BMAA-Producing Cyanobacteria and Fish Contamination in Italy: An Emerging One Health Concern
Received: 13 November 2025 Revised: 10 December 2025 Accepted: 28 January 2026 Published: 04 February 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Cyanobacteria are widely present in aquatic environments and are known for their ability to produce neurotoxic compounds, including microcystins and BMAA [1]. Bioaccumulation of these toxins in fish poses potential risks to human health, particularly through dietary exposure. The increasing frequency of cyanobacterial blooms, exacerbated by climate change and anthropogenic activities, has raised concerns about their impact on food safety and ecosystem stability.
The production of freshwater fish in Italy has been estimated at 32,000 tons/year (7% of the total production, year 2005). Some studies have been carried out in our country to investigate the presence of cyanotoxin (mainly microcystins) contamination in commercial freshwater fish, e.g., [2,3] using different immunochemical and instrumental detection methods. In one of these, 155 samples of fish and crustaceans collected from five contaminated lakes were analyzed, showing levels of microcystin contamination ranging from 0.21 μg/kg to 14.6 mg/kg [4,5]. Considering blooms of 100 million cells/L, the detection of microcystins in the internal organs and muscles of fish had been simultaneous with the onset of the bloom.
β-N-methylamino-L-alanine (BMAA) is a non-proteinogenic amino acid synthesized by various cyanobacteria commonly found in freshwater, marine, and terrestrial environments. Once ingested, BMAA has been shown to inflict progressive damage on motor neurons within the spinal cord, replicating the neuropathological effects observed in individuals diagnosed with amyotrophic lateral sclerosis (ALS). Additionally, BMAA promotes the formation of neurofibrillary tangles in both the spinal cord and the brain, as seen in Alzheimer’s disease. In the last few years, the role of BMAA as a potential contributing factor for ALS has been deeply investigated, and evidence supporting a causal relation with the disease onset was observed [6,7,8,9,10]. This correlation was first observed on the island of Guam (Pacific Ocean) in the local Chamorro population because of BMAA accumulation in the food chain. The source of BMAA was the seeds and fruits of the cycad plant, which hosts symbiotic cyanobacteria in its roots [11]. The primary route of exposure for the local population is believed to have been the consumption of flying foxes and other wild animals that fed on cycad seeds [12,13].
The consumption of contaminated aquatic organisms and seafood represents a potential route of human exposure to BMAA [14,15]. Possible associations between sporadic amyotrophic lateral sclerosis (ALS) and BMAA presence in aquatic organisms have been proposed in Chesapeake Bay, Maryland, USA [16] and in the Thau Lagoon, France [9] using derivatization-based analytical methods.
Several investigations into the neurotoxic potential of BMAA have revealed different plausible mechanisms of action. BMAA exerts excitotoxic effects on central nervous system (CNS) neurons, both in vitro and in vivo [17]. It may mimic glutamate by binding to glutamate receptors, such as NMDA (non–N-methyl-D-aspartate), AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid), and mGluRs, leading to dysregulated ion fluxes (e.g., Ca2+, Na+, Cl−, K+, Mg2+), prolonged neuronal depolarization, intracellular calcium overload, and activation of apoptotic pathways.
In animal studies, such as those by Spencer et al. [18], repeated oral dosing of L-BMAA in macaques led to neurodegenerative behaviors and CNS histopathology, effects partially reversible with NMDA receptor antagonists. However, not all studies are consistent: some research on dietary BMAA exposure in mice did not reveal significant behavioral or pathological outcomes [19,20].
Rats administered BMAA via intracerebroventricular injections exhibited temporary motor disturbances and cortical dysfunction, believed to stem from AMPA receptor overactivation [21]. BMAA also demonstrated cytotoxicity in cultured cortical neurons, with NMDA antagonists like D-APV mitigating its effects [22]. Low BMAA doses (e.g., 0.3 mM) preferentially targeted vulnerable neurons via non-NMDA mechanisms.
BMAA has been shown to selectively harm spinal motor neurons in culture via AM-PA/kainate receptor pathways, confirmed by NBQX inhibition [23]. It raises intracellular Ca2+ and reactive oxygen species (ROS) preferentially in motor neurons, and while NMDA blockers reduce toxicity, full protection requires mGluR5 antagonists and antioxidants [24]
Beyond receptor binding, BMAA may integrate into nascent proteins as a serine analogue, disrupting folding and promoting aggregation, a hallmark of neurodegenerative disease [25,26]. Acid hydrolysis reveals bound BMAA in tissues, supporting the misincorporation theory. L-serine has shown promise as a therapeutic agent, countering BMAA-induced damage in vitro and in vivo [27].
Behavioral studies using honeybees found reduced learning ability and elevated mortality following BMAA exposure, with ROS and calcium imbalance implicated [28]. Furthermore, BMAA inhibits catalase activity and melatonin synthesis, possibly through mGluR3 modulation and protein kinase C pathways, suggesting alternative toxicity routes [29].
Collectively, these findings indicate that BMAA’s neurotoxicity arises from a complex interplay of excitotoxicity, oxidative stress, receptor overstimulation, and protein misfolding mechanisms with profound implications for neurodegenerative disease research [30].
In the Thau Lagoon study—located in the Languedoc-Roussillon region on the southern coast of France—total BMAA concentrations in mussels showed a time-dependent increase during the summer of 2009, whereas concentrations remained more stable in oysters. Total 2,4-diaminobutyric acid (DAB) concentrations were similar in both mollusk species, ranging from 3.4–9.7 and 3.3–8.8 µg/g dry weight (DW) in mussels and oysters, respectively. Regarding BMAA, total concentrations of its isomer AEG in mussels increased between 20 June and 7 September. The higher accumulation of BMAA and its isomers in mussels may be attributed to their generally higher filtration activity compared to oysters [9].
During the summer of 2009, two phytoplankton blooms dominated by diatoms (e.g., Chaetoceros spp.) were observed in early July and late August in the Thau Lagoon [31]. Diatoms have recently been identified as potential BMAA producers [32], suggesting that the proliferation of such microalgae, alongside picocyanobacteria [8], could explain the presence of BMAA in mollusks during that summer.
In the Baltic Sea, cyanobacterial blooms in summer (June–August) are typically dominated by the genera Nodularia and Aphanizomenon, both of which have demonstrated the ability to produce BMAA [11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34].
Larger diatom blooms occur in spring (March–May). Jonasson et al. [34] reported that zooplankton, which naturally feed on cyanobacteria, contained significantly higher levels of BMAA than the cyanobacteria themselves. Moreover, various fish tissues showed up to 200-fold higher BMAA concentrations compared to the original cyanobacterial producers. These findings provided the first evidence of BMAA biomagnification in an aquatic ecosystem outside of Guam.
While the Baltic Sea is a major source of commercial fish in Sweden, the country is also home to numerous lakes, including some of Europe’s largest. BMAA has been detected in several commercial fish species from the Baltic Sea, prompting concern over possible bioaccumulation in Swedish lake systems.
In the Finjasjön Lake, the presence of BMAA was confirmed in water samples, and bioaccumulation patterns were identified in both planktivorous/benthivorous and piscivorous fish species [35]. Variables such as fish species, total body weight, sex, and sampling season were evaluated. For the first time, a large sample of fish individuals was used to statistically assess BMAA bioaccumulation in a closed ecological community.
Findings suggest that feeding behavior (planktivorous/benthivorous) and increasing fish age are associated with higher BMAA tissue concentrations. The cyanobacterium Aphanizomenon klebahnii, previously known as a BMAA producer [11], was a likely source of BMAA in Finjasjön Lake during the sampling period. However, several reports identifying BMAA production by diatoms and dinoflagellates [31,34] raise the possibility that other phytoplankton species—such as Ceratium hirundinella, Cryptomonas spp., Fragilaria crotonensis, and Asterionella formosa [36]—may also contribute to BMAA presence.
Two of the highest BMAA concentrations were found in brain samples of female Perca fluviatilis specimens weighing approximately 200 g, presumably older individuals. These elevated levels likely reflect cumulative dietary exposure—initially from zooplankton and benthos, followed by predation on BMAA-contaminated fish prey. Among the factors significantly associated with BMAA concentration were species and total body weight; heavier and presumably older fish exhibited higher BMAA tissue concentrations.
Thus, in systems where BMAA remains bioavailable over time, organisms are likely to accumulate BMAA throughout their lifespan, with older individuals harboring greater concentrations. The influence of organism weight may also explain observed variations related to sex and season. Females, which often have faster growth rates and larger body sizes, may ingest larger quantities of BMAA-contaminated prey, potentially explaining their higher brain BMAA concentrations. As most fish sampled in spring were female, this season also yielded higher BMAA levels. In general, BMAA concentrations in fish muscle tissues were lower and less frequent than in brain tissues.
Overall BMAA concentrations in fish tissues from Finjasjön Lake were comparable to those observed in the Baltic Sea [33], though lower than levels reported in fish from southern Florida’s coastal waters (muscle: 58 ± 41 µg/g wet weight; liver: 588 ± 81 µg/g; kidney: 1450 ± 687 µg/g; [37] or from Lake Taihu in China (0.07–35.91 µg/g dry weight in muscle; [38]). This variability may reflect biological differences, although methodological inconsistencies between studies are likely to contribute more significantly.
The detection of both BMAA and mercury in the muscle tissues of Abramis brama and Sander lucioperca is of particular concern. BMAA, even at sub-toxic concentrations, has been shown to enhance mercury neurotoxicity [39]. The synergistic neurotoxic effect of BMAA and mercury may thus pose a human health risk, even when mercury levels remain below current European regulatory limits. Collectively, the data indicate the widespread natural occurrence of BMAA in Finjasjön Lake. The co-detection of BMAA and mercury in fish species commonly consumed by residents near Hässleholm raises critical questions regarding potential public health risks.
The suggested link between BMAA exposure and neurodegenerative disease onset, coupled with the identification of this cyanotoxin in aquatic organisms, has prompted global investigation into the potential human health risks associated with food sourced from eutrophic water bodies.
The Hartbeespoort Dam reservoir in Northwest Province, South Africa, experiences persistent cyanobacterial blooms and is widely used by local fishers, many of whom consume their catch. The commercial sale of fish species harvested from this reservoir as part of a recent biomanipulation remediation strategy may pose an additional threat. Both BMAA and microcystins (MCs) have been detected in fish from the reservoir. BMAA concentrations reached up to 1.630 ng/g dry weight, while MCs reached up to 29.44 ng/g during an extensive bloom. A clear correlation was found between total BMAA levels in fish muscle tissues and trophic position in the Hartbeespoort Dam food web. Interestingly, fish collected during winter—when dense cyanobacterial blooms were absent—still exhibited BMAA concentrations as high as 3.055 ng/g dry weight.
Research has also demonstrated that BMAA accumulates in the central nervous system of patients suffering from neurodegenerative diseases, such as Alzheimer’s disease and amyotrophic lateral sclerosis (ALS), supporting its potential role as an environmental risk factor for these conditions [37,40]. Additionally, chronic exposure to BMAA has been associated with an increased risk of developing neurodegenerative disorders [41,42]. These findings highlight the urgent need to monitor BMAA contamination in fish intended for human consumption and to investigate further the mechanisms through which this neurotoxin may contribute to disease onset. In this frame, the Italian lakes of different sizes were investigated for BMAA presence in fish.
2. Materials and Methods
2.1. Sample Collection and Study Sites
Fish and water samples were collected from several Italian lakes known for hosting recurrent cyanobacterial blooms, including Lake Occhito, Lake Vico, Lake Albano, and Lake Averno (Figure 1).
Lake Vico is a large volcanic basin within a crater depression of the Cimino Volcano Complex, located 60 km north of Rome in Central Italy. It has a surface area of 12.09 km2, a volume of 260.7 × 106 m3, and reaches a maximum depth of 48.5 m (with an average depth of 21.6 m). The total water turnover time is approximately 17 years [43]. The lake’s total catchment area is 41 km2. It is part of a Regional Wildlife Park and is designated as a wetland of international importance under the Ramsar Convention. The surrounding area features extensive hazel woods, and from the 1300s until the early 1960s, several paper mills operated near the lake. Lake Vico serves as a drinking water reservoir for around 10,000 residents of Ronciglione and Caprarola and is a popular recreational area throughout the year.
Over the past few decades, the lake has undergone progressive eutrophication [44,45]. Lake Vico hosts annual blooms of Planktothrix rubescens, occurring from winter through late spring [46]. During summer, the populations rest at approximately 25 m in depth, gradually ascending in autumn to form blooms that reach the surface in winter. Phyto-planktivorous fish feed on these blooms, accumulating the associated toxins. In recent years, our group has studied the population dynamics of P. rubescens and the production of related toxins in this lake [3,4,47,48].
Lake Albano, another volcanic basin within a crater depression, has an ellipsoidal shape and has been studied by our research group for several years [49]. The lake perimeter is 12 km, with a maximum depth of 175 m. The average renewal time exceeds 67 years. Lacking tributary outlets, the lake features an ancient Roman channel carved into the lava rocks of the crater, which historically regulated the water level. Since the 1960s, uncontrolled water extraction for human use has caused the lake level to drop nearly four meters below the artificial outflow, shifting it from an oligotrophic to a eutrophic state.
P. rubescens population dynamics in Lake Albano show surface stratification with blooms in winter and metalimnic stratification during summer, peaking at depths of 25–30 m. However, the overall population declines by 80–86% from late April to early November [50,51]. These findings are consistent with observations from Lake Zurich [52] and Lac du Bourget [53]. In spring, with frequent cold and rainy weather, the thermal stratification of the upper layers is sensitive to climatic fluctuations, occasionally leading to unusual rises in P. rubescens populations and surface scums even in April–May.
Lake Averno is a volcanic lake (quiescent volcano) located in the municipality of Pozzuoli, precisely between the hamlet of Lucrino and the archaeological site of Cuma, in the metropolitan city of Naples. It rises inside a crater of an extinct volcano, born 4000 years ago, and is the second largest, after Lake Fusaro, among all the lakes in the Campi Flegrei. It has an elliptical shape with a surface area of 0.55 km2 and a maximum depth of 34 m.
Enclosed by steep crater walls and surrounded by lush vegetation, the lake spans approximately 0.6 km2. It has been renowned since antiquity for its mythical association with the underworld, given its sulfurous emissions from surrounding geothermal activity. In recent decades, anthropogenic activities have contributed to the ecological degradation of Lake Avernus: agricultural runoff and wastewater discharge (which, in this specific case, are not regulated due to their illegality) introduce high levels of nitrogen and phosphorus into the lake, as also reported by the local Agency for Environmental Protection. These nutrients promote excessive algal growth, leading to periodic reddish-brown algal blooms sustained by the toxic cyanobacterium P. rubescens [54,55], which occur in the winter months, especially between late January and early February, or in conjunction with cold night temperatures.
Lake Occhito, located on the border between the Italian regions of Molise and Apulia, is the largest artificial reservoir in Italy. Constructed for potable water supply by damming the Fortore River, the lake has an average depth of approximately 90 m, a surface area of 13 km2, and extends longitudinally for 12 km. The Fortore River continues downstream of the dam, ultimately discharging into the Adriatic Sea after a course of roughly 67 km. This river outflow reaches the northern Apulian coastline near significant mussel farming operations, including those situated in two coastal lagoons known as Lesina and Varano.
During the winter of 2009, an extensive bloom of Planktothrix rubescens developed in Lake Occhito, forming thick, reddish surface scums that persisted from January through April. Although local observers had sporadically reported similar red blooms in previous years, the 2009 event was notably widespread. By January, the bloom had enveloped the entire lake surface, and in the following months, it infiltrated the regional water distribution network.
In March and April 2009, a series of intense rainfall events prompted the opening of the dam’s spillways to relieve critically high-water levels. As a result, lake water—heavily contaminated with P. rubescens biomass—was discharged into the Fortore River. Aerial footage recorded during helicopter surveillance documented the transport of these red surface scums downstream, ultimately reaching the Adriatic Sea. This freshwater “red tide” thus traveled from Lake Occhito through the Fortore River, potentially impacting coastal ecosystems and aquaculture zones [5].
2.2. BMAA Extraction and Quantification
BMAA extraction and quantification from water samples were performed as explained in Messineo et al. [56]: Water samples were processed to quantify both intracellular and extracellular BMAA. Fresh phytoplankton biomass (10–130 mg) was obtained from water samples by centrifugation and resuspended in 2 mL of sterile bidistilled water. The suspension was homogenized by vortex mixing and subsequently subjected to ultrasonic disruption for 5 min at 30–40 °C using an ultrasonic bath (Elgasonic, Switzerland). The samples were then centrifuged for 10 min at 11,000 rpm using a Beckman L5–55 ultracentrifuge to remove cellular debris. The supernatant was collected, and the pellet was resuspended in sterile bidistilled water. This extraction procedure was repeated twice, and the resulting supernatants were pooled and used to determine intracellular BMAA concentrations.
Extracellular BMAA concentrations were determined directly from the centrifuged water samples obtained before phytoplankton extraction.
BMAA extraction from fish tissues was performed following Clausi et al. [57]: Tissue samples were processed according to the following extraction procedure. Samples were immediately homogenized for 15 min using an Ultra-Turrax T8 homogenizer (IKA Werke, Staufen, Germany) in 10 mL of HPLC-grade methanol (100% MeOH). The homogenates were centrifuged at 5000× g for 5 min, and the supernatants were collected. The extraction was repeated on the resulting pellets under identical conditions, and the supernatants were combined and filtered through paper filters. The pooled extracts were adjusted to a final volume of 25 mL with 100% MeOH. An aliquot of 5 mL was then diluted with 5 mL of distilled water and loaded onto an Oasis HLB 3 cc solid-phase extraction (SPE) cartridge (Waters Corporation, Milford, MA, USA) or a C18 SPE cartridge (Isolute International Sorbent Technology, Mid-Glamorgan, UK), previously conditioned with 20 mL of 100% MeOH followed by 20 mL of distilled water. The cartridge was washed sequentially with 30 mL of distilled water and 30 mL of 20% MeOH. Analytes were eluted with 50 mL of 100% MeOH. The methanolic eluate was evaporated to dryness, and the residue was reconstituted in 2 mL of distilled water. Samples were stored at −30 °C until further analysis. The extracted compounds were analyzed using the ELISA method. The currently available test (ABRAXIS, Horsham, PA, USA) is a direct competitive ELISA based on the recognition of BMAA by specific antibodies. If present in a sample, BMAA competes with a BMAA-HRP conjugate for binding sites on anti-BMAA rabbit antibodies in solution. These antibodies are subsequently captured by a secondary antibody immobilized on the wells of a microtiter plate. Following a washing step and the addition of a substrate solution, a colorimetric signal is generated. The intensity of the resulting blue color is inversely proportional to the concentration of BMAA in the sample. The color reaction is stopped after a specified incubation time, and the absorbance is measured using an ELISA microplate reader. Sample concentrations are determined by interpolation from a standard curve constructed during each assay. The coefficients of variation (CV%) for the standards are below 10%; for the samples, below 15%. The method’s limit of detection (LOD) was 4 μg/L, ensuring precise quantification of BMAA in fish muscle tissue and water samples [56,58].
2.3. Cyanobacteria Identification and Quantification
Water samples for toxicological and biological analyses were collected monthly using a 2.5 L Ruttner bottle from four surface stations near the lake shore and one central station. Samples were stored in ice chests and transported to the laboratory for analysis.
Microscopic examinations, toxin extraction, and quantification were conducted in the laboratory. For microscopic observations, subsamples were analyzed with an inverted microscope [Leitz Labovert FS] within 48 h from sampling, following the methods of [59,60], using 25 mL sedimentation chambers for phytoplankton identification and cell density estimation.
3. Results
3.1. BMAA Detection and Cyanobacteria Identification
3.1.1. Vico
Lake Vico (Viterbo) was monitored for about 11 years (2005–2009, 2012–2013, 2018–2019, 2021–2022) collecting samples from different sites (Table 1), investigating the species of cyanobacteria present, population dynamics, contamination in fish fauna, in the lake’s aquifer where artesian wells deepened, and in public drinking water fountains. Blooms of the cyanobacterium Planktothrix rubescens regularly took place in this lake during the autumn-winter months, and during the study period reached maximum values of 72.5 million cells/L (January 2007) [47], with microcystin production (maximum value of 350 µg/L in March 2008) [3,4]. The highest detected BMAA level in water samples was 718 µg/L in November 2019. From March 2006 to March 2009, the study of the lake also included toxic contamination in fish fauna with particular attention to the most commercially appreciated species, the whitefish Coregonus lavaretus, which exhibited BMAA concentrations reaching 26.56 ng/g, raising concerns about bioaccumulation in commercially significant species. Hazelnut samples from the surrounding area were also tested, with BMAA levels of 2.3 ng/g in 2010, indicating possible contamination beyond aquatic ecosystems.
Table 1. GPS coordinates of the sampling sites in Lake Vico and Albano.
|
Lake Vico |
Latitude |
Longitude |
|---|---|---|
|
Promontorio |
42.32044 |
12.17381 |
|
Emissario |
42.31053 |
12.19032 |
|
Bella Venere |
42.32857 |
12.1913 |
|
Ultima spiaggia |
42.31033 |
12.15584 |
|
Center |
42.31917 |
12.16722 |
|
Lake Albano |
||
|
Molo |
41.75327 |
12.65891 |
|
Hotel |
41.7507 |
12.6751 |
|
Bosco |
41.74021 |
12.678 |
|
Castello |
41.75041 |
12.6618 |
|
Center |
41.74305 |
12.67482 |
3.1.2. Albano
Over the past two decades, studies on the population dynamics of toxic cyanobacteria in the lake from different sampling sites (Table 1) and the associated production of biotoxins have revealed the occurrence of periodic winter blooms of Planktothrix rubescens [51]. These blooms typically peak in January, with the highest recorded concentration reaching 298 × 106 cells/L in January 2008. The simultaneous presence of microcystins and BMAA accompanied these events. During the last sampling campaign in 2018 winter and 2019 spring, the analysis of the water samples revealed BMAA levels ranging from a minimum of 96.1 g/L (March 2019, Lake center) to a maximum of 2109.96 μg/L in the lake center in December (Figure 2a) The seasonal variation in BMAA levels suggests that the intensity of cyanobacterial blooms significantly influences toxin accumulation in aquatic fauna. Moreover, the toxins can remain available in the water, although the P. rubescens population was still growing (early winter) or had receded during the summer season (Figure 2b). Additionally, microcystin levels were found to be elevated during bloom peaks, indicating a combined toxin exposure risk for aquatic organisms and humans.
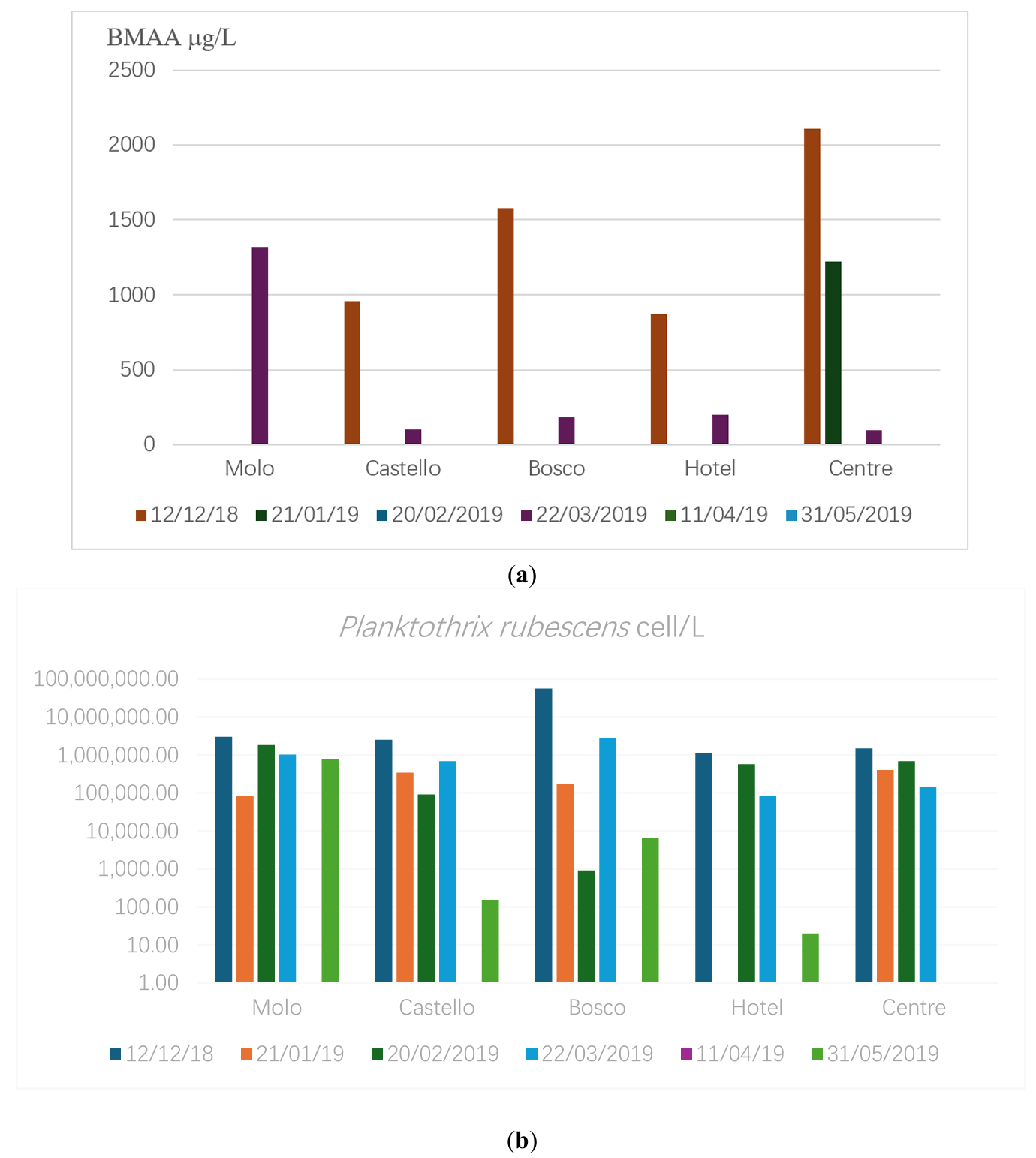
Figure 2. (a) BMAA (average surface value, μg/L) in Albano during 2018–2019; (b) P.rubescens amount (logarithmic scale) in Albano during 2018–2019 (cell/L).
3.1.3. Averno
In March 2022, Lake Averno and the nearby Lake Lucrino (Campania region) has been interested by a massive bloom of P. rubescens. In the meantime, the phenomenon has spread even along the stretch between Baia and Lucrino that connects the lake with the sea, and has taken on a reddish color. This area is used by many mussel farms that reach the food markets of central Italy. For this reason, mussels (Mytilus galloprovincialis) samples from the mussel farms area were analyzed for BMAA contamination.
Based on the laboratory analyses, it was observed that during the two sampling campaigns between April 4 and May 5, the algal biotoxins detected were β-N-methylamino-L-alanine (BMAA) and microcystins (MC). The latter was found in a single sample unit, specifically in the tissues of a mussel, where BMAA was also present. Both intracellular and extracellular values of BMAA and MC were detected, with the former corresponding to the mussel tissues and the latter to the pellets obtained from water samples collected from Lakes Averno and Lucrino.
On 8 April 2022, a sample of mussels was collected from the area of Baia Gulf, which is connected by a channel to Averno and Lucrino lakes, and three mussels were randomly selected. The obtained values indicated the presence of intracellular BMAA and MC. The first sample contained 0.862 ng/g of BMAA and, in addition, 0.047 ng/g of MC, the second 5.687 ng/g, and the third 4.373 ng/g of BMAA.
On 20 April, three water aliquots were collected from the lakes, and three pellets were obtained in the laboratory. The first sample showed a BMAA concentration of 0.965 ng/g, no biotoxins were detected in the second and third samples.
On 3 May 2022, three water aliquots were collected, yielding three laboratory-processed pellets. The first sample showed a BMAA concentration of 4.89 ng/g, the second 1.85 ng/g, and the third 1.55 ng/g. On 5 May, mussel samples were collected, and four mussels were randomly selected. The first sample revealed a BMAA value of 0.24 ng/g, while the second contained 0.067 ng/g of BMAA, the third 0.53 ng/g, and the fourth 0.302 ng/g of BMAA (Table 2).
Table 2. Lake Averno: BMAA amount (wet weight) in water samples and in mussels from the contaminated mussel farm around Baia.
|
SAMPLE |
BMAA (ng/g) in Mytilus galloprovincialis |
BMAA in P. rubescens Pellet from Lake Averno (ng/g) |
|
|---|---|---|---|
|
April 2022 |
M1 |
0.862 |
0.965 |
|
M2 |
5.687 |
0 |
|
|
M3 |
4.373 |
0 |
|
|
May 2022 |
M4 |
0.24 |
4.89 |
|
M5 |
0.067 |
1.853 |
|
|
M6 |
0.53 |
1.55 |
|
|
M7 |
0.302 |
0 |
3.1.4. Lake Occhito
Sampling activities, which began on 4 November 2013, and concluded on 26 November 2014, involved both water and fish samples when environmental conditions permitted. Sampling locations included Lake Occhito and the confluence of the Fortore River. The sampling frequency was monthly, except from November to April 2014, when it was conducted bimonthly. A total of 42 water samples and 226 fish specimens were analyzed, including largemouth bass (Micropterus salmoides), carp (Cyprinus carpio), crucian carp (Carassius carassius), chub (Squalius cephalus), and Mediterranean bleak (Alburnus alburnus).
Taxonomic analysis revealed the presence of algal species, among which cyanobacteria accounted for 13.1% of the total community, with Oscillatoria brevis being predominant. This species exhibited a bloom of 21 × 106 cells/L in November 2013 and a concentration of 571.6 × 103 cells/L in September 2014. Planktothrix rubescens was detected in April 2014 at a concentration of 34,800 cells/L.
Regarding BMAA presence in water samples, the highest levels were observed in March (24.14 ng/mL), with peak concentrations recorded at the sampling site located on the right bend of the Occhito reservoir. Comparable concentrations of the toxin were found in spring samples from both 2013 and 2014, with slightly higher average levels observed in the latter year (12.20 ng/mL in 2013 vs. 16.37 ng/mL in 2014), particularly in March. No BMAA was detected in samples collected from May through November (Figure 3).
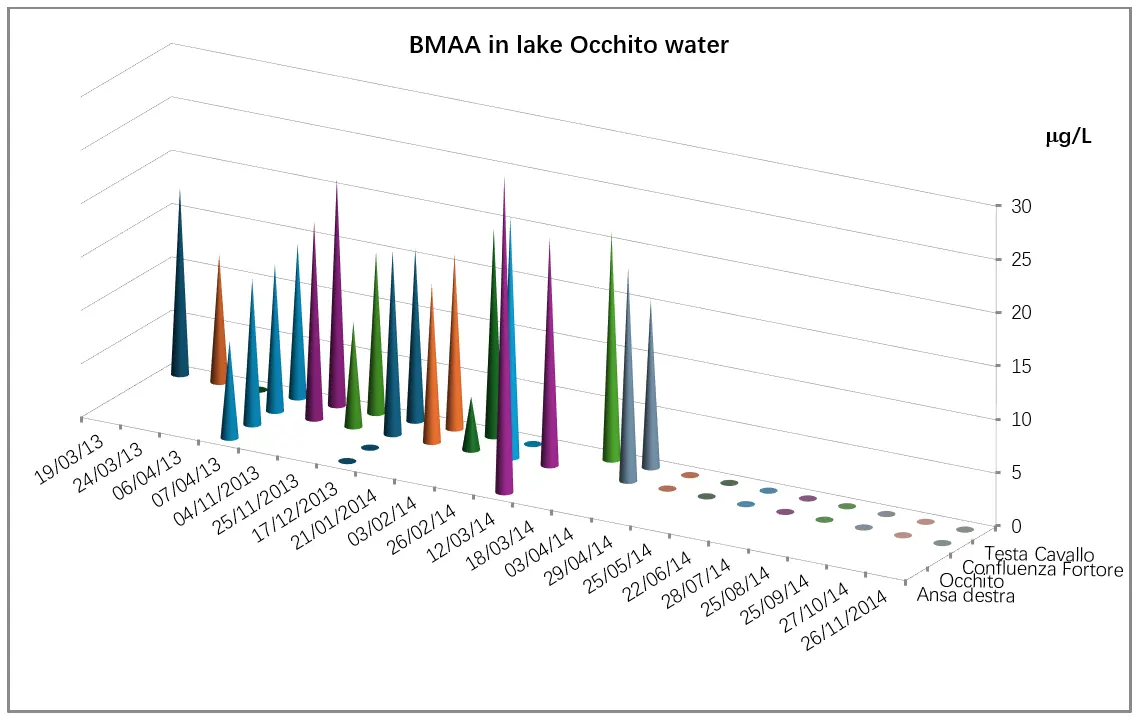
Figure 3. Occhito Lake: BMAA concentration levels detected using the ELISA method in the Occhito basin at sampling sites 1 and 2 (right bend and Fortore confluence, respectively), and at the sites Testa di Cavallo locality and Occhito basin.
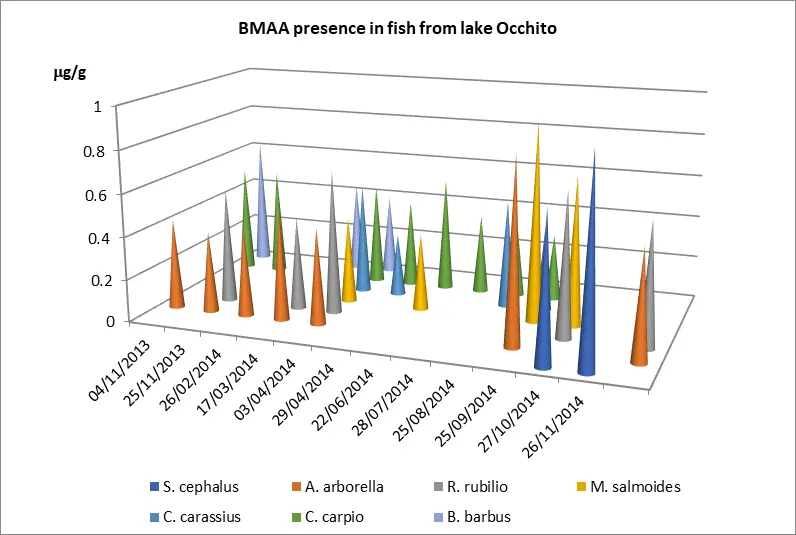
Fish samples analyzed using the ELISA immunoenzymatic technique revealed BMAA concentrations ranging from 1.44 to 3.93 μg/g dry weight in muscle tissue (Table 3). The highest levels of the toxin were found between September and November 2014, with a mean concentration of 2.01 μg/g dry weight. All fish species analyzed showed increased contamination levels during October–November 2014 (Figure 4).
International epidemiological studies suggest that BMAA presence influences the neuronal pathology in human populations surrounding water bodies with toxic-producing blooms [61].
Table 3. Lake Occhito: BMAA in fishes (ng/g) wet weight.
|
Species |
4 Nov. 2013 |
23 Nov. 2013 |
26 Feb. 2013 |
17 Mar. 2013 |
3 Apr. 2014 |
29 Apr. 2014 |
22 Jun. 2014 |
28 Jul. 2014 |
25 Aug. 2014 |
25 Sep. 2014 |
27 Oct. 2015 |
26 Jan. 2015 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
S. cephalus |
687 |
947 |
||||||||||
|
A. arborella |
427 |
383 |
423 |
518 |
453 |
86 |
||||||
|
R. rubilio |
543 |
435 |
677 |
678 |
||||||||
|
M. salmoides |
41 |
362 |
937 |
707 |
||||||||
|
C. carassius |
52 |
297 |
509 |
408 |
||||||||
|
C. carpio |
512 |
511 |
485 |
415 |
543 |
377 |
344 |
316 |
||||
|
B. barbus |
617 |
434 |
383 |
Epidemiological data up to 2013 on the incidence of diseases such as Alzheimer’s and dementia were provided by the epidemiological centers of the Puglia and Molise regions, covering municipalities in the provinces of Foggia and Campobasso, as shown in Figure 4. Among the larger municipalities in the province of Foggia (Foggia, Lucera, Manfredonia, San Severo), the highest number of cases was recorded in the provincial capital. Comparable incidence rates were also observed in municipalities located near the Occhito reservoir (Figure 5). The municipalities listed first in the legend are those closest to the Occhito basin.
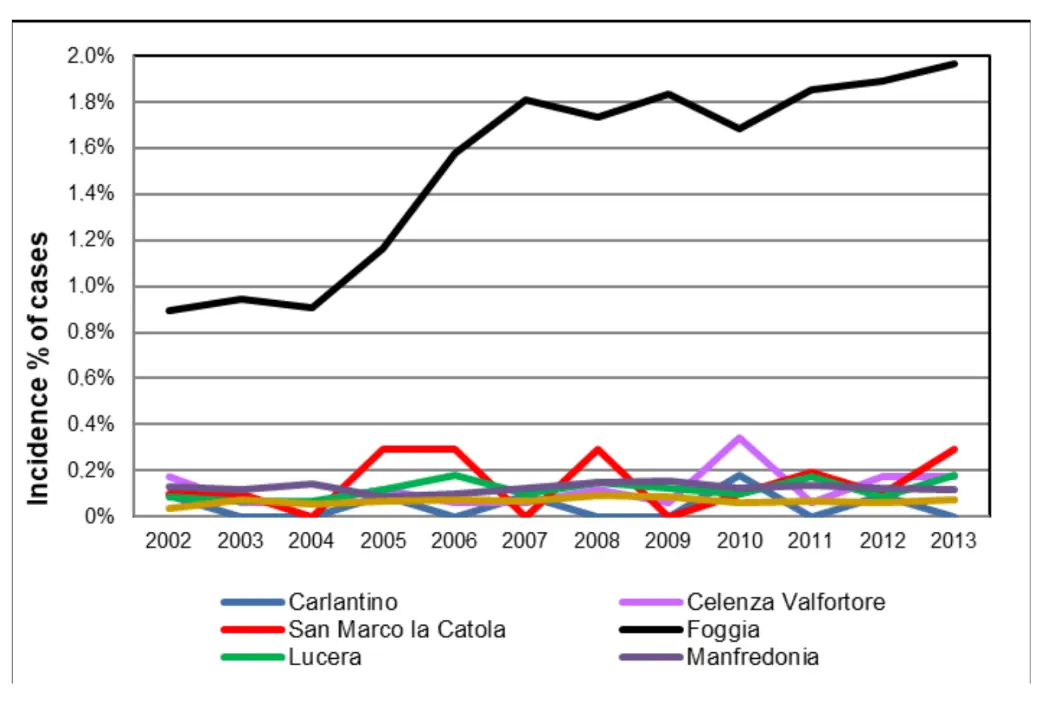
Figure 5. Cases of Alzheimer’s/dementia (% of the population) in Apulian municipalities during 2002–2013.
Moreover, a marked increase in the incidence of neurological diseases in the city of Foggia was observed beginning in 2007. Regarding the province of Campobasso, epidemiological data were available only for the year 2010. Within this province, municipalities located near the reservoir showed a higher number of neurological disease cases, with percentage values exceeding those of other municipalities (Figure 6).
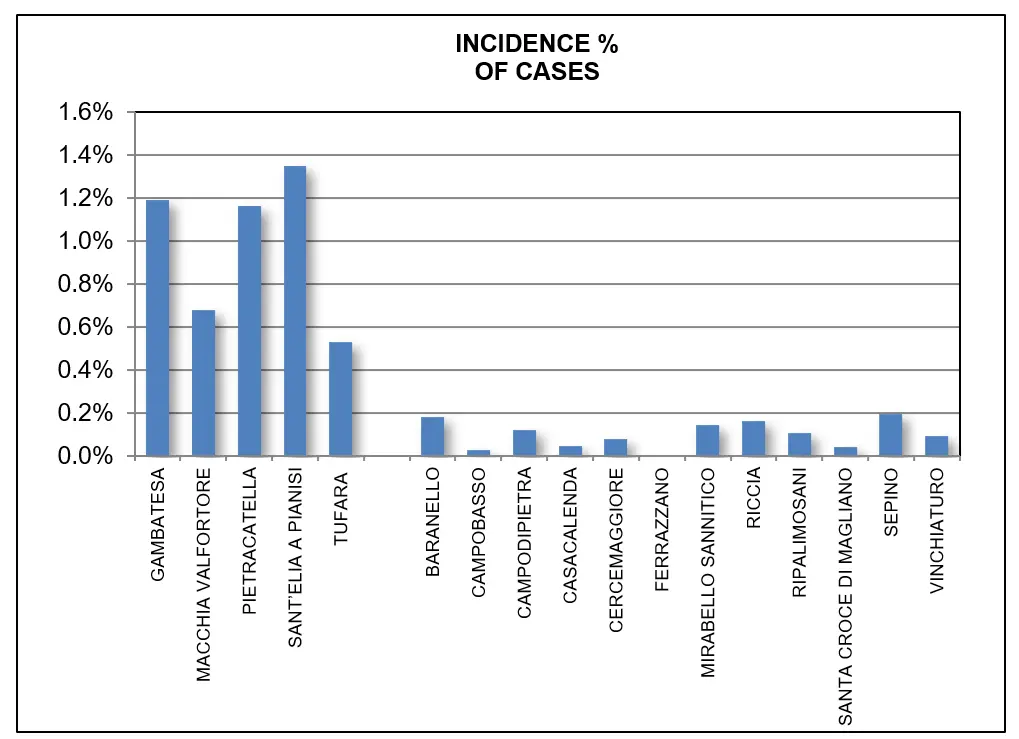
Figure 6. Percentage of the population affected by Alzheimer’s/dementia in Molise municipalities in 2010. The municipalities shown at the beginning of the graph are those situated near the Occhito basin.
4. Discussion
The findings of this study underscore the increasing ecological and public health concerns associated with cyanobacterial blooms, particularly in freshwater ecosystems experiencing nutrient enrichment and climate-driven perturbation. The detection of β-N-methylamino-L-alanine (BMAA) and microcystins in water, fish tissues, and mussels across multiple Italian lakes illustrates the risk of toxin bioaccumulation and potential human exposure through the food chain.
These findings demonstrate that even contaminated marine mussels may pose a health risk to consumers and result in economic losses if eutrophic inland waters—especially those connected to coastal areas—are not managed through targeted remediation plans.
Lake Occhito showed extensive Planktothrix rubescens blooms during the winter months, with concurrent detection of BMAA in water (up to 24.14 ng/mL) and in fish muscle tissue (up to 3.93 μg/g dry weight). Similar patterns were observed in Lakes Albano and Averno, where seasonal blooms were accompanied by the simultaneous presence of BMAA and microcystins in aquatic organisms. This temporal overlap raises concerns about combined toxin exposure, especially among vulnerable populations that rely on local water and food resources.
Of particular significance is the potential for synergistic neurotoxicity between BMAA and microcystins. Although these toxins act through distinct mechanisms—BMAA primarily through excitotoxicity and misincorporation into proteins, and microcystins via hepatotoxicity and inhibition of protein phosphatases—recent evidence suggests that co-exposure may amplify neurotoxic outcomes. The presence of both compounds in fish and mussels thus represents a compounded risk that single toxin evaluations may not adequately address.
Mixed contamination of inland waters represents an emerging risk perspective increasingly observed in field investigations and environmental monitoring [62,63]. Evidence indicates that the detection of mixed contaminants complicates both comprehensive human health risk assessment and the timely implementation of countermeasures. Although traditional risk assessment methods typically assume that the components of a toxic mixture conform to the concentration addition model—thus allowing prediction of total toxicity as the sum of the individual toxicities [64,65,66]—studies have demonstrated that interactions may occur which modify the overall toxicity, making it either lower or higher than expected [67,68]. Synergistic mechanisms, not yet fully understood, may involve enhanced affinity for target sites, increased persistence of toxic effects, or the generation of variable toxic responses.
Moreover, epidemiological data revealing elevated incidence rates of neurological disorders, including Alzheimer’s disease and dementia, in municipalities adjacent to Lake Occhito further highlight the need to investigate long-term health effects of chronic exposure to cyanotoxin mixtures. It is important to note that the observed association between elevated BMAA concentrations in aquatic environments and higher incidence rates of neurodegenerative diseases in nearby populations is purely observational; as such, it does not establish a causal relationship, but it is consistent with a few previous studies, which found a higher ALS risk in subjects living in the proximity of lakes containing algal blooms [69,70,71].
BMAA can be effectively removed from water intended for human consumption using treatment methods similar to those used for microcystins, such as activated carbon filtration. Consequently, it is strongly recommended that water from the studied lakes be subjected to targeted treatment before distribution through public water supply systems.
This precautionary strategy underscores the importance of stringent water quality management and continuous monitoring to safeguard drinking water and public health against the potential risks posed by BMAA and other toxins linked to harmful algal blooms.
From a One Health perspective, these results advocate for integrated, multisectoral monitoring strategies that address not only individual toxin presence but also combined and cumulative exposures. Given the persistence of cyanotoxins in aquatic environments, their capacity for bioaccumulation and trophic transfer, and their potential to act synergistically, the development of regulatory thresholds and early-warning systems becomes imperative.
While this study provides evidence of BMAA contamination in fish and mussels, it does not include a quantitative dietary exposure assessment or comparisons with established safety thresholds, largely because no official tolerable intake values for BMAA exist. Future research should aim to estimate human exposure levels based on local consumption patterns and toxin concentrations, to inform preliminary risk characterizations and support the development of evidence-based regulatory guidelines.
5. Conclusions
In conclusion, this study reinforces the urgent need for comprehensive risk assessments that consider the co-occurrence of multiple cyanotoxins and their interactive effects. Future research should focus on the mechanistic basis of BMAA and microcystin co-toxicity, refined biomonitoring protocols, and the establishment of protective guidelines to safeguard environmental and public health.
The presence of BMAA, often associated with microcystins in the aquatic fauna, is a widespread phenomenon in various freshwater bodies across Italy, highlighting the need for appropriate and multifaceted analytical techniques for effective detection, as well as proactive measures to prevent these cyanotoxins from reaching water sources and accumulating in fish and mussel tissue at levels hazardous to aquatic life and human consumers.
Moreover, the results from Lake Occhito underscore the wider implications of cyanobacterial blooms for water safety, food security, and public health. The detection of toxins in water sources and food items such as fish and mussels highlights the need for continuous monitoring and proactive risk management. Furthermore, the potential long-term health effects suggested by epidemiological data on neurological diseases in areas impacted by BMAA and microcystin contamination emphasize the urgency of comprehensive research efforts and targeted policy interventions, as well as, in the regulatory frameworks, the development and reinforcement of regulations and guidelines for water quality management, including acceptable levels of algal toxins in water and food products, to protect public health and the environment.
Currently, there are no specific regulatory limits for BMAA in drinking water or food products within the European Union, and this absence of legal thresholds reflects both the emerging nature of the toxin and the limited availability of standardized monitoring data. The findings presented in this study highlight the widespread occurrence of BMAA in freshwater ecosystems and its bioaccumulation in aquatic organisms consumed by humans. These results underscore the need for regulatory agencies to consider the inclusion of BMAA in water quality monitoring programs and risk assessment frameworks, particularly within a One Health context. Establishing provisional guideline values could serve as an important step toward mitigating potential health risks and informing future environmental and public health policies.
By prioritizing water safety and public health, communities can better deal with the challenges posed by these environmental phenomena and work towards sustainable water management and health outcomes.
6. Limitations of the Study
A limitation of this work is the lack of direct analytical confirmation of BMAA by LC-MS/MS in the biological samples discussed. Future studies integrating targeted LC-MS/MS analyses will be essential to further strengthen the causal link between cyanobacterial blooms, BMAA production, and bioaccumulation in fish.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this manuscript, the authors used ChatGPT in order to improve the use of English language. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the published article.
Author Contributions
All authors have read and agreed to the published version of the manuscript.
Ethics Statement
The use of publicly available data does not fall under the scope of human subject research requiring ethical approval.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data supporting the findings of this study are included within the article. Additional data related to environmental monitoring and analytical measurements are available from the corresponding author upon reasonable request.
Funding
This research received no external funding.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
-
Chorus I, Welker M. Toxic Cyanobacteria in Water—HA Guide to Their Public Health Consequences, Monitoring and Management, 2nd ed.; CRC Press: London, UK, 2021. DOI:10.1201/9781003081449 [Google Scholar]
-
Bogialli S, Bruno M, Curini R, Di Corcia A, Laganà A, Mari B. Simple assay for analyzing five microcystins and nodularin in fish muscle tissue: Hot water extraction followed by liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem. 2005, 53, 6586–6592. DOI:10.1021/jf050257m [Google Scholar]
-
Bruno M, Melchiorre S, Messineo V, Volpi F, Di Corcia A, Aragona I, et al. Microcystin detection in contaminated fish from italian lakes using ELISA immunoassays and LC-MS/MS analysis. In Handbook on Cyanobacteria: Biochemistry, Biotechnology and Applications; Nova Science Publishers: New York, NY, USA, 2009; pp. 191–210. [Google Scholar]
-
Bruno M, Gallo P, Messineo V, Melchiorre S. Health risk associated with microcystin presence in the environment: The case of an Italian lake (Lake Vico, Central Italy). Int. J. Environ. Prot. 2012, 2, 34–41. Available online: https://www.researchgate.net/publication/274719831 (accessed on 2 February 2026).
-
De Pace R, Vita V, Bucci MS, Gallo P, Bruno M. Microcystin Contamination in Sea Mussel Farms from the Italian Southern Adriatic Coast following Cyanobacterial Blooms in an Artificial Reservoir. J. Ecosyst. 2014, 2014, 374027. DOI:10.1155/2014/374027 [Google Scholar]
-
Banack SA, Caller T, Henegan P, Haney J, Murby A, Metcalf JS, et al. Detection of cyanotoxins, β-N-methylamino-L-alanine and microcystins, from a lake surrounded by cases of amyotrophic lateral sclerosis. Toxins 2015, 7, 322–336. DOI:10.3390/toxins7020322 [Google Scholar]
-
Cox PA, Kostrzewa RM, Guillemin GJ. BMAA and neurodegenerative illness. Neurotox. Res. 2018, 33, 178–183. DOI:10.1007/s12640-017-9753-6 [Google Scholar]
-
Delcourt N, Claudepierre T, Maignien T, Arnich N, Mattei C. Cellular and molecular aspects of the β-N-Methylamino-l-alanine (BMAA) mode of action within the neurodegenerative pathway: Facts and controversy. Toxins 2018, 10, 6. DOI:10.3390/toxins10010006 [Google Scholar]
-
Masseret E, Banack S, Boumédiène F, Abadie E, Brient L, Pernet F, et al. The French Network on A.L.S.: Investigation, dietary BMAA exposure in an amyotrophic lateral sclerosis cluster from Southern France. PLoS ONE 2013, 8, e83406. DOI:10.1371/journal.pone.0083406 [Google Scholar]
-
Davis DA, Cox PA, Banack SA, Lecusay PD, Garamszegi SP, Hagan MJ, et al. L-serine reduces spinal cord pathology in a vervet model of preclinical ALS/MND. J. Neuropathol. Exp. Neurol. 2020, 79, 393–406. DOI:10.1093/jnen/nlaa002 [Google Scholar]
-
Cox PA, Banack SA, Murch SJ, Rasmussen U, Tien G, Bidigare RR, et al. Diverse taxa of cyanobacteria produce beta-N-methylamino-L-alanine, a neurotoxic amino acid. Proc. Nat. Acad. Sci. USA 2005, 102, 5074–5078. DOI:10.1073/pnas.0501526102 [Google Scholar]
-
Cox PA, Banack SA, Murch SJ. Biomagnification of cyanobacterial neurotoxins and neurodegenerative disease among the Chamorro people of Guam. Proc. Nat. Acad. Sci. USA 2003, 100, 13380–13383. DOI:10.1073/pnas.2235808100 [Google Scholar]
-
Cox PA, Sacks OW. Cycad neurotoxins, consumption of flying foxes, and ALSPDC disease in Guam. Neurology 2002, 58, 956–959. DOI:10.1212/wnl.58.6.956 [Google Scholar]
-
Lage S, Annadotter H, Rasmussen U, Rydberg S. Biotransfer of β-N-Methylamino-l-alanine (BMAA) in a Eutrophicated Freshwater Lake. Mar. Drugs 2015, 13, 1185–1201. DOI:10.3390/md13031185 [Google Scholar]
-
Faassen EJ. Presence of the neurotoxin BMAA in aquatic ecosystems: What do we really know? Toxins 2014, 6, 1109–1138. DOI:10.3390/toxins6031109 [Google Scholar]
-
Field NC, Metcalf JS, Caller TA, Banack SA, Cox PA, Stommel EW. Linking beta-methylamino-L-alanine exposure to sporadic amyotrophic lateral sclerosis in Annapolis, MD. Toxicon 2013, 70, 179–183. DOI:10.1016/j.toxicon.2013.04.010 [Google Scholar]
-
Ross SM, Seelig M, Spencer PS. Specific antagonism of excitotoxic action of ‘uncommon’ amino acids assayed in organotypic mouse cortical cultures. Brain Res. 1987, 425, 120–127. DOI:10.1016/0006-8993(87)90490-2 [Google Scholar]
-
Spencer PS, Nunn PB, Hugon J, Ludolph AC, Ross SM, Roy DN, et al. Guam amyotrophic lateral sclerosis-parkinsonism-dementia linked to a plant excitant neurotoxin. Science 1987, 237, 517–522. DOI:10.1126/science.3603037 [Google Scholar]
-
Cruz-Aguado R, Winkler D, Shaw CA. Lack of behavioral and neuropathological effects of dietary β-methylamino-L-alanine (BMAA) in mice. Pharmacol. Biochem. Behav. 2006, 84, 294–299. DOI:10.1016/j.pbb.2006.05.012 [Google Scholar]
-
Perry TL, Bergeron C, Biro AJ. Hansen S. β-N-Methylamino-L-alanine chronic oral administration is not neurotoxic to mice. J. Neurol. Sci. 1989, 94, 173–180. DOI:10.1016/0022-510X(89)90227-X [Google Scholar]
-
Rakonczay Z, Matsuoka Y, Giacobini E. Effects of L-beta-N-methylamino-L-alanine (L-BMAA) on the cortical cho-linergic and glutamatergic systems of the rat. J Neurosci Res. 1991, 29, 121–126. DOI:10.1002/jnr.490290114 [Google Scholar]
-
Weiss JH, Koh JY, Choi DW. Neurotoxicity of β-N-methylamino-L-alanine (BMAA) and β-N-oxalylamino-L-alanine (BOAA) on cultured cortical neurons. Brain Res. 1989, 497, 64–71. DOI:10.1016/0006-8993(89)90970-0 [Google Scholar]
-
Rao SD, Banack SA, Cox PA, Weiss JH. BMAA selectively injures motor neurons via AMPA/kainate receptor activation. Exp. Neurol. 2006, 201, 244–252. DOI:10.1016/j.expneurol.2006.04.017 [Google Scholar]
-
Lobner D, Piana PMP, Salous A, Peoples RW. β-N-methylamino-l-alanine enhances neurotoxicity through multiple mechanisms. Neurobiol. Dis. 2007, 25, 360–366. DOI:10.1016/j.nbd.2006.10.002 [Google Scholar]
-
Dunlop RA, Cox PA, Banack SA, Rodgers KJ. The non-protein amino acid BMAA is misincorporated into human proteins in place of L-serine causing protein misfolding and aggregation. PLoS ONE 2013, 8, e75376. DOI:10.1371/journal.pone.0075376 [Google Scholar]
-
Albano R, Lobner D. Transport of BMAA into neurons and astrocytes by system xc. Neurotox. Res. 2018, 33, 1–5. DOI:10.1007/s12640-017-9739-4 [Google Scholar]
-
Main BJ, Dunlop RA, Rodgers KJ. The use of L-serine to prevent betamethylamino-L-alanine (BMAA)-induced proteotoxic stress in vitro. Toxicon 2018, 109, 7–12. DOI:10.1016/j.toxicon.2015.11.003 [Google Scholar]
-
Okle O, Rath L, Galizia CG, Dietrich DR. The cyanobacterial neurotoxin beta-N-methylamino-L-alanine (BMAA) induces neuronal and behavioral changes in honeybees. Toxicol. Appl. Pharmacol. 2013, 270, 9–15. DOI:10.1016/j.taap.2013.04.003 [Google Scholar]
-
Pierozan P, Andersson M, Brandt I, Karlsson O. The environmental neurotoxin β-N-methylamino-L-alanine (BMAA) inhibits melatonin synthesis in primary pinealocytes and a rat model. J. Pineal Res. 2018, 65 (Suppl. S1), e12488. DOI:10.1111/jpi.12488 [Google Scholar]
-
Li M, Qiu J, Yan G, Zheng X, Li A. How does the neurotoxin β-N-methylamino-L-alanine exist in biological matrices and cause toxicity? Sci. Total Environ. 2024, 922, 171255. DOI:10.1016/j.scitotenv.2024.171255 [Google Scholar]
-
Pernet F, Malet N, Pastoureaud A, Vaquer A, Quéré C, Dubroca L. Marine diatoms sustain growth of bivalves in a Mediterranean lagoon. J. Sea Res. 2012, 68, 20–32. DOI:10.1016/j.seares.2011.11.004 [Google Scholar]
-
Jiang LY, Eriksson J, Lage S, Jonasson S, Shams S, Mehine M, et al. Diatoms: A novel source for the neurotoxin BMAA in aquatic environments. PLoS ONE 2014, 9, e84578. DOI:10.1016/j.seares.2011.11.004 [Google Scholar]
-
Degerholm J, Gundersen K, Bergman B, Söderbäck E. Phosphorus-limited growth dynamics in two Baltic Sea cyanobacteria, Nodularia sp. and Aphanizomenon sp. FEMS Microbiol. Ecol. 2006, 58, 323–332. DOI:10.1111/j.1574-6941.2006.00180.x [Google Scholar]
-
Jonasson S, Eriksson J, Berntzon L, Spacil Z, Ilag LL, Ronnevi LO, et al. Transfer of a cyanobacterial neurotoxin within a temperate aquatic ecosystem suggests pathways for human exposure. Proc. Natl. Acad. Sci. USA 2010, 107, 9252–9257. DOI:10.1073/pnas.0914417107 [Google Scholar]
-
Lage S, Costa PR, Moita T, Eriksson J, Rasmussen U, Rydberg SJ. BMAA in shellfish from two Portuguese transitional water bodies suggests the marine dinoflagellate Gymnodinium catenatum as a potential BMAA source. Aquat. Toxicol. 2014, 152, 131–138. DOI:10.1016/j.aquatox.2014.03.029 [Google Scholar]
-
Annadotter H, Forssblad J. Limnological investigation of Lake Finjasjön (Technical Report, pp. 1–71). Hässleholm Municipality, Technical Office & RegitoAB. Cited in Lage, S. Biotransfer of β-N-Methylamino-l-alanine (BMAA) in a Temperate Aquatic Ecosystem—Biotransfer of β‑N‑Methylamino‑l‑alanine (BMAA) Study. 2011. Available online: https://pmc.ncbi.nlm.nih.gov/ (accessed on 2 February 2026).
-
Mondo K, Glover WB, Murch SJ, Liu G, Cai Y, Davis DA, et al. Environmental neurotoxins β-N-methylamino-L-alanine (BMAA) and mercury in shark cartilage dietary supplements. Food Chem. Toxicol. 2014, 70, 26–32. DOI:10.1016/j.fct.2014.04.015 [Google Scholar]
-
Jiao Y, Chen Q, Chen X, Wang X, Liao X, Jiang L, et al. Occurrence and transfer of a cyanobacterial neurotoxin β-methylamino-L-alanine within the aquatic food webs of Gonghu Bay (Lake Taihu, China) to evaluate the potential human health risk. Sci. Total Environ. 2014, 468–469, 457–463. DOI:10.1016/j.scitotenv.2013.08.064 [Google Scholar]
-
Rush T, Liu X, Lobner D. Synergistic toxicity of the environmental neurotoxins methylmercury and be-ta-N-methylamino-l-alanine. Neuroreport 2012, 23, 216–219. DOI:10.1097/WNR.0b013e32834fe6d6 [Google Scholar]
-
Cox PA, Davis DA, Mash DC, Metcalf JS, Banack SA. Dietary exposure to an environmental toxin triggers neurofibrillary tangles and amyloid deposits in the brain. Proc. R. Soc. B Biol. Sci. 2016, 283, 20152397. DOI:10.1098/rspb.2015.2397 [Google Scholar]
-
Lopicic S, Svirčev Z, Palanački Malešević T, Kopitović A, Ivanovska A, Meriluoto J. Environmental Neurotoxin β-N-Methylamino-L-alanine (BMAA) as a Widely Occurring Putative Pathogenic Factor in Neurodegenerative Dis-eases. Microorganisms 2022, 10, 2418. DOI:10.3390/microorganisms10122418 [Google Scholar]
-
Xie X, Basile M, Mash DC. Cerebral uptake and protein incorporation of cyanobacterial toxin β-N-methylamino-L-alanine. Neuroreport 2013, 24, 779–784. DOI:10.1097/WNR.0b013e328363fd89 [Google Scholar]
-
Carollo A, Barbanti L, Gerletti M, Chiaudani G, Ferrari I. Quaderni IRSA; CNR: Rome, Italy, 1974. [Google Scholar]
-
Barbanti L, Bonomi G, Carollo A, Chiudani G, Ferrari I, Gerletti H, et al. Limnologia ed ecologia dei laghi di Bolsena, Bracciano, Trasimeno e Vico: situazione attuale e prevedibili conseguenze derivanti da una loro utilizzazione multipla; Istituto Italiano di Idrobiologia: Pallanza, Italy, 1971; pp. 1–26. [Google Scholar]
-
Gelosi E, Bazzanti M, Colombari P, Ferrara O, Mastrantuono L, Nicotra G, et al. Physico-Chemical and Biological Characteristics of Lake Vico, Central Italy. Preliminary Results. In International Symposium on Environmental Biogeochemistry, 1985, p. 42. Available online: https://www.bolsenalagodeuropa.net/wp-content/uploads/2021/08/stato-trofico-del-lago-di-Vico.pdf (accessed 3 February 2026). [Google Scholar]
-
Dyer M. The water quality at Lago di Vico during 1992–1993. Sci. Total Environ. 1995, 171, 77–83. DOI:10.1016/0048-9697(95)04677-8 [Google Scholar]
-
Mazza R, Capelli G, Teoli P, Bruno M, Messineo V, Melchiorre S, et al. Toxin contamination of surface and subsurface water bodies connected with Lake Vico’s watershed (Central Italy). In Drinking Water: Contamination, Toxicity and Treatment; Romero JD, Molina PS, Eds.; Nova Publishers Inc.: New York, NY, USA, 2008; pp. 1–100
.[Google Scholar] -
Laneve G, Téllez A, Kallikkattil Kuruvila A, Bruno M, Messineo V. Eutrophication and HAB Occurrence Control in Lakes of Different Origins: A Multi-Source Remote Sensing Detection Strategy. Remote Sens. 2024, 16, 1792. DOI:10.3390/rs16101792 [Google Scholar]
-
Bruno M, Messineo V, Mattei D, Melchiorre S. Dinamica di specie algali tossiche nei laghi di Albano e di Nemi; Rapporti ISTISAN; Istituto Superiore di Sanità: Rome, Italy, 2004; 04/32, 55p. [Google Scholar]
-
Messineo V, Bogialli S, Melchiorre S, Sechi N, Lugliè A, Casiddu P, et al. Cyanobacterial toxins in Italian freshwaters. Limnologica 2009, 39, 95–106. DOI:10.1016/j.limno.2008.09.001 [Google Scholar]
-
Messineo V, Mattei D, Melchiorre S, Salvatore G, Bogialli S, Salzano R, et al. Microcystin diversity in a Planktothrix rubescens population from Lake Albano (Central Italy). Toxicon 2006, 48, 160–174. DOI:10.1016/j.toxicon.2006.04.006 [Google Scholar]
-
Walsby AE, Avery A, Schanz F. The critical pressure of gas vesicles in Planktothrix rubescens in relation to the depth of winter mixing in Lake Zurich, Switzerland. J. Plank. Res. 1998, 20, 1357–1375. DOI:10.1093/plankt/20.7.1357 [Google Scholar]
-
Jacquet S, Briand JF, Leboulanger C, Avois-Jacquet C, Oberhaus L, Tassin B, et al. The proliferation of the toxic cyanobacterium Planktothrix rubescens following res-toration of the largest natural French lake (Lac du Bourget). Harmful Algae 2005, 4, 651–672. DOI:10.1016/j.hal.2003.12.006 [Google Scholar]
-
Lega M, Medio G, Endreny T, Casazza M, Esposito G, Costantino V, et al. Cyanobacterial biomonitoring in Lake Avernus during the COVID-19 pandemic: Integrating remote sensing and field data for pollution source detection. Int. J. Comput. Methods Exper. Meas. 2023, 11, 135–141. DOI:10.18280/ijcmem.110301 [Google Scholar]
-
Ferranti P, Fabbrocino S, Cerulo MG, Bruno M, Serpe L, Gallo P. Characterisation of biotoxins produced by a cyanobacteria bloom in Lake Averno using two LC–MS-based techniques. Food Addit. Contam. Part A 2008, 25, 1530–1537. DOI:10.1080/02652030802136196 [Google Scholar]
-
Bruno M, Messineo V. Extraction and Quantification of BMAA from Water Samples. In Protocols for Cyanobacteria Sampling and Detection of Cyanotoxin; Thajuddin N, Sankara Narayanan A, Dhanasekaran D, Eds.; Springer: Singapore, 2023. DOI:10.1007/978-981-99-4514-6_25 [Google Scholar]
-
Clausi MT, Vita V, Bruno M, Franchino C, Trifirò G, Palumbo MP, et al. Validation of ELISA methods for search and quantification of β-N-methylamino-l-alanine in water and fish tissue. Int. J. Environ. Anal. Chem. 2016, 96, 1290–1299. DOI:10.1080/03067319.2016.1249480 [Google Scholar]
-
Bruno M, Messineo V. Extraction and Quantification of BMAA from Fish Tissue. In Protocols for Cyanobacteria Sampling and Detection of Cyanotoxin; Thajuddin N, Sankara Narayanan A, Dhanasekaran D, Eds.; Springer: Singapore, 2023. DOI:10.1007/978-981-99-4514-6_26 [Google Scholar]
-
Utermöhl H. Neue Wege in der quantitativen Earfassung des Planktons (mit besonderer Berücksichtigung des Ultraplanktons). Internationale Vereinigung für theoretische und angewandte Limnologie Verhandlunge 1931, 5, 567–596. DOI:10.1080/03680770.1931.11898492 [Google Scholar]
-
Lund JWG, Kipling C, Le Cren E. The inverted microscope method of estimating algal numbers and the statistical basis of estimations by counting. Hydrobiology 1958, 11, 143–170. DOI:10.1007/BF00007865 [Google Scholar]
-
Fiore M, Parisio R, Filippini T, Mantione V, Platania A, Odone A, et al. Living near waterbodies as a proxy of cyanobacteria exposure and risk of amyotrophic lateral sclerosis: A population-based case-control study. Environ Res. 2020, 186, 109530. DOI:10.1016/j.envres.2020.109530 [Google Scholar]
-
Akporido SO, Onianwa PC. Heavy metals and total petroleum hydrocarbon concentrations in surface water of Esi River, western Niger Delta. Res. J Environ. Sci. 2015, 9, 88–100. DOI:10.3923/rjes.2015.88.100 [Google Scholar]
-
Wang DQ, Yu YX, Zhang XY, Zhang SH, Pang YP, Zhang XL, et al. Polycyclic aromatic hydrocarbons and organochlorine pesticides in fish from Taihu Lake: Their levels, sources, and biomagnification. Ecotoxicol. Environ. Saf. 2012, 82, 63–70. DOI:10.1016/j.ecoenv.2012.05.010 [Google Scholar]
-
Ostgaard K. The oil, the water and the phytoplankton. Adv. Limnol. 1994, 42, 167–193. [Google Scholar]
-
Feron VJ, Groten JP, Van Bladeren PJ. Exposure of Humans to Complex Chemical Mixtures: Hazard Identification and Risk Assessment. In Diversification in Toxicology—Man and Environment; Seiler JP, Autrup JL, Autrup H, Eds.; Springer: Berlin/Heidelberg, Germany, 1998; pp. 363–373. [Google Scholar]
-
Almeda R, Cosgrove S, Buskey EJ. Oil spills and dispersants can cause the initiation of potentially harmful dinoflagellate blooms (“red tides”). Environ. Sci. Technol. 2018, 52, 5718–5724. DOI:10.1021/acs.est.8b00335 [Google Scholar]
-
Krewski D, Thorslund T, Withey J. Carcinogenic Risk Assessment of Complex Mixtures. Toxicol. Ind. Health 1989, 5, 851–867. DOI:10.1177/074823378900500520 [Google Scholar]
-
Heys KA, Shore RF, Pereira MG, Jones KC, Martin FL. Risk assessment of environmental mixture effects. RSC Adv. 2016, 6, 47844–47857. DOI:10.1039/C6RA05406D [Google Scholar]
-
Caller TA, Doolin JW, Haney JF, Murby AJ, West KG, Farrar HE, et al. A cluster of amyotrophic lateral sclerosis in New Hampshire: A possible role for toxic cyanobacteria blooms. Amyotroph. Lateral Scler. 2009, 2 (Suppl. S2), 101–108. DOI:10.3109/17482960903278485 [Google Scholar]
-
Mello FD, Braidy N, Marçal H, Guillemin G, Nabavi SM, Neilan BA. Mechanisms and effects posed by neurotoxic products of cyanobacteria/microbial eukaryotes/dinoflagellates in algae blooms: A review. Neurotox. Res. 2018, 33, 153–167. DOI:10.1007/s12640-017-9780-3 [Google Scholar]
-
Torbick N, Ziniti B, Stommel E, Linder E, Andrew A, Caller T, et al. Assessing cyanobacterial harmful algal blooms as risk factors for amyotrophic lateral sclerosis. Neurotox. Res. 2018, 33, 199–212. DOI:10.1007/s12640-017-9740-y [Google Scholar]