The Denture Mycofilm Plaque: A Literature Review
Pierre Le Bars 1,* Ayépa Alain Kouadio 1,2 Octave Nadile Bandiaky 1,3 Yves Amouriq 1
Received: 20 October 2025 Revised: 08 December 2025 Accepted: 16 March 2026 Published: 26 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
A removable prosthesis provides a rapidly colonized substrate for a specific microbial plaque often referred to as denture biofilm (or denture mycofilm plaque) (DMP) [1,2]. Interactions between this microbial community and the physicochemical properties of denture materials quickly lead to a new ecological equilibrium [3,4].
Thus, the DMP is likely to disperse and convey its microbial flora in continuity with the microbiome of the aerodigestive tract. The latter can cause dysbiosis, leading to harmless and/or pathogenic infections [5,6,7,8]. In addition, recently the impact of the material, the denture base in polymethyl methacrylate, of micro and nano size on the intestinal microbiota and the lipid metabolism of the liver has been highlighted [9].
This removable prosthesis is removed from the patient’s mouth after each meal; for hygiene reasons, it brings two types of surfaces into contact [10]. One surface is inert, or abiotic, consisting of a polymer (polymethyl methacrylate (PMMA), polyamides (nylon), polyether ether ketone (PEEK), polycarbonate, polyester), or metal part of a prosthesis base and the other is alive, or biotic, essentially in the form of different oral mucous membranes (representing a complex, genetic, biological, and biochemical entity), composed of host cells in the presence of an extracellular matrix (ECM). The DMP generally grows more easily on the intaglio surface than on the cameo surface of the prosthesis [11]. The intaglio or intrados denture adjustment area is deliberately not polished in order to best preserve the prosthetic adaptation while retaining the relief of the palatal vault. In addition, the mandibular movements exercised during chewing, speaking, and swallowing subject this ecosystem to pressures generated by the kinematics of the contact between the denture and the oral mucosa. These pressures (force: N/mm2, expressed in Pascal) are broken down for educational purposes into vertical forces and horizontal tangential shear forces. In reality, the pressures are exerted in both directions concomitantly.
Other mechanical and chemical stresses are exerted on the denture plaque, such as variations in the composition and flow rate of saliva [12,13,14]. Given the frequency of denture stomatitis (affecting three-quarters of patients) and the lack of consensus regarding hygienic, mechanical, and chemical methods for removing denture mycofilm, our objective is to understand its resistance better. This characteristic is related to the microbial composition and architectural adaptability of the denture mycofilm. This knowledge can influence the preventive and curative management of denture stomatitis.
2. Materials and Methods
This literature review was conducted in accordance with the PRISMA 2020 (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and was prospectively registered in PROSPERO 2025 (International Prospective Registry of Systematic Reviews) under the identification number CRD420251052219. The PICO framework was used to formulate the research question: What are the characteristics of Denture Mycofilm Plaque (DMP)? (P: human subjects; I: partial removable dentures; C: complete dentures; O: prevalence and mechanical/physical characteristics of biofilms).
An electronic search was conducted in PubMed/Medline, Scopus, Web of Science Core Collection, Embase, and the Cochrane Library, without date restriction, until 2025.
The search strategy incorporated both free-text terms and controlled vocabulary (MeSH/Emtree), adapted to each database. The primary search terms included: “denture biofilm”, “denture mycofilm”, “denture plaque”, “Candida biofilm”, “denture stomatitis”, “oral biofilm”, “prosthesis-related infections”.
Eligible studies included: original research articles (clinical, in vitro, or in vivo), observational studies (cross-sectional, case–control, or cohort designs), clinical trials investigating denture-associated biofilms, microbiological studies assessing the composition or properties of denture mycofilm.
Studies were included if they met the following criteria: (1) if they were conducted on human subjects or on biological samples directly related to the use of human dental prostheses, (2) if the participants wore partial or complete removable dental prostheses, (3) if the studies characterized the mycofilm plaque of dental prostheses, including its composition, structural characteristics, adhesion mechanisms or mechanical and physical properties, (4) if the articles were written in English, (5) if the articles provided extractable and analyzable data relevant to the research question.
Studies were excluded if: (1) they focused solely on dental biofilms unrelated to dentures, (2) they used animal models or were exclusively theoretical. (3) they lacked sufficient data for extraction or presented incomplete or non-quantifiable results. (4) they were narrative reviews, editorials, letters to the editor, or conference abstracts. (5) Full-text access was unavailable, or the article was not written in English.
Two independent reviewers (P.L.B. and O.B.) carried out the entire review process, including database searching, study screening (titles, abstracts, and full-text assessment), data extraction, and evaluation of methodological quality and risk of bias. Discrepancies were resolved through discussion or consultation with a third reviewer when necessary.
3. Results
3.1. Composition of Microbial Denture Plaque
Based on modern genomic analyses, it has been established that the composition of the oral microbiome includes more than 700 species of bacteria, archaea, fungi, viruses, and protozoa [15,16,17]. Bacterial and fungal sequencing studies of the various denture colonization sites revealed different habitats sheltering microbial communities that are dependent on access to oxygen and nutritional products available in saliva. This condition, essentially of infectious origin, has given rise to numerous microbiological analyses.
The results of studies suggest the existence of distinct biofilms in healthy subjects and in patients suffering from denture stomatitis (DS) [18]. However, this difference in biofilms [19,20,21] appears only at the species level, not at the genus level. Another particularity of the richness and diversity of the denture microbiome is the influence of the presence of natural teeth. For O’Donnell [22], the presence of natural teeth with periodontal disease can increase DS more than in totally edentulous patients.
Thus, the denture constitutes a microbial reservoir for numerous bacteria with pathogenic potential [23]. The same can be said regarding the constant presence of Candida spp. [18,19,22,23] but the absence of Archaea [4]. The more abundant bacterial genera were reported to be Actinomyces, Capnocytophaga, Streptococcus, and Veillonella in both groups, i.e., in individuals with DS and without DS (independently of the surface). These are the major colonizers of dentures [18,22,24].
Finally, regarding prosthetic hygiene, biofilm development is faster and more complex on natural teeth than on dentures [25]. Between a clean prosthesis and an unclean one, there is no statistically significant difference in the overall microbiome (uniformity and richness). Study shows an increase in the microbial index of pathogenic bacteria (PIMs), which are generally harmless microorganisms associated with a potential microbiological risk. The PIMs (Streptococcus oralis, Streptococcus mutans, Streptococcus sobrinus, Ralstonia mannitolilytica, Haemophilus parainfluenzae, Klebsiella varicola, and Porphyromonas gingivalis) were observed in the non-hygienic group compared to the hygienic group [4]. One more multi-kingdom analysis revealed additional microbial species beyond Candida spp. providing new insights into the DS-associated shifts [26] (Table 1).
Table 1. Different characteristics of prosthetic biofilms.
|
Authors |
Denture Wearers Population Origine |
Location of Samples |
Culture or NGS |
Study Objectives |
Species Level |
Results |
|---|---|---|---|---|---|---|
|
Campos et al., 2008 [18] |
10 healthy denture wearers/10 DS (USA) |
Samples on the denture |
Culture-independent method, obtained by PCR |
Comparison of microbial composition between healthy denture wearers and DS. |
29 bacterial species exclusively on DS individuals. Candida sp. was detected from the ITS, in both biofilms. |
Existence of distinct biofilms in healthy subjects and in patients with DS. |
|
Teles et al., 2012 [25] |
62 Edentulous full denture wearers/55 dentate patients: periodontally healthy (N = 38) and chronic periodontitis (N = 17) (USA) |
Supragingival plaque samples were taken from dentate subjects and the denture teeth before and after professional cleaning |
Analyzed using checkerboard DNA–DNA hybridization. Counts and proportions of 41 bacterial taxa |
The aim of this study was to compare the re-development of biofilms on natural teeth and dentures. |
Pre-cleaning edentate Biofilms had higher counts and proportions of Streptococcus mitis, Streptococcus oralis and Streptococcus mutans, whereas dentate subjects had higher proportions of Tannerella forsythia, Selenomonas noxia and Neisseria mucosa |
Post-cleaning Biofilm re-development is more rapid and more complex on the natural teeth than on dentures. |
|
O’Donnell et al., 2015 [22] |
123 Denture wearers with partial or complete maxillary denture and at least one tooth. 45 DS versus 78 no DS (Scotland) |
Three samples, on the palatal mucosa, on the denture and the supragingival mucosa |
Illumina MiSeq detection (bacteria). Culture found 72% prevalence of Candida species on dentures. |
Can the presence of periodontal disease influence the development of (DS)? |
Patients with teeth showing microbiota diversity > Edentulous patients. |
The presence of natural teeth with periodontal diseases can increase DS more than in totally edentulous patients. |
|
Shi B et al., 2016 [19] |
19 patients with a complete removable denture and with a minimum of four remaining teeth (9 DS vs. 10 no DS) (China) |
Two samples, denture and teeth for groups with DS and no DS. |
454 FLX titanium platform. Detection of Candida via PCR. |
Is there a difference between the microbiome of a denture and that of the natural teeth of the same patient? |
Fusobacterium nucleatum subsp. Animalis is always present on the intrados of the denture with DS. |
Significant differences exist between DS and no DS at the species level. |
|
Morse DJ et al., 2019 [21] |
8 DS versus 11 no DS Complete unimaxillary prosthesis (England) |
3 samples per patient (tongue, palatal mucosa, intrados of the denture). |
Illumina MiSeq + detection of Candida by culture and by PCR |
Is there a difference between microbiota of the tongue with DS versus healthy palatal mucosa? |
On the denture fitting surface of NDS Acinetobacter johnsonii and Streptococcus mitis were nearly threefold higher compared with DS, and Actinomyces odontolyticus was sixfold higher |
Significant differences exist between no DS and DS at the species level. |
|
Fujinami et al., 2021 [23] |
18 healthy, denture-wearing individuals with no DS; 2 with no remaining teeth wore full dentures, 7 with implants or root caps wore full over dentures, and nine participants wore partial removable dentures with some remaining teeth (Japan) |
Samples:
|
llumina MiSeq platform |
What is the prevalence of Candida albicans in plaque Samples? |
Correlation between bacterial genera and C. albicans. Lactobacillus, followed by Scardovia and Bifidobacterium, showed the most significant positive correlation. By contrast, Leptotrichia exhibited the most negative correlation |
The prevalence of Candida albicans is: 100% outside the prosthesis, 93% inside the prosthesis, and 94% on teeth/implants. Dentures may be signifiant reservoirs of pathogen. |
|
Lim TW et al., 2024 [4] |
16 individuals with complete and 81 with partial dentures with half PMM resin and other metallic framework (MIP between clean and unclean) (Hong-Kong) |
Quantification of prosthetic plaque was determined via planimetric evaluation. |
2bRAD metagenomic sequencing. Then, the purified products were sequenced using the Illumina Novaseq 6000 platform |
Does prosthetic hygiene influence the composition of the microbiome? |
Among 1989 species in the saliva, Streptococcus mutans, Streptococcus sobrinus, Ralstonia mannitolilytica, Haemophilus parainfluenzae, Klebsiella variicola, and Porphyromonas gingivalis were the most abundantly detected pathogenic bacteria. |
The overall microbial evenness and richness in both groups (clean and unclean denture) showed no statistically significant difference. However, there is an increase in MIP in the unclean group compared to the clean group. |
|
Manzoor et al., 2025 [26] |
28 with DS and 28 without signs of DS (Finland) |
Microbial sampling was performed from the denture fitting surface using a sterile curette. |
Shotgun sequencing was performed on an Illumina NovaSeq 6000 instrument (Helsinki, Finland) with 150-bp paired-end sequencing. Helsinki. Finland. |
Is there a difference in the evenness and richness (alpha diversity) of microbial communities at the species level? Is there a difference between DS and non-DS groups? |
There are no significant differences in microbial community evenness and richness (alpha diversity) at the species level. In contrast, beta diversity was significantly different between the DS and non-DS groups. |
Results indicate that DS is associated with significant alterations in the denture-associated oral microbiome. Consideration of bacterial and fungal interactions sheds light on the pathogenesis of DS. |
Microbial characteristics of prosthetic biofilms. In summary, despite the heterogeneity of investigative methods, most studies confirm a difference in microbiome composition between (NDS) and (DS) [18,19,21]. The presence of natural teeth influences this composition [22], as does oral hygiene [4,25,26]. The presence of Candida albicans is also consistently noted [23,26].
NGS: next-generation sequencing; PCR: polymerase chain reaction; DS: denture stomatitis; NDS: absence of denture stomatitis; ITS: internal transcribed spacer; MIP: microbial index of pathogenic bacteria.
A new microbiological investigation of plaque using Type IIB Restriction-site Associated DNA for Microbiome (2bRAD-M) sequencing, provides access to the species taxonomy level for prosthesis biofilms [27].
Among the top 30 species colonizing prosthesis surfaces, Streptococcus oralis, Streptococcus mutans, Streptococcus sobrinus, Ralstonia mannitolilytica, Haemophilus parainfluenzae, Klebsiella variicola, and Porphyromonas gingivalis were recognized pathogens [4].
Knowledge of the well-established chronology of the maturation of the DMP allows for better control in terms of hygiene [28].
3.2. Chronology of the Maturation of Denture Biofilm
Some authors support the hypothesis that biofilm formation, at least in its initial phase, depends primarily on the phenomenon of adhesion to the various colonized surfaces [29,30] Figure 1. This phenomenon is particularly evident with regard to the adhesion of C. albicans to the denture base. Adhesion also depends on the surface roughness of the materials; it is greater with thermosetting PMMA than with CAD/CAM-pre-cured PMMA. Thus, it can be concluded that new-generation prosthetic base materials, in terms of adhesion, are better suited for clinical use [31,32].
An explanation may lie in the role of factors specific to each individual, such as saliva, which can determine the microbial composition of oral bacterial biofilm in relation to surfaces [19]. Previous research has confirmed that saliva is an important variable among individuals [33,34]. Indeed, saliva is the predominant host parameter involved in adhesion [35]. Saliva contributes to maintaining good oral health. It protects, lubricates, and hydrates the denture base. The physicochemical properties of resins offer varying surface textures that can influence the adhesion of Candida albicans. Thus, milled acrylic bases present a less favorable environment for adhesion than conventionally manufactured materials. During mastication with a denture, the increase in the amount of saliva secreted but also the composition of salivary metabolites, is implicated. A recent study shows that improved masticatory function following restoration of occlusion with a denture can affect salivary metabolites. Metabolomic analysis demonstrated a significant decrease in four substances in resting saliva (N1-acetyl spermidine, betaine, malate, and 2-hydroxy-4-methyl pentanoate) [36].
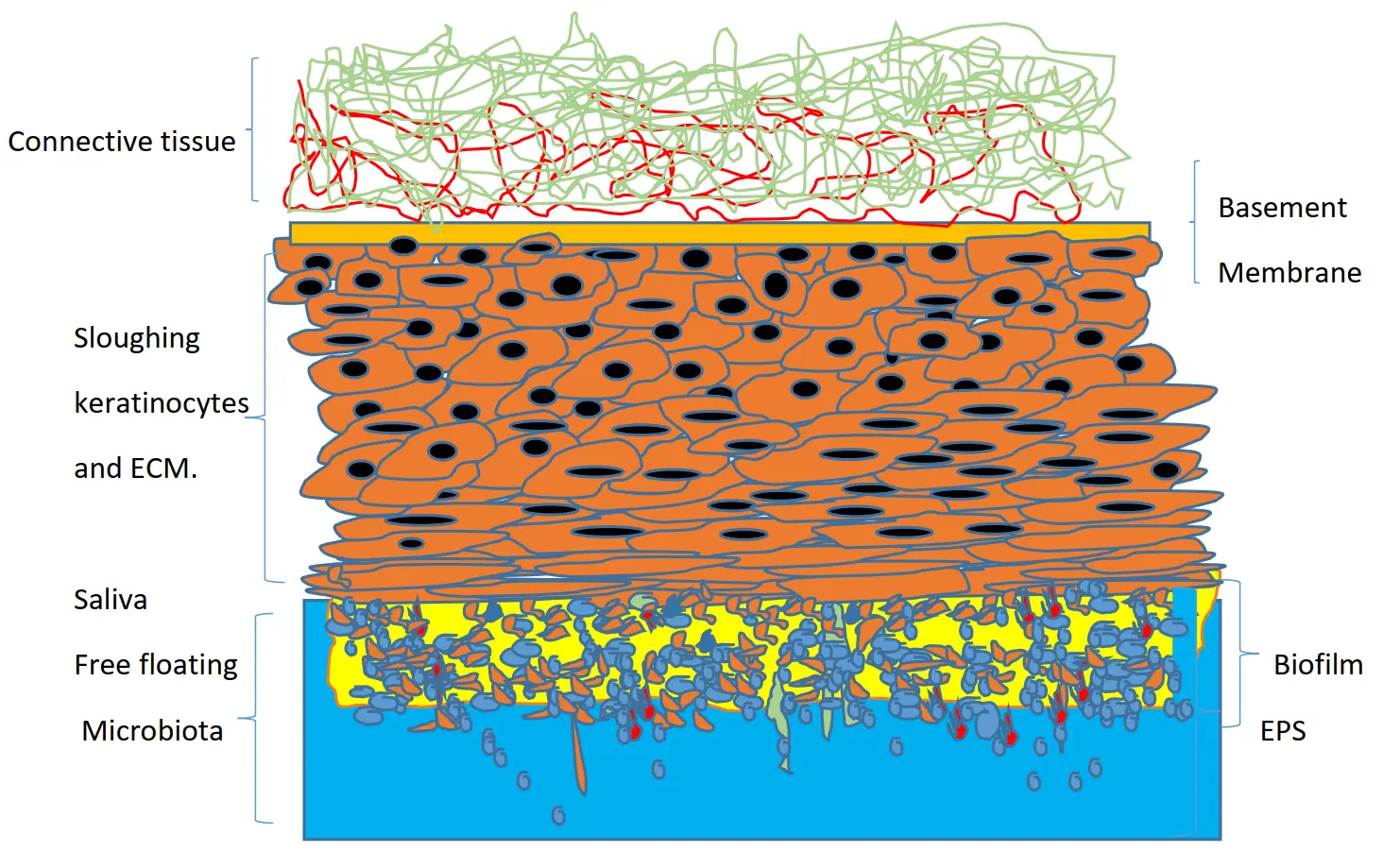 |
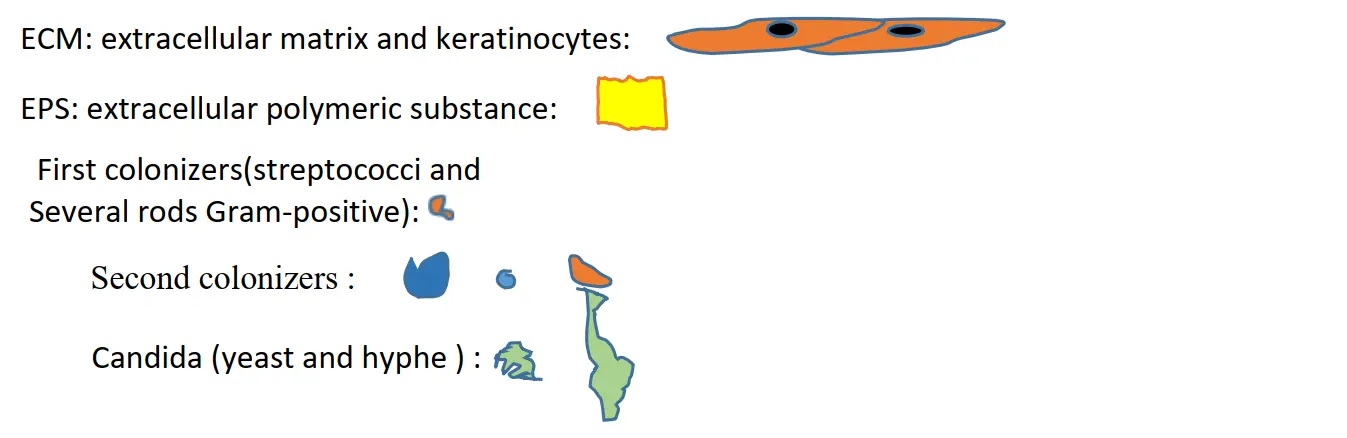 |
Figure 1. Proposal for a schematic representation of the mature biofilm on the healthy mucosal surface. Saliva and free floating microbiota, nutrients, and the presence of commensal morphotype bacteria and fungi influence the architecture and characteristics of the biofilm.
3.3. Salivary Films Instantly Coat Dentures
The first step in the formation of a denture biofilm is acquired pellicles (salivary-conditioning film; SCF). Saliva interacts with the complex environment of the oral cavity. The SCF comprises a first thin, dense, and resistant layer that forms a network rich in proteins (statins, histatins, prolines, mucins), in direct contact with the material. The upper layer is thicker but less dense and resistant [37].
The adsorption of glycoproteins and mucins during SCF formation promotes water retention, thereby influencing the surface properties of the oral mucosa and biomaterials. This phenomenon explains why these hydrophobic surfaces tend to become hydrophilic [38].
Then, the formation process, including many bacterial species, follows a temporal sequence [39].
Thus, concomitantly, passive microbial adhesion to the surface of the denture quickly follows physicochemical interactions such as Brownian motion as well as hydrophobic and electrostatic interactions, accompanied by van der Waals forces. Subsequently, adhesion of the first bacterial colonizers to prosthetic surfaces occurs through molecular and cellular interactions [40].
Concerning microbial adhesion to the denture base, this phenomenon depends on the roughness, the surface energy of the different materials used, and the contact angle of saliva, rather than on selective phenomena at the biotic surface of the mucosa [41,42]. Streptococcal species are the first colonizers and will prepare the microenvironmental conditions to facilitate the settlement of subsequent colonizers. Among the latter, Fusobacterium nucleatum is considered to be the link between the first and second colonizers, including Aggregatibacter actinomycetemcomitans, Prevotella intermedia, Eubacterium spp., Treponema spp., and Porphyromonas gingivalis [41].
At this stage, if the prosthetic biofilm is not eliminated, its maturation can gradually result in the appearance of a microenvironment that favors the growth and resistance of the biofilm. Under the denture, the depletion of oxygen and nutrients promotes the growth of anaerobic species capable of attaching to and colonizing the biofilm [18] .
In addition to the majority of prokaryotic organisms, the contribution of Candida species frequently constitutes a feature of the denture biofilm. It should be noted that the presence of non-albicans Candida species in people using removable dental prostheses has recently been implicated as a potential challenge in the management of DS [42,43].
In fact, Candida can attach directly to PMMA surfaces but also to the denture covered with bacterial biofilm, including various species of Streptococci and to Fusobacterium nucleatum [43,44,45] (Figure 2).
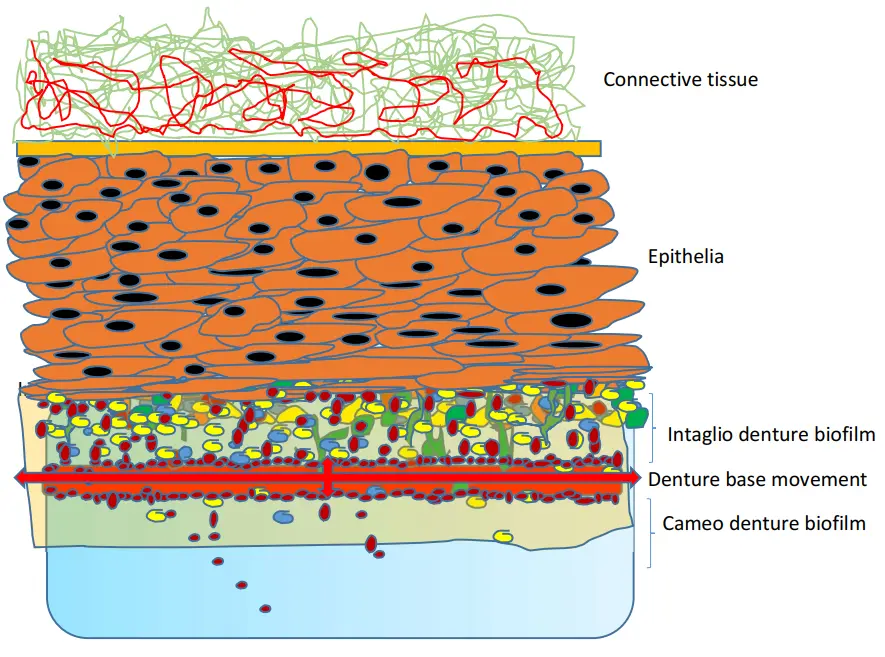 |
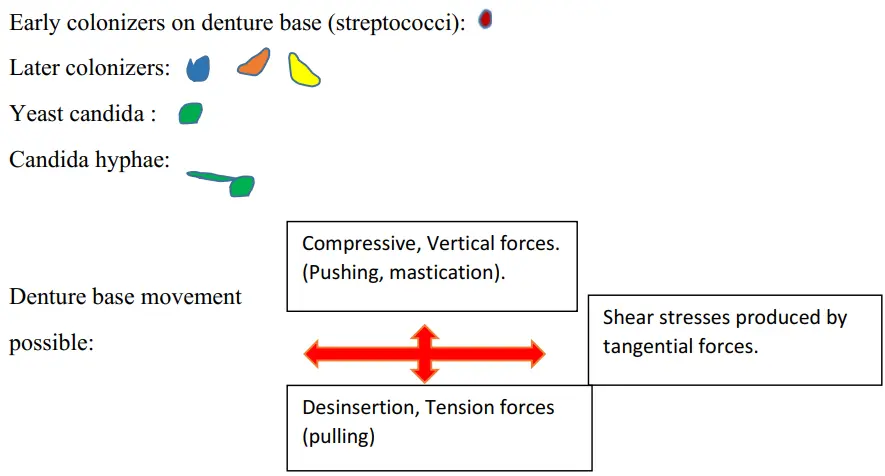 |
Figure 2. Proposition schematic of palatal mucosa in the presence of early denture microbial-related stomatitis. The denture promotes a dysbiosis of oral microbiome with an overgrowth of C. albicans and numerous commensal bacterial species. Biofilm generally grows more on the intaglio surface than on the surface of the cameo prosthesis. The instantaneous resistance of the mucosa results from both the rigid matrix structure (epithelial layer, fibrous network, blood vessel) and fluids. Bacterial species most frequently isolated with Candida albicans from these specific niches of the oral cavity (Streptococcus spp.: S. gordonii, S. mutans, S. salivaris). Saliva, moisture, nutrient, hyphal Candida morphotype and presence of commensal bacteria influence the architecture and virulence characteristics of mucosal fungal biofilms, according to Bertolini et al., 2015 [45].
Another particularity is the occurrence of biofilm, characterized by the transition from planktonic, individual behavior of a microorganism to a fixed, community trait. A change in microbial phenotypes accompanies this shift. At the genetic level, this translates into adaptations in activation mechanisms, enabling modification of cellular functions within the biofilm [46].
An additional aspect of saliva is the recent notion of salivary biofilm [47]. Microorganisms can spontaneously aggregate in saliva without support; these aggregates can also attach to biofilms on the prosthesis surface. Thus, certain salivary glycoproteins and proteins of the host mucosa are included in the extracellular polymeric substances (EPS).
This different information on the complexity of the oral ecosystem in relation to the biofilm attached to the surface of the denture can guide the choices regarding daily hygiene strategies (mouthwashes, toothpaste, tablets).
3.4. Microbial Denture Plaque and Hygiene Strategy
In the absence of hygiene, the denture, by promoting microbial colonization and mechanical interactions between non-pathogenic species and their host, contributes to epithelial damage and thus to the maintenance of chronic inflammation such as denture stomatitis (DS) [48].
This mucosal inflammation in the presence of saliva promotes an immune response. Saliva contributes to the release of anti-inflammatory cytokines but also to the cleavage of chemokines mediated by MMPs (matrix metalloproteinases) and to the remodeling of the ECM of the oral mucosa. Every night, once the mucosa is relieved of the prosthetic covering, the host–microbial balance tends to be restored. This phenomenon is attenuated thanks to salivary, but also by the transformation of proinflammatory macrophages of an M1 phenotype to an M2 phenotype, which secrete growth factors to stimulate ECM remodeling mediated by fibroblasts.
However, this hypothesis is called into question by authors who emphasize that the differences between the bacteria causing acute infections and chronic diseases are due to the metabolic activity of microorganisms rather than to cellular aggregation [49,50]. One more, dead and viable microbial cells can survive in the biofilm [51] even after cleaning.
The presence of C. albicans in biofilms significantly conditions the competition and structure of the interbacterial community on both abiotic and biotic substrates [52]. Candida spp. present as commensal microorganisms in the oral cavity that proliferate and promote the appearance of DS. If daily maintenance of prosthetic hygiene is insufficient, several microorganisms, such as Candida albicans, can survive within the base material [49,53,54].
Removal of denture microbial plaque also depends on understanding the mechanical characteristics of biofilms.
3.5. III-Physical Properties of Mucosa, Saliva, and Mycofilm under Dentures
Understanding the simultaneous biomechanical behaviors of the mucosa, saliva, and mycofilm will help to better optimize the prosthodontic treatment. Thus, the stability of a removable, mucosal-supported prosthesis depends on the viscoelastic properties of the mucosa. The quantity and quality of saliva will influence prosthetic adhesion and friction on the mucosa. Finally, the presence of a mycofilm can, by promoting inflammatory processes, disrupt prosthesis adhesion.
3.6. Mucosa
Historically, the physiological changes in the mucous membranes under prostheses have been described by various authors [55,56,57,58]. Many parameters influence the episodic daily cohabitation of the denture in the mouth, including the following: the proximity of the denture base to the epithelial cells and the teeth, the salivary pH (unstimulated 6.8, stimulated 7.8), the oral temperature (35.5 to 37.5 °C), the hygiene procedure, and also the diet, the air breathed, and the general condition of the patient (immune competence, diabetes, cancer) [59].
These different parameters can be analyzed through the following properties:
- (a)
-
Concerning the static response, the elastic modulus from the hard palate mucosa is 18.1 ± 4.5 MPa, and the tensile strength is 1.70 ± 0.87 MPa [60].
- (b)
-
For the dynamic response, the viscoelasticity is around (8.0 ± 3.0) × 10−5 Gpa—The biofilm on the surface of the epithelium undergoes the effects of viscosity, permeability, and the elasticity of the mucosa.
- (c)
-
Poisson’s ratios (ʋ) of the biofilm are estimated in vitro by compressing the biofilm in the vertical direction and measuring its modulus of elasticity.
Experimentally, it was found that the modulus K of a biofilm is around 130 kPa, which is much higher than the modulus of shear G′; the calculation of Poisson’s ratio is then ʋ = (3K − 2G′)/2(3K + G′) = 0.495, which is close to the incompressible limit of ʋ = 0.5 [61].
The friction of the prosthesis on the mucosa promotes and maintains chronic pathologies such as inflammation, angular cheilitis, and epithelial dysplasia. The commonly accepted coefficient ranges from 0 (no friction) to 0.75 (damaging contact). Saliva modulates this coefficient; thus, the coefficient of friction is around 0.213 for the hydrated mucosa. The lubricating function of saliva is closely related to the glycosylation of many salivary proteins, such as mucins. On the other hand, mucins, by facilitating humidity, provide saliva with its viscoelastic and rheological properties [62,63,64,65].
3.7. Viscosity Is a Property That Also Characterizes Saliva
The salivary flow that increased immediately following denture insertion returned to almost normal levels 6 months later [66]. In the presence of a good prosthetic fit only, salivary flow contributes to retention, thanks to surface tension forces at the periphery of the complete denture [67].
Regarding the oral epithelium, the salivary mucus film that covers it has a thickness estimated at 70–100 μm [7]. Salivary molecules selectively adsorbed to the mucosal surface facilitate a change from hydrophobic to hydrophilic properties. This is the same for acrylic surfaces, which are easily wetted by saliva containing proteins and mucopolysaccharides absorbed onto the surface. Hydrophilicity facilitates microbial adhesion, such as that of yeasts, to prosthetic materials, but this phenomenon remains poorly understood [68,69,70]. From a mechanical point of view, the contact between the prosthetic base and the oral mucosa is dependent on tribology (a discipline that studies the principles of friction, lubrication, and wear of interacting surfaces) [71].
In reality, biofilm formation varies little depending on the prosthetic surfaces (contact angle and free energy). The properties specific to biofilms, such as viscosity and lubrication, characterize the sliding of the denture but have little influence on retention [72].
Between the two surfaces, the rheology (deformation and flow of biological lubricants) of saliva under a force depends inversely on viscosity. The fact that saliva’s viscosity is much higher than that of air explains the effectiveness of adhesion when the fluid fills the space. Under these conditions, the denture base coating with saliva and in contact with soft surfaces modifies the salivary and sessile physiology of the oral flora. Consequently, the coating and pressure of the denture influence the mechanics of the soft surfaces (e.g., epithelium, connective tissue). However, our knowledge remains limited regarding the spatial organization of the biofilm and, in particular, its mechanical behavior during morphogenesis.
3.8. Biofilm
Other features characteristic of the ill-fitting surface of denture mycofilm include mechanical, chemical, and physicochemical parameters. The biomechanics of biofilms between the mucosa and the denture base involve the static, dynamic, and volumetric aspects.
Concerning viscoelasticity, various results show that biofilms have the ability to resist mechanical stress by dissipating energy from the outside [73]. The mechanical adaptation properties of the biofilm result from both its viscous and elastic character, which promote its strengthening [74,75].
Thus, the physical phenomenon of viscoelasticity finds its application at the level of the denture mycofilm. From the start of its development, the microbial biofilm of an ill-fitting denture surface, subjected to the external forces of the oral cavity and its ecosystem, adapts its microbial structure. For didactic reasons, we consider that the biofilm of the intrados denture is subjected to normal compressive vertical forces, measurable (elasticity, rigidity) thanks to Young’s modulus (E) compression (pushing), tension (pulling), or dynamic (cycling of compression and tension) modes but also to shear stresses produced by tangential forces (measured with rheology systems). Another advantage of viscoelasticity is the recovery capacity of the biofilm after removal of the stress [76]. In vivo, these different stresses occur concomitantly during the day, particularly during chewing. Removal of the denture at night to eliminate the stress imprinted on the mucosa facilitates the return of the biofilm to normal. In these conditions, viscoelasticity contributes to biofilm survival by promoting its expansion under mechanical stress [73].
The viscoelastic behavior of the mycofilm comes in part from its composition. The microorganisms within the biofilm multiply while embedding themselves in a matrix of extracellular polymeric substances (EPS), which forms a crosslinked polymer [77].
Concerning viscoelasticity, EPS is a structure with a dual role: it establishes the link between cells within the biofilm and can also resist and adapt to the various pressures in the oral cavity. Additionally, EPS promotes bacterial aggregation on both natural and artificial surfaces. EPS also resists the flow of saliva and the host’s immune defenses [78,79].
The microbial composition influences the viscoelastic behavior of biofilm. Polymicrobial biofilms have a low Young’s modulus, of the order of Pa to kPa [80]. Likewise, it was observed for biofilms of Pseudomonas fluorescens, most commonly found in natural water systems (present (7%) on the intrados of the denture with DS) [81], that having more EPS than cells confers a more ductile behavior [82]. On the contrary, biofilms made up of Streptococcus mutants only and presenting a low quantity of EPS are rigid [83]. In vitro, staphylococcal biofilms show their ability to adapt to the shear force of fluids. This adaptation occurs at the level of the EPS, and more particularly in the rigidity of the molecular bonds [84]. On the other hand, EPS components enhance biofilm cohesion [85].
3.9. Shear Stress and Biofilm
The fluid that occupies the space between the denture base and the palatal mucosa is subjected, among other things, to shear forces. This situation can be reproduced in vitro.
In these experimental conditions, biofilms could generate significant mechanical stresses of up to 100 kPa. Also, in laboratory studies, biofilms can deform and even damage soft tissue engineered epithelia. Some authors explain this phenomenon by the opposition between the growth of the biofilm and friction with the surface, which generates internal pressure within the biofilm, oriented towards its center [86].
This suggests that the forces produced by biofilms under prostheses are likely to both influence their own architecture and be subject to pressure, thereby compromising the mucosal physiology.
In this condition, via denture pressures, biofilms could deform the mucous membranes by transmission of internal stresses. Thus, the question is:
Do denture biofilms subject the mucosa to pressure to facilitate their growth? We know that different epithelial cell–cell junctions break under the effect of a few kilopascals [87].
Experimentally, after 12 h of growth, the reaction forces are surprisingly large, reaching 100 kPa in the center of the biofilm. Consequently, biofilms can deform the surface of the soft materials on which they grow. This characteristic of biofilm growth occurs independently of the bacterial species tested [88].
3.10. Adaptive Capacity of Biofilm under Dentures
In the space between the mucosa and the denture base, the microorganisms included in these biofilms can react to surrounding mechanical instabilities. By changing their position, they can adapt to the verticalization of pressure, thereby overcoming horizontal cell-surface adhesion. This phenomenon can modify cell growth that is oriented differently [89].
In vitro, viscoelasticity facilitates the survival of biofilms when exposed to shear strength. It promotes fluid behavior and viscous flow. In this situation, the consistency of the biofilm favors its resistance to mechanical and chemical hygiene means [90,91]. Thus, the viscoelastic behavior of the biofilm could increase its virulence and promote chronic infections such as DS.
The growth of biofilms on the surface of soft tissues can exert pressures that deform the epithelia (Figure 3). On the other hand, epithelial integrity can be compromised by the pathogenic flora of inflammatory diseases such as periodontitis, caries, mucosal pathologies, or DS; the process of which depends on the composition of the microbiota. For example, bacteria such as Pseudomonas aeruginosa are likely to form biofilms, which have the particularity of modifying the architecture of the host’s tissue surfaces [92]. Thus, in the near future, several studies suggest significant advances concerning the physiology of the oral microecology [93].
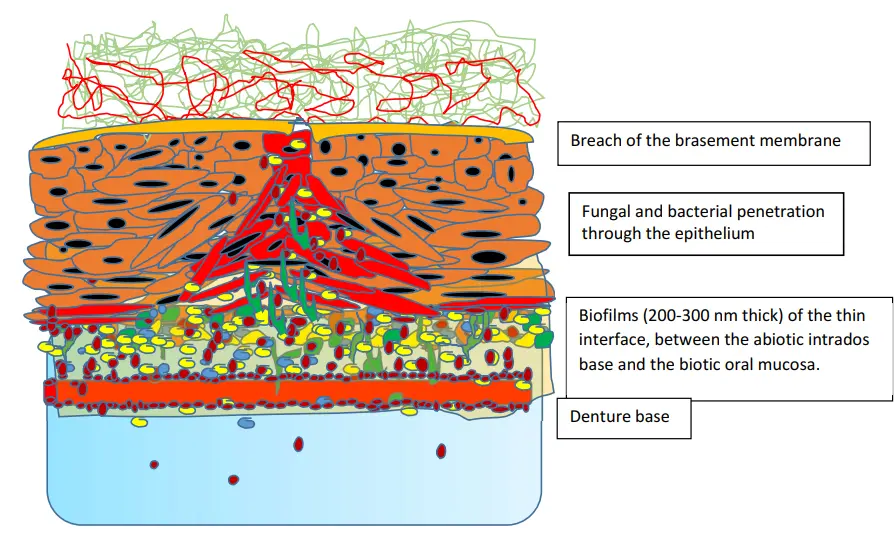 |
 |
Figure 3. Proposal for a schematic representation of palatal mucosa in the presence of denture microbial-related stomatitis. The rigidity of the denture base facilitates microbial penetration into the epithelium. The growth of the biofilm favored by the covering of the prosthesis promotes microbial penetration. The epithelial cells and ECM—under denture pressure—modify their shape and arrangement, facilitating bacterial penetration. In vitro, research shows that soft substrates represent a much more favorable environment than solid substrates with respect to microbial invasion.
4. Conclusions
The recent in vitro advances on the deformation of biofilms attached to soft surfaces and the modification of epithelia attached to the biofilm have provided new insights. Understanding the interactions between biofilm, saliva, immune competence, and the denture involves the following fields: (1) histological, (2) mechanical, and (3) biological. This can shed light on the global approach to microbial colonization, dysbiosis, and stomatitis within the oral ecosystem of denture wearers. Among the many parameters influencing denture biofilm, the mechanics and morphogenesis, viscoelasticity, interfacial energies, and the phenomena of sliding and friction on the substrates (between mucosa, denture base, and saliva) constitute a little-exploited field of investigation. Given the lack of in vivo experiments to confirm in vitro studies concerning these different parameters, reservations are necessary pending further investigations.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
In preparing this manuscript, the authors conducted an electronic search in PubMed/Medline, Scopus, Web of Science Core Collection, Embase and the Cochrane Library, up to 2025. After using this tool/service, the authors reviewed and corrected the content as needed and assume full responsibility for the content of the published article.
Acknowledgments
The authors thank Word technology for making the illustrations possible.
Author Contributions
P.L.B.: article conception, writing, and illustration. A.A.K.: conceptualization, literature review, proofreading, and editing. O.N.B.: conceptualization, methodology, validation, writing (first draft), proofreading, and editing. Y.A.: proofreading.
Ethics Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Readers can access all original articles cited in this review. Access to this article-related data is available on PubMed.
Funding
This research received no external funding.
Declaration of Competing Interest
The four authors declare that there are no financial or personal relationships with other persons or organizations that could unduly influence (bias) their work.
References
-
Nikawa H, Hamada T, Yamamoto T. Denture plaque—Past and recent concerns. J. Dent. 1998, 26, 299–304. DOI:10.1016/S0300-5712(97)00026-2 [Google Scholar]
-
Coulthwaite L, Verran J. Potential pathogenic aspects of denture plaque. Br. J. Biomed. Sci. 2007, 64, 180–189. DOI:10.1080/09674845.2007.11732784 [Google Scholar]
-
Redfern J, Tosheva L, Malic S, Butcher M, Ramage G, Verran J. The denture microbiome in health and disease: An exploration of a unique community. Lett. Appl. Microbiol. 2022, 75, 195–209. DOI:10.1111/lam.13751 [Google Scholar]
-
Lim TW, Huang S, Jiang Y, Zhang Y, Burrow MF, McGrath C. Characterization of pathogenic microbiome on removable prostheses with different levels of cleanliness using 2bRAD-M metagenomic sequencing. J. Oral. Microbiol. 2024, 16, 2317059. DOI:10.1080/20002297.2024.2317059 [Google Scholar]
-
Escapa IF, Chen T, Huang Y, Gajare P, Dewhirst FE, Lemon KP. New insights into human nostril microbiome from the expanded Human Oral Microbiome Database (eHOMD): A Resource for the Microbiome of the Human Aerodigestive Tract. mSystems 2018, 3, e00187-18. DOI:10.1128/mSystems.00187-18 [Google Scholar]
-
Dyrhovden R, Eagan TM, Fløtten Ø, Siljan W, Leegaard TM, Bø B, et al. Pleural empyema caused by Streptococcus intermedius and Fusobacterium nucleatum: A distinct entity of pleural infections. Clin. Infect. Dis. 2023, 77, 1361–1371. DOI:10.1093/cid/ciad378 [Google Scholar]
-
Chopra A, Franco-Duarte R, Rajagopal A, Choowong P, Soares P, Rito T, et al. Exploring the presence of oral bacteria in non-oral sites of patients with cardiovascular diseases using whole-metagenomic data. Sci. Rep. 2024, 14, 1476. DOI:10.1038/s41598-023-50891-x [Google Scholar]
-
Bradić-Vasić M, Pejčić A, Kostić M, Obradović R, Minić I, Stanković I, et al. Influence of local denture-related factors on inflammatory marker levels in patients with denture stomatitis. BMC Oral. Health. 2025, 25, 744. DOI:10.1186/s12903-025-06160-6 [Google Scholar]
-
Zheng PC, Pan XQ, Zhou YJ, Lai KP, Li R, Zhang XX. Unraveling the impact of micro- and nano-sized polymethyl methacrylate on gut microbiota and liver lipid metabolism: Insights from oral exposure studies. Environ. Pollut. 2025, 373, 126157. DOI:10.1016/j.envpol.2025.126157 [Google Scholar]
-
Gendreau L, Loewy ZG. Epidemiology and etiology of denture stomatitis. J. Prosthodont. 2011, 20, 251–260. DOI:10.1111/j.1532-849X.2011.00698.x [Google Scholar]
-
Ikeya K, Iwasa F, Inoue Y, Fukunishi M, Takahashi N, Ishihara K, et al. Inhibition of denture plaque deposition on complete dentures by 2-methacryloyloxyethyl phosphorylcholine polymer coating: A clinical study. J. Prosthet. Dent. 2018, 119, 67–74. DOI:10.1016/j.prosdent.2017.02.012 [Google Scholar]
-
Yurdukoru B, Terzioğlu H, Yilmaz T. Assessment of whole saliva flow rate in denture-wearing patients. J. Oral. Rehabil. 2001, 28, 109–112. DOI:10.1046/j.1365-2842.2001.00624.x [Google Scholar]
-
Mary KM, Cherian B. Evaluation of oral stereognosis, masticatory efficiency, and salivary flow rate in complete denture wearers. J. Indian Prosthodont. Soc. 2020, 20, 290–296. DOI:10.4103/jips.jips_453_19 [Google Scholar]
-
Song EAC, Chung SH, Kim JH. Molecular mechanisms of saliva secretion and hyposecretion. Eur. J. Oral. Sci. 2024, 132, e12969. DOI:10.1111/eos.12969 [Google Scholar]
-
Dewhirst FE, Chen T, Izard J, Paster BJ, Tanner ACR, Yu WH, et al. The human oral microbiome. J. Bacteriol. 2010, 192, 5002–5017. DOI:10.1128/JB.00542-10 [Google Scholar]
-
Diaz PI, Xie Z, Sobue T, Thompson A, Biyikoglu B, Ricker A, et al. Synergistic Interaction between Candida albicans and Commensal Oral Streptococci in a Novel In Vitro Mucosal Model. Infect. Immun. 2012, 80, 620–632. DOI:10.1128/IAI.05896-11 [Google Scholar]
-
Sultan AS, Kong EF, Rizk AM, Jabra-Rizk MA, Sheppard DC. The oral microbiome: A lesson in coexistence. PLoS Pathog. 2018, 14, e1006719. DOI:10.1371/journal.ppat.1006719 [Google Scholar]
-
Campos MS, Marchini L, Bernardes LA, Paulino LC, Nobrega FG. Biofilm microbial communities of denture stomatitis. Oral. Microbiol. Immunol. 2008, 23, 419–424. DOI:10.1111/j.1399-302X.2008.00445.x [Google Scholar]
-
Shi B, Wu T, McLean J, Edlund A, Young Y, He X, et al. The denture-associated oral microbiome in health and stomatitis. mSphere 2016, 1, e00215-16. DOI:10.1128/mSphere.00215-16 [Google Scholar]
-
Morse DJ, Wilson MJ, Wei X, Lewis MAO, Bradshaw DJ, Murdoch C, et al. Denture-associated biofilm infection in 3D mucosal tissue models. J. Med. Microbiol. 2018, 67, 364–375. DOI:10.1099/jmm.0.000677 [Google Scholar]
-
Morse DJ, Smith A, Wilson MJ, Marsh L, White L, Posso R, et al. Molecular community profiling of the bacterial microbiota associated with denture-related stomatitis. Sci. Rep. 2019, 9, 10228. DOI:10.1038/s41598-019-46494-0 [Google Scholar]
-
O’Donnell LE, Robertson D, Nile CJ, Cross LJ, Riggio M, Sherriff A, et al. The oral microbiome of denture wearers is influenced by levels of natural dentition. PLoS ONE 2015, 10, e0137717. DOI:10.1371/journal.pone.0137717 [Google Scholar]
-
Fujinami W, Nishikawa K, Ozawa S, Hasegawa Y, Takebe J. Correlation between the relative abundance of oral bacteria and Candida albicans in denture and dental plaques. J. Oral. Biosci. 2021, 63, 175–183. DOI:10.1016/j.job.2021.02.003 [Google Scholar]
-
Theilade E, Budtz-Jørgensen E, Theilade J. Predominant cultivable microflora of plaque on removable dentures in patients with healthy oral mucosa. Arch. Oral. Biol. 1983, 28, 675–680. DOI:10.1016/0003-9969(83)90101-2 [Google Scholar]
-
Teles FR, Teles RP, Sachdeo A, Uzel NG, Song XQ, Torresyap G, et al. Comparison of microbial changes in early redeveloping biofilms on natural teeth and dentures. J. Periodontol. 2012, 83, 1139–1148. DOI:10.1902/jop.2012.110506 [Google Scholar]
-
Manzoor M, Pussinen PJ, Saarela RKT, Hiltunen K, Mäntylä P. Metagenomic analysis of the denture-associated oral microbiome in patients with denture stomatitis. Sci. Rep. 2025, 15, 31913. DOI:10.1038/s41598-025-16915-4 [Google Scholar]
-
Sun Z, Huang S, Zhu P, Tzehau L, Zhao H, Lv J, et al. Species-resolved sequencing of low-biomass or degraded microbiomes using 2bRAD-M. Genome Biol. 2022, 23, 36. DOI:10.1186/s13059-021-02576-9 [Google Scholar]
-
Gomes ACG, Maciel JG, Garcia AAMN, Coelho LAS, Rodrigues GM, Porto VC, et al. Anti-biofilm effectiveness of protocols for cleaning complete dentures in hospitalized patients: A randomized controlled trial. J. Appl. Oral. Sci. 2024, 32, e20230381. DOI:10.1590/1678-7757-2022-0381 [Google Scholar]
-
Teughels W, Van Assche N, Sliepen I, Quirynen M. Effect of material characteristics and/or surface topography on biofilm development. Clin. Oral. Implants Res. 2006, 17 (Suppl. S2), 68–81. DOI:10.1111/j.1600-0501.2006.01353.x [Google Scholar]
-
Khan SA, Mirani ZA, Khalid T, Khan EMWA, Choudhary Z, Kazmi SMR. Effect of polishing methods on Candida albicans adhesion and contributing factors in heat-cured acrylic dentures: An in-vitro comparative study. BMC Oral. Health. 2025, 25, 197. DOI:10.1186/s12903-025-05436-1 [Google Scholar]
-
Eren AM, Borisy GG, Huse SM, Mark Welch JL. Oligotyping analysis of the human oral microbiome. Proc. Natl. Acad. Sci. USA 2014, 111, E2875–E2884. DOI:10.1073/pnas.1409644111 [Google Scholar]
-
Arslan E, Akay C, Erdönmez D, Avukat EN. Evaluation of the effect of new-generation denture base materials aged in artificial saliva at different pH levels on surface roughness and Candida albicans adhesion. BMC Oral. Health. 2025, 25, 356. DOI:10.1186/s12903-025-05700-4 [Google Scholar]
-
Nobbs AH, Jenkinson HF, Jakubovics NS. Stick to your gums: Mechanisms of oral microbial adherence. J. Dent. Res. 2011, 90, 1271–1278. DOI:10.1177/0022034511399096 [Google Scholar]
-
Jehmlich N, Dinh KH, Gesell-Salazar M, Hammer E, Steil L, Dhople VM, et al. Quantitative analysis of the intra- and inter-subject variability of the whole salivary proteome. J. Periodontal Res. 2013, 48, 392–403. DOI:10.1111/jre.12025 [Google Scholar]
-
Lindh L, Aroonsang W, Sotres J, Arnebrant T. Salivary pellicles. Monogr. Oral. Sci. 2014, 24, 30–39. DOI:10.1159/000358782 [Google Scholar]
-
Ichigaya N, Kawanishi N, Adachi T, Sugimoto M, Kimoto K, Hoshi N. Effects of denture treatment on salivary metabolites: A pilot study. Int. J. Mol. Sci. 2023, 24, 13959. DOI:10.3390/ijms241813959 [Google Scholar]
-
Boyd H, Gonzalez-Martinez JF, Welbourn RJ, Gutfreund P, Klechikov A, Robertsson C, et al. A comparison between the structures of reconstituted salivary pellicles and oral mucin (MUC5B) films. J. Colloid. Interface Sci. 2021, 584, 660–668. DOI:10.1016/j.jcis.2020.10.124 [Google Scholar]
-
Ma G, Tang Y, Zeng Q, Zheng J. On adhesion mechanism of salivary pellicle–PDMS interface. Biosurf. Biotribol. 2019, 5, 93–96. DOI:10.1049/bsbt.2019.0014 [Google Scholar]
-
Welch JLM, Rossetti BJ, Rieken CW, Dewhirst FE, Borisy GG. Biogeography of a human oral microbiome at the micron scale. Proc. Natl. Acad. Sci. USA 2016, 113, E791–E800. DOI:10.1073/pnas.1522149113 [Google Scholar]
-
Katsikogianni M, Missirlis YF. Concise review of mechanisms of bacterial adhesion to biomaterials and of techniques used in estimating bacteria–material interactions. Eur. Cell Mater. 2004, 8, 37–57. DOI:10.22203/eCM.v008a05 [Google Scholar]
-
Arzani S, Khorasani E, Mokhlesi A, Azadian S, Ghodsi S, Mosaddad SA. Do 3D-printed and milled denture bases differ in microbial activity and adhesion? A systematic review and meta-analysis. Int. Dent. J. 2025, 75, 100857. DOI:10.1016/j.identj.2025.100857 [Google Scholar]
-
Singh B, Jain S, Bhasin N, Kaur J, Borse P, Longkumer P. Comparative evaluation of surface roughness, wettability, and hardness of conventional, heat-polymerized, CAD–milled, and 3D-printed PMMA denture base resins: An in vitro study. Cureus 2025, 17, e85008. DOI:10.7759/cureus.85008 [Google Scholar]
-
Benyounes O, Bekri S, Belgacem S, Labidi A, Khemis M, Mansour L. Oral colonization by different Candida species: First comparative study between denture and nondenture wearers in Tunisia. Eur. J. Dent. 2025, 19, 206–213. DOI:10.1055/s-0044-1787819 [Google Scholar]
-
Sterzenbach T, Helbig R, Hannig M. Bioadhesion in the oral cavity and approaches for biofilm management by surface modifications. Clin. Oral. Investig. 2020, 24, 4237–4260. DOI:10.1007/s00784-020-03646-1 [Google Scholar]
-
Pereira-Cenci T, Deng DM, Kraneveld EA, Manders EM, Cury AA, Ten Cate JM, et al. The effect of Streptococcus mutans and Candida glabrata on Candida albicans biofilms on different surfaces. Arch. Oral. Biol. 2008, 53, 755–764. DOI:10.1016/j.archoralbio.2008.02.015 [Google Scholar]
-
Bor B, Cen L, Agnello M, Shi W, He X. Morphological and physiological changes induced by contact-dependent interaction between Candida albicans and Fusobacterium nucleatum. Sci. Rep. 2016, 6, 27956. DOI:10.1038/srep27956 [Google Scholar]
-
Bertolini MM, Xu H, Sobue T, Nobile CJ, Del Bel Cury AA, Dongari-Bagtzoglou A. Candida–streptococcal mucosal biofilms display distinct structural and virulence characteristics depending on growth conditions and hyphal morphotypes. Mol. Oral. Microbiol. 2015, 30, 307–322. DOI:10.1111/omi.12095 [Google Scholar]
-
Sauer K, Stoodley P, Goeres DM, Hall-Stoodley L, Burmølle M, Stewart PS, et al. The biofilm life cycle: Expanding the conceptual model of biofilm formation. Nat. Rev. Microbiol. 2022, 20, 608–620. DOI:10.1038/s41579-022-00767-0 [Google Scholar]
-
Simon-Soro A, Ren Z, Krom BP, Hoogenkamp MA, Cabello-Yeves PJ, Daniel SG, et al. Polymicrobial aggregates in human saliva build the oral biofilm. mBio 2022, 13, e0013122. DOI:10.1128/mbio.00131-22 [Google Scholar]
-
Perić M, Miličić B, Kuzmanović Pfićer J, Živković R, Arsić Arsenijević V. A systematic review of denture stomatitis: Predisposing factors, clinical features, etiology, and global Candida spp. distribution. J. Fungi. 2024, 10, 328. DOI:10.3390/jof10050328 [Google Scholar]
-
Zhao A, Sun J, Liu Y. Understanding bacterial biofilms: From definition to treatment strategies. Front. Cell Infect. Microbiol. 2023, 13, 1137947. DOI:10.3389/fcimb.2023.1137947 [Google Scholar]
-
Kolpen M, Kragh KN, Enciso JB, Faurholt-Jepsen D, Lindegaard B, Egelund GB, et al. Bacterial biofilms predominate in both acute and chronic human lung infections. Thorax 2022, 77, 1015–1022. DOI:10.1136/thoraxjnl-2021-217576 [Google Scholar]
-
Chatzigiannidou I, Teughels W, Van de Wiele T, Boon N. Oral biofilms exposure to chlorhexidine results in altered microbial composition and metabolic profile. NPJ Biofilms Microbiomes 2020, 6, 13. DOI:10.1038/s41522-020-0124-3 [Google Scholar]
-
Cheong JZA, Johnson CJ, Wan H, Liu A, Kernien JF, Gibson ALF, et al. Priority effects dictate community structure and alter virulence of fungal-bacterial biofilms. ISME J. 2021, 15, 2012–2027. DOI:10.1038/s41396-021-00901-5 [Google Scholar]
-
Nett J, Lincoln L, Marchillo K, Massey R, Holoyda K, Hoff B, et al. Putative role of β-1,3-glucans in Candida albicans biofilm resistance. Antimicrob. Agents Chemother. 2007, 51, 510–520. DOI:10.1128/AAC.01056-06 [Google Scholar]
-
Latib YO, Owen CP, Patel M. Viability of Candida albicans in Denture Base Resin After Disinfection: A Preliminary Study. Int J Prosthodont. 2018, 31, 436–439. DOI:10.11607/ijp.5653 [Google Scholar]
-
Neppelenbroek KH, Falcão Procópio AL, Gurgel Gomes AC, Campos Sugio CY, Maia Neves Garcia AA, Porto VC, et al. A modified Newton classification for denture stomatitis. Prim Dent J. 2022, 11, 55–58. DOI:10.1177/20501684221101095 [Google Scholar]
-
Kydd WL, Daly CH. The biologic and mechanical effects of stress on oral mucosa. J. Prosthet. Dent. 1982, 47, 317–329. DOI:10.1016/0022-3913(82)90162-7 [Google Scholar]
-
Altarawneh S, Bencharit S, Mendoza L, Curran A, Barrow D, Barros S, et al. Clinical and histological findings of denture stomatitis as related to intraoral colonization patterns of Candida albicans, salivary flow, and dry mouth. J. Prosthodont. 2013, 22, 13–22. DOI:10.1111/j.1532-849X.2012.00906.x [Google Scholar]
-
Kamio ABS, da Silva Barboza A, da Silva MEB, Soto AF, de Andrade JSR, Duque TM, et al. Disinfection strategies for poly(methyl methacrylate): Method sequence, solution concentration, and intraoral temperature on antimicrobial activity. Polymers 2024, 17, 8. DOI:10.3390/polym17010008 [Google Scholar]
-
Koehler J, Ramakrishnan AN, Ludtka C, Hey J, Kiesow A, Schwan S. The influence of oral cavity physiological parameters: Temperature, pH, and swelling on the performance of denture adhesives—in vitro study. BMC Oral Health 2024, 24, 206. DOI:10.1186/s12903-024-03967-7 [Google Scholar]
-
Choi JJE, Zwirner J, Ramani RS, Ma S, Hussaini HM, Waddell JN, et al. Mechanical properties of human oral mucosa tissues are site dependent: A combined biomechanical, histological and ultrastructural approach. Clin. Exp. Dent. Res. 2020, 6, 602–611. DOI:10.1002/cre2.305 [Google Scholar]
-
Yan J, Fei C, Mao S, Moreau A, Wingreen NS, Košmrlj A, et al. Mechanical instability and interfacial energy drive biofilm morphogenesis. Elife 2019, 8, e43920. DOI:10.7554/eLife.43920 [Google Scholar]
-
Dorey JL, Blasberg B, MacEntee MI, Conklin RJ. Oral mucosal disorders in denture wearers. J. Prosthet. Dent. 1985, 53, 210–213. DOI:10.1016/0022-3913(85)90111-8 [Google Scholar]
-
Emami E, De Grandmont P, Rompré PH, Barbeau J, Pan S, Feine JS. Favoring trauma as an etiological factor in denture stomatitis. J. Dent. Res. 2008, 87, 440–444. DOI:10.1177/154405910808700505 [Google Scholar]
-
Jainkittivong A, Aneksuk V, Langlais RP. Oral mucosal lesions in denture wearers. Gerodontology 2010, 27, 26–32. DOI:10.1111/j.1741-2358.2009.00289.x [Google Scholar]
-
Ince D, Lucas TM, Malaker SA. Current strategies for characterization of mucin-domain glycoproteins. Curr. Opin. Chem. Biol. 2022, 69, 102174. DOI:10.1016/j.cbpa.2022.102174 [Google Scholar]
-
Darvell BW, Clark RK. The physical mechanisms of complete denture retention. Br. Dent. J. 2000, 189, 248–252. DOI:10.1038/sj.bdj.4800734 [Google Scholar]
-
Konstantinova V, Ibrahim M, Lie SA, Birkeland ES, Neppelberg E, Marthinussen MC, et al. Nano-TiO2 penetration of oral mucosa: In vitro analysis using 3D organotypic human buccal mucosa models. J. Oral. Pathol. Med. 2017, 46, 214–222. DOI:10.1111/jop.12469 [Google Scholar]
-
Kang SH, Lee HJ, Hong SH, Kim KH, Kwon TY. Influence of surface characteristics on the adhesion of Candida albicans to various denture lining materials. Acta Odontol. Scand. 2013, 71, 241–248. DOI:10.3109/00016357.2012.671360 [Google Scholar]
-
Chladek G, Zmudzki J, Kasperski J. Long-term soft denture lining materials. Materials 2014, 7, 5816–5842. DOI:10.3390/ma7085816 [Google Scholar]
-
Sarkar A, Andablo-Reyes E, Bryant M, Dowson D, Neville A. Lubrication of soft oral surfaces. Curr. Opin. Colloid. Interface Sci. 2019, 39, 61–75. DOI:10.1016/j.cocis.2019.01.008 [Google Scholar]
-
Ranc H, Elkhyat A, Servais C, Mac-Mary S, Launay B, Humbert P. Friction coefficient and wettability of oral mucosal tissue: Changes induced by a salivary layer. Colloids Surf. A Physicochem. Eng. Asp. 2006, 276, 155–161. DOI:10.1016/j.colsurfa.2005.10.033 [Google Scholar]
-
Peterson BW, He Y, Ren Y, Zerdoum A, Libera MR, Sharma PK, et al. Viscoelasticity of biofilms and their recalcitrance to mechanical and chemical challenges. FEMS Microbiol. Rev. 2015, 39, 234–245. DOI:10.1093/femsre/fuu008 [Google Scholar]
-
Klapper I, Rupp CJ, Cargo R, Purvedorj B, Stoodley P. Viscoelastic fluid description of bacterial biofilm material properties. Biotechnol. Bioeng. 2002, 80, 289–296. DOI:10.1002/bit.10376 [Google Scholar]
-
Di Stefano A, D’Aurizio E, Trubiani O, Grande R, Di Campli E, Di Giulio M, et al. Viscoelastic properties of Staphylococcus aureus and Staphylococcus epidermidis mono-microbial biofilms. Microb. Biotechnol. 2009, 2, 634–641. DOI:10.1111/j.1751-7915.2009.00120.x [Google Scholar]
-
Vincent JF. Structural Biomaterials; Princeton University Press: Princeton, NJ, USA, 2012. [Google Scholar]
-
Körstgens V, Flemming HC, Wingender J, Borchard W. Uniaxial compression measurement device for investigation of the mechanical stability of biofilms. J. Microbiol. Methods 2001, 46, 9–17. DOI:10.1016/S0167-7012(01)00248-2 [Google Scholar]
-
Mah TF, O’Toole GA. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 2001, 9, 34–39. DOI:10.1016/S0966-842X(00)01913-2 [Google Scholar]
-
Flemming HC, Wingender J, Szewzyk U, Steinberg P, Rice SA, Kjelleberg S. Biofilms: An emergent form of bacterial life. Nat. Rev. Microbiol. 2016, 14, 563–575. DOI:10.1038/nrmicro.2016.94 [Google Scholar]
-
Even C, Marliere C, Ghigo JM, Allain JM, Marcellan A, Raspaud E. Recent advances in studying single bacteria and biofilm mechanics. Adv. Colloid. Interface Sci. 2017, 247, 573–588. DOI:10.1016/j.cis.2017.07.026 [Google Scholar]
-
Morse DJ, Wilson MJ, Wei X, Bradshaw DJ, Lewis MAO, Williams DW. Modulation of Candida albicans virulence in in-vitro biofilms by oral bacteria. Lett. Appl. Microbiol. 2019, 68, 337–343. DOI:10.1111/lam.13145 [Google Scholar]
-
Cao H, Habimana O, Safari A, Heffernan R, Dai Y, Casey E. Revealing region-specific biofilm viscoelastic properties via micro-rheology. NPJ Biofilms Microbiomes 2016, 2, 5. DOI:10.1038/s41522-016-0005-y [Google Scholar]
-
Palmer SR, Ren Z, Hwang G, Liu Y, Combs A, Soderstrom B, et al. Streptococcus mutans yidC1 and yidC2 impact cell-envelope biogenesis, biofilm matrix, and biophysical properties. J. Bacteriol. 2018, 200, e00396-18. DOI:10.1128/jb.00396-18 [Google Scholar]
-
Hou J, Veeregowda DH, van de Belt-Gritter B, Busscher HJ, van der Mei HC. Extracellular polymeric matrix production and relaxation under shear and mechanical pressure in Staphylococcus aureus biofilms. Appl. Environ. Microbiol. 2018, 84, e01516-17. DOI:10.1128/AEM.01516-17 [Google Scholar]
-
Cense AW, Peeters EA, Gottenbos B, Baaijens FP, Nuijs AM, van Dongen ME. Mechanical properties and failure of Streptococcus mutans biofilms studied using micro-indentation. J. Microbiol. Methods. 2006, 67, 463–472. DOI:10.1016/j.mimet.2006.04.023 [Google Scholar]
-
Charras G, Yap AS. Tensile forces and mechanotransduction at cell–cell junctions. Curr. Biol. 2018, 28, R445–R457. DOI:10.1016/j.cub.2018.02.003 [Google Scholar]
-
Cont A, Rossy T, Al-Mayyah Z, Persat A. Biofilms deform soft surfaces and disrupt epithelia. Elife 2020, 9, e56533. DOI:10.7554/eLife.56533 [Google Scholar]
-
Beroz F, Yan J, Sabass B, Stone HA, Bassler BL, Wingreen NS, et al. Verticalization of bacterial biofilms. Nat. Phys. 2018, 14, 954–960. DOI:10.1038/s41567-018-0170-4 [Google Scholar]
-
Gloag ES, Fabbri S, Wozniak DJ, Stoodley P. Biofilm mechanics: Implications in infection and survival. Biofilm 2020, 2, 100017. DOI:10.1016/j.bioflm.2019.100017 [Google Scholar]
-
Ibáñez-Cortés M, Martín-Piedra MÁ, Blanco-Elices C, García-García ÓD, España-López A, Fernández-Valadés R, et al. Histological characterization of human masticatory oral mucosa. Microsc. Res. Tech. 2023, 86, 1712–1724. DOI:10.1002/jemt.24398 [Google Scholar]
-
Pestrak MJ, Chaney SB, Eggleston HC, Dellos-Nolan S, Dixit S, Mathew-Steiner SS, et al. Pseudomonas aeruginosa rugose small-colony variants evade host clearance and persist in multiple host environments. PLoS Pathog. 2018, 14, e1006842. DOI:10.1371/journal.ppat.1006842 [Google Scholar]
-
Yi X, Liang X, Li Z, Gong T, Ren B, Li Y, et al. Omics for deciphering oral microecology. Int. J. Oral. Sci. 2024, 16, 2. DOI:10.1038/s41368-023-00264-x [Google Scholar]
