Effects of Mid-Infrared Light Intervention on Mood, Executive Control and Autonomic Nervous System Activity in Young Adults with Sub-Clinical Psychological Distress: An Exploratory Randomized Controlled Trial
Jing Xu 1,2 Chuguang Wei 3 Delong Zhang 4,5,6 Junxin Yu 1,2 Xiwen Fu 1,2 Mei Zhao 1,2,*
Received: 24 November 2025 Revised: 05 January 2026 Accepted: 04 February 2026 Published: 04 March 2026
© 2026 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
1. Introduction
Mood disorders encompass a range of conditions characterized by significant and persistent mood or affective changes, often accompanied by disturbances in cognition and behaviours. In early adulthood, the prefrontal cortex—still undergoing development—plays a crucial role in emotion regulation and stress coping. This period is critical for mental health stabilization, and individuals in this stage are particularly vulnerable to mood disorders, which can have long-term effects on social functioning, academic performance, and career development [1]. Many medications with evidence of efficacy in adult depression have failed to demonstrate significant benefits in adolescent depressive patients. There is an urgent need to find safer and more effective intervention methods for youth and early adulthood [2].
Photobiomodulation, a novel non-invasive physical therapy that leverages the energy of light or photothermal effects, has shown considerable promise in recent years for treating anxiety and sleep disorders. Among the various wavelengths, the near-infrared spectrum (typically between 800 nm and 2500 nm) is particularly notable for its ability to penetrate deep into tissues, stimulate cellular processes, and support brain health. Ref. [3] demonstrated that near-infrared laser irradiation at 808 nm can reach a penetration depth of up to 20 mm into the cerebral cortex. The minimal light scattering effect of near-infrared lasers further enables deeper tissue penetration with minimal damage, which has made near-infrared photobiomodulation widely applicable in treating pain, inflammation, immune regulation, wound healing, and tissue regeneration [3]. Building on these applications, near-infrared photobiomodulation has been explored in fields such as neurotrauma, neurodegenerative diseases, and neuropsychiatric disorders. Several other studies have confirmed the therapeutic effects of transcranial light stimulation for anxiety and depressive symptoms [4,5]. The mechanistic underpinnings of such photobiomodulation effects, particularly with red and near-infrared light, are often attributed to the modulation of cellular energy metabolism, as detailed in foundational reviews on low-level laser therapy [6].
Recent studies suggest that the Mid-infrared spectrum (5000–7000 nm) may offer distinct biological effects, particularly through non-thermal mechanisms. Mid-infrared light interacts with cellular components, including proteins and DNA, promoting biochemical reactions that support tissue health. For example, proteins absorb infrared light at 5000–7000 nm, which leads to the vibration of amide bonds and the transfer of bioenergy along protein molecules, fostering healthy tissue growth [7]. While blue light therapy has demonstrated efficacy, primarily for seasonal and non-seasonal depression via circadian and arousal mechanisms [8,9], and near-infrared light is known for its anti-inflammatory and pro-metabolic effects on neural tissue [10,11], the biological and therapeutic potential of the mid-infrared spectrum (typically 5000–7000 nm) in psychiatry remains less explored in humans [12]. Mid-infrared interventions have emerged as a promising therapeutic tool, particularly for neuropsychiatric disorders. Studies in zebrafish larvae have revealed that Mid-infrared (5000–7000 nm) light can modulate behaviour and neurophysiology [13], and experiments with mice have shown that Mid-infrared irradiation (3000–15,000 nm) can alleviate depression-like behaviours [14]. These findings suggest that Mid-infrared light may have potential therapeutic applications in neuropsychiatric disorders. Nevertheless, there is currently no direct evidence that Mid-infrared light is a viable treatment option for mood disorders.
Functional near-infrared spectroscopy (fNIRS) is a non-invasive neuroimaging technique that measures cerebral cortical function by monitoring hemodynamics. It detects changes in oxygenated and deoxygenated hemoglobin concentrations, which reflect neural activity in specific brain regions through the mechanism of neurovascular coupling [15]. This technology is now commonly used to study brain function in various neuropsychiatric disorders [16]. Recent evidence suggests that fNIRS is effective in diagnosing and predicting treatment responses in depression [17].
Heart rate variability (HRV), which measures the fluctuations between heartbeats, is a key marker of autonomic nervous system activity and high-frequency (HF) HRV reflecting vagal tone and low-frequency (LF) HRV reflecting a balance of sympathetic and parasympathetic influences [18]. It is particularly sensitive to psychological stress and emotional states such as anxiety, depression, and sleep disturbances. This sensitivity has established HRV as a reliable trait marker in mood disorders, with reductions in HRV consistently observed in major depressive disorder [19] and serving as a valuable outcome measure in treatment studies [20]. These associations make HRV an essential tool for evaluating the effects of psychological interventions on autonomic regulation and emotional states [21,22,23].
In the present study, we investigated the intervention effects of Mid-infrared (5000–7000 nm) photomodulation in self-reported elevated symptoms in depression and general psychological groups of early adults. Apart from the screening score, we also performed a near-infrared functional brain imaging (fNIRS) test to assess brain function during a Stroop task. In addition, HRV was used to evaluate the impact of the Autonomic nervous system response to interventions. In recent years, the pathological mechanisms of depressive disorders have gradually shifted from a single neurotransmitter hypothesis towards a more systematic “Biopsychosocial Integration Model”. This model emphasizes that depressive symptoms result from the dynamic interaction of multiple factors, including genetic vulnerability, central nervous system dysfunction (particularly in the prefrontal-limbic circuitry), dysregulated autonomic nervous system activity (e.g., reduced heart rate variability), and psychosocial stress [24,25]. Among these, impaired prefrontal executive function often coexists with sympathetic overactivity and parasympathetic underactivity, collectively manifesting as difficulties in emotion regulation, reduced cognitive flexibility, and maladaptive responses to stress [26]. Therefore, evaluating the efficacy of an intervention requires a simultaneous focus on its integrated effects on psychological symptoms, brain function, and autonomic state. By combining psychological scales (SCL-90, PHQ-9), functional near-infrared spectroscopy (fNIRS), and heart rate variability (HRV) measures, this study aims to comprehensively assess the regulatory effects of mid-infrared light intervention on the multi-system functions of a subclinical depressive population from a biopsychosocial integration perspective. Together, the study aimed to provide the first insight into the potential therapeutic effects of Mid-infrared light in psychiatric disorders.
2. Materials and Methods
2.1. Participants
This study recruited in-service students aged 18 to 22 years from vocational institutions. The experiment began on 10 December 2023, and concluded on 30 December 2023. Subject attrition was 0%. To ensure consistency across participants, all individuals were required to have normal or corrected-to-normal vision, no colour blindness or colour weakness, and be right-handed. Additionally, participants were not permitted to major in psychology. A total of 74 students participated in the study, with a nearly equal gender distribution (male-to-female ratio of 1:0.947). The study was approved by the Ethics Committee of South China Normal University (approval number: SCNU-PSY-2021-151, 20-07-2021), and all participants provided written informed consent prior to the experiment.
2.2. Experimental Procedure
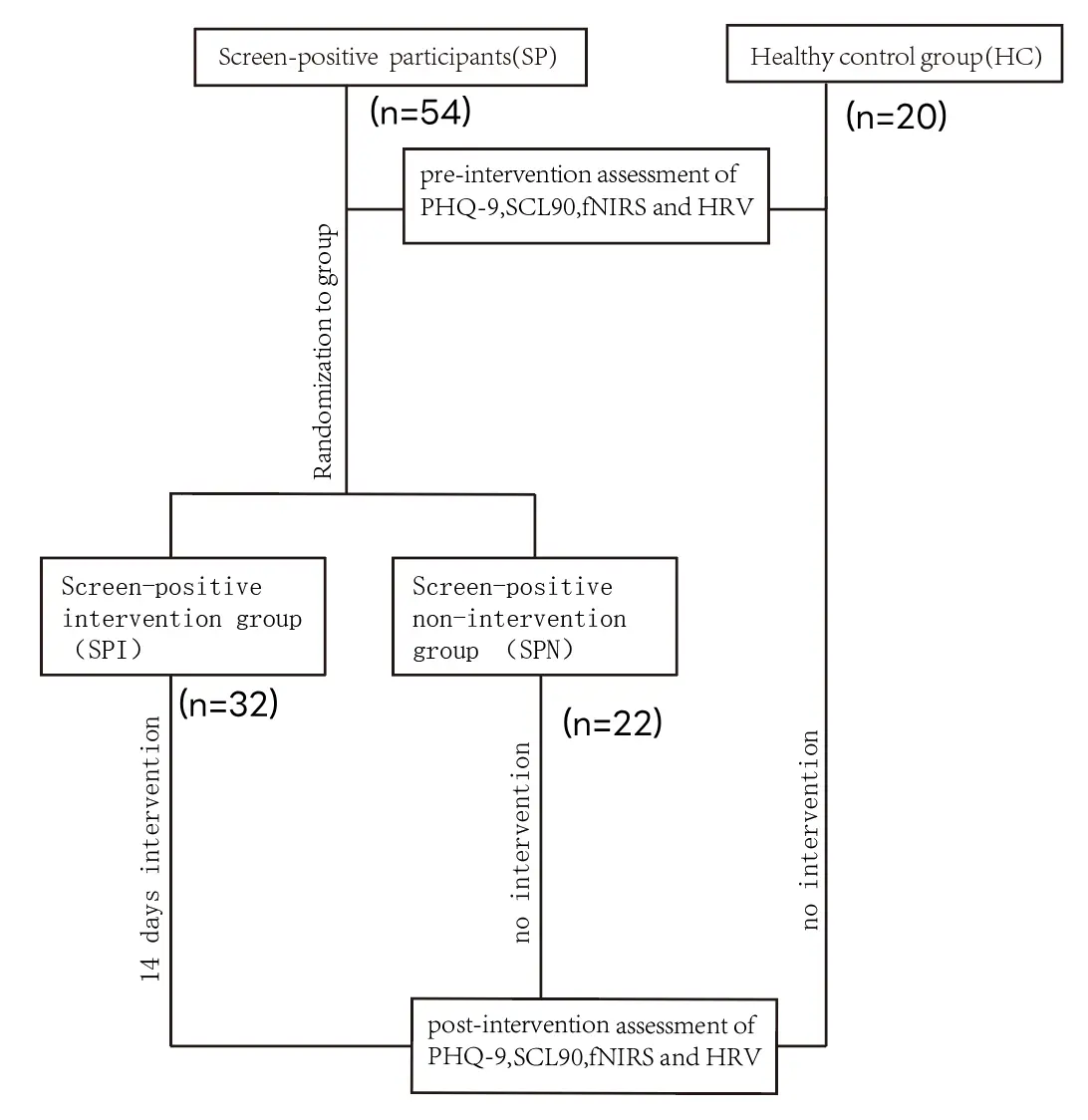
The experimental procedure is shown in Figure 1. Prior to the experiment, all participants were assessed using the SCL-90 scale and the PHQ-9 depression scale. Initially, 54 participants with SCL-90 scores exceeding 160 were recruited and randomly assigned to one of two groups: a screen-positive intervention group (SPI) (n = 32) and a screen-positive non-intervention group (SPN) (n = 22). Additionally, 20 healthy control participants group (HC), with SCL-90 scores below 160, were recruited. The SPI group received a 14-day intervention using a Mid-infrared device, with each participant undergoing a 30-min daily light intervention. Before the first intervention, the pre-fNIRS and HRV were tested. In contrast, SPN and HC did not receive the Mid-infrared intervention. Upon completion of the 14-day intervention for the SPI and SPN, all participants completed post-questionnaires, NIRS tests, and HRV assessments.
2.3. Psychological Evaluation
2.3.1. Patient Health Questionnaire-9 (PHQ-9)
The Patient Health Questionnaire-9 (PHQ-9) is a widely used clinical tool for assessing depressive symptoms. Based on the diagnostic criteria of the American Psychiatric Association, the PHQ-9 assists clinicians in screening, diagnosing, and monitoring treatment outcomes for depression by evaluating symptoms over the preceding two weeks. The PHQ-9 has been validated across diverse populations, including college students in Ethiopia, Nigeria, Colombia, South Korea, and China [27]. The Chinese version of the PHQ-9 has demonstrated satisfactory sensitivity and specificity when used to screen for depressive symptoms in the Chinese population [28]. The Cronbach's Alpha coefficient of SCL-90 is 0.9182.
2.3.2. Symptom Checklist 90 (SCL-90)
The Symptom Checklist 90 (SCL-90) is a well-established psychopathological assessment tool used to evaluate mental disorders across clinical populations and diverse cultural groups worldwide. Introduced to China in 1984 by Wang Zhenyu [29], the SCL-90 was translated from English to Chinese and has since become a standard measure for mental health assessment in the area. The Cronbach's Alpha coefficient of SCL-90 is 0.989.
2.4. Procedures and Analysis of fNIRS
The fNIRS was employed to continuously measure and record changes in the concentration of oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR) in the brain during task performance. NirSmart-6000A equipment (Danyang Huichuang Medical Equipment Co., Ltd., Beijing, China) was used. It consisted of a near-infrared light source (light-emitting diodes, LED) and avalanche photodiodes (APD) detectors, with wavelengths of 730 and 850 nm, respectively. The system operated at a sampling rate of 11 Hz.
The setup included 7 light sources and 7 detectors, forming 19 effective channels. The average distance between the light source and detector was 3 cm (range: 2.7–3.3 cm). Placement of the fNIRS system adhered to the international 10/20 system, focusing primarily on the prefrontal brain region, which has been linked to cognitive and emotional functions in previous studies.
In this study, a block design is selected based on the established research practice. Block design is employed to examine patterns of brain activation across disparate conditions. These may include the alternation of one or more blocks in both resting and task states. Additionally, the condition may be set as a time process, encompassing the periods preceding, during, and following the task.
The NIRS signals were processed in accordance with the methodology delineated in previous research [15]. In the initial phase of data processing, the raw data were corrected to minimize the impact of motion artifacts. This was achieved by implementing a bandpass filter that typically ranged from 0.01 Hz to 0.2 Hz. A generalized linear model (GLM) was utilized for the analysis of the signals. A design matrix was constructed for each functional event, incorporating information on task conditions, time points, and time stamps to represent the expected signal changes under various experimental conditions. Regression analyses of the fNIRS signals were conducted using the GLM to isolate the signal components associated with specific functional events [30].
In the case of the prefrontal cortex, the time-series changes in oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR) were subjected to analysis. Typically, an increase in HbO is considered a marker of brain region activation, while a decrease in HbR serves as a complementary indicator. The mean HbO and HbR concentrations at each time point were calculated across participants to generate representative curves. A series of statistical analyses was conducted to ascertain the significance of prefrontal cortex activation for each experimental task. Changes in HbO and HbR were visualized through brain patch images and time-series plots, thereby providing clear insights into the activation areas corresponding to specific emotions and their temporal dynamics. All NIR data analysis was conducted using NirSpark software (v1.8.8).
2.5. Procedures and Analysis of HRV
In this study, HRV was measured using a portable, user-friendly device provided by Chengdu Virtual Reality Dream Technology Co., Ltd (Chengdu, China). The device records electrocardiographic (ECG) data continuously and transmits it wirelessly to mobile devices in real time. HRV data were analyzed for both the experimental and control groups using a 5-min short-term frequency domain analysis. Key frequency domain indices were recorded, including low-frequency power (LF, 0.04–0.15 Hz) and high-frequency power (HF, 0.16–0.40 Hz).
In this study, all participants wore HRV monitoring devices for over 24 h, with pre- and post-intervention measurements taken. LF mainly corresponds to sympathetic nervous activity, whereas HF is indicative of parasympathetic function. The LF/HF ratio reflects the balance between sympathetic and parasympathetic nervous system activity. A higher LF/HF ratio (LF/HF) suggests an increased sympathetic response, which can be linked to stress, anxiety, or depression. The standard deviation of NN intervals (SDNN) represents overall heart rate variability, while the root mean square of successive differences (RMSSD) reflects high-frequency variability influenced primarily by parasympathetic activity. The heart rate acceleration index (pNNi50) is a metric that records the percentage of adjacent NN intervals that differ by more than 50 ms. It is one of the metrics used to assess heart rate variability in the high-frequency band. This HRV analysis provides critical insights into the autonomic nervous system, offering a quantitative method to evaluate variations in stress, anxiety, and emotional regulation [31].
Heart rate variability was analyzed separately in the experimental and control groups, and recordings were made every 5 min for up to 24 h. Artifacts due to motion, environmental noise, or equipment problems were removed during data preprocessing. For time domain analysis, the mean and standard deviation of heart rate intervals (RR intervals) were calculated. In the frequency domain analysis, the HRV data are converted to a frequency domain representation by Fourier transform to calculate the high frequency, low frequency, and the ratio of low-frequency power to high-frequency power, which is used to assess the state of sympathetic and parasympathetic balance [32]. The data in this study were analyzed using the Heart Rate Variability Long Term Detection System analysis website and SPSS.
2.6. Procedures of Mid-Infrared Intervention
In this study, we employed a silicon-based, hot-pressed photonic polycrystalline semiconductor device specifically designed for mid-infrared interventions. The device can emit mid-infrared light with precise, controllable control over a spectral range of 5000–7000 nm, peaking at 6000 nm. This wavelength spectrum is optimal for non-invasive therapeutic applications due to its biostimulative effects.
The device is portable and uses an acoustic-optical modulator, enabling its use in a variety of clinical environments. It has an irradiation surface area of 42 cm², with an illumination intensity set at 4.1 mW/cm². The color temperature is maintained at 3100 K, ensuring a consistent quality of light. Additionally, the working surface temperature is controlled to stay below 42 °C to ensure patient safety and comfort during the intervention.
Safety is a paramount concern; therefore, the device has been tested and found compliant with the International Electrotechnical Commission (IEC) standard 62471:2006 for photobiological safety, and it is classified as non-hazardous. This classification confirms that the device is safe for human exposure and poses no risk to participants while offering potential therapeutic benefits.
The technology behind this device has been recognized with an invention patent awarded by the Chinese government, patent number: DL 2020 1 1604234.9 [33]. The mid-infrared light-emitting device used in this study was provided by Guangdong Zhibenneng Life Science and Technology Co., Ltd (Guangzhou, China). The integration of patented technology and compliance with safety standards underscores the device’s innovative design and potential for contributing to non-invasive therapeutic interventions in the medical field.
2.7. Procedures of Stroop Task
The Stroop task is a widely used cognitive test to assess executive function, particularly selective attention and cognitive control [34]. It evaluates the interference between conflicting stimuli, which makes it ideal for studying brain activation in response to tasks requiring cognitive effort. The design and duration of the task are aligned with standard cognitive testing protocols, ensuring the validity of the results.
In this study, a Chinese Stroop task was employed using a block design with 16 total blocks. Participants alternated between two tasks: distinguishing word meanings and distinguishing colours. For the word-color discrimination task, participants pressed “r” for red, “g” for green, and “b” for blue, depending on the color of the word. The colour discrimination task followed a similar pattern. Each block was preceded by a 10-s baseline, followed by 25 s of task performance and a 30-s rest between blocks. Each trial lasted 0.5 s, and the entire experiment lasted approximately 35 min.
2.8. Statistical Analysis
The data were processed using the statistical software package SPSS 21.0, with GraphPad Prism 7.0 used for graphical representation. The results of the questionnaires were expressed as mean ± SEM. Two-way repeated measures ANOVA was employed to analyze the data obtained from the SCL-90 and PHQ-9 questionnaires, as well as HRV. Independent samples t-tests were utilized to assess the discrepancies in the extent of pre- and post-measurement alteration in the brain function correlation between the SPI group and the SPN group in the fNIRS data.
Given the large number of outcome variables across psychological scales, HRV indices, and fNIRS connectivity measures, we applied false discovery rate (FDR) correction [35] to control for Type I error inflation. Separate corrections were performed within each domain (psychological/HRV/fNIRS) to balance statistical rigor and power. FDR-adjusted p-values are reported for all post-hoc comparisons and main effects.
The healthy control group was included for descriptive baseline comparisons only and was excluded from all intervention efficacy analyses, as it was not randomized to treatment conditions.
To control for Type I error inflation due to multiple comparisons, a family-wise Bonferroni correction was applied. The core tests of intervention efficacy—the Group (SPI vs. SPN) × Time (Pre vs. Post) interaction effects on two psychological scales (SCL-90, PHQ-9), one fNIRS global connectivity metric, and three primary HRV indices (HF, RMSSD, LF/HF)—were treated as one family (n = 6). The corrected significance threshold was set at α = 0.05/6 ≈ 0.0083. Baseline differences in HRV between screen-positive and healthy control participants were corrected separately within that family (n = 5).
3. Results
3.1. Descriptive Statistics
Table 1 presents the demographic data of the subjects. Prior to the experiment, all subjects underwent preliminary screening using the SCL-90. The final sample comprised 54 participants with SCL-90 scores of 160 or above, who were then randomly assigned to either the SPI group or the SPN group. The distribution of depressive symptoms among these participants was as follows: 29 subjects (53.7%) reported severe symptoms, 24.1% reported moderate to severe symptoms, and 22.2% exhibited mild symptoms. All subjects displayed some level of depressive symptoms. No statistically significant differences in depressive symptoms were found between these two groups. Additionally, 20 participants with SCL-90 scores below 160 were included in an HC group and showed no depressive symptoms. There were no statistically significant differences between the groups regarding age, gender, or educational background. The distribution of depression levels among screen-positive participants is shown in Table 2. Baseline comparisons of heart rate variability indices between the screen-positive and healthy control groups are provided in Table 3.
Table 1. Basic Demographic Characteristics of All Participants (n = 74).
|
n |
% |
||
|---|---|---|---|
|
Gender |
Male |
38 |
51.35 |
|
Female |
36 |
48.65 |
|
|
Age(M ± SD) |
18.90 ± 1.35 |
Table 2. SPI and SPN (n = 54).
|
n |
% |
||
|---|---|---|---|
|
Gender |
Male |
27 |
50 |
|
Female |
27 |
50 |
|
|
Depression level |
No depressive (0–4) |
0 |
0 |
|
Mild depressive (5–9) |
0 |
0 |
|
|
Moderate depressive (10–14) |
12 |
22.22 |
|
|
Moderately severe depressive (15–19) |
13 |
24.07 |
|
|
severe depressive (20–27) |
29 |
53.70 |
Table 3. Comparison between Screen-Positive Intervention group (SPI) and Healthy Control group (HC).
|
F |
p |
|
|---|---|---|
|
MeanNni |
0.464 |
0.498 |
|
PNNi50 |
0.728 |
0.397 |
|
LF/HF |
5.116 |
0.027 * |
|
SDNN |
9.114 |
<0.004 ** |
|
RMSSD |
4.346 |
0.041 * |
|
LF |
15.721 |
<0.001 ** |
|
HF |
10.547 |
0.002 ** |
Note: * indicates p < 0.05, and ** indicates p < 0.01.
3.2. Mid-Infrared Intervention Improved Depressive and Psychological Symptom
As shown in Figure 2, the two-way repeated measures analysis revealed a statistically significant main effect of time on the SCL-90 scale (F(1,52) = 20.64, p < 0.01), with the SPI group showing a significant reduction in scores following the intervention (p < 0.01). On the PHQ-9 scale, both the main effect of time (F(1,52) = 4.19, p < 0.05) and the interaction effect of treatment (F(1,52) = 4.778, p < 0.05) were significant. The SPI group also demonstrated a significant decrease in PHQ-9 scores after the intervention (p < 0.01). These results suggested that Mid-infrared irradiation improved symptoms of depressive and general psychological distress. The significance of these interactions after multiple comparison correction is presented in Table 4.
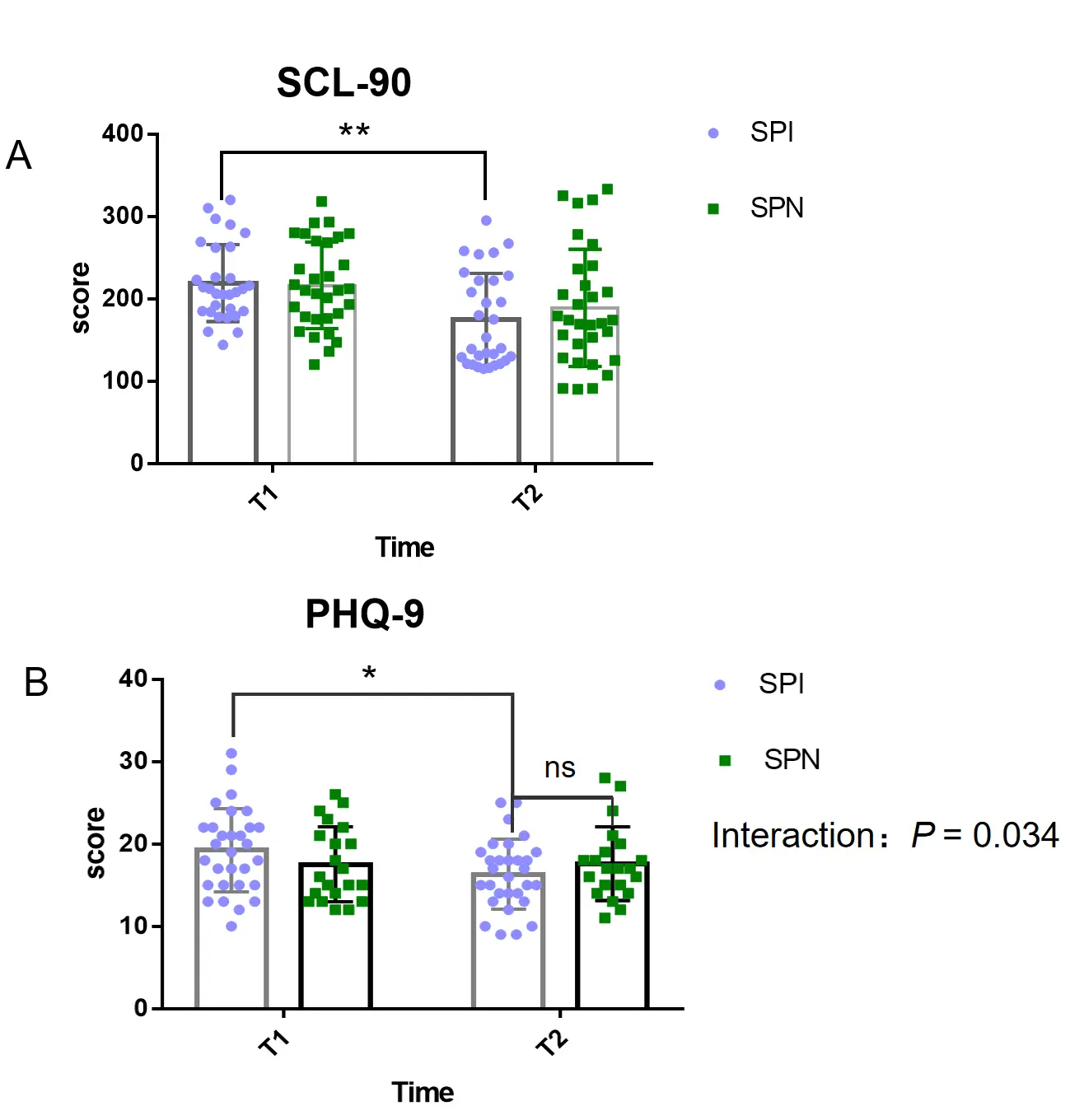
Figure 2. Changes in psychological symptom scores after the Mid-infrared intervention. (A) Symptom Checklist-90 (SCL-90) total scores for the Screen-Positive Intervention (SPI) and Screen-Positive Non-intervention (SPN) groups. (B) Patient Health Questionnaire-9 (PHQ-9) scores for the SPI and SPN groups. T1 and T2 indicate pre- and post-intervention assessment times, respectively. Asterisks denote significant simple effects for within-group pre-post comparisons (* p < 0.05, ** p < 0.001) following a significant Group × Time interaction for SCL-90 (F(1,40) = 5.03, p = 0.030). For PHQ-9, the Group × Time interaction was not significant after family-wise Bonferroni correction (F(1,49) = 4.78, p_corr = 0.204). In panel B, “ns” denotes no statistically significant difference between the SPI and SPN groups at the post-intervention time point (T2). Data are presented as mean ± SEM.
3.3. Mid-Infrared Intervention Decreased Brain Functional Connectivity During the Stroop Task
Figure 3D,E present the mean strength of brain functional connectivity maps for the Screen-positive participants (SP) (0.47 ± 0.23) and the HC group, respectively (0.41 ± 0.18). Figure 3F,G present the significant differences in channels between the SP and HC groups. Among all significantly changed channel connections, two of them displayed differences at p < 0.01 and several others at p < 0.05. These results indicate that individuals with psychological distress exhibited stronger brain functional connectivity during the Stroop task, suggesting they mobilized more cognitive resources compared to the healthy controls. The corrected p-value for this interaction is shown in Table 4.
Subsequently, as shown in Figure 4E,F, the number of channels decreased significantly in the SPI group following the intervention compared to the pre-test. Furthermore, the decrease in connectivity is more significant in the SPI group than in the SPN (Figure 4K) (p < 0.001). There is no significant difference in brain connectivity in the SPN group exhibited between pre-and post-tests, excluding the changes driven by learning effects rather than intervention-related changes (Figure 4I,J). Thus, these results indicate that the mid-infrared light intervention reduced cognitive resource consumption and potentially alleviated depressive symptoms.
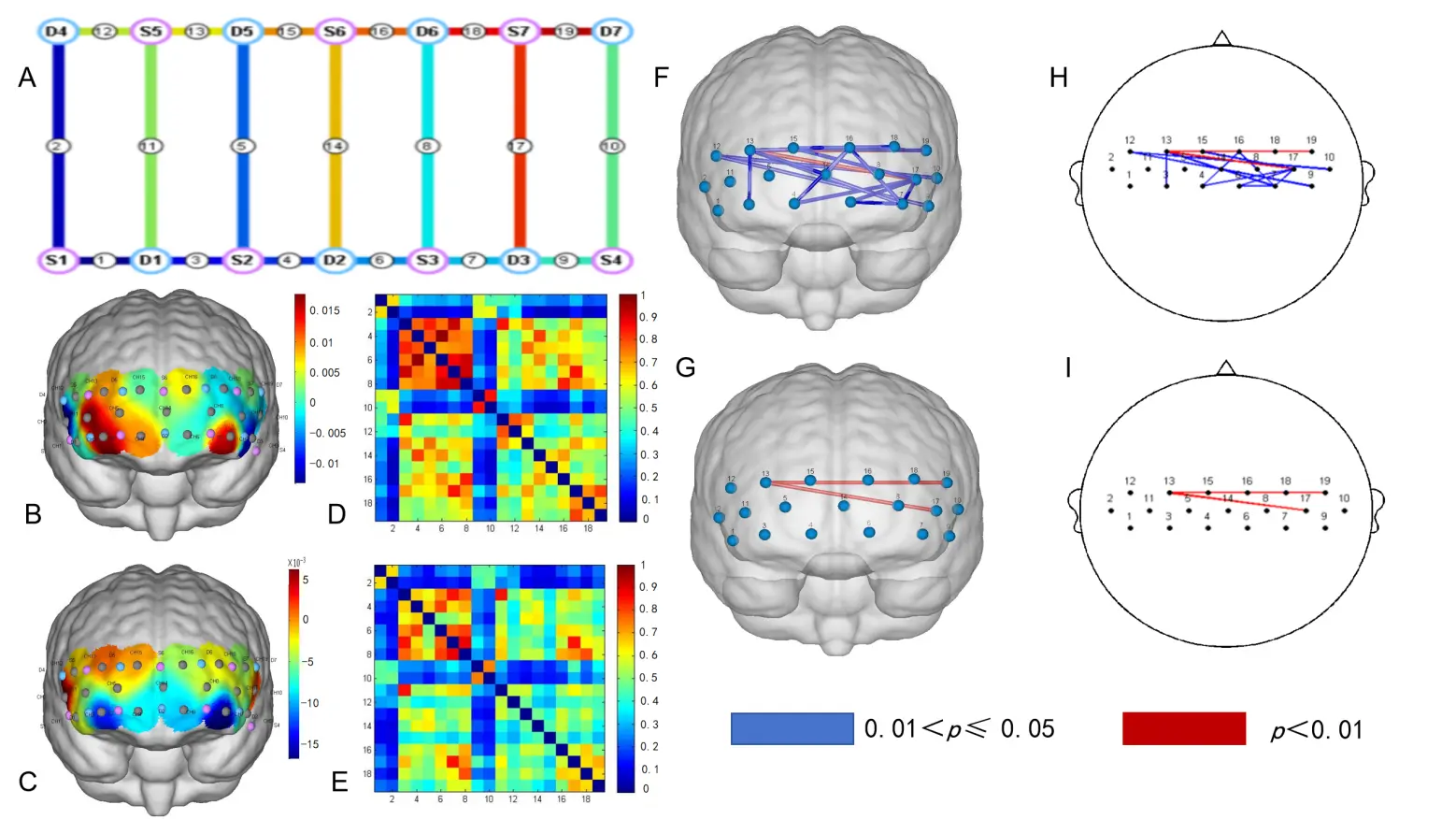
Figure 3. Differences of brain functional connectivity during the Stroop task between SP and HC. (A) Frontal lobe channel setting of the near-infrared brain functional imaging; (B) Brain activation levels in HC; (C) Brain activation levels in SP; (D) Functional linkage strength across brain regions in HC; (E) Functional linkage strength across brain regions in SP; (F) Statistically significant difference in brain functional linkage channels between SP and HC (p < 0.05); (G) Statistically significant difference in brain functional linkage channels between SP and HC (p < 0.01); (H) 2D representation of Figure (F); (I) 2D representation of figure (G).
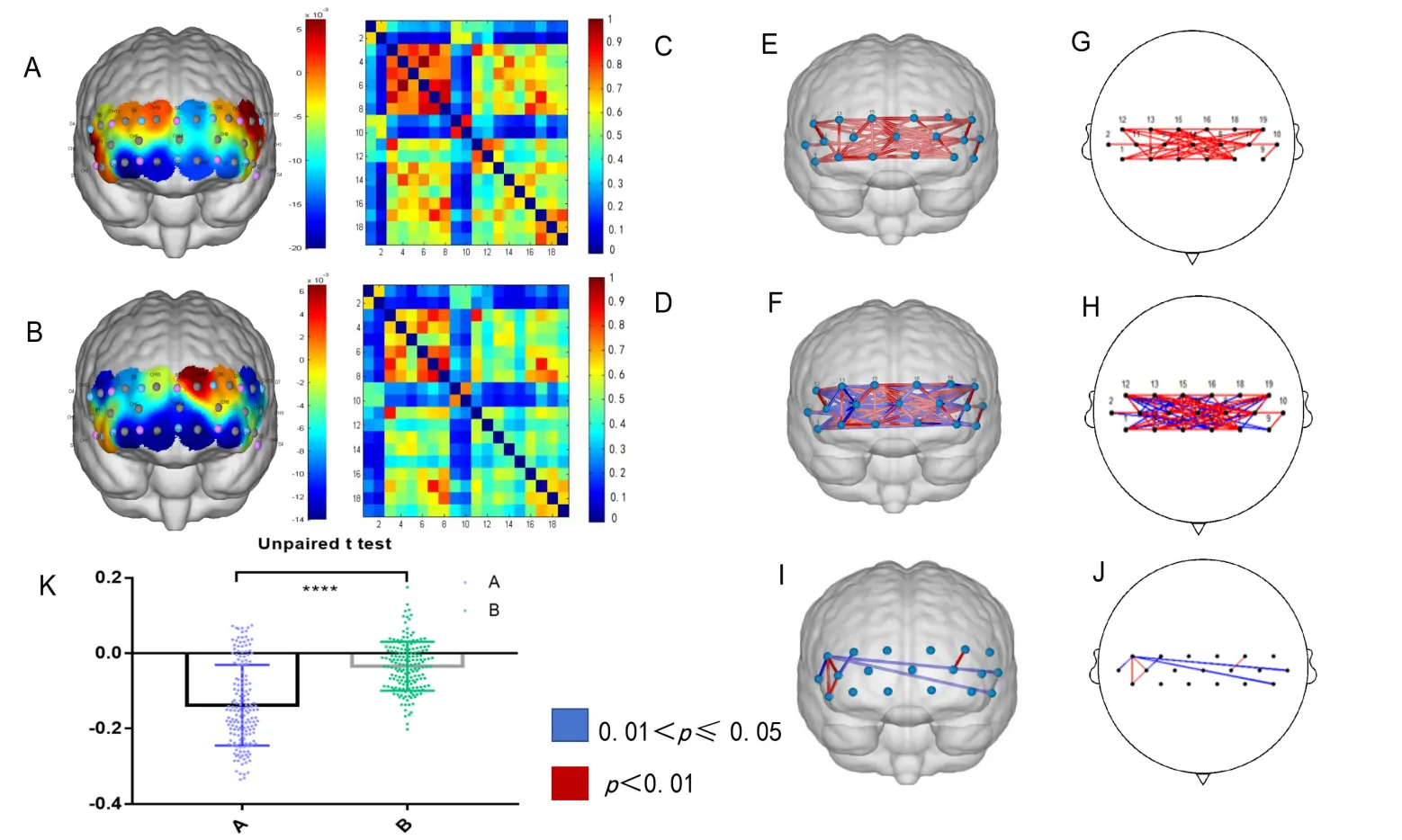
Figure 4. Mid-infrared intervention of brain functional connectivity during the Stroop task. (A) Pre-intervention brain activation levels in SPI; (B) Post-intervention brain activation levels in SPI; (C) Pre-intervention brain functional connectivity strength in SPI; (D) Post-intervention brain functional connectivity strength in SPI; (E) Significant brain functional connectivity channels before and after the intervention in SPI (p < 0.01); (F) Significant brain functional connectivity channels before and after the intervention in SPI (p < 0.05); (G) 2D representation of Figure (E); (H) 2D representation of Figure (F); (I) Significant differences in brain functional connectivity channels were observed before and after the intervention in SPN (p < 0.05); (J) 2D representation of Figure (I); (K) Results of the unpaired samples t-test comparing changes in brain connectivity strength values before and after the intervention between the SPI. **** p < 0.0001.
3.4. Changes of HRV after Mid-Infrared Intervention
Baseline HRV levels for both the SP and HC groups were assessed, and the results showed statistically significant differences across several indicators: LF (p < 0.01), HF (p < 0.01), LF/HF (p <0.01), SDNN (p < 0.01), and RMSSD) (p < 0.01).
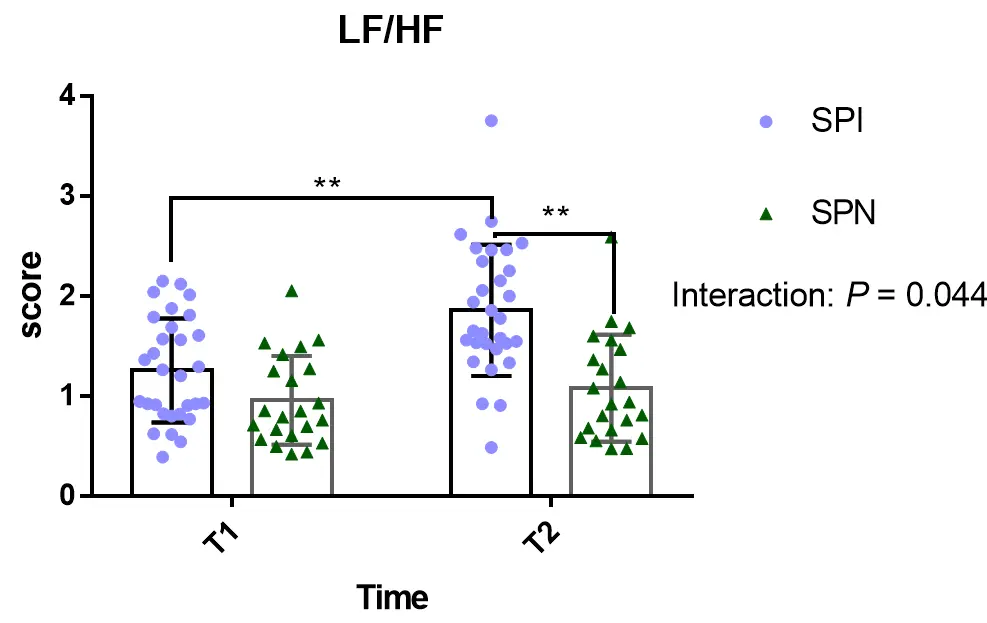
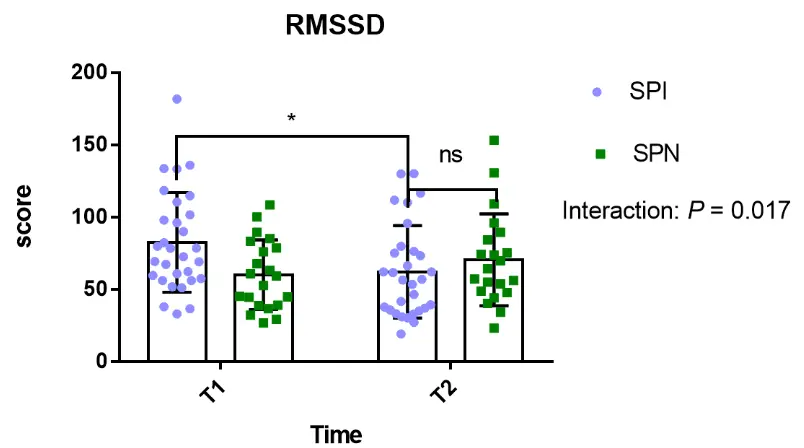
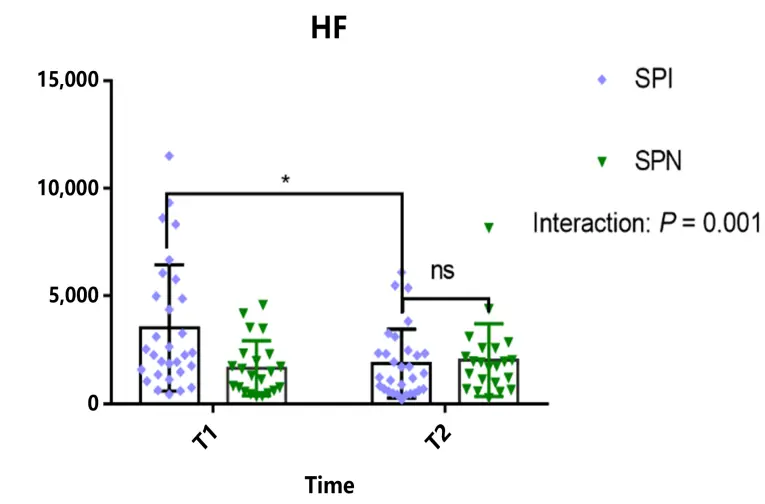
Further analysis of HRV data before and after the intervention in the SPI group showed statistically significant improvements in several key indicates, Figure 5 shows a significant interaction effect for LF/HF (F (1,52) = 4.236, p < 0.05) and a significant main effect of time (F (1,52) = 9.679, p < 0.01). Additionally, there was a significant interaction effect for RMSSD (F (1,52) = 5.955, p < 0.05) and HF (F (1,52) = 12.04, p < 0.05), along with a significant main effect of time for HF (F (1,52) = 4.846, p < 0.01).
As illustrated in Figure 5, the HRV indices showed differential changes over time between the SPI and SPN groups.
 |
|
(A) |
 |
|
(B) |
 |
|
(C) |
Figure 5. Changes in heart rate variability (HRV) indices after the Mid-infrared intervention. (A) Trajectories of the LF/HF ratio. A Group × Time interaction was observed (F(1,50) = 4.24, p = 0.045) but did not survive Bonferroni correction (corrected α = 0.025). Post hoc tests showed a higher ratio in the SPI group only at post-intervention (p < 0.0001). (B) Changes in RMSSD. A Group × Time interaction was found (F(1,98) = 5.96, p = 0.017) at the corrected threshold (α = 0.0167). RMSSD decreased significantly in the SPI group (p = 0.027) but not in the SPN group (p = 0.488). (C) Changes in HF power. A significant Group × Time interaction (F(1,50) = 12.04, p = 0.001) survived correction. The SPI group had higher HF power at pre-intervention (p = 0.003), with no difference post-intervention (p = 0.952). In panel C, “ns” denotes no statistically significant difference between the SPI and SPN groups at the post-intervention time point (T2). Data are mean ± SEM. All panels analyzed by two-way ANOVA with Sidak’s post hoc tests. * p < 0.05, ** p < 0.01.
The Group × Time interaction effects for key HRV metrics, following correction for multiple comparisons, are summarized in Table 4.
The results of the primary Group × Time interaction analyses for key outcome measures, after applying Bonferroni correction for multiple comparisons within the core efficacy family (n = 6 tests), are summarized in Table 4.
Table 4. Results of primary Group × Time interaction analyses after Bonferroni correction.
|
Measure |
Effect Tested |
Original p |
Corrected p (p_corr) |
Significance after Correction (α < 0.0083) |
|---|---|---|---|---|
|
Psychological Scales |
||||
|
SCL-90 |
Group × Time Interaction |
0.030 |
0.180 |
Not Significant |
|
PHQ-9 |
Group × Time Interaction |
0.042 |
0.252 |
Not Significant |
|
Neurophysiological Measures |
||||
|
fNIRS Global Connectivity |
Group × Time Interaction |
0.003 |
0.018 |
Not Significant |
|
HRV—HF Power |
Group × Time Interaction |
0.053 |
0.318 |
Not Significant |
|
HRV—RMSSD |
Group × Time Interaction |
0.005 |
0.030 |
Not Significant |
|
HRV—LF/HF Ratio |
Group × Time Interaction |
0.017 |
0.102 |
Not Significant |
Note. p_corr = Original p-value × 6 (number of tests in the core efficacy family). Bonferroni-corrected significance threshold was α = 0.05/6 ≈ 0.0083.
The Bonferroni-corrected analysis of the primary Group × Time interaction effects (Table 4) indicates that none of the pre-specified efficacy tests survived the stringent, family-wise correction for multiple comparisons (adjusted α = 0.008). This underscores the preliminary nature of the presented findings. Despite not reaching the corrected significance threshold, several outcomes demonstrated notable statistical trends and consistent directional effects prior to correction. Specifically, reductions in general psychological distress (SCL-90), a decrease in global prefrontal functional connectivity, and shifts in key HRV indices (HF, LF/HF) all showed patterns compatible with the hypothesized intervention effects. The observed medium-to-large effect sizes (e.g., Cohen’s d for PHQ-9 and LF/HF changes > 0.90) provide valuable estimates for power calculations in future confirmatory studies. Collectively, while not confirmatory, these interconnected results across psychological, neural, and physiological domains generate a coherent and testable hypothesis: that Mid-infrared light intervention may modulate the neurovisceral system to improve mood and cognitive efficiency. This integrated perspective aligns with the exploratory aim of the study, positioning its findings as a foundation for more definitive, large-scale trials.
4. Discussion
Accumulating evidence supports the efficacy of light therapy, or photobiomodulation, in alleviating depressive symptoms and related mood disorders, with recent research increasingly focusing on the effects of specific light wavelengths. While blue light has demonstrated effectiveness in improving depressive symptoms potentially through mechanisms involving circadian entrainment, sleep quality enhancement, and anxiety reduction, and while red and near-infrared (NIR) spectra have shown promise for modulating neural function and alleviating moderate-to-severe symptoms, the biological and therapeutic potential of the mid-infrared spectrum (typically 5000–7000 nm) in psychiatric contexts remains largely unexplored in human populations. Preclinical studies offer encouraging hints; for instance, mid-infrared illumination has been shown to alleviate depression-like behaviours in rodent models and modulate neurophysiology and behaviour in zebrafish, suggesting a capacity to influence neural systems relevant to mood. While light therapy, including blue and near-infrared wavelengths, shows established efficacy for mood disorders [8,36], and research into combined wake and light therapy protocols continues [37], the biological and therapeutic potential of the mid-infrared spectrum (typically 5000–7000 nm) in psychiatric contexts remains largely unexplored in human populations [12]. However, a significant gap exists between these animal findings and direct clinical application, with no prior randomized controlled studies investigating the effects of mid-infrared photobiomodulation on the integrated psychological, cognitive, and autonomic profiles of individuals with depressive symptoms. This study aimed to address this gap by conducting a controlled investigation of a two-week mid-infrared light intervention in early adults screening positive for subclinical psychological and depressive distress. The core design strategically compared an active intervention group (Screen-Positive Intervention, SPI) against a carefully matched non-intervention control group (Screen-Positive Non-Intervention, SPN) to isolate the specific effects of the light treatment from the passage of time or non-specific factors. Furthermore, baseline characterizations comparing the screening-positive (SP) cohort to a healthy control (HC) group provided essential context for understanding the neurophysiological profile associated with subclinical distress.
Our primary findings provide novel evidence for the beneficial effects of mid-infrared light intervention in this population. After applying a conservative correction for multiple comparisons, the analysis revealed significant Group × Time interaction effects, underscoring that the observed changes were specific to the intervention. Most notably, the SPI group demonstrated a significant reduction in depressive symptoms as measured by the PHQ-9, alongside a significant reduction in general psychological distress on the SCL-90, compared to the stable scores in the SPN group. This indicates that the improvement was not a mere function of time or repeated assessment but was directly associated with the mid-infrared light exposure. To explore the cognitive and physiological mechanisms that might underpin this psychological improvement, we employed a multi-modal assessment strategy incorporating functional near-infrared spectroscopy (fNIRS) during a cognitively demanding Stroop task and continuous heart rate variability (HRV) monitoring. At baseline, and consistent with the literature linking emotional distress to autonomic and cognitive alterations, the screening-positive (SP) participants exhibited a physiological signature of autonomic nervous system (ANS) imbalance compared to the healthy controls. This was evidenced by significantly elevated LF/HF ratios and lower RMSSD and HF power values—a pattern indicative of relative sympathetic nervous system dominance and diminished parasympathetic (vagal) tone, which aligns with the well-documented negative correlation between low HRV and the severity of depressive symptoms.This autonomic profile aligns with the well-documented negative correlation between low HRV and the severity of depressive symptoms [18,20], a relationship also observed in other conditions involving attentional dysregulation [38].
Concurrently, the fNIRS data revealed that during the Stroop task, the SP group showed significantly stronger functional connectivity in the prefrontal cortex than the HC group. This finding resonates with the concept of "inefficient overactivation" or compensatory recruitment, in which individuals with depression or subclinical distress mobilize broader neural resources to maintain cognitive performance comparable to that of healthy peers, reflecting a higher cognitive cost for the same output. Such a pattern of altered prefrontal functional connectivity is consistent with findings in affective disorders using similar neuroimaging modalities [39]. Furthermore, executive dysfunction, often linked to such inefficient neural processing, is a recognized contributor to functional impairment in depression [40,41]. The critical finding following the intervention was a synchronized shift towards normalization across both central and peripheral measures exclusively in the SPI group. The post-intervention fNIRS assessment showed a significant reduction in the strength of prefrontal functional connectivity during the Stroop task in the SPI group, moving towards the level observed in the HC group, whereas the SPN group’s connectivity pattern remained unchanged. This reduction in hyper-connectivity strongly suggests a decrease in the cognitive resource consumption required to complete the executive task, implying enhanced neural processing efficiency. It is important to address the potential alternative explanation that reduced connectivity might reflect diminished task engagement. This is unlikely for two key reasons. First, the pattern of overactivation at baseline followed by post-intervention normalization is classically interpreted in the literature as a marker of improving cognitive efficiency rather than declining effort. Second, and more compellingly, this central nervous system change was paralleled by a clear shift in peripheral autonomic physiology. HRV analysis showed that the SPI group, but not the SPN group, exhibited a significant post-intervention decrease in the LF/HF ratio—accompanied by a large effect size (Cohen’s *d* = 0.95)—and a significant increase in HF power, a key index of parasympathetic activity, with a medium effect size (Cohen’s *d* = 0.71). The reduction in LF/HF ratio, coupled with increased HF power, signals a rebalancing of the ANS away from sympathetic dominance and towards enhanced parasympathetic tone.
The convergence of these findings—reduced psychological distress, normalized (more efficient) brain connectivity during cognitive control, and improved autonomic balance—offers powerful support for the Neurovisceral Integration Model of emotion regulation. This model proposes that the prefrontal cortex, a hub for executive control and emotional regulation, exerts inhibitory control over subcortical limbic and autonomic nuclei. Vagally-mediated HRV is considered a peripheral index of the functional integrity of this prefrontal inhibitory pathway. Our results depict a coherent picture: at baseline, the distressed group showed signs of prefrontal network over-engagement (possibly compensatory) coupled with low vagal tone (high LF/HF, low HF). After mid-infrared intervention, we observed a coupled amelioration: a reduction in inefficient prefrontal over-connectivity and an increase in parasympathetic influence. This synchronized improvement across brain and body systems suggests the intervention may have facilitated a higher-order integration within the neurovisceral network, potentially by enhancing the regulatory capacity of the prefrontal cortex over limbic-autonomic circuits, thereby producing concurrent benefits for mood, cognition, and physiological arousal.
The question of mechanism, while beyond the definitive scope of this initial clinical study, invites speculation based on the known biophysics of mid-infrared light and existing photobiomodulation literature. Unlike the shorter wavelengths used in most photobiomodulation research (e.g., 810 nm NIR, which acts primarily on mitochondrial cytochrome c oxidase), mid-infrared light (5000–7000 nm) is strongly absorbed by vibrational modes of chemical bonds in water and organic molecules like proteins. The non-thermal biological effects of this spectrum are thought to involve the direct transfer of resonant energy, potentially promoting the vibrational excitation of amide bonds in proteins and activating structured water molecules within cells. This could influence protein conformation, enzymatic activity, and cellular signalling cascades in ways distinct from NIR mechanisms. While no direct evidence yet links these specific bioeffects to psychiatric outcomes, it is plausible that such fundamental cellular actions could enhance bioenergy metabolism and cellular resilience in neural and glial tissues. Given the growing recognition of metabolic and inflammatory dysregulation in the pathophysiology of mood disorders, mid-infrared light may exert its beneficial effects by acting on these upstream, shared physiological pathways, thereby creating a cellular environment more conducive to neural plasticity, efficient network communication, and autonomic stability. This putative mechanism aligns with our observed cross-system benefits and warrants dedicated future investigation.
When considering the translational potential of this intervention, its safety and practicality profile are noteworthy. The device used emitted non-ionizing radiation within a controlled spectral band, complied with international photobiological safety standards (IEC 62471:2006), and maintained a safe, non-thermal surface temperature. No adverse events were reported, consistent with the generally favourable safety record of non-thermal photobiomodulation. Furthermore, the intervention was non-invasive, portable, and required only a moderate time commitment (30 min daily), suggesting good potential acceptability, particularly in young adult populations who may be hesitant towards pharmacotherapy. Regarding durability, while this study was not designed to assess long-term effects, preliminary evidence from studies of NIR photobiomodulation suggests that psychological and physiological benefits can persist for weeks after the cessation of treatment. Whether mid-infrared light induces similarly sustained changes is a crucial question for future research with extended follow-up periods.
It is essential to acknowledge the limitations of this exploratory trial. The sample was recruited from a single educational institution and was restricted to a narrow age range (18–22 years), which may limit the generalizability of the findings to other age groups or clinical populations with diagnosed major depressive disorder. The intervention period was relatively short at two weeks, sufficient to demonstrate acute effects but not to evaluate long-term efficacy or relapse prevention. The study also lacked detailed behavioural performance metrics from the Stroop task (e.g., conflict-specific reaction time costs), which would have strengthened the interpretation of the fNIRS connectivity data by directly linking neural efficiency changes to behavioural outcomes. Furthermore, while the multi-modal approach is a strength, the precise biological cascade initiated by mid-infrared light in the human brain remains to be elucidated and would benefit from complementary methodologies such as metabolomic profiling or advanced neuroimaging.Future trials would also benefit from incorporating more detailed cognitive and behavioural measures, such as those outlined in comprehensive trial protocols for light-based interventions [42], to directly link the observed neural changes to performance outcomes.
In conclusion, this randomized controlled study provides the first integrated empirical evidence that mid-infrared light intervention can positively impact the complex interplay of mood, cognition, and autonomic function in young adults with subclinical psychological distress. By demonstrating specific improvements in depressive symptoms, a normalization of task-related prefrontal connectivity suggestive of enhanced cognitive efficiency, and a rebalancing of autonomic activity toward greater parasympathetic tone, the findings position mid-infrared photobiomodulation as a promising novel, non-invasive therapeutic modality. The coherent pattern of results across disparate measurement domains strongly supports the Neurovisceral Integration Model and suggests the intervention may work by promoting system-wide functional integration. While further research is necessary to confirm these effects in larger, more diverse samples, to establish optimal dosing parameters, to elucidate the molecular mechanisms, and to evaluate long-term outcomes, this study successfully bridges a gap between preclinical research and human application, offering a compelling foundation for the continued investigation of mid-infrared light as a potential tool for mental health and cognitive enhancement.
5. Conclusions
This randomized controlled trial provides initial evidence that a two-week Mid-infrared light intervention is associated with beneficial changes across psychological, neurocognitive, and physiological domains in young adults exhibiting subclinical psychological distress. The findings indicate that participants in the Screen-Positive Intervention group demonstrated significant reductions in self-reported depressive symptoms and general psychological distress compared to the attention-matched control group. Notably, these subjective improvements were paralleled by objective neurobiological changes: a post-intervention reduction in prefrontal functional connectivity during a cognitively demanding Stroop task—suggesting a normalization towards more efficient neural processing—and a shift in heart rate variability metrics (e.g., increased HF, RMSSD; decreased LF/HF ratio) indicative of enhanced parasympathetic tone and improved autonomic nervous system balance.
Taken together, this integration of mood assessment, functional near-infrared spectroscopy, and HRV offers the first multimodal, preliminary support for the biological and psychological effects of Mid-infrared photobiomodulation in a non-clinical population. The concordance across self-report, central neural, and peripheral autonomic measures strengthens the plausibility of a genuine intervention effect. Our study therefore, establishes a novel proof of principle, suggesting that Mid-infrared light may positively modulate the neurovisceral integration pathways implicated in emotional regulation and cognitive control.
Nevertheless, the exploratory nature of this study, constrained by its modest sample size, single-institution recruitment, and the limitations of the active control condition, necessitates that these conclusions be viewed as preliminary and hypothesis-generating. The healthy control group data, while helpful for characterizing baseline atypicality, cannot be used to infer efficacy. Crucially, our findings do not support direct clinical application at this stage.
Future research must prioritize confirmatory studies with larger, more diverse cohorts and longer follow-up periods. Pre-registration, fully blinded sham-controlled designs (using inert devices), and the inclusion of clinically diagnosed populations are essential next steps to validate and extend these results. Furthermore, mechanistic investigations should aim to elucidate the specific molecular and cellular pathways through which Mid-infrared light influences brain function and autonomic activity. By addressing these directions, the promising potential of this non-invasive intervention for mental health and cognitive performance can be rigorously evaluated.
Statement of the Use of Generative AI and AI-Assisted Technologies in the Writing Process
During the drafting of this manuscript, the author utilized Deepseek and ChatGPT to refine the article’s content and structure. Following the use of these tools, the author has thoroughly reviewed and edited the content and assumes full responsibility for the published material.
Acknowledgments
We would like to express our sincere gratitude to the teachers and students of the Rainbow Bridge Talent Program schools for their invaluable support in participant recruitment and study coordination. We are also thankful to the technical team involved in the development of “A sustainable far-infrared emitting rare-earth material and its preparation method”, whose foundational technology contributed to the mid-infrared intervention device. Furthermore, we acknowledge HuiChuang Medical Equipment Co., Ltd. for providing the functional near-infrared spectroscopy (fNIRS) system, and Chengdu Virtual Reality Dream Technology Co., Ltd. for supplying the portable, user-friendly heart rate variability (HRV) monitoring devices used in this study.
Author Contributions
Concept Design: M.Z., D.Z. and J.X.; Methodology: C.W. and D.Z.; Software Development: J.X. and C.W.; Validation Work: J.X., J.Y. and X.F.; Research Investigation: J.X., X.F. and J.Y.; Data Organization: J.X.; Drafting—Initial Draft Preparation: J.X.; Drafting—Review and Editing: J.X.
Ethics Statement
This study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of South China Normal University (protocol code: SCNU-PSY-2021-151, date of approval: 20 July 2021).
Informed Consent Statement
Informed consent has been obtained from all research subjects.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Funding
This research received no external funding.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
-
McLaughlin KA, King K. Developmental Trajectories of Anxiety and Depression in Early Adolescence. J. Abnorm. Child Psychol. 2015, 43, 311–323. DOI:10.1007/s10802-014-9898-1 [Google Scholar]
-
Dwyer JB, Stringaris A, Brent DA, Bloch MH. Annual Research Review: Defining and treating pediatric treatment-resistant depression. J. Child Psychol. Psychiatry 2020, 61, 312–332. DOI:10.1111/jcpp.13202 [Google Scholar]
-
Zivin JA, Albers GW, Bornstein N, Chippendale T, Dahlof B, Devlin T, et al. Effectiveness and Safety of Transcranial Laser Therapy for Acute Ischemic Stroke. Stroke 2009, 40, 1359–1364. DOI:10.1161/STROKEAHA.109.547547 [Google Scholar]
-
Schiffer F, Johnston AL, Ravichandran C, Polcari A, Teicher MH, Webb RH, et al. Psychological benefits 2 and 4 weeks after a single treatment with near infrared light to the forehead: a pilot study of 10 patients with major depression and anxiety. Behav. Brain Funct. 2009, 5, 46. DOI:10.1186/1744-9081-5-46 [Google Scholar]
-
Cassano P, Cusin C, Mischoulon D, Hamblin MR, De Taboada L, Pisoni A, et al. Near-Infrared Transcranial Radiation for Major Depressive Disorder: Proof of Concept Study. Psychiatry J. 2015, 2015, 352979. DOI:10.1155/2015/352979 [Google Scholar]
-
Chung H, Dai T, Sharma SK, Huang YY, Carroll JD, Hamblin MR. The Nuts and Bolts of Low-level Laser (Light) Therapy. Ann. Biomed. Eng. 2011, 40, 516–533. DOI:10.1007/s10439-011-0454-7 [Google Scholar]
-
Vatansever F, Hamblin MR. Far infrared radiation (FIR): Its biological effects and medical applications. Photonics Lasers Med. 2012, 1, 255–266. DOI:10.1515/plm-2012-0034 [Google Scholar]
-
Do A, Li VW, Huang S, Michalak EE, Tam EM, Chakrabarty T, et al. Blue-Light Therapy for Seasonal and Non-Seasonal Depression: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Can. J. Psychiatry 2022, 67, 745–754. DOI:10.1177/07067437221097903 [Google Scholar]
-
Meesters Y, Dekker V, Schlangen LJ, Bos EH, Ruiter MJ. Low-intensity blue-enriched white light (750 lux) and standard bright light (10 000 lux) are equally effective in treating SAD. A randomized controlled study. BMC Psychiatry 2011, 11, 17. DOI:10.1186/1471-244X-11-17 [Google Scholar]
-
Hamblin MR. Mechanisms and applications of the anti-inflammatory effects of photobiomodulation. AIMS Biophys. 2017, 4, 337–361. DOI:10.3934/biophy.2017.3.337 [Google Scholar]
-
Lapchak PA. Taking a light approach to treating acute ischemic stroke patients: Transcranial near-infrared laser therapy translational science. Ann. Med. 2010, 42, 576–586. DOI:10.3109/07853890.2010.532811 [Google Scholar]
-
Tsai SR, Hamblin MR. Biological effects and medical applications of infrared radiation. J. Photochem. Photobiol B Biol. 2017, 170, 197–207. DOI:10.1016/j.jphotobiol.2017.04.014 [Google Scholar]
-
Liu X, Qiao Z, Chai Y, Zhu Z, Wu K, Ji W, et al. Nonthermal and reversible control of neuronal signaling and behavior by midinfrared stimulation. Proc. Nat. Acad. Sci. USA 2021, 118, e2015685118. DOI:10.1073/pnas.2015685118 [Google Scholar]
-
Tsai JF, Hsiao S, Wang SY. Infrared irradiation has potential antidepressant effect. Progr. Neuro-Psychopharmacol. Biol. Psychiatry 2007, 31, 1397–1400. DOI:10.1016/j.pnpbp.2007.06.006 [Google Scholar]
-
Takizawa R, Fukuda M, Kawasaki S, Kasai K, Mimura M, Pu S, et al. Neuroimaging-aided differential diagnosis of the depressive state. NeuroImage 2014, 85, 498–507. DOI:10.1016/j.neuroimage.2013.05.126 [Google Scholar]
-
Boas DA, Elwell CE, Ferrari M, Taga G. Twenty years of functional near-infrared spectroscopy: introduction for the special issue. NeuroImage 2014, 85, 1–5. DOI:10.1016/j.neuroimage.2013.11.033 [Google Scholar]
-
Ho CSH, Lim LJH, Lim AQ, Chan NHC, Tan RS, Lee SH, et al. Diagnostic and Predictive Applications of Functional Near-Infrared Spectroscopy for Major Depressive Disorder: A Systematic Review. Front. Psychiatry 2020, 11, 378. DOI:10.3389/fpsyt.2020.00378 [Google Scholar]
-
Billman GE. The LF/HF ratio does not accurately measure cardiac sympatho-vagal balance. Front. Physiol. 2013, 4, 26. DOI:10.3389/fphys.2013.00026 [Google Scholar]
-
Brunoni AR, Kemp AH, Dantas EM, Goulart AC, Nunes MA, Boggio PS, et al. Heart rate variability is a trait marker of major depressive disorder: evidence from the sertraline vs. electric current therapy to treat depression clinical study. Int. J. Neuropsychopharmacol. 2013, 16, 1937–1949. DOI:10.1017/S1461145713000497 [Google Scholar]
-
Kemp AH, Quintana DS, Gray MA, Felmingham KL, Brown K, Gatt JM. Impact of Depression and Antidepressant Treatment on Heart Rate Variability: A Review and Meta-Analysis. Biol. Psychiatry 2010, 67, 1067–1074. DOI:10.1016/j.biopsych.2009.12.012 [Google Scholar]
-
Chen W, Zhong Q, Chen H, Chen S. Heart rate variability in children and adolescents with major depressive disorder: A systematic review and meta-analysis. J. Affect. Disord. 2023, 335, 204–215. DOI:10.1016/j.jad.2023.05.022 [Google Scholar]
-
Sakakibara E, Satomura Y, Matsuoka J, Koike S, Okada N, Sakurada H, et al. Abnormality of Resting-State Functional Connectivity in Major Depressive Disorder: A Study with Whole-Head Near-Infrared Spectroscopy. Front. Psychiatry 2021, 12, 664859. DOI:10.3389/fpsyt.2021.664859 [Google Scholar]
-
Mulcahy JS, Larsson DEO, Garfinkel SN, Critchley HD. Heart rate variability as a biomarker in health and affective disorders: A perspective on neuroimaging studies. NeuroImage 2019, 202, 116072. DOI:10.1016/j.neuroimage.2019.116072 [Google Scholar]
-
Thayer JF, Lane RD. Claude Bernard and the heart–brain connection: Further elaboration of a model of neurovisceral integration. Neurosci. Biobehav. Rev. 2009, 33, 81–88. DOI:10.1016/j.neubiorev.2008.08.004 [Google Scholar]
-
Kemp AH, Quintana DS, Felmingham KL, Matthews S, Jelinek HF, Hashimoto K. Depression, Comorbid Anxiety Disorders, and Heart Rate Variability in Physically Healthy, Unmedicated Patients: Implications for Cardiovascular Risk. PLoS ONE 2012, 7, e30777. DOI:10.1371/journal.pone.0030777 [Google Scholar]
-
Appelhans BM, Luecken LJ. Heart Rate Variability as an Index of Regulated Emotional Responding. Rev. General Psychol. 2006, 10, 229–240. DOI:10.1037/1089-2680.10.3.229 [Google Scholar]
-
Wang Y, Liang L, Sun Z, Liu R, Wei Y, Qi S, et al. Factor structure of the patient health questionnaire-9 and measurement invariance across gender and age among Chinese university students. Medicine 2023, 102, e32590. DOI:10.1097/MD.0000000000032590 [Google Scholar]
-
Group BMJP. Correction: Reliability and validity of the Chinese version of the Patient Health Questionnaire-9 (C-PHQ-9) in patients with psoriasis: A cross-sectional study. BMJ Open 2020, 10, e033211corr1. DOI:10.1136/bmjopen-2019-033211corr1 [Google Scholar]
-
Derogatis LR, Cleary PA. Confirmation of the dimensional structure of the SCL-90: A study in construct validation. J. Clin. Psychol. 1977, 33, 981–989. DOI:10.1002/1097-4679(197710)33:4<981::AID-JCLP2270330412>3.0.CO;2-0 [Google Scholar]
-
Pinti P, Merla A, Aichelburg C, Lind F, Power S, Swingler E, et al. A novel GLM-based method for the Automatic IDentification of functional Events (AIDE) in fNIRS data recorded in naturalistic environments. NeuroImage 2017, 155, 291–304. DOI:10.1016/j.neuroimage.2017.05.001 [Google Scholar]
-
Gullett N, Zajkowska Z, Walsh A, Harper R, Mondelli V. Heart rate variability (HRV) as a way to understand associations between the autonomic nervous system (ANS) and affective states: A critical review of the literature. Int. J. Psychophysiol. 2023, 192, 35–42. DOI:10.1016/j.ijpsycho.2023.08.001 [Google Scholar]
-
Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. Circulation 1996, 93, 1043–1065. DOI:10.1161/01.CIR.93.5.1043 [Google Scholar]
- Zhang D, Shen G. A Device for Promoting Human Oxygen Metabolism Based on Semiconductor Polycrystalline Material Acousto-Optical Energy Supply. CN202011288167.4, 23 July 2024. [Google Scholar]
-
Scarpina F, Tagini S. The Stroop Color and Word Test. Front. Psychol. 2017, 8, 557. DOI:10.3389/fpsyg.2017.00557 [Google Scholar]
- Benjamini Y, Hochberg Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. DOI:10.1111/j.2517-6161.1995.tb02031.x [Google Scholar]
-
Tao L, Jiang R, Zhang K, Qian Z, Chen P, Lv Y, et al. Light therapy in non-seasonal depression: An update meta-analysis. Psychiatry Res. 2020, 291, 113247. DOI:10.1016/j.psychres.2020.113247 [Google Scholar]
-
Danilenko KV, Lebedinskaia MY, Gadetskaia EV, Markov AA, Ivanova YA, Aftanas LI. A 6-day combined wake and light therapy trial for unipolar depression. J. Affect. Disord. 2019, 259, 355-361. DOI:10.1016/j.jad.2019.08.051 [Google Scholar]
-
Griffiths KR, Quintana DS, Hermens DF, Spooner C, Tsang TW, Clarke S, et al. Sustained attention and heart rate variability in children and adolescents with ADHD. Biol. Psychol. 2017, 124, 11–20. DOI:10.1016/j.biopsycho.2017.01.004 [Google Scholar]
-
Zhu H, Xu J, Li J, Peng H, Cai T, Li X, et al. Decreased functional connectivity and disrupted neural network in the prefrontal cortex of affective disorders: A resting-state fNIRS study. J. Affect. Disord. 2017, 221, 132–144. DOI:10.1016/j.jad.2017.06.024 [Google Scholar]
-
Gotlib IH, Joormann J. Cognition and Depression: Current Status and Future Directions. Ann. Rev. Clin. Psychol. 2010, 6, 285–312. DOI:10.1146/annurev.clinpsy.121208.131305 [Google Scholar]
-
Kiosses DN, Klimstra S, Murphy C, Alexopoulos GS. Executive Dysfunction and Disability in Elderly Patients With Major Depression. Am. J. Geriatr. Psychiatry 2001, 9, 269–274. DOI:10.1097/00019442-200108000-00011 [Google Scholar]
-
Li SX, Cheung FTW, Chan NY, Chan JWY, Zhang J, Li AM, et al. Effects of cognitive behavioural therapy and bright light therapy for insomnia in youths with eveningness: study protocol for a randomised controlled trial. Trials 2024, 25, 246. DOI:10.1186/s13063-024-08090-0 [Google Scholar]