Found 1 results
Open Access
Article
20 October 2025Divergent Aging Mechanisms of Calcium Arsenic Residue under Dry-Wet and Freeze-Thaw Cycles: Toxic Metal Mobility, Multiscale Physicochemical Characterization, and Escalated Ecological Risks
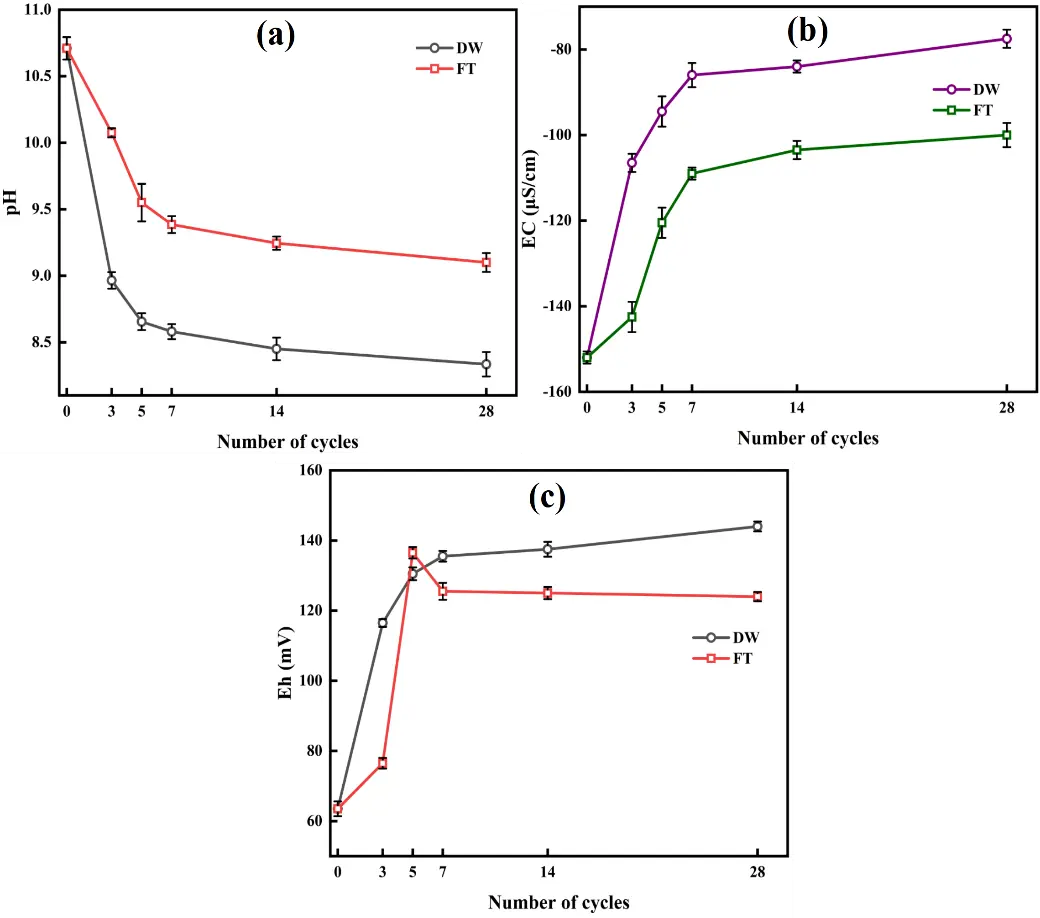
This study investigates the long-term mobility and ecological risks of As, Zn, and Cd in calcium arsenic residue (CAR) under simulated dry-wet (DW) and freeze-thaw (FT) cycles. Accelerated aging experiments, combined with multiscale characterization (XRD, XPS, SEM, FTIR), revealed distinct transformation mechanisms. DW cycles promoted carbonate-driven dissolution, As(III) oxidation to As(V) (resulting in an 18.4% increase in As(V) as shown by XPS), and sulfide oxidation (with reductions of 47.7% in ZnS and 15.08% in CdS). These processes increased the acid-soluble metal fractions (F1: As by 11.3%, Zn by 6.0%, and Cd by 8.7%) and metal release rates (52.39% for As, 42.63% for Zn, and 68.55% for Cd under DW conditions). In contrast, FT cycles induced mechanical fracturing and ice-mediated stabilization, which limited ion migration, partially amorphized ZnO, and promoted the precipitation of Cd(OH)2. Ecological risk assessments indicated rising risks, with integrated potential ecological risk indices (IPER) reaching 11,187.85 under DW conditions and 10,668.29 under FT conditions, with arsenic contributing over 80%. The Risk Assessment Code (RAC) reclassified all metals into moderate-risk categories (As: 11.9–19.7%, Zn: 9.4–15.2%, Cd: 12.1–18.6%). Weibull modeling (α = 6.98–10.98, R2 > 0.96) described the nonlinear kinetics, showing that cadmium aged the fastest (λ: Cd > As > Zn), with delayed but persistent risks under FT conditions. These results underscore the importance of developing climate-resilient stabilization strategies. The integrated framework combining mineral evolution, kinetics, and risk forecasting offers significant insights for managing legacy CAR pollution under changing climate conditions.