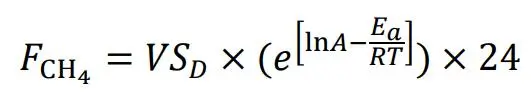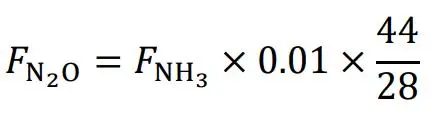*1.1. Sludge Management—Theory and Current Practice*
Denmark produced 5.6 million m
3 of milk in 2020, with each m
3 milk resulting in between 0.75 and 1.5 m
3 effluent depending on the product [
1]. Most of this effluent is treated by the activated sludge treatment, whereby multiple treatment tanks with high concentrations of microorganisms convert organics and nutrients in the wastewater into microbial biomass, which can then be separated from the water [
2]. The product of this treatment is an activated sludge, which can be separated from the wastewater by filtration, while the present ‘clean’ wastewater is discharged to a local water body. In Denmark, approximately two thirds of the activated sludge is applied to the cultivated land, with the remaining third utilized in biogas production [
1]. The amount of sludge produced depends on the milk derived product at the dairy, and quantities produced depend on the biological oxygen demand (BOD) of the effluent. Roughly one-kilogram BOD generates 0.6 kg dry activated sludge [
3], and the average, mixed-product dairy produces 0.2–6.0 kg BOD per m
3 effluent [
4].
According to current EU practices, dairy sludge is considered as a biosolid and therefore can be spread on the agricultural land as it is rich in plant nutrients required for healthy plant and animal growth [
2]. This is specifically the case for phosphorus (P) with dairy effluent being one of the main sources of P-rich industrial effluent in EU [
4,
5,
6]. P is a finite non-renewable resource, and the EU is highly dependent on external imports. The P in dairy sludge is therefore included in the Circular Economy Policy of the EU [
7], as by recycling to the fields it is possible to substitute mineral P fertilizer from mined rock phosphate [
8]. While the principle of recycling dairy sludge is noble, the applications of excessive amounts of manure, slurries and sludge to soil bring about the accumulation of nutrients in soils that can go on to pollute surface waters and groundwater [
9]. In Denmark, national P application limits impose restrictions on P application to land, with these values varying depending on current P loading and soil type [
10]. The high nitrogen (N) content of the sludge (nitrate (NO
3−), nitrite (NO
2−), ammonia (NH
3) and organically bound N) also impose limits due to EU Nitrates Directive 91/676/EEC. To limit runoff, agricultural best practice states that biosolids should not be applied to the agricultural land if the soil is waterlogged, flooded or snow covered, or if it has been frozen for more than 12 h out of the previous 24 h. This limits sludge application during winter months, while the crop cover prevents application during summer months. This therefore limits land application during the spring and autumn, but given that the sludge is produced continually, it brings about a requirement to store the material for the interim period. Furthermore, dairies are situated in areas with high animal density, and with the likely tightening of national P and N limits in accordance with the water framework (2000/60/EC), application to land in the immediate vicinity of the dairy could pose challenges when moving forward. This will most likely result in transport of the material to areas dominated by arable farming, for example from Jutland to Zealand in Denmark, but transport of this sludge would be costly and carbon (C) intensive as the material often has a high-water content (>85%), even when filtered by the high end technology such as decanter centrifuges.
When transporting and storing dairy sludge, the moisture content is of high importance. In activated sludge the present water can be distributed in four different forms: (i) free water which can typically be removed by gravity settling, (ii) interstitial water which is the water trapped between particles within the flocculated sludge structure and within microbial cells, (iii) vicinal water which is water held tightly to sludge surfaces via hydrogen bonding, and (iv) chemically bound water (metal hydroxides etc.) [
11]. Free and interstitial water can be removed by mechanical dewatering using technology such as centrifuges, screw presses or filter presses [
12] to increase dry matter to approximately 15% (i.e., 85% water) but will often involve considerable expense for the plant operator [
13]. Further reductions in moisture is, however, challenging as activated sludge has a very high surface area and a prevalence of hydroxyl functional groups which form hydrogen bonds with water [
14], resulting in a high vicinal water content, which can usually only be removed by thermal drying [
11]. Drying sludge is energy intensive, requiring 2.3 MJ of energy to raise 1 kg of water from 20 to 101 °C (g). Therefore, to dry one cubic meter of dewatered sludge to 15% dry matter (DM), 2000 MJ of energy is required to produce just 150 kg of dried sludge and is therefore best avoided. Issues, however, arise when the material is stored since the high moisture content within the sludge after mechanical compression brings about anaerobic conditions and the formation of methane (CH
4), a potent greenhouse gas (34 kg CO
2eq), and NH
3 as an indirect source of nitrous oxide (N
2O) (298 kg CO
2eq) in the atmosphere [
15]. Furthermore, when transporting sludge for the purpose of recycling P, haulage is constrained by both mass and volume, and carbon dioxide (CO
2) is emitted from fossil fuel used for transport of the sludge, which may be six times higher on account of the moisture.
. Schematic of water types in activated sludge taken from Syed-Hassan et al. [
11].
Besides the application to land without treatment, anaerobic digestion (AD) of dairy sludge for biogas production is common in Denmark [
1]. The advantages of AD include sludge volume reduction, savings on solid management costs, and production of CH
4 for energy recovery which will reduce fossil fuel consumption and CO
2 emissions. Personal communications with Arla Foods wastewater facility at Vium (Denmark), however, suggest that the sludge produced at this facility contains low concentrations of degradable organic matter and therefore has limited biogas potential. The sludge from Arla Foods Vium Dairy is primarily utilized by biogas plants as it has a high iron content, desirable for abating hydrogen sulfide emissions [
16]. Therefore, the sludge has reasonably limited biogas potential. This low biogas potential is not necessarily surprising given that the activated sludge process uses aerobic micro-organisms that can digest organic matter in sewage, flocculating together as they do so. This will remove most of the easily digestible material which anaerobic microorganisms would otherwise convert to CH
4. Gavala et al. [
17] estimated that CH
4 would yield 2.5 times lower than the equivalent volatile solids loading in primary sludge (organic matter which can be recovered without the activated sludge process). The low CH
4 potential and increasing handling costs of digestate (slurry from the biogas plant) due to tightening restrictions on environmentally friendly management in accordance with the water framework directive (2000/60/EC), may reduce the longer-term viability of this treatment method.
*1.2. Hydrothermal Carbonisation*
In this work we propose a third treatment method, Hydrothermal Carbonization (HTC). HTC works using pressurized high temperature liquid water (up to 250 °C) to mimic natural coal formation, and convert organic material within the sludge to a coal like the material called hydrochar [
18]. For high moisture slurries such as dairy sludge, the HTC process enables a hydrothermal dewatering process, due to the reduced polarity of the hydrochar, making it hydrophobic and water-repelling. In the process, water within the sludge will deoxygenate, flocculate and mineralize C. Therefore, organic compounds are fragmented by hydrolysis, and the compounds become aromatic through intermolecular dehydration and aldol condensation. The aromatic components then go through intramolecular dehydration and keto-enol tautomerism to become a large aromatic macromolecule with a hydrophobic core [
19]. These macromolecules, known as a hydrochar, have a smaller surface area and the dominant dehydration reaction removes a large portion of the hydroxyl groups [
20], which form hydrogen bonds with water [
14]. Therefore, through reduced surface area and reduced hydrogen bonding, vicinal water content is reduced [
11]. This means most of the interstitial and vicinal water within the dewatered sludge becomes free water in the sludge derived hydrochar, enabling easy removal of 80–90% of the remaining water.
To achieve this, HTC requires reasonably limited energy input as the system uses elevated pressure to raise the boiling point of water and avoid latent heat of vaporization. The energy requirement is only 0.9 MJ/kg water to heat the water to 250 °C, and 80–90% of this energy input can be recovered via exchangers and used in the process [
21]. Therefore, treatment requires 70–140 MJ/ton heat input as opposed to 2000 MJ/ton for thermal drying. The net carbonization reaction should also be exothermic, albeit specific heat values for dairy sludge are currently unavailable. Nonetheless several studies have been undertaken by high pressure Differential Scanning Calorimetry (DSC). Funke and Ziegler [
22] measured at 240 °C and found 1.06, 1.07 and 0.76 MJ/kg heat release for glucose, cellulose and wood. Pecchi et al., [
23] reported 1.31, 0.96 and 0.49 MJ/kg heat release for cellulose and wood and digestate at 250 °C. Ischia et al., [
24] noted 0.86, 1.33, 1.53 MJ/kg heat release for glucose, cellulose and grape seed. Thus, energy losses through heat loss should be offset by the heat of the reaction.
In addition to hydrochar, HTC also produces aqueous co-products within the additional free water liberated by the HTC process (aqueous fraction) and a gaseous phase which typically is made up of CO
2. The amount of C within this aqueous fraction depends on the feedstock and the processing parameters but typically contains between 10% and 20% of the original C. HTC is routinely undertaken on sewage sludge, but given that dairy sludge is almost purely derived from milk, extrapolating sewage data to dairy should be done with caution. To the best of our knowledge, treating dairy sludge via HTC is limited to Atallah et al. [
25]. In this study, 22% of the feedstock C ends in the aqueous phase while undertaking HTC at 225 °C with two-hour retention time and a water content of 82%. 70% of the C is transferred to the hydrochar. In this run the aqueous product is found to be made up of acetic acid, propanoic acid, hexanoic acid, octanoic acid, pentanamine, butanoic acid and a large portion of unknowns which are likely furfural and furfural derivatives [
26]. In the envisioned setup, HTC would be undertaken at the water treatment plant, so this aqueous fraction will be recirculated back into the buffer tanks before being digested in the activated sludge tanks. Nonetheless this aqueous fraction could have potential inhibitory effects on the activated sludge treatment and will likely contain approximately 50% of the N from the original feedstock [
27]. In the activated sludge tank ammonia-oxidizing bacteria may transform a portion of N to N
2O [
28], albeit the acidic conditions and polyphenols present may limit any release [
29,
30]. To overcome this, it is proposed that the process water be treated via a post carbonization wet air oxidation reaction, where high pressure air is injected into a hydrothermal reaction. Under these conditions oxygen dissolves into the water and reacts with organic substances to produce CO
2 via organic acid intermediates in an exothermic reaction, destroying potentially problematic compounds and generating heat [
26,
31,
32]. By treating the process water with this method, it is expected that any additional heat requirements of the process will be met [
31,
32], and complex and potentially inhibitory organic compounds will be degraded to smaller and less problematic intermediates that can be treated via activated sludge treatment [
26]. During wet oxidation a large proportion of ammonium within the process water, will be released as NH
3 [
33]. Incorporation of N-scrubber as a form of gas treatment enables this NH
3 to react with a sulphuric acid solution, forming ammonium sulphate, which could constitute a marketable product (base fertilizer) [
34]. A schematic of the proposed reactor setup is given in .
. Process schematic of HTC plant.
The hydrochar in this application is intended to be used as a P-fertilizer and a soil conditioner due to the hydrochar retaining most of the P, (due to its high iron content [
35,
36]), 50% of the N [
27] and most of the C. Soil applications of hydrochar are not new; the technology obtains notoriety as an alternative biochar production route to pyrolysis due to higher C yields and greater feedstock flexibility, including the use of wet feedstocks, while still being simple, cheap and scalable. Pyrolytic biochar typically requires dry and lignin-rich feedstocks for good quality char, while HTC utilizes more carbohydrate chemistry [
18,
37]. Nonetheless, phytotoxic compounds generated as reaction intermediaries result in poor performance of crops in initial growth trails and taint this application [
38,
39]. These phytotoxic properties are now known to be short lived [
40], and with improved understanding of the process, can be removed by either simple post-treatments, such as wet oxidation or optimized process control [
39,
40,
41]. Nonetheless, the C in hydrochar is typically less recalcitrant than biochar produced by pyrolysis. During pyrolysis a dry biomass or biosolid is heated in the absence of oxygen, which leads to the thermal decomposition of non-aromatic compounds such as cellulose and hemicellulose, which are lost as flammable volatile compounds. The biochar fraction is largely formed from aromatic fragments from the lignin (80%), albeit with some cellulose derived aromatics produced due to dehydration of compounds such as levoglucosan (20%). This gives biochar a largely aromatic structure [
42,
43]. In hydrochar, a greater proportion of the C is reformed C from the aliphatic C. Aromatization reactions are often time limited, with shorter retention times and lower reaction temperatures imposing kinetic limitations that limit aromatization and repolymerisation [
27,
44]. This means that hydrochars typically have a less stable structure, with reduced aromaticity and presence of alkyl moieties, whereas biochars are dominated by aromatics. Therefore, it is highly probable that the residence time of hydrochars in soil is low compared to biochars [
45]. In contrast, nutrient retention capacity in the soil is higher in hydrochars compared to biochars due to the presence of the alkyl moieties which give surface functionality, whereas the highly aromatic biochar has almost complete loss of functionality [
45]. Therefore, while a biochar may be better for C sequestration, the hydrochar will offer benefits in terms of soil functioning and nutrient retention.
*1.3. Predictions of Carbon Recalcitrance in Dairy Derived Biosolids*
Predictions of C recalcitrance in the soil is difficult with few studies analyzing C, because the annual changes of biochar content are too small for any practical experimental period (months to few years) [
46]. However, the aromatic structure is quite closely correlated to the materials fixed C content when analyzed by proximate analysis. Thus, the fixed C is closely related to stable C content [
47]. Therefore, the fixed C yield of biochar is often used within biochar literature to show biochar's potential as a C sequestration material from the environmental point of view; higher yield means its higher potential as a climate change mitigation tool [
47]. Results presented in Atallah et al. [
25] show the fixed C content of dairy sludge was very low, 1.5%, which would imply very low C sequestration potential. Depending on HTC processing temperatures, the fixed C can be increased to 32%, which would imply a 20 fold increase in C sequestration potential, while the overall C content is only reduced by 30% and the dry matter (DM) by 50%.
The use of proximate analysis originates from combustion research, from which most of the pyrolysis research originate. Therefore, it is less commonly used in biosolids and the direct comparison between sequestration potential of biosolids and biochar is difficult. In addition, the role and function of the C is considered very differently. Biochar is intentionally inert and while it brings advantages to the soil, the advantages are mostly associated with the component’s porosity, which are beneficial in terms of water retention, nutrient cycling, aeration, and alkaline pH [
48]. Bioslurries are not inert and their C sequestration mechanism involves increasing the liable C pool resulting from the addition of fresh residues such as plant roots and living organisms. This increases soil organic matter and C cycling, but the biosolids-derived C in soils is still unlikely to stay for long periods. Therefore, it is not sequestered in the same sense as biochar. Assessing sequestering potential is very challenging without techniques such as
14C isotope labelling [
46]. Furthermore, obtaining sequestration values beyond a few years is problematic, with many longer-term sites in the literature former mining sites which have limited biological activity before biosolids. Long-term studies on stable organic soils are more limited. Li and Evenylo [
49] observed two sites with a single application of anaerobically stabilized biosolids on stable clay loam (1984) and sandy loam (1996) agricultural soils and found 5% to 10% C retention after 27 and 16 years respectively. AD appears to increase the C sequestration potential of biosolids, although the results presented in Thomsen et al. [
50] suggest that increases in C sequestration after AD are proportional to decrease in organic matter. Thus, it is suggested that AD converts the more liable C in the original feedstock to CH
4. If the material is added to land, it would be simply converted to CO
2 in the soil. The organic material used by Thomsen et al. [
50] has a relatively high lignin content in the feeds and digestate, with lignin content closely correlated to fixed C content when undertaking proximate analysis [
51] and fixed C is closely related to stable C content [
47]. Nonetheless, the lignin portion of the C retention reported in Thomsen et al. [
50] will only account for approximately 50% of the C fraction, so the cycling of the volatile fraction is also present. Based on these studies, it is possible to state that C stability is as follows: raw organic material > biosolids > anaerobically digested biosolids > hydrochar > biochar.
*1.4. Study Aims, Objectives and Assumptions*
This study is a simulation study which models three hypothetical treatments of GHG emissions in the context of Arla Foods wastewater facility at Vium, Denmark. Within these three scenarios CH
4 and N
2O are produced during sludge management and the ammonia (NH
3) is emitted when deposited to the land which contributes to N
2O in the atmosphere. The CO
2 is emitted from fossil fuel used for transporting and processing the sludge. And biogas production contributes to reducing fossil fuel consumption and thus CO
2 emission. Recalcitrant C added to the soil in sludge, digestated from AD or in biochar will reduce atmospheric CO
2 as shown in .
. Flowchart conceptualizing the management of secondary sludge from a dairy sewage treatment plant.
Hydrochar produced by hydrothermal treatment will enhance dewatering with low energy consumption and the large reduction in content of water will reduce transport energy consumption. Emission of CH
4 and N
2O during storage and from the applied hydrochar may be a source of GHG, but most studies known to us indicate that the stored biochar is not a source of GHG emissions, such as N
2O emission, when field is applied [
52,
53,
54]. This management chain will be compared to GHG emissions and balances of the two reference systems where the sludge is managed untreatedly or used for biogas production. The procedure of Danish Centre of Environment and Energy is used when the emission of GHG from livestock manure is calculated.
*2.1. Nitrous Oxide and Methane Emissions*
In the scenario figure, the flow of C and N is presented in three sludge management systems (). CH
4, NH
3 and N
2O are emitted in these processes. After deposition, NH
3 contribute to N
2O emissions, and this fraction is included in the calculation of GHG emission. One-hundred-year global warming potentials (GWP
100) for CH
4 are set to 34 kg CO
2eq per kg CH
4 and N
2O to 298 kg CO
2eq per kg N
2O [
55]. In addition, the C being stored in soil is included in the calculations.
In the calculation, it’s assumed that one ton of annual produced sludge (Untreated or anaerobic digested) in each month (1/12) is transferred to a store and the temperature is like the monthly average air temperature. The tanks are emptied in the start of April and the sludge is applied to the fields.
*2.2. Methane Emission and Org-C Transformation during Anaerobic Digestion and Storage*
Methane production during AD is calculated as given by Baral et al. [
55], and shown in Equation (1) where *P*
CH4 is the CH
4 production (kg). The input of volatile solids *VS* to a digester is set to 73% (SD = 5) of DM ( and ), *B*
0 (BMP) is 270 mLCH
4 g·VS
−1 (min = 97 mLCH
4 g·VS
−1, max = 427 mLCH
4 g·VS
−1). The ε is a conversion efficiency factor set to 0.9 to account for the fact that actual CH
4 yields are typically lower than *B*
0, and 0.67 converts from volume (m
3) to mass (kg) of CH
4 [
55]. Digester leakage is assumed to be 3% of CH
4 produced:
The amount of VS transformed during AD is calculated based on the assumption given in Equation (2), where *B*
CH4 is the methane produced in gram. By the use of Bushwell stoichiometric calculation of anaerobic transformation of organic components, it is calculated that 2.64 g VS is transformed into per 1 g CH
4 produced [
56]. Using this equation the amount of VS degraded during AD can be calculated.
Methane emissions from stored liquid sludge are also calculated according to Baral et al. [
55], as shown in Equation (3), where *F*
CH4 is CH
4 emission rate (g CH
4 kg
−1 VS day
−1), A is the pre-exponential factor (g CH
4 kg
−1 VS h
−1) and *E
a* is the apparent activation energy (J·mol
−1) giving the temperature response of CH
4 production. Then, *R* is the gas constant (8.314 J·mol
−1·K
−1), *T* is teperature (K), and *E
a* value is set to 81 kJ·mol
−1. For secondary sludge ln(*A*) value is set to 31.2 g CH
4 kg *VS
D*
−1 h
−1 [
55] and *VS
D* is 56% of VS (). For digested sludge, ln(*A*) is set to 27.9 and VS concentration is used in the calculation [
57], because to our knowledge no model can assess transformation of *VS
D* during AD of sludge.
The assumption is that no CH
4 is emitted from stored biochar however this assumption requires experimental validation and at present literature on this appears unavailable.
Table 1. Composition of sludge from Arla Foods Fællesrenseanlæg’s.
Table 2. Greenhouse gas balance for the three systems being analyzed, where one-ton sludge from the centrifuge is managed during one year (Negative values are net emission and positive numbers are net reduction in emissions).
*2.3. Fossil Fuel Substitution and CO
2 Emission Transport*
Methane produced during AD is converted to energy, assuming 53.43 MJ/kg CH
4 [
55]. Biogas will be introduced into the natural gas grid, and it is assessed that burning of the substituted natural gas produces 0.058 kg CO
2/MJ (47 MJ per kg gas, 1 kg gas ≈ 62.5 mol, this corresponds to 2750 g CO
2, i.e., 2.750 kg CO
2/47MJ = 0.058 kg CO
2/MJ).
Emissions of CO
2 during one-way transportation (2.7 kg CO
2/L diesel) of digestates were calculated, assuming a fuel consumption of 0.0265 L diesel per Mg and km [
55]. Our assumption is that the one-way transport distance of sludge is 50 km.
The sludge is heated in HTC due to the wet oxidation of C within the process water, in which it is estimated that 30% of C [
25] is lost in form of CO
2. The CO
2 is part of the plant uptake, feeding animal, milk etc. circulation and is assumed not to contribut to net climate warming.
*2.4. Nitrogen Transformation and NH
3 Emission*
During anaerobic degradation of the sludge, 34% of the proteins are transformed [
58] and it is assumed that it is transformed to total ammoniacal nitrogen (TAN = NH
4+ + NH
3).
Emission of NH
3 from stores are set to 2.6% of TAN and of sludge injected into soil to 2% of TAN [
57]. The negative charge of biochar will contribute to reducing NH
3 emission soil to which hydrochar is applied [
53], but we don’t account for this process.
During HTC treatment, 50% of N is retained in the char and 50% enters into aqueous phase [
27] with subsequent recovery as ammonium sulphate. Gaseous NO
x or NH
3 will either bind to the char or end in the water.
*2.5. Carbon Retention*
The C retention for biosolids is set at a timescale of 20 years, so 7% C retention of sludge-C is chosen based on values in Li and Evanylo, [
49]. A value of 10% is given for the AD treated biosolids as the recalcitrant fraction will likely increase with the volatile solid’s reduction [
50].
Carbon in hydrochar is less recalcitrant than biochar, but more functional in the soil. The general assumption is that 50% will be long-term recalcitrant and the remaining 50% will degrade quite slowly through 10–20 year. In the calculation, it is assumed that 60% of C added to soil is recalcitrant. This value is slightly higher than the 50% recalcitrance in Naisse et al. [
59], and however the hydrochar should be more recalcitrant due to the higher fixed C contents yielded by higher processing temperature. 70% of the biochar carbon yields were assumed based on the findings of Atallah et al. [
25].
*2.6. N
2O Emission*
At deposition on land or in water a fraction of NH
3 will be transformed to N
2O and be emitted to the atmosphere. This indirect N
2O emission is estimated using the Equation (4), where *F*
N2O is given in kg N
2O, *F*
NH3 is given in kg NH
3-N emitted and 0.01 is a default factor given by the IPCC for calculation of the transformation of NH
3 to N
2O, and 44/28 is to be calculated from the concentration given in 2 × N g·mol
−1 to 2 × (N + O) g·mol
−1 [
60]:
In the Danish GHG emission inventory (Nielsen et al. 2018), it is assumed that 0.5% of total N is emitted in form of N
2O from stored liquid manure and sludge and 1% of total-N is emitted from sludge and digestate applied to soil, i.e., based on the standard emission factors given by IPCC (2006). We assume that N
2O emissions is 0.7% of total-N contained in hydrochar applied to soil. In recent studies, biochar and hydrochar have not increased N
2O from the “fertilized soil” when applied to field, neither do they when applied to composts [
52,
54,
61,
62]. There is no emission of N
2O during storage of biochar as it is proven in compost studies that biochar reduces N
2O production and emission [
63,
64].
*How Current Dairy Sludge Management Practices Influence Net GHG Emissions*
The results of this desktop study are shown in . The results demonstrate that treatment of sludge via AD can considerably reduce the GHG emission potential of the sludge, reducing the GHG footprint from 420 kg CO
2/ton to 160 kg CO
2/ton. This is principally due to reductions in the emissions of CH
4 from stored sludge, which is 7.3 kg from the untreated sludge () and 0.3 kg from the stored AD treated digestate, which contains less degradable C.
. Methane emissions arising during year-long storage of untreated dairy secondary sludge.
In AD this easily degradable C within the sludge is converted to CH
4, with one ton of sludge yielding 20.2 tons of CH
4 (). In the biogas plant, the CH
4 is combusted releasing power for the electricity grid and heat for district heating, and this substitutes 48.6 kg (CO
2) of fossil CH
4, which is regarded as a net reduction in emissions. A 3% CH
4 leakage is assumed for the anaerobic digester plant (0.6 kg), offering a penalty of 20.6 kg CO
2eq. Nitrous oxide emissions during storage and after application in the field are near similar for the two processes, as it is assumed that only limited N is removed in the anaerobic digestion process, but more NH
3 is emitted from the stored and applied digestate which contributes to the slightly lower emission than that from the untreated sludge emissions in storage (0.203 kg and 0.205 kg N
2O respectively, digestate and sludge) and 0.403 kg and 0.408 kg N
2O from field applied digestate and sludge, respectively. Emission of N
2O from deposition of emitted NH
3 is low, 0.005 kg N
2O from digestate and 0.001 kg N
2O from untreated sludge.
Nitrous oxide emission from hydrochar applied in the field is lower than that from sludge because N is reduced with 50%. In addition, the N
2O emission factor applied to the hydrochar is 0.7% as opposed to 1% used for the digestate and untreated sludge. This lower value is used as hydrochars and biochars can retain NH
3 making it inaccessible for nitrifiers, and studies have shown that hydrochar used as additives in composting can reduce N
2O emission [
52,
54,
61,
62]. The model assumes no storage emissions of CH
4 and N
2O as the material would be processed immediately after it passes through the centrifuge decanter. Reduction in mass from 1000 kg in sludge to 150 kg of hydrochar due to hydrothermal dewatering and deoxygenation reduces the emissions associated with transport.
. Methane yields, fugitive emissions and energy production arising from anaerobic digestion of dairy secondary sludge.
In terms of C sequestration, a higher recalcitrant fraction (10%) is awarded to the digestate, compared to the unprocessed sludge (7%), based on the findings of Li and Evanylo, [
49]. This is primarily because a larger portion of the biogas is produced through transformation of easily degradable C, and this increases the recalcitrant fraction within the remaining volatile solids [
50]. Therefore, a greater portion of the digestate will be recalcitrant in the reduced mass of VS to be added to the field. The unprocessed sludge will have a smaller recalcitrant C fraction, but a greater mass will be applied to a field. Therefore, the model predictes both applications would sequester almost similar masses of C, with 3.8 kgC/m
3 sludge and 3.6 kgC/m
3 sludge for the standard and AD treatments respectively. This is equated to 14.0 and 13.2 CO
2eq/ton respectively. For the hydrochar, it is assumed a 70% carbon yield as reported in Atallah et al. [
25] and there would be a greater recalcitrant fraction. Due to the aromatization reactions, as part of HTC chemistry, the amount of fixed C increases 20-fold (1.5% to 32.0% in Atallah et al. [
25]). Fixed C is closely related to stabile or recalcitrant C content [
47]. Therefore, in the calculation it is assumed that 60% of C added to soil in hydrochar is recalcitrant. Therefore 27.9 kg C/m
3 sludge will be long-term sequestered, offering a greenhouse gas reduction of 102.4 kg CO
2eq/ton of feed. This results in a higher C sequestration for the hydrochar than the two treatments where sludge is applied to the field. The hydrothermal treatment gives a positive GHG balance of 63.4 kg CO
2eq/ton of feed, unlike the other two treatments with negative footprints of 419.8 kg CO
2/ton and 156 kg CO
2/ton. In our calculation we do not include emissions from the wastewater treatment plant.
The HTC is a technologically simple sludge treatment that could lower the cost for dewatering dairy sludge, forming a biochar like material known as a hydrochar. That can be applied to land, for the purpose of carbon sequestration, P and N recycling. In this work, HTC is compared to two alternative sludge handling strategies; (i) whereby sludge is stored, transported to a field and then directly applied to land, and (ii) sludge is co-digested in AD and its digestate is applied to land. Our calculations indicate that GHG balances of HTC sludge management sequesters 63.4 kg CO2eq per ton sludge, and by contrast standard sludge management emits 420 kg CO2eq per ton sludge, and AD treatment 156 kg CO2eq per ton sludge. The HTC offers the greatest potential reduction in GHG emissions, primarily due to a reduction in predicted N2O emissions and a higher carbon stability of the biosolid. Without HTC, AD still offers considerable GHG reductions over direct application of the sludge to land. This is primarily due to AD reducing methane release during storage.
The authors would greatly acknowledge the inputs of Alan Rasmussen of Arla Foods, Søren O. Petersen for Aarhus University, and Ingeborg Frøsig Pedersen from Aarhus University, in the development of this concept.
Conceptualization, A.M.S., S.G.S. and A.T.-T.; Methodology, A.M.S., S.G.S. and A.T.-T.; Validation, A.M.S., S.G.S. and A.T.-T.; Formal Analysis, A.M.S. and S.G.S.; Investigation, A.M.S. and S.G.S.; Resources, X.X.; Data Curation, S.G.S.; Writing—Original Draft Preparation, A.M.S.; Writing—Review & Editing, A.M.S., S.G.S. and A.T.-T.; Visualization, S.G.S.
Not applicable.
Not applicable.
This research has received no direct funding. AMS hours while preparing this manuscript was funded by the Poul Due Jensen Fond rePair.
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.