing the harmful effects of oxygen on food. Aromatic hydrocarbon–based antioxidants are generally classified into two main groups: synthetic and natural antioxidants. Synthetic antioxidants include compounds such as butylated hydroxytoluene (BHT) and butylated hydroxyanisole (BHA), whereas natural antioxidants include vitamins C and E. In addition, several other antioxidant substances can be incorporated into edible coating formulations, including organic acids such as oxalic, malic, succinic, and citric acids, as well as phenolic compounds like tannins and phenolic acids. Plant extracts rich in polyphenols, alkaloids, and flavonoids, as well as essential oils derived from cinnamon, ginger, and clove, are widely used for their antioxidant properties. Furthermore, agro-industrial by-products such as grape peel, mango peel, pomegranate peel, peanut shell, and rice husk have been identified as promising sources of natural antioxidants for edible coating applications. According to [56], chitosan films incorporated with mango leaf extract were developed to improve the oxidative stability of cashew nuts, which are rich in lipids. The results demonstrated that mango leaf extract acted as an effective antioxidant, maintaining the quality of cashew nuts for up to 28 days compared with conventional packaging materials. The primary role of antioxidants is to inhibit oxidative processes by neutralizing free radicals, complexing oxidation-promoting catalysts, preventing hydroperoxide decomposition, and blocking the initiation of oxidation reactions [57,58]. The application of edible materials as coatings on fruits and vegetables has been widely explored to extend their shelf life and preserve key quality attributes such as firmness, color, and overall freshness. According to recent studies, Arabic edible gum coatings with tulsi extract, which has a higher concentration of flavonoids and polyphenols, were effective in reducing the ripening rate in guava [59]. The packaging matrix, active chemicals, and food components are the three major components of antioxidant packaging films or coatings [60].
Plasticizers: Plasticizers are tiny, relatively stable particles, having low molecular weight, which provide strength and elasticity to coatings while simultaneously improving their capacity to withstand gases and water vapor. Typical plasticizers include polyols that can be used as plasticizers, including glycerol, mannitol, propylene glycol, honey, sucrose, and fatty acid esters. Agar-based films are made with glycerol, the most widely used plasticizer among others, because of their structural resemblance. Sorbitol, glycerol, and polyethylene glycol are examples of polyols that can be utilized as food-based plasticizers are sugars (glucose, fructose, and sucrose) and carbohydrates [61].
3.2. Coating Methods
Dipping: Dipping is the most common method of coating a food product. It is the process of immersing a food sample in a coating-forming dispersion [62]. The dipping method for applying edible coating to food products consists of three steps: immersion and dwelling, deposition, and solvent evaporation [63]. In the initial step, the substrate is immersed in the coating solution or emulsion at a uniform rate, allowing it to become completely wetted. This full immersion ensures sufficient contact and effective interaction between the coating matrix and the substrate surface [64]. The characteristics of the coating solution, particularly its density and viscosity, play an important role in determining the thickness of the coating layer. However, coating freshly cut fruits and vegetables can be difficult because of poor surface adhesion. Despite this limitation, the dipping method remains the most widely used technique due to its effectiveness in improving microbial stability and controlling browning in cut produce (Table 2). In this method, fruits and vegetables are immersed in a coating solution containing antimicrobial or antioxidant agents for about 5–30 s. Although it is simple, economical, and consistently applicable, the dipping process has several drawbacks, including potential respiration suppression in coated products, dilution and accumulation of waste in the dipping vat, microbial growth in the solution, and removal of the natural wax layer from the produce surface [5]. Contact time with the coating solution is a key factor; prolonged immersion may lead to moisture uptake, whereas short exposure can produce uneven coverage. Applying the coating in foam form can improve surface uniformity and help achieve a consistent layer thickness [8].
According to a study by [65], strawberries coated using the dipping method with CMC showed better control over weight and decay loss. Similarly, ref. [66] reported that garlic coated with an agar-agar-based coating containing chitosan and citric acid showed reduced respiration rate and moisture loss.
Spraying: The spraying technique is the best method for low-viscosity coating solutions that can be applied under high pressure. In this technique, an atomizer is used to spray the coating solution on the food sample. The food sample is covered with tiny droplets that range in size from micrometers to nanometers as the solution is sprayed at high pressure. The fluid’s rheological characteristics, such as temperature, viscosity, and surface temperature, determine how effectively spray coating is performed [5]. Therefore, a coating mixture with a proper fluid rheology is prepared and passed through the atomizer. After spraying, the coated sample is dried. The coating efficiency is determined by the drying technique, duration, and temperature [67]. In this method, the development of a polymeric coating is influenced by factors such as drying time and temperature [62,68]. The efficiency of spray-coating systems is strongly influenced by nozzle design and configuration. A variety of atomization techniques are applied in industrial processes, including pressure atomization, air-spray atomization, and air-assisted airless atomization, each characterized by distinct operational mechanisms and spray properties. Among these, pneumatic and hydraulic atomizing nozzles are the most widely adopted. More recently, electrospraying has attracted considerable attention as an advanced technique that employs a strong electric field to produce electrically charged droplets with precise size control, enabling the generation of micro- and submicron-scale droplets [8]. The findings of [69] indicated that the spray-coating technique facilitates the formation of a thin, homogeneous coating on mozzarella cheese, thereby improving surface coverage. The coated samples exhibited enhanced oxygen transfer rates, better control of water vapor transmission, and improved flavor retention. The main advantage of spraying is its ability to increase the coating’s surface area by generating fine droplets that evenly distribute over the product surface. Ref. [70] reported that fruits coated with pea starch and guar gum showed a reduced level of ethylene production. In addition, compared with uncoated fruit samples, the coated fruits exhibited significantly lower weight loss and better firmness retention.
Fluidized bed processing method: The first fluidized bed was discovered in the 1950s and named the “Wurster process”. It is a modified version of the fluidized bed dryer, which is a common drying method used in the food industry. This technique generally requires a larger quantity of coating solution compared with other coating methods. Among the different fluidized-bed coating approaches, including top-spray, rotary, and bottom-spray systems, the top-spray method is considered the most efficient [71]. However, despite its effectiveness, it is generally more expensive than alternative coating techniques. Its primary advantages include minimizing agglomeration and allowing precise control over the release rate of active compounds, thereby improving product quality and extending shelf life [72]. According to [73], fluidized bed coating was found to lower the amount of weight loss of ready-to-eat food while increasing its crispness and resistance to attrition. There is no cluster formation, and processing time is reduced when using a fluidized bed processing method.
Panning method: In this technique, a large rotating, pan-like container is utilized to coat the food material. Food products are placed in a pan, and the coating solution is applied to their surface using a spray gun installed within the coating chamber. During the process, the pan rotates continuously at a controlled speed to ensure uniform distribution of the coating material [5]. Following the coating stage, the products are transferred to a drying phase, where hot air is circulated to achieve complete drying. This method is particularly effective for coating food items with round or oval shapes [71].
Table 2. Polymers and their application methods.
|
Polymers |
Food Products |
Coating Method |
References |
|---|---|---|---|
|
Mango kernel starch |
Tomato |
Dipping |
[74] |
|
Chitosan nanoparticles |
Tomato, chilly, brinjal |
Dipping |
[75] |
|
Pea starch/Guar gum |
Valencia’ orange |
Spraying |
[70] |
|
Chitosan + Oxidised starch |
Papaya |
Dipping |
[76] |
|
Tapioca starch/decolourized Hsian-tsao leaf gum |
Fruit based salads, romaine hearts and pork slices |
Spraying |
[77] |
|
Aloe Vera |
Tomato |
Dipping |
[78] |
|
Alginate |
Watermelon |
Dipping |
[58] |
|
Hydrolysed collagen |
Rice crisp balls |
Panning |
[79] |
|
Alginate and chitosan |
Bell pepper |
Spraying |
[80] |
4. Functional Properties of Starch-Based Coatings
Barrier Properties: The efficiency of an edible coating’s barrier properties is important for maintaining the quality and safety of a food product. The efficiency of an edible coating’s barrier properties is important for maintaining the quality and safety of a food product. Commonly used polysaccharides for edible coatings are chitosan, starch, alginate, pectin, and cellulose. Starch mainly comprises amylose and amylopectin, making it suitable for the production of edible coatings due to its mechanical and barrier properties (Figure 7). The hydrogen bonding of starch shows an excellent gas barrier property for both CO2 and O2 [81].
Moisture Barrier: Starch based edible films and coatings improve the texture and appearance of food products and extend their shelf life by forming semi-permeable barriers that stop moisture loss, aroma loss, water absorption by the food material, or even oxygen penetration. Due to the hydrophilic nature of starch, they have a poor moisture barrier property [82]. According to the study of [83], the efficiency and functional behaviour of polysaccharide-based coatings mainly depend on the use of plasticizers, relative humidity, and the pH level.
Oxygen Barrier: Starch-based coatings are characterized by their efficient oxygen barrier property because of their hydrogen-bonding structure, their colorless and oil-free appearance, and their minimum caloric content, making it suitable for the surface application on fruits & vegetables [84]. Therefore, the presence of oxygen can have detrimental effects on some packaged fresh food products. It has been found that certain edible films and coatings include humidity control systems and oxygen scavengers, both play an essential role in reducing the gases that cause fruits and vegetables to deteriorate. According to [85], the coating of chitosan/cellulose nano-fibril minimizes the oxygen diffusion, decreases respiration, and delays strawberry oxidation by ascorbic acid reaction. For example: according to study of [86], coating of perishable fruits (orange, mango, strawberry, and cherry) by the dipping method with a biomimetic hybrid material derived from shellac membrane by incorporating chitosan/poly(L-lactic acid) PLLA microspheres could improve the fruit’s shelf life and control gas permeability of oxygen, carbon dioxide.
Optical and Surface Properties: Consumers intention to purchase and the product’s appearance can both be significantly affected by the optical properties of food packaging materials [87]. Evaluation of optical properties is crucial in the development of food packaging materials. Among these properties, color and transparency are particularly important for biopolymer-based packaging systems. The color of films and coatings is usually expressed using color parameters (L*, a*, and b*) and the total color difference (ΔE), which represents the overall change in color calculated from these parameters (Lee et al., 2020). The incorporation of plant oils (POs) often results in edible films and coatings that are more coloured and less transparent. For instance, the addition of walnut and almond oils increased both the opacity and total color difference of films made from whey protein isolate [88]. Moreover, ref. [89] reported that surface modification of paperboard using a mineral–starch-based solution applied through an electro-spraying method, along with an additional layer of polylactide (PLA), resulted in microscale surface roughness.
Biodegradability and Environmental Impact: The starch films and coatings showed high biodegradability under burial conditions in soil. Pseudomonas aeruginosa and Bacillus subtilis are two examples of environmental bacterial strains that can efficiently degrade starch molecules [90]. According to some studies, the addition of phenolic extracts and compounds increased the biodegradability of starch films and coatings. For example, the addition of purple passion fruit peel anthocyanin extracts had little effect on the biodegradability of Pouteria campechiana seed starch-based films [91]. However, the addition of phenolic extracts and compounds to starch-based films and coatings enhances their biodegradability, making them suitable for environmentally friendly applications. Microbe-film interactions may be facilitated by loosened structures with broken surfaces caused by phenolic addition, whereas compacted structures with smooth surfaces caused by the addition may retard the interaction. However, it can be said that starch-based films and coatings with added phenolic compounds and extracts exhibit high biodegradability for environmentally friendly applications.
5. Applications in Food Systems
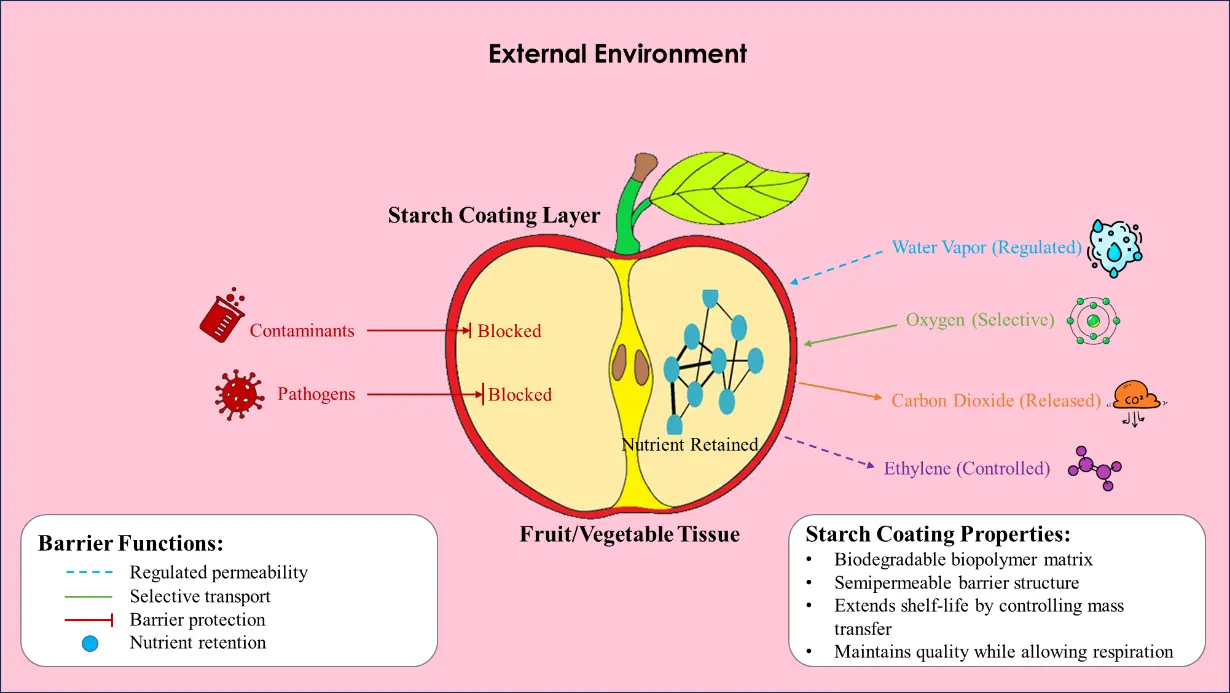
Fruits and vegetables: Starch coatings applied to the surface of fruits and vegetables act as a barrier, selectively regulating the transfer of substances between the produce and the external environment (Figure 8). These coatings adhere well to fruit surfaces and do not undergo fermentation, coagulation, or negatively affect their edible quality. Freshly harvested fruits and vegetables are prone to varying degrees of microbial spoilage, often caused by mechanical damage and handling during transportation. Numerous studies have shown that applying antifungal edible coatings can effectively reduce microbial infection in fresh produce. Ref. [81] recently reported that an antifungal starch-based coating significantly reduced black spot rot caused by Alternaria alternata in Asian pears stored for 21 days, which was attributed to the antimicrobial components present in the coating formulation. In addition, the stomata densities and physiological activities of ‘early crisp’ were preserved after being treated with starch-based coating. The authors suggested that the favourable outcomes were due to the starch coating’s ability to regulate substance exchange along with the moisture-barrier effect exerted by the incorporated antimicrobial agents. Ref. [28] studied that incorporating functional ingredients into biopolymers is an innovative approach to reduce hygroscopic character while enhancing antimicrobial performance. Overall, the use of antimicrobial edible coatings offers multiple benefits and can help reduce reliance on agrochemicals and other synthetic substances for preserving fruits and vegetables. Preserving the quality of freshly harvested produce is difficult because of rapid senescence and ongoing biochemical changes. Applying starch-based coatings to fruits and vegetables can improve their storage life and help minimize postharvest losses. These studies demonstrate that antimicrobial starch coatings can effectively control physicochemical changes and metabolic processes in fruits and vegetables, making this technology valuable for fresh produce growers and distributors. Sensory characteristics such as color, taste, aroma, texture, and overall quality acceptability are key factors that influence consumer preference and purchasing decisions [92]. Starch edible coatings are colorless, odourless, and tasteless, and they do not adversely affect the quality or commercial value of the product. Moreover, starch-based coatings possess a strong ability to carry functional ingredients, which can deliver additional nutritional benefits to consumers when the coated product is consumed. A study evaluated the impact of coating formulated from bacterial cellulose and starch based composite coatings on tomatoes. A storage study of coated tomato samples demonstrated enhanced firmness and sensory attributes. In addition, titrable acidity, ascorbic acid content, total plate count, total soluble solids, and weight loss showed better retention in coated samples than in non-coated (control) samples. The study showed that all the starch-based coating formulations could increase the shelf-life and preserve the quality parameters of tomatoes [93]. However, careful consideration must be given to the type and concentration of antimicrobial agents used in edible coatings, as some possess strong odors and may negatively affect the sensory qualities of fruits and vegetables if not properly optimized.
Meat and Seafood Products: The incorporation of biopolymers containing active components such as natural extracts and essential oils can significantly improve the preservation of fresh meat and meat products. These active compounds help reduce moisture loss while inhibiting microbial growth and lipid oxidation. Ref. [94] developed maize starch films incorporated with grape juice at volume ratios of grape juice to water ranging from 10:90 to 50:50. The results indicated that the film effectively reduced lipid oxidation and extended the shelf life of chicken breast fillets. Microbiological analysis supported these findings, showing that the Enterobacteriaceae count in coated chicken breast fillets was 7.21 ± 0.04 log CFU g−1, whereas uncoated samples recorded 8.06 ± 0.23 log CFU g−1. The lower microbial growth during eight days of storage at 4 °C was attributed to the barrier and antioxidant properties of the grape-juice-incorporated film. Ref. [95] also incorporated natural extracts into rice starch films by adding curcumin extract. These films were applied as coatings for chicken and fish meat to detect hypoxanthine (HX), a biochemical indicator used to assess meat spoilage. The results revealed that fish contained a higher HX concentration (52.84 µM) compared to chicken meat (30.25 µM). It was also reported that the storage stability of the films improved with increasing curcumin concentrations (1, 2, and 3%). This finding suggests that curcumin-containing films can serve as indicators of meat freshness, although low-temperature storage is necessary to maintain their physicochemical stability. In another study, ref. [96] incorporated apple peel pectin extract, ZrO2 nanoparticles, and Zataria multiflora essential oil into potato starch composite films. These films were used as packaging for quail meat, and microbial counts increased during 12 days of storage at 4 °C. However, the potato starch–pectin film without essential oil (MEO) and ZrO2 nanoparticles showed a higher microbial count compared with films containing these additives. Among the tested films, the one containing MEO exhibited the strongest antimicrobial effect and provided greater microbial stability for quail meat. These findings demonstrate that the addition of essential oils enhances the performance of active films due to their strong antibacterial properties. Similar observations were also reported by [97]. They added Artemisia annua essential oil on coix seed starch-based films to test pork preservation. Edible coatings have been developed from sweet potato starch and thyme essential oils for coating shrimp, and the physical properties (color and texture) of the products were determined. The L value significantly increased with the increase of EOs concentration and decreased during the storage period. However, samples coated with EOs maintained higher L values than the control at the end of the storage period. Moreover, incorporation of EOs resulted in higher hardness, cohesiveness, gumminess, springiness, chewiness, and resilience values throughout the storage time than the uncoated samples [98]. Shrimp preservation using cassava starch-chitosan composite coatings has shown remarkable results in maintaining texture and preventing melanosis (black spot formation). These coatings create an effective barrier against oxygen and moisture while providing antimicrobial protection through incorporated natural preservatives.
Bakery and Confectionery: Starch-based coatings have shown significant potential in increasing the specific volume of bread, a major quality issue in the bakery industry. Modified corn starch coatings with ascorbic acid and tomato powder were applied to the dough. Ascorbic acid in the modification of the starch contributed to the increase of the specific volume of bread, while the tomato powder showed greater influence on crust browning index [99]. Gluten-free bakery products particularly benefit from starch-based coatings, as they naturally lack the protein network that provides structure and moisture retention in conventional breads. Rice starch-xanthan gum coatings have demonstrated effectiveness in maintaining product integrity and extending shelf life in gluten-free applications. The addition of xanthan gum decreased crumb hardness of fresh and stored rice flour-based breads, and the combination of xanthan and rice starch increased peak viscosities while improving textural properties [100]. These hydrocolloid combinations help compensate for the lack of gluten’s structure-forming properties in bakery applications. Candy coating applications represent a growing segment of the starch-based edible coating market. These coatings provide alternatives to shellac-based glazes traditionally used in confectionery. Modified corn starch formulations can achieve desired gloss and barrier properties while meeting clean-label requirements increasingly demanded by consumers [101]. Chocolate applications focus on preventing fat bloom by using starch coatings that create barriers that control fat migration to product surfaces [102]. Sugar confectionery benefits from starch coatings that prevent moisture uptake and subsequent texture changes during storage.
Dairy Products: Starch-based edible coatings present sustainable alternatives to synthetic wax coatings traditionally used in cheese preservation. Modified corn starch formulations have been successfully applied to semi-hard cheeses, providing effective moisture barrier properties while allowing controlled gas exchange necessary for proper aging processes [103]. Another research demonstrates that edible films and coatings reinforced with rice bran and containing nisin and natamycin are an effective method to control the post-process and external contamination of Argentinian Port Salut cheese. These matrices can successfully contribute to improving the cheese microbial stability, offering the consumer a safer food product. The developed films and coatings can be incorporated into the end of the manufacturing process, thus contributing to better cheese preservation during storage by applying an environmentally friendly treatment [104]. Yogurt container applications utilize starch-based edible films as sustainable alternatives to conventional plastic linings. These applications require careful pH compatibility considerations, as dairy products’ acidic nature can affect coating integrity [105].
Commercial Applications: Edible coating can be applied on the fruits and vegetables at different levels in the supply chain at commercial level. Multiple scenarios are possible, for example, edible coating can be applied at the production level prior to collection or at the packaging level prior to export of the food product. It may be beneficial to apply the coating before exportation in order to reduce spoilage during transit. If the exportation of food products is relatively short and quick and does not show high risk of post-harvest damage, the coating may be applied at the retailer level to extend shelf-life of the product. Investment in the application of edible coating technology greatly depends on the processing level, i.e., large or semi-industrial scale industries, while scaling up edible coating application at the industrial level involves a number of challenges that must be resolved [106]. For example, washing fruits in water containing detergent before coating might remove the natural protective layer present on the surface of some fruits, and surface dehydration and moisture loss might happen during drying [107]. According to a comprehensive review recently published by [108] showed that dipping was the most commonly used technique for the application of edible coatings. However, at the commercial level, the spraying method is regularly used for application. Therefore, scientific studies play an important role in the assessment of edible coatings application during scaling up. It might be argued that the lack of scientific information regarding the use of spraying methods for the application of edible coatings on fruits and vegetables represents a research gap. A number of biopolymers have received scientific consideration for the development of edible coatings. Moreover, several companies providing postharvest solutions, such as Fomesa Fruitech and Decco, have launched new edible coatings. For example, GreenSeal, Naturcover, and GreenGard-LE, based on biopolymers, hydrocolloids, edible emulsifiers, fatty acids, and natural waxes, have been commercialized [109]. The cost of production of edible coating depends on a number of factors, including the source of biopolymer, extraction method, supply, storage, and drying of raw materials, and standardization of the biopolymer [110]. The benefits of uncoated and coated fruits or vegetables are fundamental to ensure sustainable growth and demand. The ability of edible coatings to preserve the sensory properties, freshness, and nutritional value of fruits, as well as extend shelf life, thus reducing waste, is a decisive factor [111].
Comparison with Other Biopolymer Coatings
Starch-based coatings have emerged as prominent candidates in sustainable food packaging, yet their comparative performance against other biopolymer systems requires systematic evaluation. Among polysaccharides, proteins, lipids, and composite materials used in food preservation, starch biopolymers stand out for their affordability, availability, and excellent film-forming properties [112]. However, starch exhibits some limitations as a stand-alone film material, such as inferior barrier and mechanical properties compared to its commercial plastic counterparts [113]. When compared to chitosan-based systems, starch coatings demonstrate distinct advantages in cost-effectiveness and processing simplicity, while chitosan composite films often exhibit superior mechanical strength and enhanced barrier performance compared to pure chitosan film [114]. Protein-based alternatives, especially soy protein films, exhibit outstanding barrier properties. Soy-protein-based films demonstrate oxygen permeability that is 260-, 540-, 500-, and 670-fold lower than that of low-density polyethylene systems, thereby outperforming starch-based coatings in this key functional attribute [115] (Jahangiri et al., 2024). Cellulose, starch, chitosan, polylactic acid, and polyhydroxyalkanoates are seen as promising alternatives to conventional plastics in food packaging. However, the application of these biopolymers in the food packaging industry on a commercial scale is limited due to their poor performance [116], highlighting the need for comprehensive comparative studies to optimize material selection for specific applications (Table 3).
Table 3. Performance Comparison Matrix of Starch-based Coating with Other Biopolymers.
|
Property |
Starch-Based |
Chitosan-Based |
Protein-Based |
Cellulose-Based |
References |
|---|---|---|---|---|---|
|
Barrier Properties |
|||||
|
Water Vapor Permeability (g·m·m−2·s−1·Pa−1) |
8 × 10−10–12 × 10−10 |
3 × 10−11–7 × 10−11 |
1.5 × 10−11–6 × 10−11 |
5 × 10−12–15 × 10−12 |
|
|
Oxygen Permeability (cm3·μm·m−2·day−1·atm−1) |
8–45 |
2–18 |
0.05–2.5 |
0.01–0.15 |
|
|
Oil/Fat Barrier |
Poor-Moderate |
Good |
Excellent |
Good-Excellent |
|
|
Mechanical Properties |
|||||
|
Tensile Strength (MPa) |
6–18 |
25–55 |
12–45 |
45–180 |
|
|
Elongation at Break (%) |
35–85 |
12–35 |
25–120 |
3–12 |
|
|
Young’s Modulus (MPa) |
120–600 |
800–2800 |
250–1200 |
1800–12,000 |
[121] |
|
Functional Properties |
|||||
|
Intrinsic Antimicrobial Activity |
None |
Excellent |
Limited |
None |
[122] |
|
Antimicrobial Agent Carrier |
Good |
Excellent |
Good-Excellent |
Moderate |
[123] |
|
Transparency |
Excellent |
Good-Very Good |
Good-Excellent |
Good |
[124] |
|
Colour |
Colorless |
Slight yellow |
Variable |
Colorless-Slight yellow |
[124] |
|
Odor/Taste |
None-Minimal |
Slight |
Variable |
None |
[124] |
|
Processing Characteristics |
|||||
|
Processing Temperature (°C) |
65–90 |
55–85 |
75–100 |
80–110 |
|
|
pH Requirements |
5.5–8.5 |
3–5 |
6–10 |
6.5–9 |
|
|
Solvent |
Water/ethanol |
Acidic water |
Water/alcohol |
Water/organic |
[127] |
|
Economic Factors |
|||||
|
Raw Material Cost ($/kg) |
0.8–2.2 |
18–35 |
4–12 |
3–9 |
[127] |
|
Availability |
Excellent |
Moderate |
Good |
Good |
[128] |
|
Environmental Impact |
|||||
|
Biodegradation Time (days) |
15–35 |
45–75 |
25–55 |
35–70 |
[127] |
|
Compostability |
Excellent |
Good |
Good-Excellent |
Excellent |
[129] |
|
Renewable Source |
Yes |
Yes |
Yes |
Yes |
[129] |
6. Advancement in Starch-Based Coating Technology
6.1. Nanocomposite Starch Coating
The integration of nanoparticles into starch-based coating matrices represents a significant advancement in biopolymer coating technology, addressing the inherent limitations of pure starch films while maintaining their biodegradable characteristics. Nanocellulose incorporation has emerged as a particularly promising approach, with its one-dimensional structure providing excellent reinforcement properties, renewability, degradability, and good biocompatibility, indicating promising applications in food packaging. Recent studies have demonstrated that starch solutions containing cellulose nanocrystals (CNC) at 0.3% concentration significantly improve paper strength and resistance to air permeation when applied through size press operations [130]. The incorporation of clay nanoparticles, particularly montmorillonite, has shown remarkable improvements in barrier properties and mechanical strength of starch-based coatings. Novel biodegradable starch/clay nanocomposite films have been successfully developed by homogeneously dispersing montmorillonite nanoparticles in starch-based materials via polymer melt processing techniques, resulting in enhanced structural and mechanical properties [131].
Beyond traditional nano-clays, the incorporation of metal oxide nanoparticles has opened new avenues for functional starch coatings with enhanced properties. Research has shown that starch films incorporating 0.01% w/w of titanium dioxide (TiO2) nanoparticles can extend the shelf life of tomatoes from 7–10 days to 21 days, demonstrating significant practical applications in active packaging systems [132]. The synergistic effects of combining different nanoparticles have also been explored, where TiO2 functionalized with alkoxysilane and decorated with silver nanodots in starch-chitosan films showed a 33% improvement in tensile strength while simultaneously reinforcing antimicrobial properties. The development of TiO2/starch-based biodegradable copolymer nanocomposites has further demonstrated successful grafting of acrylamide over starch, with enhanced thermal and biological properties. These nanocomposite approaches effectively address the traditional weaknesses of starch coatings, including poor mechanical properties and high-water sensitivity, while introducing additional functionalities such as antimicrobial activity, UV protection, and controlled release properties, positioning nanocomposite starch coatings as next-generation sustainable packaging solutions for diverse food applications.
6.2. pH-Sensitive and Intelligent Coating
pH-sensitive starch-based coatings have emerged as revolutionary intelligent packaging systems capable of real-time food quality monitoring. The film containing 15 mg/g of betacyanin showed a color change from light pink to yellow as a response to pH adjustment between 2 to 13. It was also able to detect the presence of ammonia in a range of 0.1 and 0.01 mg of ammonia per ml of water [133]. This pioneering work demonstrates the dual functionality of starch-based systems in monitoring both pH changes and specific volatile compounds associated with food spoilage. Recent developments have focused on novel starch sources for enhanced pH sensitivity. A pH-sensitive intelligent packaging film harnessing Dioscorea zingiberensis starch and anthocyanin for meat freshness monitoring represents a significant advancement in utilizing underexploited starch resources [134]. This approach not only provides effective pH monitoring but also promotes sustainable utilization of alternative starch sources. The incorporation of natural pH-sensitive pigments into starch matrices has become a cornerstone of intelligent coating technology. Recently, the assimilation of pH-sensitive natural pigments into biopolymers has shown promising prospects for pH-reactive based smart packaging material. Unlike synthetic pigments, natural alternatives offer enhanced safety profiles and biodegradability [135]. Manufacturing advances have enabled scalable production of these intelligent systems. Production of sustainable smart packaging based on cassava starch and a natural pH indicator (anthocyanin) by a twin-screw extrusion process demonstrates industrial feasibility for commercial applications [136]. This extrusion-based approach ensures the uniform distribution of pH-sensitive components while maintaining cost-effectiveness. With the increasing demand for environmentally friendly, safe, preservative, and intelligent food packaging, there is a growing trend towards using plant-derived natural colorants that possess green, non-toxic, antioxidant, antibacterial, and pH-sensitive properties, positioning starch-based pH-sensitive coatings at the forefront of sustainable intelligent packaging solutions.
6.3. Starch Blends with Other Polymers
The incorporation of starch into polymer blends has emerged as a pivotal strategy for advancing biodegradable coating technology in food packaging applications, addressing both environmental sustainability and functional performance requirements. Starch-polyvinyl alcohol (PVA) blend films demonstrate enhanced barrier properties, water resistance, oxidation resistance, and antibacterial properties, making them promising candidates for food preservation applications [137]. The synergistic effects of these blends have been particularly evident in ternary systems, where composite films based on maize starch, low molecular weight chitosan, and PVA have shown improved structural features and functional characteristics for multi-purpose food packaging applications [138]. Recent investigations into starch-chitosan combinations have revealed that increasing chitosan content in glutinous rice starch/chitosan blends enhances mechanical strength, hydrophobicity, and resistance to Colletotrichum gloeosporioides infection, effectively overcoming the inherent limitations of pure starch films [139]. Moreover, combining poly (lactic acid) with starch has demonstrated significant potential, as such polymer blends can address the poor degradability of conventional synthetic polymers in food packaging while still preserving suitable performance properties [140]. The advancement of these starch-based coating technologies has been further enhanced by innovative approaches such as electrospinning, in which starch blended with polymers like polycaprolactone, polyvinyl alcohol, and polyglycolide forms nanofibers with applications in food packaging [141]. These developments collectively demonstrate that strategic polymer blending represents a viable pathway for creating high-performance, environmentally sustainable coating solutions that address the growing demand for biodegradable alternatives in food packaging applications.
6.4. Active Packaging and Smart Functionalities
The integration of active and smart functionalities into starch-based coating systems represents a transformative advancement in food packaging technology, providing dynamic interaction with packaged foods through controlled release mechanisms and real-time quality monitoring capabilities. Active packaging interacts with the contents and the environment by acting as a scavenger (O2, CO2, ethylene) or releasing agent (antimicrobial substance and antioxidant), effectively extending shelf life while maintaining food quality and safety [142,143]. Recent developments have demonstrated that starch-based films incorporating phenolic compounds not only provide enhanced antioxidant properties but also serve as intelligent packaging systems when formulated with anthocyanins to reflect the degree of food freshness through visual color changes [144]. The emergence of pH-sensitive intelligent packaging films utilizing natural pigments, particularly anthocyanins integrated into starch matrices, has enabled real-time monitoring of food deterioration through distinct colorimetric responses that correlate with spoilage indicators [145]. The smart functionalities have been successfully applied across diverse food categories, with multifunctional intelligent films incorporating purple sweet potato anthocyanin and quercetin-loaded chitosan nanoparticles demonstrating exceptional performance in monitoring and maintaining the freshness of perishable products such as shrimp [135]. Furthermore, innovative approaches have led to the development of high-sensitivity intelligent packaging systems that harness rose anthocyanins combined with hydrophilic silica aerogel, significantly enhancing the visual detection capabilities for food freshness monitoring applications [146]. The advancement of these intelligent packaging technologies has been particularly notable in meat and seafood applications, where natural pigment-based pH/gas-sensitive films provide comprehensive freshness monitoring while ensuring consumer safety and reducing economic losses associated with food spoilage [147]. These developments collectively position starch-based active and smart packaging as a sustainable and technologically advanced solution that addresses the growing consumer demand for real-time food quality assurance while contributing to waste reduction in the global food supply chain.
7. Safety Concerns, Regulatory Aspects, and Consumer Acceptability of Starch-Based Coating Technology
Advancement in starch-based coating technology has transformed the safety and regulatory landscape for food packaging applications. Modern starch coatings demonstrate superior barrier properties against oxygen and moisture while maintaining complete biodegradability within 90 days under industrial composting conditions. Regulatory bodies, including the FDA and European Food Safety Authority, have streamlined approval processes for starch-based materials, recognizing their proven safety profile through extensive migration testing that shows zero harmful compound transfer into food products. However, consumer acceptance remains the critical bottleneck, as 68% of consumers express concern about altered food texture and reduced shelf life, despite technological improvements that now achieve 95% moisture barrier efficiency, comparable to that of conventional plastic films. Advanced cross-linking techniques have eliminated the brittleness issues that plagued earlier starch coatings, while new antimicrobial additives extend product freshness by 40% compared to uncoated alternatives. The convergence of enhanced performance characteristics, streamlined regulatory pathways, and growing environmental awareness has positioned advanced starch coatings as commercially viable alternatives, though widespread market adoption hinges on continued consumer education and demonstration of long-term food safety benefits that match or exceed traditional packaging performance standards.
8. Challenges and Limitations
Despite the promising potential of starch-based coatings for sustainable food packaging applications, several significant challenges and limitations continue to impede their widespread commercial adoption and industrial implementation. Starch exhibits inherent mechanical weakness and permeability to moisture and gases, with inferior barrier and mechanical properties compared to conventional plastic counterparts, fundamentally limiting its standalone application in demanding packaging environments [148]. The most critical limitations include low mechanical strength, moisture sensitivity, and limited barrier properties, which compromise the protective function essential for food preservation [112]. The inherent brittleness, fragility, and water solubility of starch-based membranes present substantial challenges in practical applications where structural integrity is paramount [149]. From an industrial scalability perspective, most studies focus on laboratory stages of formulation and plasticization, leaving significant gaps in understanding key phases such as raw material conditioning, industrial-scale molding, post-production processes, and storage [150]. Starch-based bioplastics face a stark economic reality: production costs are 40–60% higher than those of traditional plastics, creating an immediate market barrier that competitors exploit ruthlessly. These industrial challenges amplify this cost crisis through unpredictably fluctuating raw material prices that sometimes double within six months, fragile supply chains with single-source dependencies that create bottlenecks, and complex regulatory frameworks demanding expensive compliance infrastructure that smaller manufacturers cannot afford. Consumers present the final hurdle as starch-based packaging deteriorates 30% faster than conventional alternatives, with altered texture and reduced durability clashing against consumer expectations built over decades, causing shoppers to abandon eco-friendly brands immediately when they encounter soggy packaging or crushed products. The convergence of these factors has trapped the industry where moisture sensitivity destroys product integrity, poor mechanical strength limits application scope, and regulatory uncertainty prevents long-term investment, making widespread adoption unlikely without breakthrough innovations in material science and strategic market positioning.
9. Conclusions
Starch-based edible coatings represent a promising and sustainable alternative to conventional synthetic packaging, aligning with the growing global emphasis on environmental preservation and circular economy principles. Their biodegradability, renewability, and non-toxic nature make them ideal candidates for reducing plastic waste and minimizing ecological impact. The incorporation of bioactive compounds and nanomaterials through green synthesis approaches has further enhanced their functional and mechanical properties, enabling improved barrier performance, antimicrobial activity, and food quality preservation. However, challenges related to moisture sensitivity, mechanical stability, and large-scale production must be addressed through innovative formulation strategies and advanced processing techniques. Future research should focus on optimizing starch modification methods, integrating sustainable cross-linking agents, and developing scalable green technologies to ensure cost-effectiveness and industrial applicability. Starch-based edible coatings hold immense potential as a key component of sustainable food packaging systems, contributing to food security, waste reduction, and a cleaner, greener future.
Author Contributions
Conceptualization, P.S.; Methodology, P.S., A.Y.; Formal Analysis, A.Y. and M.A.; Investigation, A.Y., M.A.; Resources A.Y., M.A., P.Y.; Data Curation, A.Y. and M.A.; Writing—Original Draft Preparation, P.Y.; Writing—Review & Editing A.Y., M.A.; Visualization, P.S. Supervision, P.S.
Ethics Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not Applicable.
Funding
This research received no external funding.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
-
Ibrahim ID, Hamam Y, Sadiku ER, Ndambuki JM, Kupolati WK, Jamiru T, et al. Need for sustainable packaging: An overview. Polymers 2022, 14, 4430. DOI:10.3390/polym14204430 [Google Scholar]
-
Thapliyal D, Karale M, Diwan V, Kumra S, Arya RK, Verros GD. Current status of sustainable food packaging regulations: Global perspective. Sustainability 2024, 16, 5554. DOI:10.3390/su16135554 [Google Scholar]
-
Ncube LK, Ude AU, Ogunmuyiwa EN, Zulkifli R, Beas IN. Environmental impact of food packaging materials: A review of contemporary development from conventional plastics to Polylactic Acid based materials. Materials 2020, 13, 4994. DOI:10.3390/ma13214994 [Google Scholar]
-
Rafey A, Siddiqui FZ. A review of Plastic Waste Management in India—Challenges and opportunities. Int. J. Environ. Anal. Chem. 2021, 103, 3971–3987. DOI:10.1080/03067319.2021.1917560 [Google Scholar]
-
Suhag R, Kumar N, Petkoska AT, Upadhyay A. Film formation and deposition methods of edible coating on food products: A review. Food Res. Int. 2020, 136, 109582. DOI:10.1016/j.foodres.2020.109582 [Google Scholar]
-
Rohasmizah H, Azizah M. Pectin-based edible coatings and nanoemulsion for the preservation of fruits and vegetables: A review. Appl. Food Res. 2022, 2, 100221. DOI:10.1016/j.afres.2022.100221 [Google Scholar]
-
Salgado PR, Ortiz CM, Musso YS, Di Giorgio L, Mauri AN. Edible Films and Coatings Containing Bioactives. Curr. Opin. Food Sci. 2015, 5, 86–92. DOI:10.1016/J.COFS.2015.09.004 [Google Scholar]
-
Gupta D, Lall A, Kumar S, Patil TD, Gaikwad KK. Plant-based edible films and coatings for food-packaging applications: Recent advances, applications, and trends. Sustain. Food Technol. 2024, 2, 1428–1455. DOI:10.1039/D4FB00110A [Google Scholar]
-
Alcazar-Alay SC, Meireles MAA. Physicochemical properties, modifications and applications of starches from different botanical sources. Food Sci. Technol. 2015, 35, 215–236. DOI:10.1590/1678-457X.6749 [Google Scholar]
-
Durrani CM, Donald AM. Physical characterization of amylopectin gels. Polym. Gels Netw. 1995, 3, 1–27. DOI:10.1016/0966-7822(94)00005-R [Google Scholar]
-
Phukan M, Nongkhlaw FT. Starch: An overview of its conventional and unconventional sources, pharmaceutical and industrial applications. Curr Trends Pharm Res 2022, 9, 140–161. [Google Scholar]
-
de Oliveira MM, Lago A, Pinto NGM, de Oliveira Araujo C, Velho JP. Systematic review of innovations in food packaging with a focus on circularity and the reduction of food loss and waste. Discov. Appl. Sci. 2025, 7, 1133. DOI:10.1007/s42452-025-07788-3 [Google Scholar]
-
Bergo P, Moraes ICF, Sobral PJA. Effects of plasticizer concentration and type on moisture content in gelatin films. Food Hydrocoll. 2013, 32, 412–415. DOI:10.1016/j.foodhyd.2013.01.015 [Google Scholar]
-
Araujo-Farro PC, Podadera G, Sobral PJA, Menegalli FC. Development of films based on quinoa (Chenopodium quinoa, Willdenow) starch. Carbohydr. Polym. 2010, 81, 839–848. DOI:10.1016/j.carbpol.2010.03.051 [Google Scholar]
-
Piyada K, Waranyou S, Thawien W. Mechanical, thermal and structural properties of rice starch films reinforced with rice starch nanocrystals. Int. Food Res. J. 2013, 20, 439. Available online: http://www.ifrj.upm.edu.my/20%20(01)%202013/61%20IFRJ%2020%20(01)%202013%20Thawien%20(369).pdf (accessed on 17 September 2025).
-
Da Rosa Zavareze E, Storck CR, de Castro LAS, Schirmer MA, Dias ARG. Effect of heat-moisture treatment on rice starch of varying amylose content. Food Chem. 2010, 121, 358–365. DOI:10.1016/j.foodchem.2009.12.036 [Google Scholar]
-
Oderinde AA, Ibikunle AA, Bakre LG, Babarinde NAA. Modification of African breadfruit (Treculia africana, Decne) kernel starch: Physicochemical, morphological, pasting, and thermal properties. Int. J. Biol. Macromol. 2020, 153, 79–87. DOI:10.1016/j.ijbiomac.2020.02.293 [Google Scholar]
-
Patino-Rodríguez O, Agama-Acevedo E, Ramos-Lopez G, & Bello-Pérez LA. Unripe mango kernel starch: Partial characterization. Food Hydrocoll. 2020, 101, 105512. DOI:10.1016/j.foodhyd.2019.105512 [Google Scholar]
-
Guo K, Lin L, Fan X, Zhang L, Wei C. Comparison of structural and functional properties of starches from five fruit kernels. Food Chem. 2018, 257, 75–82. DOI:10.1016/j.foodchem.2018.03.004 [Google Scholar]
-
Fontes SM, Cavalcanti MT, Candeia RA, Almeida EL. Characterization and study of functional properties of banana starch green variety of Mysore (Musa AAB—Mysore). Food Sci. Technol. 2017, 37, 224–231. DOI:10.1590/1678-457X.18916 [Google Scholar]
-
Khawas P, Deka SC. Effect of modified resistant starch of culinary banana on physicochemical, functional, morphological, diffraction, and thermal properties. Int. J. Food Prop. 2017, 20, 133–150. DOI:10.1080/10942912.2016.1147459 [Google Scholar]
-
Jan KN, Panesar PS, Rana JC, Singh S. Structural, thermal and rheological properties of starches isolated from Indian quinoa varieties. Int. J. Biol. Macromol. 2017, 102, 315–322. DOI:10.1016/j.ijbiomac.2017.04.027 [Google Scholar]
-
Li L, Yuan TZ, Setia R, Raja RB, Zhang B, Ai Y. Characteristics of pea, lentil and faba bean starches isolated from air-classified flours in comparison with commercial starches. Food Chem. 2019, 276, 599–607. DOI:10.1016/j.foodchem.2018.10.064 [Google Scholar]
-
Zheng M, You Q, Lin Y, Lan F, Luo M, Zeng H, Zheng B, Zhang Y. Effect of guar gum on the physicochemical properties and in vitro digestibility of lotus seed starch. Food Chem. 2019, 272, 286–291. DOI:10.1016/j.foodchem.2018.08.029 [Google Scholar]
-
Ai Y, Gong L, Reed M, Huang J, Zhang Y, Jane J-L. Characterization of starch from bamboo seeds. Starch 2016, 68, 131–139. DOI:10.1002/star.201500206 [Google Scholar]
-
Felisberto MHF, Beraldo AL, Costa MS, Boas FV, Franco CML, Clerici MTPS. Physicochemical and structural properties of starch from young bamboo culm of Bambusa tuldoides. Food Hydrocoll. 2019, 87, 101–107. DOI:10.1016/j.foodhyd.2018.07.032 [Google Scholar]
-
Felisberto MHF, Beraldo AL, Costa MS, Boas FV, Franco CML, Clerici MTPS. Bambusa vulgaris starch: Characterization and technological properties. Food Res. Int. 2020, 132, 109102. DOI:10.1016/j.foodres.2020.109102 [Google Scholar]
-
Sapper M, Chiralt A. Starch-Based Coatings for Preservation of Fruits and Vegetables. Coatings 2018, 8, 152. DOI:10.3390/coatings8050152 [Google Scholar]
-
Falguera V, Quintero JP, Jiménez A, Muñoz JA, Ibarz A. Edible films and coatings: Structures, active functions and trends in their use. Trends Food Sci. Technol. 2011, 22, 292–303. DOI:10.1016/j.tifs.2011.02.004 [Google Scholar]
-
Flores-López ML, Cerqueira MA, de Rodríguez DJ, Vicente AA. Perspectives on utilization of edible coatings and nano-laminate coatings for extension of postharvest storage of fruits and vegetables. Food Eng. Rev. 2016, 8, 292–305. DOI:10.1007/s12393-015-9135-x [Google Scholar]
-
Hirsch JB, Kokini JL. Understanding the mechanism of cross-linking agents (POCl3, STMP, and EPI) through swelling behavior and pasting properties of cross-linked waxy maize starches. Cereal Chem. 2002, 79, 102–107. DOI:10.1094/CCHEM.2002.79.1.102 [Google Scholar]
-
Betancur AD, Chel GL, Cañizares HE. Acetylation and characterization of Canavalia ensiformis starch. J. Agric. Food Chem. 1997, 45, 378–382. DOI:10.1021/jf960272e [Google Scholar]
-
Cui SW. Food Carbohydrates: Chemistry, Physical Properties, and Applications; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
-
Blennow A. Phosphorylation of the starch granule. In Starch: Metabolism and Structure; Springer: Tokyo, Japan, 2015; pp. 399–424. [Google Scholar]
-
Kittipongpatana OS, Kittipongpatana N. Preparation and physico chemical properties of modified jackfruit starches. LWT-Food Sci. Technol. 2011, 44, 1766–1773. DOI:10.1016/j.lwt.2011.03.023 [Google Scholar]
-
Otache MA, Duru RU, Achugasim O, Abayeh OJ. Advances in the modification of starch via esterification for enhanced properties. J. Polym. Environ. 2021, 29, 1365–1379. DOI:10.1007/s10924-020-02006-0 [Google Scholar]
-
Chen Q, Yu H, Wang L, ul Abdin Z, Chen Y, Wang J, et al. Recent progress in chemical modification of starch and its applications. RSC Adv. 2015, 5, 67459–67474. DOI:10.1039/C5RA10849G [Google Scholar]
-
Ariyantoro AR, Katsuno N, Nishizu T. Effects of dual modification with succinylation and annealing on physicochemical, thermal and morphological properties of corn starch. Foods 2018, 7, 133. DOI:10.3390/foods7090133 [Google Scholar]
-
Pandiselvam R, Manikantan MR, Divya V, Ashokkumar C, Kaavya R, Kothakota A, et al. Ozone: An advanced oxidation technology for starch modification. Ozone Sci. Eng. 2019, 41, 491–507. DOI:10.1080/01919512.2019.1577128 [Google Scholar]
-
Djordjevic S, Nikolic L, Kovacevic S, Miljkovic M, Djordjevic D. Graft copolymerization of acrylic acid onto hydrolyzed potato starch using various initiators. Period. Polytech. Chem. Eng. 2013, 57, 55. DOI:10.3311/PPch.2171 [Google Scholar]
-
Jyothi AN. Starch graft copolymers: Novel applications in industry. Compos. Interfaces 2010, 17, 165–174. DOI:10.1163/092764410X490581 [Google Scholar]
-
Singh J, Kaur L, McCarthy OJ. Factors influencing the physico-chemical, morphological, thermal and rheological properties of some chemically modified starches for food applications—A review. Food Hydrocoll. 2007, 21, 1–22. DOI:10.1016/j.foodhyd.2006.02.006 [Google Scholar]
-
Iqbal U, Saini P, Ahmed M. Effect of Heat Moisture Treatment, Annealing, and Gelatinization-Retrogradation Modifications on Physico-Chemical, Functional, and Structural Properties of Starch from Elephant Foot Yam (Amorphophallus paeoniifolius). Starch-Stärke 2024, 76, 2300220. DOI:10.1002/star.202300220 [Google Scholar]
-
Wang S, Copeland L. Effect of acid hydrolysis on starch structure and functionality: A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 1081–1097. DOI:10.1080/10408398.2012.684551 [Google Scholar]
-
Spychaj T, Wilpiszewska K, Zdanowicz M. Medium and high substituted carboxymethyl starch: Synthesis, characterization and application. Starch-Stärke 2013, 65, 22–33. DOI:10.1002/star.201200159 [Google Scholar]
-
Ayoub AS, Rizvi SS. An overview on the technology of cross-linking of starch for nonfood applications. J. Plast. Film Sheeting 2009, 25, 25–45. DOI:10.1177/8756087909336493 [Google Scholar]
-
Hong J, Zeng XA, Brennan CS, Brennan M, Han Z. Recent advances in techniques for starch esters and the applications: A review. Foods 2016, 5, 50. DOI:10.3390/foods5030050 [Google Scholar]
-
Fonseca LM, Gonçalves JR, El Halal SLM, Pinto VZ, Dias ARG, Jacques AC, et al. Oxidation of potato starch with different sodium hypochlorite concentrations and its effect on biodegradable films. LWT-Food Sci. Technol. 2015, 60, 714–720. DOI:10.1016/j.lwt.2014.10.052 [Google Scholar]
-
Chen P, Xie F, Zhao L, Qiao Q, Liu X. Effect of acid hydrolysis on the multi-scale structure change of starch with different amylose content. Food Hydrocoll. 2017, 69, 359–368. DOI:10.1016/j.foodhyd.2017.03.003 [Google Scholar]
-
Compart J, Singh A, Fettke J, Apriyanto A. Customizing starch properties: A review of starch modifications and their applications. Polymers 2023, 15, 3491. DOI:10.3390/polym15163491 [Google Scholar]
-
Musarurwa H, Tavengwa NT. Application of carboxymethyl polysaccharides as bio-sorbents for the sequestration of heavy metals in aquatic environments. Carbohydr. Polym. 2020, 237, 116142. DOI:10.1016/j.carbpol.2020.116142 [Google Scholar]
-
Chaudhary S, Kumar S, Kumar V, Sharma R. Chitosan nanoemulsions as advanced edible coatings for fruits and vegetables: Composition, fabrication and developments in last decade. Int. J. Biol. Macromol. 2020, 152, 154–170. DOI:10.1016/j.ijbiomac.2020.02.276 [Google Scholar]
-
Chiralt A, González-Martínez C, Vargas M, Atarés L. Edible films and coatings from proteins. In Proteins in Food Processing; Woodhead Publishing: Sawston, UK, 2018; pp. 477–500. [Google Scholar]
-
Jampilek J, Krialov K. Nanoantimicrobials: Activity, benefits, and weaknesses. In Nanostructures for Antimicrobial Therapy; Elsevier: Amsterdam, The Netherlands, 2017; pp. 23–54. [Google Scholar]
-
Salsabiela S, Sukma Sekarina A, Bagus H, Audiensi A, Azizah F, Heristika W, et al. Development of edible coating from gelatin composites with the addition of black tea extract (Camellia sinensis) on minimally processed watermelon (Citrullus lanatus). Polymers 2022, 14, 2628. DOI:10.3390/polym14132628 [Google Scholar]
-
Rambabu K, Bharath G. Mango leaf extract incorporated chitosan antioxidant film for active food packaging. Int. J. Biol. Macromol. 2019, 126, 1234–1243. DOI:10.1016/j.ijbiomac.2018.12.196 [Google Scholar]
-
Jamroz E, Kopel P. Polysaccharide and protein films with antimicrobial/antioxidant activity in the food industry: A review. Polymers 2020, 12, 1289. DOI:10.3390/polym12061289 [Google Scholar]
-
Sipahi RE, Castell-Perez ME, Moreira RG, Gomes C, Castillo A. Improved multilayered antimicrobial alginate-based edible coating extends the shelf life of fresh-cut watermelon (Citrullus lanatus). LWT-Food Sci. Technol. 2013, 51, 9–15. DOI:10.1016/j.lwt.2012.11.013 [Google Scholar]
-
Murmu SB, Mishra HN. Optimization of the arabic gum based edible coating formulations with sodium caseinate and tulsi extract for guava. LWT 2017, 80, 271–279. DOI:10.1016/j.lwt.2017.02.018 [Google Scholar]
-
Kaur J, Singh J, Rasane P, Gupta P, Kaur S, Sharma N, et al. Natural additives as active components in edible films and coatings. Food Biosci. 2023, 53, 102689. DOI:10.1016/j.fbio.2023.102689 [Google Scholar]
-
Mostafavi FS, Zaeim D. Agar-based edible films for food packaging applications—A review. Int. J. Biol. Macromol. 2020, 159, 1165–1176. DOI:10.1016/j.ijbiomac.2020.05.123 [Google Scholar]
-
Senturk Parreidt T, Schmid M, Müller K. Effect of dipping and vacuum impregnation coating techniques with alginate based coating on physical quality parameters of cantaloupe melon. J. Food Sci. 2018, 83, 929–936. DOI:10.1111/1750-3841.14091 [Google Scholar]
-
Andrade RD, Skurtys O, Osorio FA. Atomizing spray systems for application of edible coatings. Compr. Rev. Food Sci. Food Saf. 2012, 11, 323–337. DOI:10.1111/j.1541-4337.2012.00186.x [Google Scholar]
-
Valdes A, Ramos M, Beltrán A, Jiménez A, Garrigós MC. State of the art of antimicrobial edible coatings for food packaging applications. Coatings 2017, 7, 56. DOI:10.3390/coatings7040056 [Google Scholar]
-
Khodaei D, Hamidi-Esfahani Z, Rahmati E. Effect of edible coatings on the shelf-life of fresh strawberries: A comparative study using TOPSIS-Shannon entropy method. NFS J. 2021, 23, 17–23. DOI:10.1016/j.nfs.2021.02.003 [Google Scholar]
-
Geraldine RM, Soares ND, Botrel DA, de Almeida Gonçalves L. Characterization and effect of edible coatings on minimally processed garlic quality. Carbohydr. Polym. 2008, 72, 403–409. DOI:10.1016/j.carbpol.2007.09.012 [Google Scholar]
-
Ju J, Xie Y, Guo Y, Cheng Y, Qian H, Yao W. Application of edible coating with essential oil in food preservation. Crit. Rev. Food Sci. Nutr. 2019, 59, 2467–2480. DOI:10.1080/10408398.2018.1456402 [Google Scholar]
-
Debeaufort F, Voilley A. Lipid-based edible films and coatings. In Edible Films and Coatings for Food Applications; Springer: New York, NY, USA, 2009; pp. 135–168. [Google Scholar]
-
Zhong Y, Cavender G, Zhao Y. Investigation of different coating application methods on the performance of edible coatings on Mozzarella cheese. LWT-Food Sci. Technol. 2014, 56, 1–8. DOI:10.1016/j.lwt.2013.11.006 [Google Scholar]
-
Saberi B, Golding JB, Marques JR, Pristijono P, Chockchaisawasdee S, Scarlett CJ, et al. Application of biocomposite edible coatings based on pea starch and guar gum on quality, storability and shelf life of ‘Valencia’ oranges. Postharvest Biol. Technol. 2018, 137, 9–20. DOI:10.1016/j.postharvbio.2017.11.003 [Google Scholar]
-
Lara G, Yakoubi S, Villacorta CM, Uemura K, Kobayashi I, Takahashi C, et al. Spray technology applications of xanthan gum-based edible coatings for fresh-cut lotus root (Nelumbo nucifera). Food Res. Int. 2020, 137, 109723. DOI:10.1016/j.foodres.2020.109723 [Google Scholar]
-
Pham TT, Nguyen LLP, Dam MS, Baranyai L. Application of edible coating in extension of fruit shelf life. AgriEngineering 2023, 5, 520–536. DOI:10.3390/agriengineering5010034 [Google Scholar]
-
Zank J, Kind M, Schlünder EU. Particle growth and droplet deposition in fluidised bed granulation. Powder Technol. 2001, 120, 76–81. DOI:10.1016/S0032-5910(01)00350-3 [Google Scholar]
-
Nawab A, Alam F, Hasnain A. Mango kernel starch as a novel edible coating for enhancing shelf- life of tomato (Solanum lycopersicum) fruit. Int. J. Biol. Macromol.2017, 103, 581–586. DOI:10.1016/j.ijbiomac.2017.05.057. [Google Scholar]
-
Divya K, Smitha V, Jisha MS. Antifungal, antioxidant and cytotoxic activities of chitosan nanoparticles and its use as an edible coating on vegetables. Int. J. Biol. Macromol. 2018, 114, 572–577. DOI:10.1016/j.ijbiomac.2018.03.130 [Google Scholar]
-
Escamilla-García M, Rodríguez-Hernández M, Hernández-Hernández H, Delgado Sánchez L, García-Almendárez B, Amaro-Reyes A, et al. Effect of an edible coating based on chitosan and oxidized starch on shelf life of Carica papaya L. and its physicochemical and antimicrobial properties. Coatings 2018, 8, 318. DOI:10.3390/coatings8090318 [Google Scholar]
-
Chiu PE, Lai LS. Antimicrobial Activities of Tapioca Starch/Decolorized Hsian-Tsao Leaf Gum Coatings Containing Green Tea Extracts in Fruit Based Salads, Romaine Hearts and Pork Slices. Int. J. Food Microbiol. 2010, 139, 23–30. DOI:10.1016/j.ijfoodmicro.2010.01.010 [Google Scholar]
-
Kumar S, Bhatnagar T. Studies to enhance the shelf life of fruits using Aloe vera based herbal coatings: A review. Int. J. Agric. Food Sci. Technol. 2014, 5, 211–218. [Google Scholar]
-
Fadini AL, Rocha FS, Alvim ID, Sadahira MS, Queiroz MB, Alves RMV, et al. Mechanical properties and water vapour permeability of hydrolysed collagen–cocoa butter edible films plasticised with sucrose. Food Hydrocoll. 2013, 30, 625–631. DOI:10.1016/j.foodhyd.2012.08.011 [Google Scholar]
-
Poverenov E, Zaitsev Y, Arnon H, Granit R, Alkalai-Tuvia S, Perzelan Y, et al. Effects of a composite chitosan–gelatin edible coating on postharvest quality and storability of red bell peppers. Postharvest Biol. Technol. 2014, 96, 106–109. DOI:10.1016/j.postharvbio.2014.05.015 [Google Scholar]
-
Oyom W, Xu H, Liu Z, Long H, Li Y, Zhang Z, et al. Effects of modified sweet potato starch edible coating incorporated with cumin essential oil on storage quality of ‘early crisp’. LWT 2022, 153, 112475. DOI:10.1016/j.lwt.2021.112475 [Google Scholar]
-
Tiwari A, Galanis A, Soucek MD. Biobased and Environmentally Benign Coatings; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
-
Ferreira AR, Alves VD, Coelhoso IM. Polysaccharide-based membranes in food packaging applications. Membranes 2016, 6, 22. DOI:10.3390/membranes6020022 [Google Scholar]
-
Hassan B, Chatha SAS, Hussain AI, Zia KM, Akhtar N. Recent advances on polysaccharides, lipids and protein based edible films and coatings: A review. Int. J. Biol. Macromol. 2018, 109, 1095–1107. DOI:10.1016/j.ijbiomac.2017.11.097 [Google Scholar]
-
Resende NS, Gonçalves GAS, Reis KC, Tonoli GHD, Boas EVBV. Chitosan/cellulose nanofibril nanocomposite and its effect on quality of coated strawberries. J. Food Qual. 2018, 2018, 1727426. DOI:10.1155/2018/1727426 [Google Scholar]
-
Zhou Z, Ma J, Li K, Zhang W, Li K, Tu X, et al. A plant leaf-mimetic membrane with controllable gas permeation for efficient preservation of perishable products. ACS Nano 2021, 15, 8742–8752. DOI:10.1021/acsnano.1c00997 [Google Scholar]
-
Singh GP, Bangar SP, Yang T, Trif M, Kumar V, Kumar D. Effect on the properties of edible starch-based films by the incorporation of additives: A review. Polymers 2022, 14, 1987. DOI:10.3390/polym14101987 [Google Scholar]
-
Galus S, Kadzińska J. Whey protein edible films modified with almond and walnut oils. Food Hydrocoll. 2016, 52, 78–86. DOI:10.1016/j.foodhyd.2015.06.013 [Google Scholar]
-
Thitsartarn W, Jinkarn T. Superhydrophobicity of paperboard packaging by hierarchical surface roughness modification of polylactic acid electrospraying. Prog. Org. Coat. 2021, 154, 106192. DOI:10.1016/j.porgcoat.2021.106192 [Google Scholar]
-
Poudel R, Dutta N, Karak N. A mechanically robust biodegradable bioplastic of citric acid modified plasticized yam starch with anthocyanin as a fish spoilage auto-detecting smart film. Int. J. Biol. Macromol. 2023, 242, 125020. DOI:10.1016/j.ijbiomac.2023.125020 [Google Scholar]
-
Jiang H, Zhang W, Jiang W. Effects of purple passion fruit peel extracts on characteristics of Pouteria campechiana seed starch films and the application in discernible detection of shrimp freshness. Food Hydrocoll. 2023, 138, 108477. DOI:10.1016/j.foodhyd.2023.108477 [Google Scholar]
-
Yousuf B, Wu S, Siddiqui MW. Incorporating essential oils or compounds derived thereof into edible coatings: Effect on quality and shelf life of fresh/fresh-cut produce. Trends Food Sci. Technol. 2021, 108, 245–257. DOI:10.1016/j.tifs.2021.01.016 [Google Scholar]
-
Ahmed M, Saini P, Iqbal U. Bio cellulose-based edible composite coating for shelf-life extension of tomatoes. Food Humanit. 2023, 1, 973–984. DOI:10.1016/j.foohum.2023.08.016 [Google Scholar]
-
Yildirim-Yalçın M, Sadıkoğlu H, Şeker M. Characterization of edible film based on grape juice and cross-linked maize starch and its effects on the storage quality of chicken breast fillets. LWT 2021, 142, 111012. DOI:10.1016/j.lwt.2021.111012 [Google Scholar]
-
Erna KH, Felicia WXL, Rovina K, Vonnie JM, Huda N. Development of curcumin/rice starch films for sensitive detection of hypoxanthine in chicken and fish meat. Carbohydr. Polym. Technol. Appl. 2022, 3, 100189. DOI:10.1016/j.carpta.2022.100189 [Google Scholar]
-
Sani IK, Geshlaghi SP, Pirsa S, Asdagh A. Composite film based on potato starch/apple peel pectin/ZrO2 nanoparticles/microencapsulated Zataria multiflora essential oil; investigation of physicochemical properties and use in quail meat packaging. Food Hydrocoll. 2021, 117, 106719. DOI:10.1016/j.foodhyd.2021.106719 [Google Scholar]
-
Zheng K, Zhang J, Yang F, Wang W, Li W, Qin C. Properties and biological activity of chitosan-coix seed starch films incorporated with nano zinc oxide and Artemisia annua essential oil for pork preservation. LWT 2022, 164, 113665. DOI:10.1016/j.lwt.2022.113665 [Google Scholar]
-
Alotaibi S, Tahergorabi R. Development of a sweet potato starch-based coating and its effect on quality attributes of shrimp during refrigerated storage. LWT 2018, 88, 203–209. DOI:10.1016/j.lwt.2017.10.022 [Google Scholar]
-
Galvao AM, Zambelli RA, Araújo AW, Bastos MS. Edible coating based on modified corn starch/tomato powder: Effect on the quality of dough bread. LWT 2018, 89, 518–524. DOI:10.1016/j.lwt.2017.11.027 [Google Scholar]
-
Salehi F. Improvement of gluten-free bread and cake properties using natural hydrocolloids: A review. Food Sci. Nutr. 2019, 7, 3391–3402. DOI:10.1002/fsn3.1245 [Google Scholar]
-
Otero-Herrera A, Fuentes-Gaviria L, Pérez-Cervera C, Andrade-Pizarro R. Development of edible films based on sweet potato (Ipomoea batatas) starch and their application in candy packaging. Int. J. Biol. Macromol. 2025, 299, 140031. DOI:10.1016/j.ijbiomac.2025.140031 [Google Scholar]
-
Kramer ME. Structure and function of starch-based edible films and coatings. In Edible Films and Coatings for Food Applications; Springer: New York, NY, USA, 2009; pp. 113–134. DOI:10.1007/978-0-387-92824-1_4 [Google Scholar]
-
Del Sol González-Forte L, Amalvy JI, Bertola N. Corn starch-based coating enriched with natamycin as an active compound to control mold contamination on semi-hard cheese during ripening. Heliyon 2019, 5, e01957. DOI:10.1016/j.heliyon.2019.e01957 [Google Scholar]
-
Berti S, Flores SK, Jagus RJ. Improvement of the microbiological quality of Argentinian Port Salut cheese by applying starch-based films and coatings reinforced with rice bran and containing natural antimicrobials. J. Food Process. Preserv. 2020, 44, e14827. DOI:10.1111/jfpp.14827 [Google Scholar]
-
Salama HH, Trif M, Rusu AV, Bhattacharya S. Application of functional and edible coatings and films as promising strategies for developing dairy functional products—A review on yoghurt case. Coatings 2022, 12, 838. DOI:10.3390/coatings12060838 [Google Scholar]
-
Lin D, Zhao Y. Innovations in the Development and Application of Edible Coatings for Fresh and Minimally Processed Fruits and Vegetables. Compr. Rev. Food Sci. Food Saf. 2007, 6, 60–75. DOI:10.1111/j.1541-4337.2007.00018.x [Google Scholar]
-
Md Nor S, Ding P. Trends and Advances in Edible Biopolymer Coating for Tropical Fruit: A Review. Food Res. Int. 2020, 134, 109208. DOI:10.1016/j.foodres.2020.109208 [Google Scholar]
-
Maringgal B, Hashim N, Mohamed Amin Tawakkal IS, Muda Mohamed MT. Recent Advance in Edible Coating and Its Effect on Fresh/fresh-cut Fruits Quality. Trends Food Sci. Technol. 2020, 96, 253–267. DOI:10.1016/j.tifs.2019.12.024 [Google Scholar]
-
Decco Argentina. Naturcover—Decco Argentina: 2021. Available online: http://www.deccolatam.com/producto/naturcover-cp/?lang=en (accessed on 28 March 2021).
-
Perussello CA, Zhang Z, Marzocchella A, Tiwari BK. Valorization of Apple Pomace by Extraction of Valuable Compounds. Compr. Rev. Food Sci. Food Saf. 2017, 16, 776–796. DOI:10.1111/1541-4337.12290 [Google Scholar]
-
Galus S, Arik Kibar EA, Gniewosz M, Kraśniewska K. Novel Materials in the Preparation of Edible Films and Coatings—A Review. Coatings 2020, 10, 674. DOI:10.3390/coatings10070674 [Google Scholar]
-
Karnwal A, Rauf A, Jassim AY, Selvaraj M, Al-Tawaha ARMS, Kashyap P, et al. Advanced starch-based films for food packaging: Innovations in sustainability and functional properties. Food Chem. X 2025, 29, 102662. DOI:10.1016/j.fochx.2025.102662 [Google Scholar]
-
Santhosh R, Ahmed J, Thakur R, Sarkar P. Starch-based edible packaging: Rheological, thermal, mechanical, microstructural, and barrier properties—A review. Sustain. Food Technol. 2024, 2, 307–330. DOI:10.1039/D3FB00211J [Google Scholar]
-
Shi B, Hao Z, Jia M, Xie S. Mechanical and barrier properties of chitosan-based composite film as food packaging: A review. BioResources 2024, 19, 4001–4014. DOI:10.15376/biores.19.2. [Google Scholar]
-
Jahangiri F, Mohanty AK, Misra M. Sustainable biodegradable coatings for food packaging: Challenges and opportunities. Green Chem. 2024, 26, 4934–4974. DOI:10.1039/D3GC02647G [Google Scholar]
-
Kumari SVG, Pakshirajan K, Pugazhenthi G. Recent advances and future prospects of cellulose, starch, chitosan, polylactic acid and polyhydroxyalkanoates for sustainable food packaging applications. Int. J. Biol. Macromol. 2022, 221, 163–182. DOI:10.1016/j.ijbiomac.2022.08.203 [Google Scholar]
-
Wang H, Qian J, Ding F. Emerging chitosan-based films for food packaging applications. J. Agric. Food Chem. 2018, 66, 395–413. DOI:10.1021/acs.jafc.7b04528 [Google Scholar]
-
Basumatary IB, Mukherjee A, Katiyar V, Kumar S. Biopolymer-based nanocomposite films and coatings: Recent advances in shelf-life improvement of fruits and vegetables. Crit. Rev. Food Sci. Nutr. 2022, 62, 1912–1935. DOI:10.1080/10408398.2020.1848789 [Google Scholar]
-
Purewal SS, Kaur A, Bangar SP, Singh P, Singh H. Protein-based films and coatings: An innovative approach. Coatings 2023, 14, 32. DOI:10.3390/coatings14010032 [Google Scholar]
-
Shen Y, Seidi F, Ahmad M, Liu Y, Saeb MR, Akbari A, et al. Recent advances in functional cellulose-based films with antimicrobial and antioxidant properties for food packaging. J. Agric. Food Chem. 2023, 71, 16469–16487. DOI:10.1021/acs.jafc.3c06004 [Google Scholar]
-
Yang Y, Fu J, Duan Q, Xie H, Dong X, Yu L. Strategies and Methodologies for Improving Toughness of Starch Films. Foods 2024, 13, 4036. DOI:10.3390/foods13244036 [Google Scholar]
-
Guerrero BG, Santos KDL, Kamimura ES, de Oliveira CAF. Application of microbial exopolysaccharides in packaging films for the food industry: A review. Int. J. Food Sci. Technol. 2024, 59, 17–29. DOI:10.1111/ijfs.16789 [Google Scholar]
-
Revutskaya N, Polishchuk E, Kozyrev I, Fedulova L, Krylova V, Pchelkina V, et al. Application of natural functional additives for improving bioactivity and structure of biopolymer-based films for food packaging: A review. Polymers 2024, 16, 1976. DOI:10.3390/polym16141976 [Google Scholar]
-
Abdullah, Cai J, Hafeez MA, Wang Q, Farooq S, Huang Q, et al. Biopolymer-based functional films for packaging applications: A review. Front. Nutr. 2022, 9, 1000116. DOI:10.3389/fnut.2022.1000116 [Google Scholar]
-
Wu Y, Wu H, Hu L. Recent advances of proteins, polysaccharides and lipids-based edible films/coatings for food packaging applications: A review. Food Biophys. 2024, 19, 29–45. DOI:10.1007/s11483-023-09794-7 [Google Scholar]
-
Pillai AR, Eapen AS, Zhang W, Roy S. Polysaccharide-based edible biopolymer-based coatings for fruit preservation: A review. Foods 2024, 13, 1529. DOI:10.3390/foods13101529 [Google Scholar]
-
Mathew M, Paroly S, Athiyanathil S. Biopolymer-based electrospun nanofiber membranes for smart food packaging applications: A review. RSC Adv. 2025, 15, 21742–21779. DOI:10.1039/D5RA02348C [Google Scholar]
-
Nasrollahzadeh M, Sajjadi M, Iravani S, Varma RS. Starch, cellulose, pectin, gum, alginate, chitin and chitosan derived (nano) materials for sustainable water treatment: A review. Carbohydr. Polym. 2021, 251, 116986. DOI:10.1016/j.carbpol.2020.116986 [Google Scholar]
-
Das A, Ringu T, Ghosh S, Pramanik N. A comprehensive review on recent advances in preparation, physicochemical characterization, and bioengineering applications of biopolymers. Polym. Bull. 2023, 80, 7247–7312. DOI:10.1007/s00289-022-04443-4 [Google Scholar]
-
Tozluoğlu A, Fidan H. Effect of Size Press Coating of Cationic Starch/Nanofibrillated Cellulose on Physical and Mechanical Properties of Recycled Papersheets. BioResources 2023, 18, 5993–6012. DOI:10.15376/biores.18.3.5993-6012 [Google Scholar]
-
Barman MK, Tamang N, Bhattarai A, Saha B. Montmorillonite-Starch based Nano-Composites and Applications. Adv. Appl. Micro Nano Clay Biopolym.-Based Compos. 2022, 125, 172–209. DOI:10.21741/9781644901915-8 [Google Scholar]
-
Jayakumar A, Radoor S, Kim JT, Rhim JW, Parameswaranpillai J, Nandi D, et al. Titanium dioxide nanoparticles and elderberry extract incorporated starch based polyvinyl alcohol films as active and intelligent food packaging wraps. Food Packag. Shelf Life 2022, 34, 100967. DOI:10.1016/j.fpsl.2022.100967 [Google Scholar]
-
Naghdi S, Rezaei M, Abdollahi M. A starch-based pH-sensing and ammonia detector film containing betacyanin of paperflower for application in intelligent packaging of fish. Int. J. Biol. Macromol. 2021, 191, 161–170. DOI:10.1016/j.ijbiomac.2021.09.045 [Google Scholar]
-
Wang L, Yang C, Deng X, Peng J, Zhou J, Xia G, et al. A pH-sensitive intelligent packaging film harnessing Dioscorea zingiberensis starch and anthocyanin for meat freshness monitoring. Int. J. Biol. Macromol. 2023, 245, 125485. DOI:10.1016/j.ijbiomac.2023.125485 [Google Scholar]
-
Ndwandwe BK, Malinga SP, Kayitesi E, Dlamini BC. Recent developments in the application of natural pigments as pH-sensitive food freshness indicators in biopolymer-based smart packaging: Challenges and opportunities. Int. J. Food Sci. Technol. 2024, 59, 2148–2170. DOI:10.1111/ijfs.16990 [Google Scholar]
-
Vedove TM, Maniglia BC, Tadini CC. Production of sustainable smart packaging based on cassava starch and anthocyanin by an extrusion process. J. Food Eng. 2021, 289, 110274. DOI:10.1016/j.jfoodeng.2020.110274 [Google Scholar]
-
Deng H, Su J, Zhang W, Khan A, Sani MA, Goksen G, et al. A review of starch/polyvinyl alcohol (PVA) blend film: A potential replacement for traditional plastic-based food packaging film. Int. J. Biol. Macromol. 2024, 273, 132926. DOI:10.1016/j.ijbiomac.2024.132926 [Google Scholar]
-
Kochkina NE, Lukin ND. Structure and properties of biodegradable maize starch/chitosan composite films as affected by PVA additions. Int. J. Biol. Macromol. 2020, 157, 377–384. DOI:10.1016/j.ijbiomac.2020.04.154 [Google Scholar]
-
Nitikornwarakul C, Wangpradid R, Rakkapao N. Impact of molar composition on the functional properties of glutinous rice starch–chitosan blend: Natural-based active coating for extending mango shelf life. Polymers 2024, 16, 1375. DOI:10.3390/polym16101375 [Google Scholar]
-
Carneiro da Silva LR, Rios ADO, Campomanes Santana RM. Polymer blends of poly(lactic acid) and starch for the production of films applied in food packaging: A brief review. Polym. Renew. Resour. 2023, 14, 108–153. DOI:10.1177/20412479231154924 [Google Scholar]
-
Kamoun EA, Kenawy ERS, Chen X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J. Adv. Res. 2017, 8, 217–233. DOI:10.1016/j.jare.2017.01.005 [Google Scholar]
-
Westlake JR, Tran MW, Jiang Y, Zhang X, Burrows AD, Xie M. Biodegradable active packaging with controlled release: Principles, progress, and prospects. ACS Food Sci. Technol. 2022, 2, 1166–1183. DOI:10.1021/acsfoodscitech.2c00070 [Google Scholar]
-
Dutta D, Sit N. Comprehensive review on developments in starch-based films along with active ingredients for sustainable food packaging. Sustain. Chem. Pharm. 2024, 39, 101534. DOI:10.1016/j.scp.2024.101534 [Google Scholar]
-
Zhu F. Starch based films and coatings for food packaging: Interactions with phenolic compounds. Food Res. Int. 2025, 204, 115758. DOI:10.1016/j.foodres.2025.115758 [Google Scholar]
-
Parlak ME, Sahin OI, Dundar AN, Saricaoglu FT, Smaoui S, Goksen G, et al. Natural colorant incorporated biopolymers-based pH-sensing films for indicating the food product quality and safety. Food Chem. 2024, 439, 138160. DOI:10.1016/j.foodchem.2023.138160 [Google Scholar]
-
Wu W, Zheng L, Yu J, Liu L, Goksen G, Shao P. High-sensitivity intelligent packaging films harnessing rose anthocyanins and hydrophilic silica aerogel for visual food freshness monitoring. Food Qual. Saf. 2024, 8, fyad051. DOI:10.1093/fqsafe/fyad051 [Google Scholar]
-
Marappan G, Tahir HE, Karim N, Lakshmanan A, Shishir MRI, Hashim SBH, et al. Natural pigment-based pH/gas-sensitive intelligent packaging film for freshness monitoring of meat and seafood: Influencing factors, technological advances, and future perspectives. Food Rev. Int. 2025, 42, 58–95. DOI:10.1080/87559129.2025.2473026 [Google Scholar]
-
Dong M, Mastroianni G, Bilotti E, Zhang H, Papageorgiou DG. Biodegradable starch-based nanocomposite films with exceptional water and oxygen barrier properties. ACS Sustain. Chem. Eng. 2024, 12, 11056–11066. DOI:10.1021/acssuschemeng.4c04198 [Google Scholar]
-
Hu G, Lan X, Peng B, Liao J, Xiong Y. Water resistant, biodegradable and flexible corn starch/carboxymethyl cellulose composite film for slow-release fertilizer coating materials. Int. J. Biol. Macromol. 2024, 260, 129476. DOI:10.1016/j.ijbiomac.2024.129476 [Google Scholar]
-
Garavito J, Peña-Venegas CP, Castellanos DA. Production of starch-based flexible food packaging in developing countries: Analysis of the processes, challenges, and requirements. Foods 2024, 13, 4096. DOI:10.3390/foods13244096 [Google Scholar]