Found 1 results
Open Access
Review
04 March 2026Advances in the Application of CRISPR/Cas Systems in Molecular Diagnostics
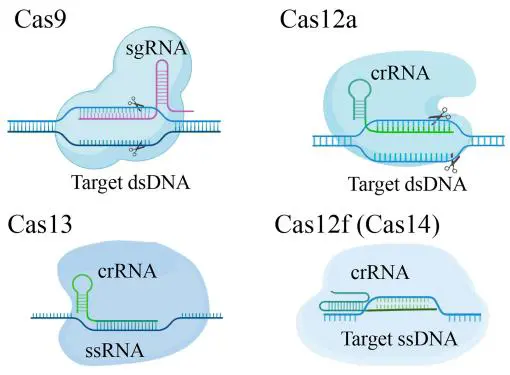
Rapid advances in CRISPR/Cas systems and the growing global demand for rapid, accurate diagnostics underscore the necessity of reviewing how these technologies are transforming molecular testing. Conventional diagnostic approaches are frequently constrained by prolonged turnaround times, complex instrumentation, and limited analytical sensitivity, and these limitations were starkly highlighted during the COVID-19 pandemic. In this context, we present a comprehensive and timely overview of CRISPR/Cas-based molecular diagnostics. We begin by summarizing the classification and molecular mechanisms of CRISPR/Cas types I–VI, followed by a detailed discussion of innovative detection strategies such as SHERLOCK, DETECTR, and amplification-free platforms that significantly enhance analytical sensitivity and specificity. We further explore clinical applications across infectious disease surveillance, antimicrobial resistance profiling, early cancer detection, genetic variant identification, and the emerging detection of non-nucleic acid biomarkers. Finally, we discuss future perspectives, including the development of miniaturized, high-throughput, and AI-assisted diagnostic platforms, their integration with microfluidics and portable readout systems for point-of-care applications, and highlight critical challenges such as standardization, automation, and cost-effectiveness that must be addressed to facilitate clinical translation.