In Vitro Bulb‑Scale Propagation of Lilium leichtlinii var. maximowiczii for Urban Landscaping
Received: 09 January 2026 Revised: 02 February 2026 Accepted: 19 March 2026 Published: 25 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Rapid urbanization in Seoul and the surrounding metropolitan area has made establishing urban green networks a key strategy for mitigating environmental degradation and the impacts of climate change [1,2,3]. In recent years, roadside flowerbeds, median strips, neighborhood green spaces, and citizen-led flower road projects have been actively expanded in Seoul [4,5]. Among these, roadside flowerbeds, which citizens encounter most frequently in their daily lives, are a core element determining the quality of urban landscapes [3,6,7]. As a vital component of green infrastructure, roadside flowerbeds serve aesthetic purposes for urban beautification and perform various ecological functions, including reducing fine dust, alleviating the urban heat island effect, and enhancing pedestrian safety [3,6,8]. However, current roadside flowerbeds in Seoul rely heavily on annual bedding plants that are replaced every season. To compensate for this, bulbous plants such as tulips and daffodils have been introduced. Still, these bulbs are largely imported, resulting in high costs and poor persistence under local environmental conditions and, consequently, requiring substantial annual budgets [5,9].
Lilium leichtlinii var. maximowiczii is a promising native bulbous geophyte that is well-adapted to the climate and soils of the Seoul region and blooms in summer, thereby complementing seasonal landscapes [10,11,12]. As a perennial bulbous plant belonging to the Liliaceae family, it is distributed throughout the Korean Peninsula, Japan, northeastern China, and the Ussuri region of Russia [11,12,13]. It develops numerous roots from an ovoid bulb (3–4 cm in diameter), with a stem that grows upright up to 1 m and branches at the top. It bears 2–10 nodding, yellowish-red flowers with strongly recurved tepals densely spotted with dark purple from July to August, providing high ornamental value for use in urban flowerbeds and roadside plantings. In Korea, this species predominantly inhabits well-drained grassy or rocky slopes and forest margins, where it grows under cool, moist conditions with full sun to light shade during the growing season. However, despite its high ornamental potential and good adaptation to local conditions, the use of L. leichtlinii var. maximowiczii in urban flowerbeds has remained limited because its bulbs must be propagated individually, making large-scale distribution difficult. The morphological characteristics and geographical distribution of L. leichtlinii var. maximowiczii in Korea are shown in Figure 1. In addition to its ornamental value, this taxon has recently been recognized as a high-value edible and medicinal lily, and colchicine-induced mutants of L. leichtlinii var. maximowiczii have been developed to improve agronomic traits [14].
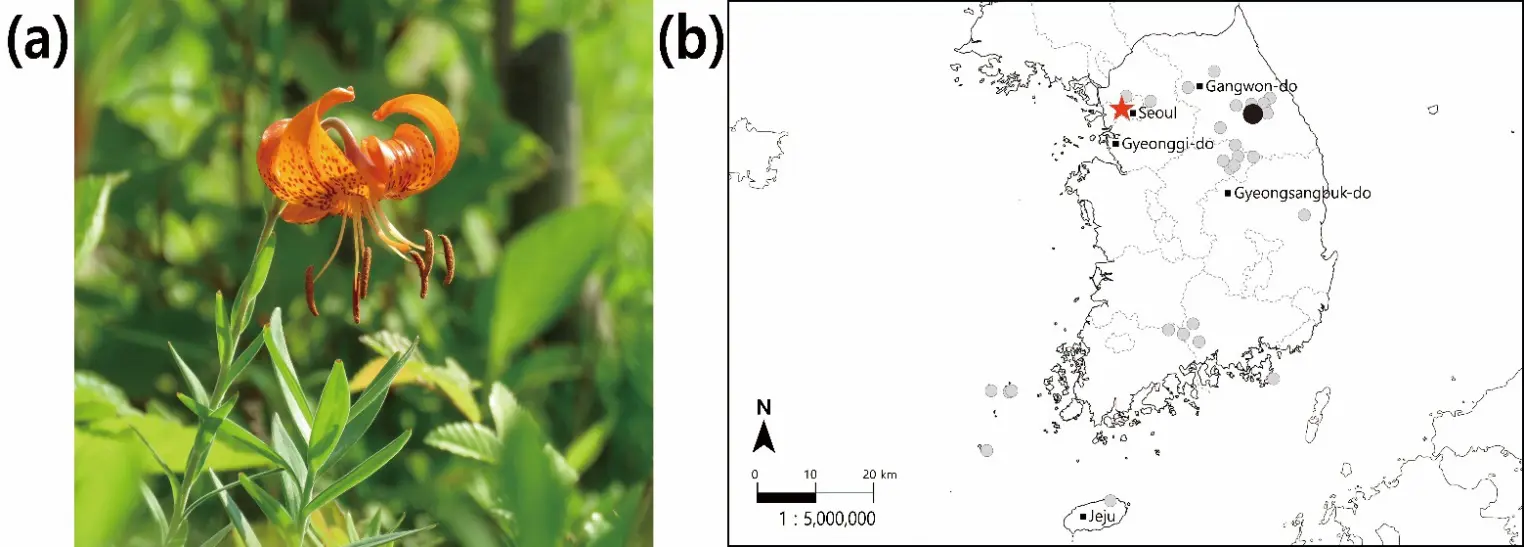
Figure 1. Morphological characteristics and geographical distribution of L. leichtlinii var. maximowiczii. (a) Flowering morphology collected and managed at Seoul Botanic Park; (b) Map indicating the natural distribution range within South Korea (●: original collection site in Gangwon-do; ★: ex situ conservation site at Seoul Botanic Park; ●: confirmed natural occurrence). Base map © National Geographic Information Institute (NGII), Korea.
For large-scale use of L. leichtlinii var. maximowiczii in roadside flowerbeds and other urban green spaces, it is essential to ensure uniformity of planting material and establish an economically viable mass-production system [2,15,16,17]. Low multiplication rates and slow growth limit natural bulb propagation. Seed propagation is unsuitable for landscaping sites because of low germination rates and considerable genetic variability among individuals, which results in differences in plant height and flowering time in the propagated material [10,12,18,19]. In vitro scale culture technology is an economical way to rapidly increase bulblet production from multiple scales of a single bulb via various organogenic or embryogenic pathways [19,20,21,22,23,24]. This method has the advantage of providing pathogen-free, high-quality, genetically uniform plantlets through clonal propagation [13,25].
In the micropropagation of Korean native lilies, regeneration pathways vary significantly depending on the species. For instance, Lilium hansonii is often propagated through indirect organogenesis using callus-inducing media [26,27]. However, L. leichtlinii var. maximowiczii exhibits a strong tendency for direct bulblet formation from bulb scales rather than callus differentiation [10,15,28]. This developmental characteristic necessitates a specialized approach to optimize combinations of plant growth regulators (PGRs), as the efficiency of direct bulblet induction and subsequent growth is highly sensitive to the specific balance of cytokinins and auxins. Therefore, establishing a tailored protocol that deviates from conventional callus-based methods is essential for the stable mass production of high-quality L. leichtlinii var. maximowiczii plantlets [19.26].
The efficiency of bulblet regeneration and proliferation in scale culture is highly dependent on the type and concentration ratio of PGRs added to the medium [15,26,29,30,31]. In tissue culture of Lilium species, the interaction between cytokinins, which promote cell division and adventitious bud formation, and auxins, which induce root formation and bulblet enlargement, plays a crucial role [1,26]. Previous studies have reported that benzyladenine (BAP) increases the number of bulblet-forming explants and that its combinations with auxins, such as naphthaleneacetic acid (NAA) or indole-3-acetic acid (IAA), can enhance the bulblet formation efficiency [16,23,29,30]. With increasing urbanization, establishing urban green networks is crucial for mitigating environmental degradation and climate change. Urban greening provides essential ecosystem services, including the mitigation of the heat island effect and the improvement of air quality [1,6,8]. However, plant species currently used for roadside greening are often limited to exotic species, necessitating the introduction of native plants adapted to local ecosystems [9,32].
2. Materials and Methods
2.1. Plant Materials
Bulb‑scales of L. leichtlinii var. maximowiczii used in this study were obtained in 2023 from plants cultivated at Seoul Botanic Park (37.78625° N, 127.7424472° E). The plant material originated from wild individuals collected in September 2014 from Gangwon-do, Korea (37°35′34.1″ N, 128°28′22.8″ E). Subsequently, it propagated at the Shingu University Botanical Garden. We acquired these plants in November 2020 and have since maintained it ex situ for conservation and propagation (manager: Hye-min Park, accession No. 10001913). This study was conducted from June 2023 to December 2024 using these ex situ conserved plants as the source of bulb materials. For experiments, healthy, vigorously growing bulbs free of visible pests and diseases were selected and used for explant preparation.
2.2. Sterilization of Scale Explants
Dry outer scales of the bulbs were removed, and the whole bulbs were rinsed under running tap water for 10 min. Individual scales were then separated and cut into segments of approximately 1.0–1.5 cm in both length and width. To determine the optimal surface sterilization method, three sterilization protocols were tested: (I) Immersion in 70% (v/v) EtOH for 30 seconds (s); followed by 2% (v/v) sodium hypochlorite (NaOCl) for 20 min; (II) Immersion in a 0.1% (w/v) benomyl solution for 1 h; (III) Immersion in 0.1% (w/v) benomyl solution for 1 h, followed by 70% (v/v) EtOH for 30 s, then sequential surface sterilization in 1% (v/v) NaOCl for 20 min and 2% (v/v) NaOCl for 10 min (Figure 2).
After sterilization, the segments were cleaned three times with sterile distilled water in a laminar airflow cabinet and blotted dry on sterile filter paper. To evaluate disinfection efficiency, the segments were cultured on Murashige and Skoog (MS) basal medium [23] supplemented with 87.6 mM sucrose (pH 5.7). Ten explants were placed in each 90 mm Petri dish, and three dishes (30 explants) were used per treatment, with each dish considered as one independent replicate (n = 3). Contamination rate and survival rate were assessed 1 week after inoculation.

Figure 2. Stepwise procedure for surface sterilization and in vitro inoculation of L. leichtlinii var. maximowiczii scale explants. (a) Mother bulbs; (b) Pretreatment with benomyl solution; (c) Sequential sterilization with NaOCl; (d) Inoculation onto the culture medium.
2.3. Bulblet Induction
Bulb-scale segments sterilized according to the optimal method identified in Section 2.2 were used for bulblet induction. Explants were placed on 90-mm Petri dishes at a density of two explants per dish, with nine dishes (18 explants) per treatment. Two groups of PGRs combinations were tested for bulblet induction, which were designed based on previous reports in Lilium species [15,26,29]: (I) BAP + IAA: 2.2 or 4.4 µM BAP combined with 1.7 or 5.7 µM IAA; (II) BAP + NAA: 4.4 or 8.9 µM BAP combined with 1.6 or 2.7 µM NAA. The MS basal medium without plant growth regulators was used as the control treatment.
All media were supplemented with 87.6 mM sucrose, 0.8% (w/v) agar, and the pH was adjusted to 5.7 before autoclaving. Unless otherwise specified, all cultures described in Sections 2.2–2.4 were maintained in a culture room at 25 ± 1 °C, 60 ± 5% relative humidity, and a photosynthetic photon flux density of 40–60 μmol·m−2·s−1 under a 16 h light/8 h dark photoperiod provided by daylight-white T5 LED lamps (15 W, 900 mm, Dooyoung Lighting Co., Ltd., Siheung-si, Gyeonggi-do, Korea) [16,33]. After 3 weeks of culture, the number of bulblets per explant was recorded.
2.4. Proliferation and Acclimatization
For further bulblet enlargement and proliferation, induced bulblets were subcultured into culture tubes (30 × 150 mm) medium containing double-strength MS mineral salts and 175.2 mM sucrose (pH 5.7) and maintained for an additional 7 weeks [19,22,30].
Plantlets with well-developed shoots and roots were removed from the culture medium, and residual agar was gently washed off with distilled water. The plantlets were then transplanted into cutting boxes filled with a 1:1:1 (v/v/v) mixture of peat moss, perlite, and commercial potting soil [19,30].
During the initial acclimatization period, the trays were covered with transparent lids and intermittently misted to maintain high humidity. After 2–3 days, the lids were gradually opened to lower humidity and increase light penetration. Plantlets were acclimatized in a greenhouse at 20–25 °C for 1 week and then slowly exposed to ambient conditions [18,30].
2.5. Statistical Analysis
Contamination rate (%) was calculated as (number of contaminated explants/total explants) × 100. Survival rate (%) was calculated as (number of survived explants/total explants) × 100.
All experiments were conducted using a completely randomized design (CRD) with three replicates (n = 3). For bulblet number and bulblet induction rate, statistical analysis was performed on the mean values, and data are presented as mean ± standard error (SE). Data were analyzed using SAS 9.1 (SAS Institute Inc., Cary, NC, USA), and differences among treatment means were determined using Duncan’s Multiple Range Test (DMRT) at a 5% significance level (p < 0.05). Means within a column followed by the same letter are not significantly different according to DMRT.
3. Results
3.1. Sterilization of Scale Explants
The effectiveness of surface sterilization treatments for in vitro establishment of L. leichtlinii var. maximowiczii scale explants differed markedly among the methods tested. The treatment combining 70% (v/v) EtOH and 1% (v/v) NaOCl, which is commonly used in lily tissue culture, resulted in a survival rate of only 33.3%, while explants treated with 0.1% (w/v) benomyl solution alone for 1 h showed the lowest survival rate of 6.7%. In the benomyl-only treatment, the contamination rate reached 93.3%. In comparison, the combined EtOH and NaOCl treatment also showed a high contamination rate of 60.0%, indicating that both conventional treatments were unsuitable for scale sterilization in this species.
In contrast, the combined protocol consisting of 0.1% (w/v) benomyl solution pretreatment followed by stepwise sterilization with 1% and 2% (v/v) NaOCl achieved the highest survival rate of 63.3% with the lowest contamination, and statistical analysis confirmed that this value was significantly higher than those of the other treatments (DMRT, p < 0.05), suggesting that this protocol is the optimal surface sterilization method for effectively reducing contamination while maintaining high explant survival in L. leichtlinii var. maximowiczii (Table 1).
Table 1. Effect of different sterilization protocols on the survival and contamination rates of L. leichtlinii var. maximowiczii scale explants after 1 week of culture.
|
Treatment |
Total Explants (No.) |
Survived Explants (No.) |
Survival Rate (%) |
Contaminated Explants (No.) |
Contamination Rate (%) |
|---|---|---|---|---|---|
|
70% (v/v) EtOH + 2%(v/v) NaOCl |
30 |
10 |
33.3 ± 3.33 b |
18 |
60.0 |
|
0.1% (w/v) Benomyl solution (1 h) |
30 |
2 |
6.7 ± 3.33 c |
28 |
93.3 |
|
0.1% (w/v) Benomyl solution + 1% & 2% (v/v) NaOCl |
30 |
19 |
63.3 ± 3.33 a |
10 |
33.3 |
Values are presented as mean ± SE (n = 3). Means within a column followed by the same letter are not significantly different according to Duncan’s Multiple Range Test (DMRT) at p < 0.05.
3.2. Bulblet Induction
Scale explants of L. leichtlinii var. maximowiczii were cultured under various combinations of PGRs to evaluate bulblet induction and proliferation. Significant differences were observed among treatments (Figure 3). Specifically, the 2.2 µM BAP + 5.7 µM IAA treatment produced the highest number of bulblets (2.5 ± 0.12 per survived explant, total 38), followed by 4.4 µM BAP + 2.7 µM NAA (2.0 ± 0.00 per survived explant, total 24). The mean number of bulblets per explant was calculated as the total number of bulblets divided by the number of surviving explants in each treatment (Table 2).
Table 2. Effect of different plant growth regulators (PGRs) combinations on bulblet induction from bulb-scale explants of L. leichtlinii var. maximowiczii after 3 weeks of culture.
|
No. |
Treatment (µM) |
Survived Explants (No.) |
Total Bulblets (No.) |
Bulblets per Explant (Mean ± SE) |
|---|---|---|---|---|
|
1 |
Control |
10 |
8 |
0.8 ± 0.17 e |
|
2 |
2.2 BAP + 2.8 IAA |
15 |
19 |
1.2 ± 0.10 c,d |
|
3 |
2.2 BAP + 5.7 IAA |
15 |
38 |
2.5 ± 0.12 a |
|
4 |
4.4 BAP + 2.8 IAA |
12 |
16 |
1.3 ± 0.04 c |
|
5 |
4.4 BAP + 5.7 IAA |
15 |
17 |
1.1 ± 0.11 d |
|
6 |
4.4 BAP + 1.6 NAA |
9 |
9 |
1.0 ± 0.00 d,e |
|
7 |
4.4 BAP + 2.7 NAA |
12 |
24 |
2.0 ± 0.00 b |
|
8 |
8.9 BAP +1.6 NAA |
12 |
12 |
1.0 ± 0.14 d,e |
Each treatment used 18 explants (6 explants × 3 replicates). All the surviving explants produced bulblets. The mean number of bulblets per explant was calculated by dividing the total number of bulblets by the number of surviving explants. Data are presented as mean ± SE (n = 3). Means within a column followed by the same letter are not significantly different according to Duncan's multiple range test (DMRT) at p < 0.05.
3.3. Proliferation and Acclimatization
After 7 weeks of subculturing the differentiated bulblets on MS medium, the plantlets developed into healthy individuals with shoot elongation, dark green foliage, and vigorous white roots (Figure 4). While the 2.2 µM BAP + 5.7 µM IAA combination was most effective in terms of the number of induced bulblets, plantlets obtained from BAP + NAA media were used for subsequent proliferation and acclimatization (Figure 4b; Table 2, treatments 6–8).
4. Discussion
4.1. Sterilization of Scale Explants
The conventional sterilization treatments reduced contamination to some extent but resulted in low survival of the scale explants, most likely because high NaOCl concentrations or benomyl applied alone imposed excessive chemical stress on the fleshy scale tissues, leading to tissue damage. As a result, the protocol combining benomyl solution pretreatment with stepwise NaOCl sterilization can be interpreted as a balanced sterilization strategy that suppresses contamination while maintaining tissue viability [23,34,35].
Similar multi-step sterilization approaches have been reported as effective for other lily species and bulbous plants, where excessive chemical stress can easily damage fleshy scale tissues [18,28]. In this context, our findings that a benomyl solution pretreatment combined with stepwise NaOCl sterilization improves explant survival while controlling contamination are in good agreement with those previous reports. Therefore, the present protocol can be considered a practical and reproducible method for in vitro introduction of L. leichtlinii var. maximowiczii, and it may also be adaptable to related Korean native lilies requiring careful control of both microbial load and explant injury.
4.2. Bulblet Induction
The results of the present study highlight the importance of the balance between cytokinins and auxins for efficient bulblet induction in L. leichtlinii var. maximowiczii. Among the treatments tested, 2.2 µM BAP + 5.7 µM IAA produced the highest bulblet induction (2.5 ± 0.12 bulblets per survived explant; Table 2), suggesting that moderate cytokinin levels combined with an appropriate auxin concentration favor direct organogenesis in this species. This pattern agrees with previous reports that BAP in combination with auxins promotes bulblet initiation in Lilium species. The enhanced shoot initiation observed under cytokinin-rich conditions in the present study is in agreement with findings reported for L. longiflorum and oriental hybrids [7,30,36].
4.3. Proliferation and Acclimatization
The combination of 4.4 µM BAP + 2.7 µM NAA was applied during the development of induced bulblets into plantlets (total 24; Table 2). This response is consistent with previous reports indicating that an appropriate cytokinin–auxin balance plays a crucial role in morphogenesis and bulb development in Lilium species [15,26,29].
Although BAP + IAA combinations, particularly 2.2 µM BAP + 5.7 µM IAA, produced the highest number of bulblets, plantlet development and subsequent acclimatization were observed under the 4.4 µM BAP + 2.7 µM NAA condition. Similar use of different cytokinin–auxin combinations during micropropagation has been reported in other Lilium species [10,11,25,28,37]. These findings suggested that the hormonal requirements for bulblet induction and later growth stages may differ in L. leichtlinii var. maximowiczii.
Regenerated plantlets were transferred to soil and maintained under nursery conditions, demonstrating their potential for further cultivation. The use of different PGR combinations during micropropagation has been widely reported in Lilium and other bulbous ornamental species [16,18,24,30].
4.4. Future Perspectives
Future research should prioritize optimizing cultivation management to promote bulblet enlargement, an essential step for successful flowering in L. leichtlinii var. maximowiczii [10,17,18]. Furthermore, it is critical to conduct field performance trials in practical settings, such as roadsides and urban parks, to evaluate the floral characteristics, viability, and long-term persistence of micropropagated plantlets [5,9,15]. Such studies will provide a more comprehensive assessment of their ecological resilience and functional value within sustainable landscape architecture [1,6,17,32]. In addition, detailed quantitative evaluations of plant growth, bulb enlargement, and flowering performance will be carried out in future field and greenhouse studies [14,25].
This in vitro bulb‑scale propagation system can also support future breeding programs and cultivar development by providing large numbers of genetically uniform bulbs as standardized material for selection and evaluation of native Lilium adapted to urban landscapes.
5. Conclusions
The present study established an in vitro propagation system for the Korean native bulbous species L. leichtlinii var. maximowiczii using bulb‑scale explants. An optimized surface-sterilization protocol with 0.1% (w/v) benomyl solution pretreatment followed by stepwise NaOCl treatment reduced contamination.
Different PGR requirements were observed for bulblet induction and subsequent growth stages. For bulblet induction, 2.2 µM BAP + 5.7 µM IAA produced the highest number of bulblets (2.5 ± 0.12 per surviving explant). Induced bulblets were subsequently developed into plantlets under the 4.4 µM BAP + 2.7 µM NAA condition.
The protocol developed in this study provides a practical framework for propagating uniform planting materials and may support the potential use of L. leichtlinii var. maximowiczii in urban greening and roadside planting.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this work, the authors used Grammarly and Perplexity (AI-assisted tools) to improve readability and language. After using these tools, the authors reviewed and edited the content as needed and took full responsibility for the publication's content.
Acknowledgments
The authors thank Wan‑Hee Lee, Head of the Plant Management & Research Division, and Su‑Mi Park, Director of Seoul Botanic Park, for administrative support and facilitation of this project. The authors also thank the staff of Seoul Botanic Park for their assistance with field sampling, cultivation and in vitro propagation of Lilium accessions.
Author Contributions
I.‑J.C.: Conceptualization, data curation, formal analysis, supervision, writing—original draft, writing—review & editing. H.‑M.P.: data curation, monitoring, ex situ conservation of Lilium, and management of the Lilium culture green house at Seoul Botanic Park. M.‑J.P.: investigation, tissue culture experiments, investigation and ongoing bulb‑scale culture and maintenance of Lilium accessions.
Ethics Statement
Not applicable. This study did not involve humans or animals. The plant materials used in this study are not listed in the IUCN Red List, and no endangered or protected species were harmed during sample collection. All plant sampling and subsequent experiments were conducted in accordance with the guidelines and regulations of Seoul Botanic Park.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data supporting the findings of this study are included in this article.
Funding
This research was funded by the Seoul Metropolitan Government.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
References
- Fadillah AR, Novianti V, Hapsari L. Analysis of the eco-physiological characteristics of street trees in urban area for sustainable urban greening. Proc. Natl. Inst. Ecol. Repub. Korea. 2025, 6, 15–27. DOI:10.22920/PNIE.2025.6.1.15 [Google Scholar]
- Liu L, Ignatieva M, Kilbane S. Urban green corridors: Plant biodiversity and pathways for nature-based solutions in Perth and Beijing. Proc. 8th Fábos Conf. Landsc. Greenw. Plan. 2023, 8, 1–10. DOI:10.7275/fabos.2404 [Google Scholar]
- Rai PK. Biodiversity of roadside plants and their response to air pollution in an Indo-Burma hotspot region: Implications for urban ecosystem restoration. J. Asia-Pac. Biodivers. 2016, 9, 47–55. DOI:10.1016/j.japb.2015.10.011 [Google Scholar]
- Han BH, Kwak JI, Park SC, Hur JY. A study on planning of roadside green for enhancing urban green network. Korean J. Environ. Ecol. 2014, 28, 128–141. DOI:10.13047/KJEE.2014.28.2.128 [Google Scholar]
- Langens-Gerrits MM, Miller WB, Croes AF, De Klerk GJ. Effect of low temperature on dormancy breaking and growth after planting in lily bulblets regenerated in vitro. Plant Growth Regul. 2003, 40, 267–275. DOI:10.1023/A:1025018728178 [Google Scholar]
- Benvenuti S. Wildflower green roofs for urban landscaping, ecological sustainability and biodiversity. Landsc. Urban Plan. 2014, 124, 151–161. DOI:10.1016/j.landurbplan.2014.01.004 [Google Scholar]
- Tunley Environmental. Native Plants in Enhancing Biodiversity. Insight Article. 2024. Available online: https://www.tunley-environmental.com/ (accessed on 18 December 2025).
- Francini A, Romano D, Toscano S, Ferrante A. The contribution of ornamental plants to urban ecosystem services. Earth 2022, 3, 1258–1274. DOI:10.3390/earth3040071 [Google Scholar]
- Chu Y, Jin SN, Son D, Park S, Cho H, Lee H. Introduction of alien plants on the fill and cut slopes of the road construction in South Korea. Ecol. Resil. Infrastruct. 2019, 6, 191–199. DOI:10.17820/eri.2019.6.4.191 [Google Scholar]
- Kim MH, Kim J, Kim KW, Oh W. Effects of the propagation method and planting season of bulblets on shoot emergence and bulb enlargement in Korean native lilies in Sinomartagon section. Hortic. Sci. Technol. 2022, 40, 447–458. DOI:10.7235/HORT20220040 [Google Scholar]
- Kim MH, Park IS, Park KI, Oh W, Kim KW. Elimination of Lily Symptomless Virus by in vitro scaling and reinfection rates under various culture conditions in Korean native lilies. Korean J. Hortic. Sci. Technol. 2015, 33, 891–899. DOI:10.7235/hort.2015.15091 [Google Scholar]
- Lee HS. A study on native wildflower planting programs for sustainable roadside vegetation in USA. J. Korean Soc. Rural Plan. 2017, 23, 157–164. DOI:10.7851/ksrp.2017.23.1.157 [Google Scholar]
- Lilies of the World: Lilium Species Database. 2025. Available online: https://liliumspeciesfoundation.org/ (accessed on 18 December 2025).
- Fu Y, Ren J, Guo R, Lu X, Lei Y, Xu W. In vitro induction and molecular and cellular identification of mutant plants of Lilium leichtlinii var. maximowiczii (Regel) Baker by colchicine. Ornamental Plant Res. 2025, 5, e014. DOI:10.48130/opr-0025-0012 [Google Scholar]
- Askari N, Visser RGF, De Klerk GJ. Growth of lily bulblets in vitro, a review. Int. J. Hortic. Sci. Technol. 2018, 5, 133–143. DOI:10.22059/IJHST.2018.268870.263 [Google Scholar]
- Han BH, Yae BW, Yu HJ, Peak KY. Improvement of in vitro micropropagation of Lilium oriental hybrid ‘Casablanca’ by the formation of shoots with abnormally swollen basal plates. Sci. Hortic. 2005, 103, 351–359. DOI:10.1016/j.scienta.2004.07.003 [Google Scholar]
- Morpurgo J. Urban Green Infrastructure for Biodiversity and Ecosystem Services. Ph.D. Thesis, Universiteit Leiden, Leiden, The Netherlands, 2025. [Google Scholar]
- Kaur R, Thakur N, Sharma DR. Low cost strategy for micropropagation of Lilium Asiatic hybrid cv. Toscana. J. Hortic. Sci. 2006, 1, 24–27. DOI:10.24154/jhs.v1i1.668 [Google Scholar]
- Ruffoni B, Mascarello C, Savona M. Strategies for Lilium propagation: Tradition vs. biotech. Acta Hortic. 2011, 900, 381–390. DOI:10.17660/ActaHortic.2011.900.44 [Google Scholar]
- Nhut DT. Micropropagation of lily (Lilium longiflorum) via in vitro stem node and shoot tip culture. Plant Cell Tissue Organ Cult. 1998, 17, 913–916. DOI:10.1007/s002990050508 [Google Scholar]
- Niimi Y, Onozawa T. In vitro bulblet formation from leaf segments of lilies, especially Lilium rubellum Baker. Sci. Hortic. 1979, 10, 379–389. DOI:10.1016/0304-4238(79)90023-2 [Google Scholar]
- Novak FJ, Havel L. Tissue culture propagation of Lilium hybrids. Sci. Hortic. 1981, 14, 191–199. DOI:10.1016/0304-4238(81)90012-1 [Google Scholar]
- Oh SC, Chung MH, Kim SW, Liu JR. High frequency bulblet formation in scale and stem thin cell layer explant cultures of Lilium oriental hybrids. Korean J. Plant Biotechnol. 2003, 30, 251–255. DOI:10.5010/JPB.2003.30.3.251 [Google Scholar]
- Rafiq S, Rather ZA, Bhat RA, Nazki IT, AL-Harbi MS, Banday N, et al. Standardization of in vitro micropropagation procedure of Oriental Lilium Hybrid cv. ‘Ravenna’. Saudi J. Biol. Sci. 2021, 28, 7581–7587. DOI:10.1016/j.sjbs.2021.09.064 [Google Scholar]
- Song JY, Yi JY, Yoon MS, Lee JR, Lee YY. An efficient in vitro micropropagation for production of disease-free bulbs in Korean native Lilium species. Korean J. Plant Res. 2019, 32, 730–734. DOI:10.7732/kjpr.2019.32.6.730 [Google Scholar]
- Ghanbari S, Fakheri BA, Naghavi MR, Mahdinezhad N. New protocol for the indirect regeneration of the Lilium ledebourii Bioss by using bulb explants. J. Plant Biotechnol. 2018, 45, 146–153. DOI:10.5010/JPB.2018.45.2.146 [Google Scholar]
- Marinangeli P. Somatic embryogenesis of Lilium from microbulb transverse thin cell layers. In Plant Tissue Culture: Methods and Protocols; Humana Press: New York, NY, USA, 2016; pp. 387–394. [Google Scholar]
- Jeong JH. In vitro propagation of bulb scale sections of several Korean native lilies. Acta Hortic. 1996, 414, 269–274. DOI:10.17660/ActaHortic.1996.414.33 [Google Scholar]
- Deswiniyanti NW, Lestari NKD. In vitro propagation of Lilium longiflorum bulbs using NAA and BAP plant growth regulator treatment. KnE Life Sci. 2020, 5, 36–45. DOI:10.18502/kls.v5i2.6437 [Google Scholar]
- Lapiz-Culqui YK, Meléndez-Mori JB, Mállap-Detquizán G, Tejada-Alvarado JJ, Vilca-Valqui NC, Huaman-Human E, et al. In vitro bulbification of five lily varieties: An effective method to produce quality seeds and flowers. Int. J. Agron. 2022, 2022, 8775989. DOI:10.1155/2022/8775989 [Google Scholar]
- Tomanguilla-Llanos O, Meléndez-Mori JB, Tejada-Alvarado JJ, Lapiz-Culqui YK, Huaman-Huaman E, Zuta-Puscan M, et al. Boosting lily bulblets production: a study on the effects of 6-benzylaminopurine and silver nanoparticles. Sci. Rep. 2025, 15, 22099. DOI:10.1038/s41598-025-06078-7 [Google Scholar]
- Mutukistna B. The benefits of urban native plant gardening: An annotated compilation of peer-reviewed literature. UBC Sustain. Sch. Program. 2023, 1–34. Available online: https://sustain.ubc.ca/about/resources/benefits-urban-native-plant-gardening (accessed on 15 February 2025).
- Murashige T, Skoog F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. DOI:10.1111/j.1399-3054.1962.tb08052.x [Google Scholar]
- Ikeuchi M, Sugimoto K, Iwase A. Plant callus: Mechanisms of induction and repression. Plant Cell. 2013, 25, 3159–3173. DOI:10.1105/tpc.113.116053 [Google Scholar]
- Kim MH, Lim YH, Oh W, Yun HK, Kim KW. Effects of Hot Water and Chilling Treatments of Bulblets Propagated by Tissue Culture on Sprouting and Bulb Development in Korean Native Lilies. Kor. J. Hortic. Sci. Technol. 2011, 29, 87–94. Available online: https://www.koreascience.or.kr/article/JAKO201122350105509.page (accessed on 15 February 2025).
- Han BH, Yu HJ, Yae BW, Peak KY. In vitro micropropagation of Lilium longiflorum ‘Georgia’ by shoot formation as influenced by addition of liquid medium. Sci. Hortic. 2004, 103, 39–49. DOI:10.1016/j.scienta.2004.04.020 [Google Scholar]
- Song JY, Kim M, Kim JB, Lee JS, Lim KB. Enhancing in vitro growth of bulbs for mass propagation of lily germplasm. Korean J. Plant Res. 2021, 34, 17–22. DOI:10.7732/kjpr.2020.34.1.017 [Google Scholar]



